Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Parasitic infections of the biliary tract are a common cause of biliary obstruction in tropical developing countries and less frequently, in developed countries. These infections are important because they can lead to serious complications such as cholelithiasis (see Chapters 33 and 39 ), recurrent pyogenic cholangitis (see Chapter 44 ), cirrhosis (see Chapter 74 ), pancreatitis (see Chapters 54 , 55 , and 57 ), and cholangiocarcinoma (see Chapters 50 and 51B ). The most common parasites of the biliary tract reported in humans are Fasciola, Opisthorchis, Clonorchis, and Ascaris spp. Opisthorchiidae (which includes species of Clonorchis and Opisthorchis ) and Fasciolidae (which includes species of Fasciola ) are two families that make up a category called liver flukes. The number of people affected by liver flukes worldwide is approximately 15.3 million for Clonorchis, 8.4 million for Opisthorchis, and 2.6 million for Fasciola . Although these flukes are unlikely to cause mortality, significant morbidity has been reported and calculated as disability-adjusted life years (DALYs) ( Clonorchis : 275,380 DALYs; Opisthorchis : 74,367 DALYs; Fasciola : 35,206 DALYs) , (see Chapter 71 ).
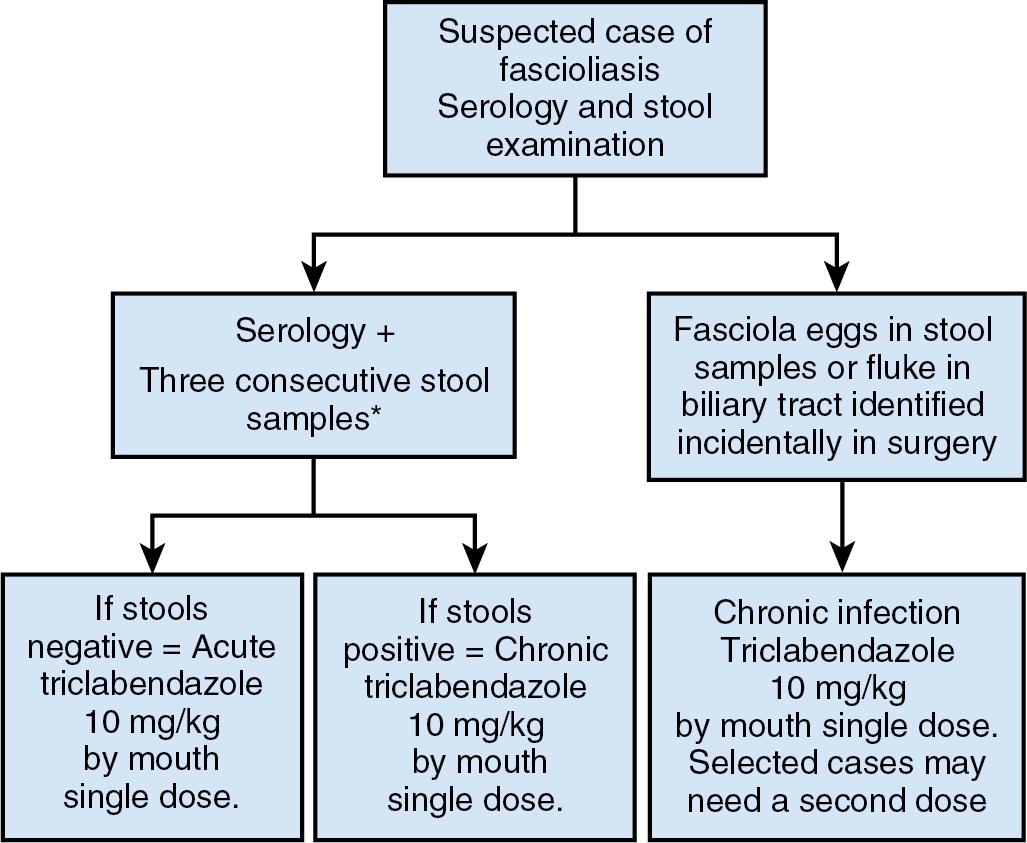
Fascioliasis, or distomatosis, is a zoonosis (disease that can be transmitted from animals to humans) caused by Fasciola hepatica or Fasciola gigantica (Trematoda: Fasciolidae). Fasciola is globally distributed and present in all continents except Antarctica, especially in sheep- and cattle-raising areas. F. hepatica and F. gigantica are mostly reported in temperate zones and tropical regions, respectively ( Fig. 45.1 ). In approaching a patient with suspected fascioliasis, epidemiologic, clinical, and imaging features can provide clues for the diagnosis before the physician orders a diagnostic test to confirm the infection. An algorithm for diagnosis and management is recommended ( Fig. 45.2 ).
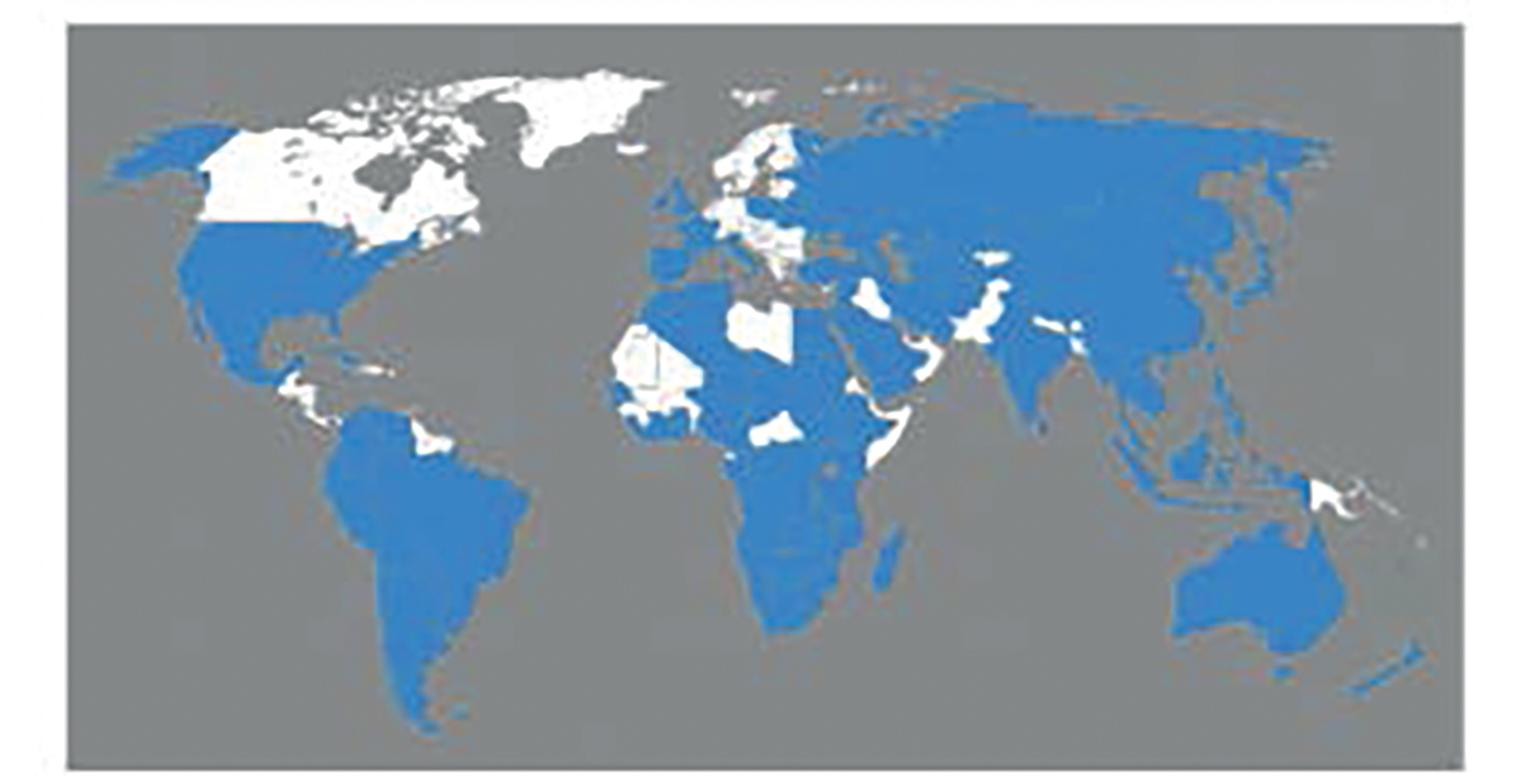
The first case of F. hepatica infection was documented in the Gallo-Roman period. Despite the recent developments in diagnostic and surveillance techniques, some countries are still lacking in data on human fascioliasis. This may be due to underreporting and underdiagnosis occurring in resource-limited regions of the world. Cases are mostly reported as a complication from the infection, and therefore the current denominator of patients with fascioliasis is unknown and likely underestimated. On the other hand, globalization and migration of populations from rural areas to large cities have led to cases of fascioliasis in areas where this fluke is typically not endemic. In fact, because of most clinicians’ lack of familiarity with this parasitic infection in nonendemic areas, the rate of complications may increase because of late diagnosis. Currently, the estimated number of human infections ranges from 35 to 72 million and 180 million are at risk of infection worldwide. In the past, fascioliasis was limited to specific and typical geographic areas, but it is now widespread throughout the world (see Fig. 45.1 ). According to the reported cases, F. hepatica transmission has increased in Europe, the Americas, and Oceania, as well as in Africa and Asia, where F. gigantica and F. hepatica overlap. The geographic distribution is determined by the intermediate host ( Lymnaea spp.) and certain other conditions, such as climate, alimentary behaviors, and poverty ( Table 45.1 ).
| GEOGRAPHIC AREA | RISK FACTORS | POPULATION AT RISK |
|---|---|---|
| Latin America (Andean region) Europe Africa Asia Australia |
Watercress Drinking alfalfa juice Green vegetables Contaminated water Travel to endemic areas Living close to irrigation canals Eating salads |
Children in endemic areas Travelers Women Vegetarians |
Examples of countries with estimates of the infected population include 830,000 in Egypt, 742,000 in Peru, 360,000 in Bolivia, 37,000 in Yemen, 20,000 in Ecuador, and 10,000 in Iran. Furthermore, other countries have reported patients with Fasciola infection, including Argentina, Venezuela, Chile, Ecuador, Mexico, Turkey, , Thailand, Japan, Korea, the United States, Tunisia, India, Lebanon, and South Africa, among others.
The Andean region of South America is likely the area most affected by the Fasciola fluke in the world, with point prevalence rates ranging from 6% to 68%. Although fascioliasis is mainly reported in rural areas, new evidence has shown that proximity of medium- to high-income industrialized cities to rural areas creates a potential source of infection because of the importation of contaminated vegetables to the high-consuming markets in large cities. Thus cases of fascioliasis may be seen in areas where this fluke is not endemic. Another factor that contributes significantly to the dissemination of the infection to new areas is the highly adaptable capacity of both the parasite and the lymnaeid snail host to challenging meteorologic conditions (e.g., 4200 m above sea level, Andean region). Paleoparasitologic studies have shown that the introduction of F. hepatica and its snail host from Europe into the Americas has been relatively recent. Given this evidence, fascioliasis is the vector-borne disease with the widest latitudinal, longitudinal, and altitudinal distribution in the world.
To facilitate the classification of fascioliasis cases based on transmission, the following epidemiologic pattern has been proposed : (1) cases imported to areas where neither human nor animal fascioliasis is transmitted; (2) autochthonous, isolated, nonconstant cases of sporadic infection in areas where animal fascioliasis is present; (3) endemic fascioliasis (hypoendemic ≤1%, mesoendemic 1%–10%, hyperendemic ≥10%); and (4) epidemic fascioliasis (a) in animal endemic areas and (b) in human endemic areas. Epidemiology is an important determinant of the initial evaluation of a patient with probable fascioliasis.
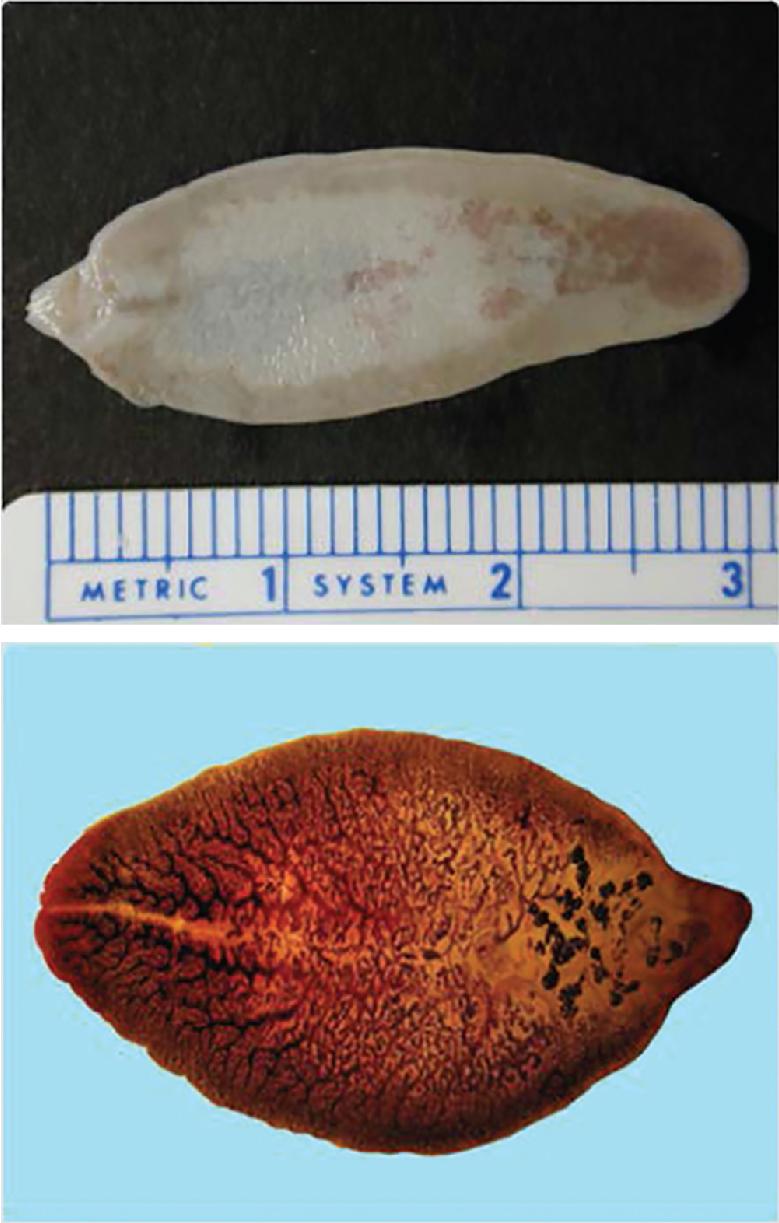
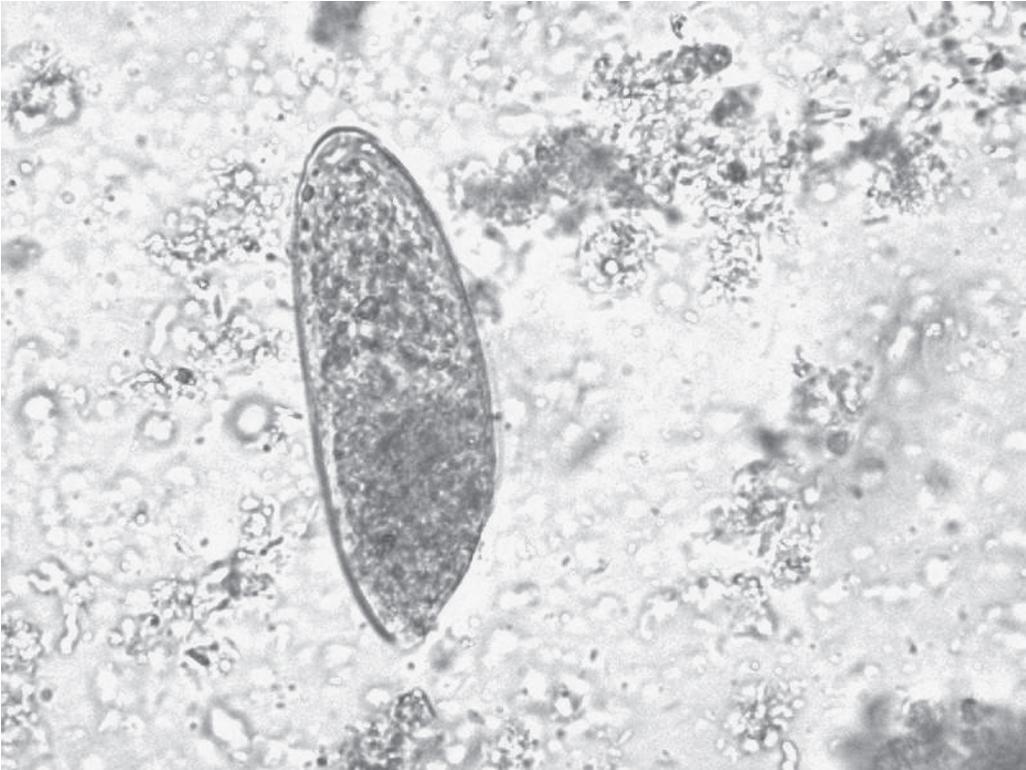
The trematodes F. hepatica (also known as the common liver fluke or the sheep liver fluke) and F. gigantica are large liver flukes ( F. hepatica: up to 30 by 15 mm; F. gigantica: up to 75 by 15 mm), which are primarily found in domestic and wild ruminants (their main definitive hosts). The adult F. hepatica flukes are large, broadly flattened, measuring 30 mm long and 15 mm wide, with a broad anterior portion covered with scale-like spines ( Fig. 45.3 ). The adult fluke lives in the common and hepatic bile ducts of the human or the animal host; the worm can live in the biliary tract for a long time—between 9 and 14 years ( Fig. 45.4 ). Animals susceptible to becoming reservoir hosts for Fasciola spp. mainly include cattle, sheep, pigs, buffaloes, and donkeys, although it has been also reported in horses, dogs, goats, llamas, alpacas, dromedaries, and camels. The eggs are oval, yellowish-brown, and measure approximately 130 to 150 by 60 to 90 μm ( Fig. 45.5 ).
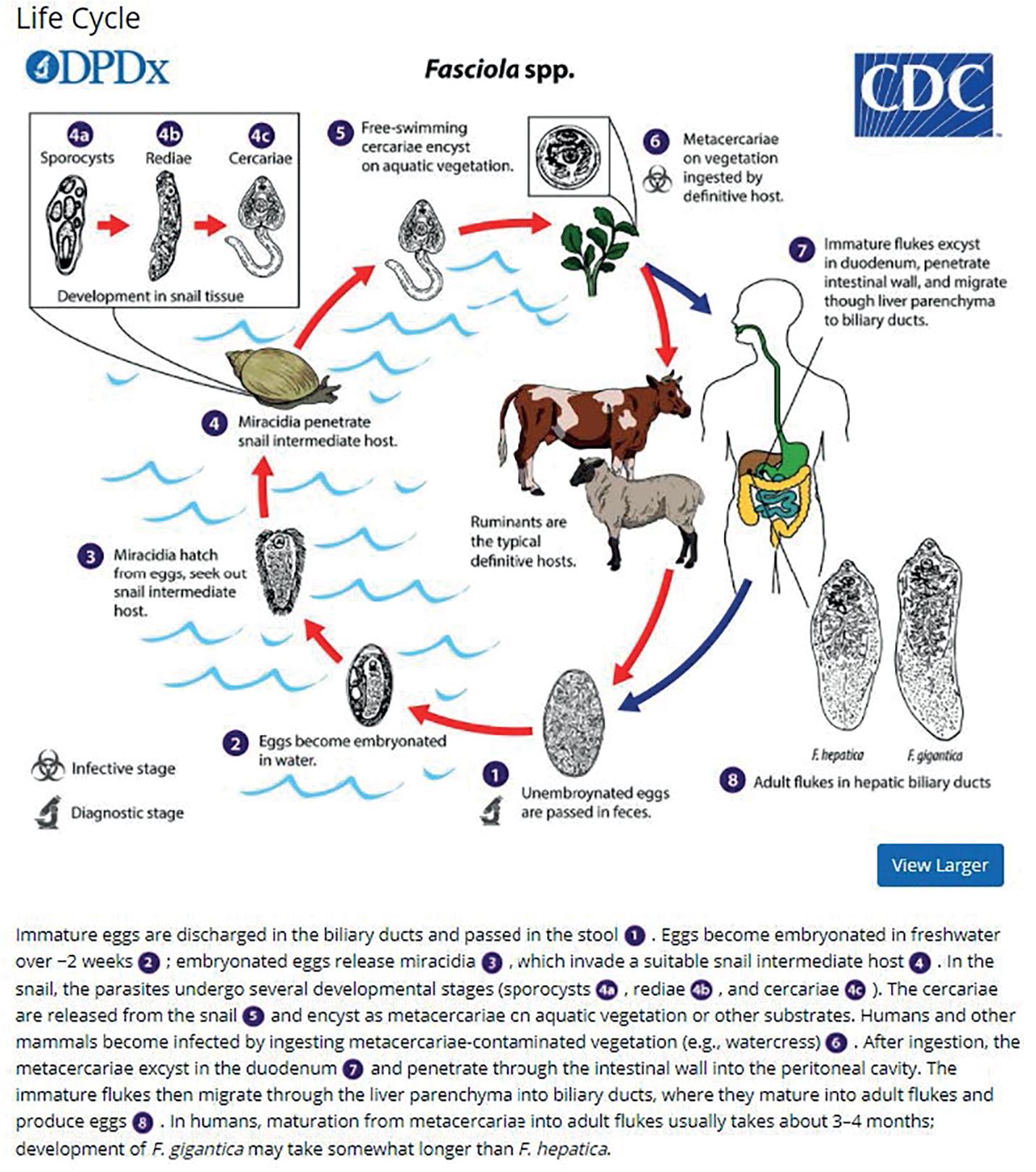
The life cycle begins when the parasite eggs in stool are deposited in tepid water (22°–26°C); miracidia appear, develop, and hatch in 9 to 14 days. These miracidia then invade many species of freshwater snails , in which they multiply as sporozoites and redia for 4 to 7 weeks. They leave as free-swimming cercariae that subsequently attach to watercress, water lettuce, alfalfa, mint, parsley, or khat. Free-swimming cercariae may remain suspended in water and encyst over a few hours, and infection of the human host begins after consumption of plants or water contaminated with the metacercariae. In the first week, the larvae excyst in the duodenum and migrate through the bowel wall and peritoneal cavity. After 4 weeks, the juvenile larvae penetrate the liver through the Glisson capsule to initiate the acute larval, hepatic, and invasive stages of human infection.
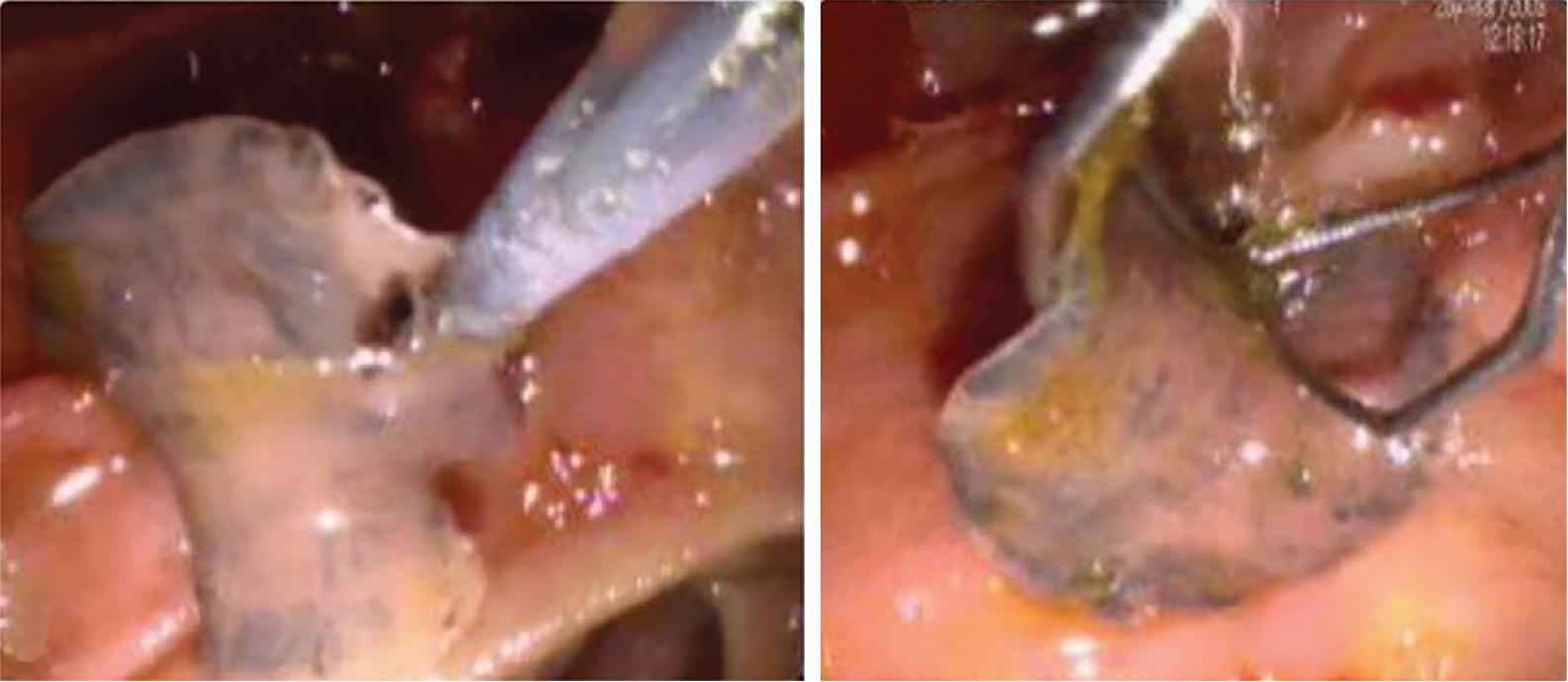
Sometimes the larvae deviate to other locations; these are called extrahepatic forms or ectopic infections. Maturation from juvenile larvae into adult flukes takes approximately 3 to 5 months, during which time the larvae mature and migrate through the liver into the large hepatic and common bile ducts ( Figs. 45.4 and 45.6 ). Mature flukes consume hepatocytes and duct epithelium and reside for years in the hepatic and common bile ducts and occasionally in the gallbladder. Adult fluke worms produce eggs within 4 months after infection (range, 3–18 months); these eggs traverse the sphincter of Oddi and intestine and then continue the cycle of infection. Interestingly, acute and chronic stages can overlap; this is often seen in endemic areas, and it is not unusual to find eggs in the stool samples of patients with acute infection.
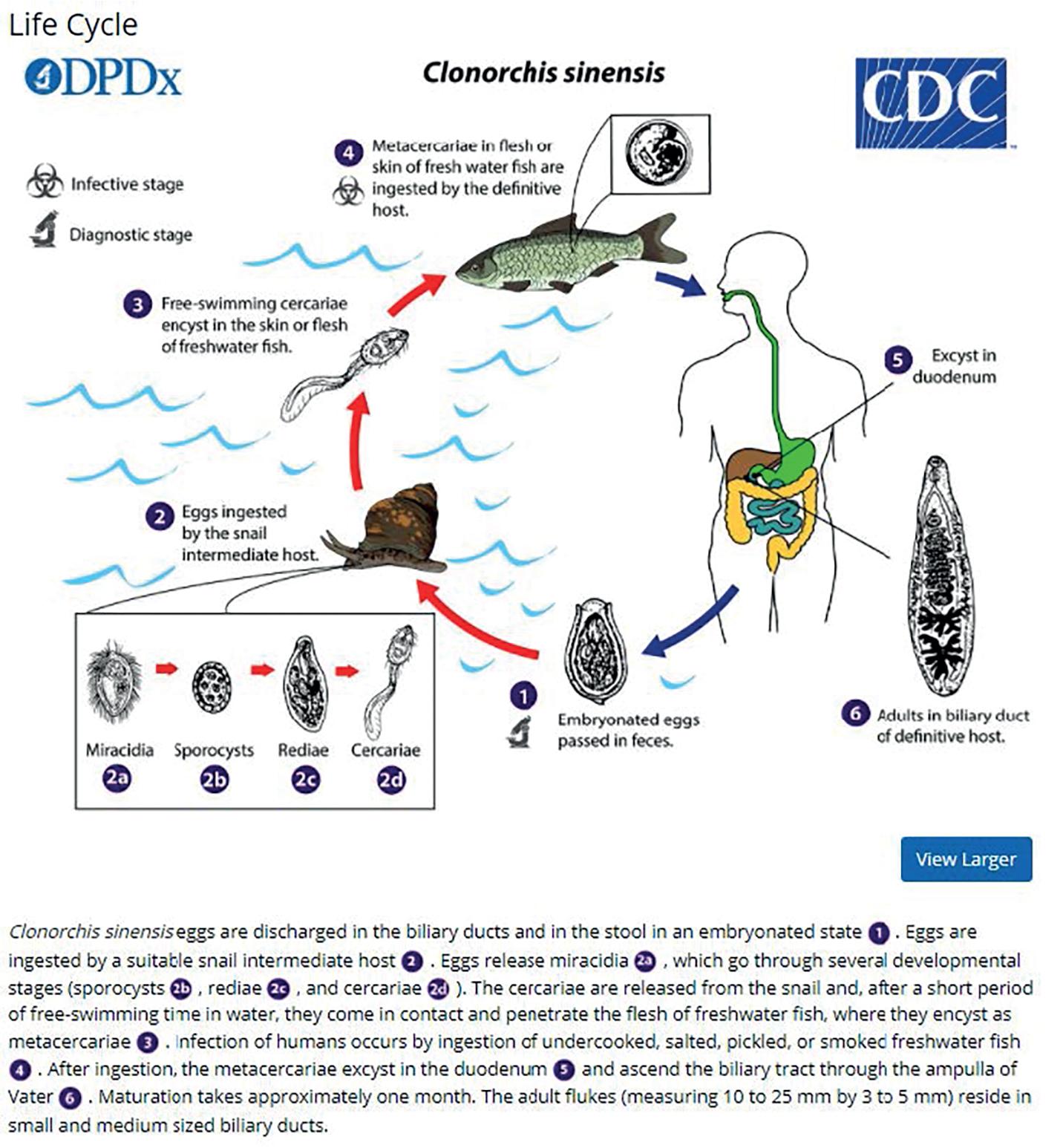
Ingestion of metacercariae in infected raw vegetables (e.g., watercress, lettuce, alfalfa juice, mixed green salads) is a major source of the infection in humans and animals. , Water contaminated with metacercariae also has been described as a potential source of infection in poor rural areas with inadequate sanitation and water supplies. People living in urban areas may be at risk for acquiring fascioliasis. Contaminated vegetables imported from endemic areas can be consumed by people living in large cities. Fascioliasis has been reported in people (e.g., tourists) who have eaten salads in luxury restaurants or hotels in endemic countries, and watercress has traditionally been the most common known source of infection. The plants that have been described associated with Fasciola infection are Medicago sativa (alfalfa in juice) in Peru; Taraxacum dens leonis (dandelion leaves), Valerianella olitoria (lamb’s lettuce), and Mentha viridis (spearmint) in France; green leafy Nasturtium spp. and Mentha spp. in Iran; Juncus andicola and Juncus ebracteatus (Juncaceae), Mimulus glabratus (Scrophulariaceae), and Nostoc spp. (Cianofitas) in the Bolivian Altiplano. , Treatment of contaminated plants with high doses of potassium permanganate decreases metacercariae viability and could be used to prevent infection.
Epidemiologic studies have been carried out in endemic areas to measure the impact of alimentary habits on the acquisition of infection. Drinking beverages made from watercress or alfalfa leaves, called emollients, and living close to irrigation channels were found to be risk factors in a multivariate analysis in the Andean region. Eating salads is the common factor among infected families, and it carries a 3.3-fold increased risk of acquiring the infection. In a logistic regression analysis, an age- and gender-matched case-control study comparing 60 infected children found that drinking alfalfa juice carries a 4.5-fold increased risk of acquiring fascioliasis and familiarity with aquatic plants a 4.3-fold increased risk. Similar risk factors have been found in other parts of the world. In Mexico, aquatic plants and alfalfa juice have been associated with fascioliasis. Socioeconomic factors and drinking untreated water may represent additional risk factors in poor people from endemic areas. Therefore aquatic plants (e.g., watercress, alfalfa) and the irrigation channels that carry the metacercariae play a key role in the transmission of fascioliasis in Andean endemic areas. Table 45.2 summarizes risk factors with studies reporting odds ratios.
| RISK FACTOR | OR (95% CI) | P VALUE | REFERENCE |
|---|---|---|---|
| Multivariate Analysis | |||
| Drinking emollients | 5.2 (1.7-15.6) | .05 | Marcos et al., 2004 |
| Living close to irrigation channels | 17.2 (2.8-106.7) | .05 | |
| Eating salads | 3.3 (1.2-9.0) | .05 | Marcos et al., 2005 |
| Drinking alfalfa juice | 4.5 (1.7-11.1) | .001 | Marcos et al., 2006 |
| Familiarity with aquatic plants | 4.3 (1.7-10.5) | .001 | |
| Univariate Analysis | |||
| Water supply from channels | 2.4 (1.1-5.3) | .03 | Marcos et al., 2006 |
| Consumption of aquatic plants | 2.5 (1.1-5.6) | .028 | |
| Breeding five or more heads of cattle | 2.5 (1.1-5.6) | .028 | |
| Owning dogs | 3.2 (1.3-8.1) | .1 | |
| Defecation site in fields | 2.6 (1.3-5.6) | .01 | |
| Familiarity with aquatic plants | 3.9 (1.8-8.3) | .0001 | |
| Breeding more than five sheep | 0.3 (0.1-0.7) | .003 | |
* These studies were performed in the Andean region of South America. Risk factors in other parts of the world may be similar.
More women are affected with fascioliasis than men. Women have higher prevalence rates, more severe infections, and more reported liver or biliary complications. Children are affected more than adults in endemic areas, which probably reflects partial acquired immunity, exposure to the metacercaria, genetic susceptibility, and other factors. Reinfection is frequently seen in highly endemic areas, and the intensity of the infection depends on the number of metacercariae ingested (this fluke does not multiply in the host).
Fascioliasis has two distinct clinical phases: acute and chronic. Signs and symptoms depend on the worm burden, duration, and phase of infection. In general, the chronic infection is usually diagnosed in epidemiologic studies in endemic areas as a cause of biliary obstruction or in routine stool tests. On the other hand, the acute infection has a more florid clinical picture that brings the patient to the emergency department ( Table 45.3 ). The clinical manifestations are so variable that mild right upper quadrant abdominal pain may call for a step-by-step workup that can lead to the final diagnosis of fascioliasis.
| CLINICAL PICTURE | IMAGING AND LABORATORY RESULTS |
|---|---|
| Acute | |
| Prolonged fever (weeks or months) Abdominal pain (mostly upper abdomen) Hepatomegaly Weight loss Urticaria Ectopic lesions * |
Eosinophilia (any cell count) Anemia Anicteric hepatitis Biliary hemorrhage or hemobilia Subscapular hematoma or hepatic rupture (seen on CT) Hepatic abscesses Track-like lesions on CT |
| Chronic | |
| Abdominal pain in right upper quadrant Biliary colic Nausea and vomiting Recurrent or intermittent jaundice Urticaria |
Eosinophilia (sometimes) Cholestasis Hepatic abscesses Liver fibrosis and ultimately cirrhosis Necrotic granuloma (increased ALT and AST levels) Cystic tumors Cholangitis caused by Klebsiella spp., Escherichia coli, Enterococcus spp. Choledocholithiasis Eosinophilic cholecystitis Acalculous cholecystitis |
* Ectopic migration and other clinical manifestations. Acute stage: Migratory nodule under the skin or peritoneal cavity, arthralgias, lymphadenopathies, hemolytic anemia, seizures, and pleural effusion. Chronic stage: Subcutaneous nodules and gastric nodules.
The first acute or invasive phase lasts from 3 to 5 months and is caused by the migration of the immature larvae from the duodenum to the liver. Finally reaching the bile ducts, parasites migrate through the liver parenchyma and digest hepatic tissue, causing intense inflammation and hemorrhage proportionate to the number of worms. Migration tracks can be observed in histologic sections, and flukes may die, leaving cavities filled with necrotic debris; these are eventually replaced by scar tissue. Symptoms include prolonged fever, hepatomegaly with abdominal pain, and mild eosinophilia (early infection) or hypereosinophilia (mid- or late-acute infection). Multiple hypodense lesions can be seen on computed tomography (CT) scan, similar to metastases. Of note, one of the most frequent manifestations in this acute phase is hypereosinophilia, which is seen in almost all cases. If no eosinophilia is detected at the initial visit, it may be too early in the acute infection; a repeat blood cell count 3 to 5 days later will detect a significant increase in the eosinophil count. Absence of persistent eosinophilia reduces the suspicion for acute infection. In summary, acute fascioliasis is a clinical syndrome similar to acute cholecystitis with significant eosinophilia.
The acute phase manifests with subcapsular hematomas, hepatic cysts, residual hepatic calcifications, and severe anemia. Hyperbilirubinemia is absent in the acute phase, which distinguishes it from other forms of acute hepatitis. Other manifestations are anorexia, weight loss, nausea, vomiting, cough, diarrhea, urticaria, lymphadenopathies, and arthralgias. Occasionally, the juvenile larvae reach other anatomic locations, such as the subcutaneous tissue, pancreas, eye, brain, and stomach wall.
The chronic phase begins approximately 3 to 6 months after the consumption of the metacercariae, when the parasite reaches the bile ducts. On macroscopic examination, the liver has large, dilated, thick-walled, and calcareous bile ducts with yellowish-brown bile. On microscopic examination, the bile ducts have a thickened hyperplastic wall with marked fibrosis. , Symptoms usually reflect biliary obstruction with colicky pain in the right upper quadrant, epigastric area, or upper abdomen. , , Fascioliasis also can be a silent, potential threat: the parasites can survive for longer than 10 years, and infection is usually asymptomatic.
Liver function tests during this phase are consistent with an extrahepatic cholestasis syndrome, which can lead to surgery to treat the biliary obstruction. Asymptomatic cholestasis can be frequently found in infected people from endemic areas. In Egypt, it was found that patients with fascioliasis had significant liver enzyme abnormalities, the most common being an increase in the γ-glutamyltransferase and alkaline phosphatase.
Imaging abnormalities can also be found on ultrasound (US), including hepatomegaly, splenomegaly, periportal fibrosis, thickened gallbladder wall, dilated common bile duct, parasites in the gallbladder and common bile duct, stones in the gallbladder, stones in bile duct, cystic lesions in the liver, focal lesions in the liver, and ascites. Fasciola may also cause acute eosinophilic cholecystitis, along with pruritus and intermittent jaundice. The parasites appear as small, intrahepatic cystic lesions or as a large, multiloculated cyst that causes abscesses. On imaging, parasites may appear very similar to echinococcosis. Bacterial superinfection of Fasciola cysts is a complication of the chronic phase. Recent studies in a rat model have shown a significantly increased risk of bacterobilia in the chronic infection and with concomitant gallstones. Even after successful treatment, abdominal pain and weight loss still may be present in approximately 2% to 4% of patients for several months, which emphasizes the risk for morbidity even after eradication of the infection.
Eosinophilia is not as common a finding in chronic fascioliasis as in the acute stage. On admission to a tertiary health center, 47% of 277 patients with complicated disease had eosinophilia. A similar percentage was found in 101 chronic cases from the Andean region and other endemic areas: 48% had eosinophilia above normal levels, and only 14% had more than 1000 eosinophils/mL. In another study, approximately half of a group of 61 children in the Peruvian Altiplano with chronic fascioliasis had eosinophilia. In Turkey, only 11% of 18 patients with fascioliasis had eosinophilia. Likewise, mild eosinophilia may be present in a minority of fascioliasis patients. Few patients in the chronic phase have high-grade eosinophilia, in contrast with the acute phase, which presents with hypereosinophilia in almost all patients. A wide variety of other infectious agents are associated with eosinophilia, such as Strongyloides stercoralis, Ascaris lumbricoides, and hookworms or other helminths. Although these are the most common parasitic causes of eosinophilia, they do not typically cause hepatic lesions and do not reach the high levels observed in patients with acute fascioliasis. In summary, chronic fascioliasis may manifest without eosinophilia.
Another presentation of the chronic infection is hemobilia as a result of ulcerative lesions in the biliary tract caused by the adult parasite (see Chapter 116 ). Severe iron deficiency anemia has been frequently reported in patients with chronic fascioliasis. , , A granulomatous chronic inflammation also may be triggered by parasite ova in the liver or other locations. ,
Chronic fascioliasis may suppress the immune system. In an animal model, persistent immune suppression has been demonstrated in advanced chronic infection, suggesting that the infected host may be susceptible during the chronic phase to any T helper cell type 2 (Th2)-suppression–dependent infection. This chronic immunosuppression may predispose to bacterial infection that can be life threatening.
An association exists between fascioliasis and liver fibrosis. It appears that hepatic fibrosis may evolve in some susceptible hosts, depending on the time and burden of infection. For example, almost 50% of cattle infected chronically by fascioliasis had cirrhosis. Data support that cathepsin L1 and its collagenolytic function are associated with tissue invasion in the pathogenesis of hepatic involvement associated with F. hepatica infection. Fascioliasis causes bile duct hyperplasia, increased levels of proline , , and types I and III collagen in the liver ; these anomalies are similar to the progression observed in cirrhosis and other pathologic conditions, including wound healing (see Chapter 7 ). However, few studies have attempted to identify factors associated with liver fibrosis, which is an important clinical outcome of the infection. , In addition, liver cirrhosis has been reported in both children and adults with fascioliasis. , In an endemic country, preliminary studies have shown that approximately 9% of cirrhotic patients have antibodies against Fasciola . In vitro and in vivo studies have shown a significant correlation between fibrogenic gene expression and both intensity and duration of infection, demonstrating that longer and higher burden of infection stimulates fibrogenesis in the infected liver. In conclusion, the chronic infection may lead to liver fibrosis (see Chapter 7 )
Table 45.4 summarizes the most common imaging findings in fascioliasis.
| ULTRASONOGRAPHY | COMPUTED TOMOGRAPHY | MAGNETIC RESONANCE IMAGING |
|---|---|---|
| Focal areas Multiple nodules Irregular lesions Variable or increased echogenicity Single complex mass Complex cystic mass Parasites moving in gallbladder |
Multiple hepatic metastatic-like lesions Change in position, attenuation, shape in time Abscess-like lesions Low-density serpiginous tortuous tunnel-like branching Subscapular hematoma Cystic calcifications Glisson capsule contrast enhancement Single non–contrast-enhanced hypodense irregular mass |
Homogeneous hyperintense T2-weighted turbo spin-echo image Subscapular multiple hypointense areas Hypointense T1-weighted three-dimensional gradient-echo image |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here