Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Although uncommon, benign tumors and pseudotumors may arise from the intrahepatic and extrahepatic biliary ducts and gallbladder. These lesions may originate from both the epithelial and nonepithelial structures that comprise the biliary ducts and gallbladder ( Box 48.1 ). Epithelial tumors include adenomas, cystadenomas, and biliary papillomatosis, and nonepithelial tumors consist of granular cell tumors, neurofibromas, and leiomyomas. Finally there are the tumor-like lesions and pseudotumors, including primary sclerosing cholangitis, sclerosing pancreatitis/autoimmune pancreatitis (AIP), and heterotopia. These entities are difficult to diagnose before surgical intervention because of their shared symptoms of obstructive jaundice and lack of specific imaging findings. These are the malignant masqueraders that create the ultimate diagnostic and therapeutic challenges.
Adenoma
Papilloma
Cystadenoma (intrahepatic)
Leiomyoma
Lipoma
Hemangioma
Lymphangioma
Granular cell tumor
Osteoma
Neurofibroma
Schwannoma
Neuroendocrine tumors
Idiopathic benign focal stricture
Lymphoplasmacytic sclerosing pancreatitis
Sclerosing cholangitis
Heterotopic tissue
Although most are benign, a few also represent premalignant lesions in which surgical intervention would be curative. Tissue biopsy does not often alter therapy in appropriate-risk patients with resectable lesions. The majority of these tumors show nonspecific clinical and radiographic features that make diagnosis difficult, if not impossible, without resection. Intraoperative frozen section will not always reveal an accurate diagnosis but is helpful for ensuring clear margins.
In this chapter, we will first review the embryology and anatomy of the biliary tract and then discuss the clinical presentation and diagnostic evaluation of patients with benign tumors and pseudotumors of the biliary tract. Benign tumors and pseudotumors of the biliary tract can broadly be classified into the following general categories: papilloma and adenoma, granular cell tumor, neurofibroma, leiomyoma, neuroendocrine tumor, and pseudotumor. Pseudotumors of the biliary tract comprise inflammatory entities such as primary sclerosing cholangitis, sclerosing pancreatitis, immunoglobulin G4 (IgG4) cholangitis, and heterotopia.
Benign tumors comprising a variety of histologic types have been observed in the extrahepatic ducts and gallbladder because of their common embryology. During the fourth week of embryogenesis, the biliary system will arise from the most caudal aspect of the foregut as a ventral bud. This small outpouching is the anlage of the liver, extrahepatic biliary ducts, gallbladder, and the ventral bud of the pancreas.
The diverticulum divides into a superior and inferior bud as it grows into the ventral mesogastrium ( Fig. 48.1 A) A ventral pancreatic bud develops from the superior surface of the diverticulum, proximal to the enlarging terminal sacculations. The cranial sacculation, the larger of the two, pushes ventrally and cranially into the septum transversum, which separates the thoracic from the celomic cavity. Composed of a solid mass of endodermal cells, the cranial sacculation spreads out into the substance of the septum transversum, eventually forming the right and left lobes of the liver. Cephalad growth and extension of the cranial sacculation results in stretching of the endodermal cell mass from the duodenum to the liver, which will form into the extrahepatic biliary tree. At approximately the seventh week of intrauterine life, vacuolization takes place within the solid mass of cells of the primitive extrahepatic biliary tree leading to a ductal lumen.
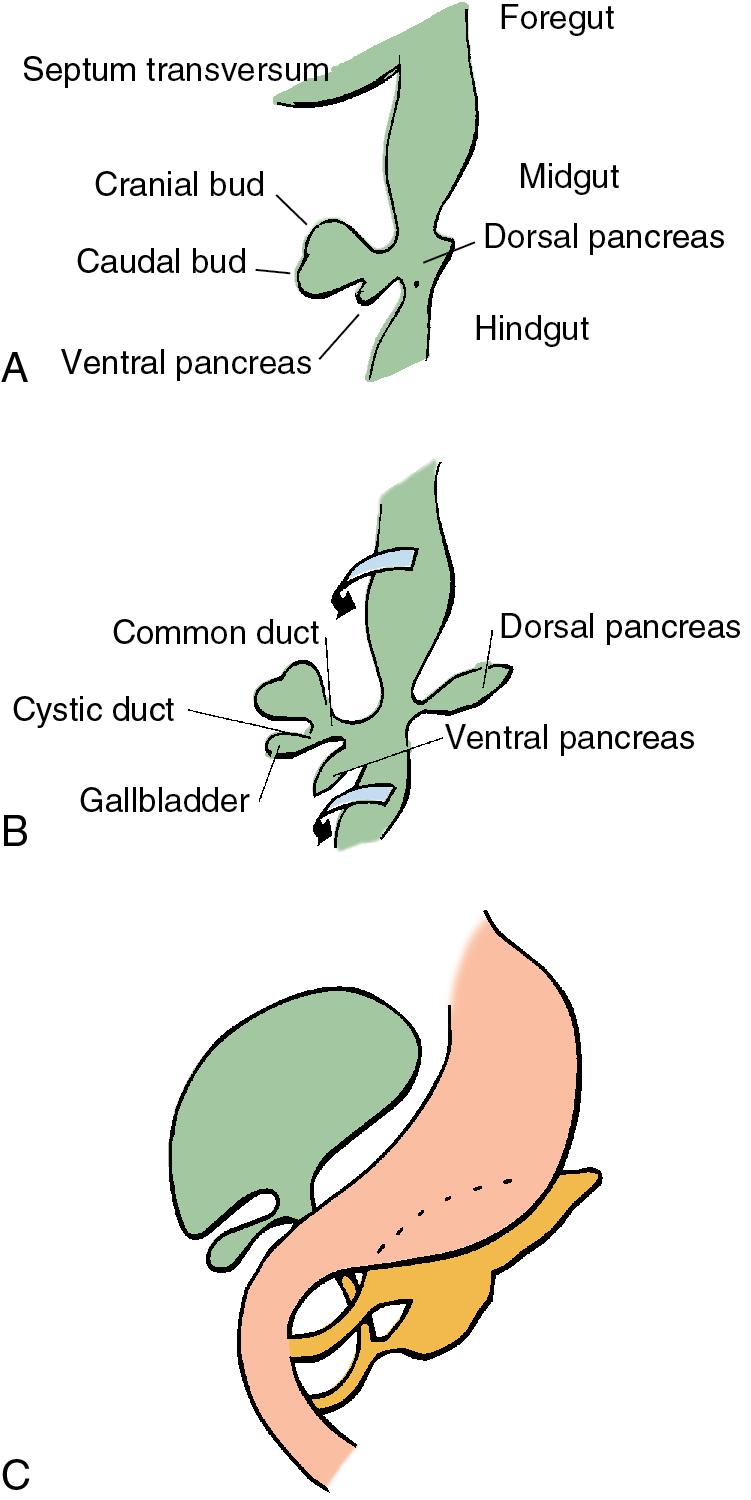
Before the 7-mm stage, the common bile duct (CBD) is attached to the ventral surface of the duodenum close to the ventral pancreatic bud. At the 7-mm stage, left-to-right rotation of the ventral pancreas and duodenum takes place so that the CBD ultimately enters the posteromedial surface of the duodenum (see Fig. 48.1 B–C). The gallbladder and cystic duct develop concurrently from the caudal portion of the primitive hepatic diverticulum during the same period.
The extrahepatic duct lies in the right border of the hepatoduodenal ligament between serosal surfaces and is composed of mucosa, fibrous tissue, and serosa. The dense connective tissue beneath the mucosa comprises collagen, elastic fibers, and scattered smooth muscle fibers, which are not a prominent component. The muscle layer becomes more prominent at the cystic duct, forming the valves of Heister and near the sphincter of Oddi. The CBD most often will join the pancreatic duct at the terminal portion but may fail to unite and enter separately. The thickness of the duct wall varies from 0.8 to 1.5 mm, with an average of approximately 1.1 mm.
The extrahepatic ducts are lined by a single layer of columnar epithelium which rests on the basement membrane and lamina propria and contains mucous glands (see Chapter 47 ). Scattered chromogranin-positive cells can be found in glands of the normal gallbladder neck, and rare cells immunoreactive for somatostatin have been found between the lining epithelium of the hepatic duct in patients with biliary disease. Chronic inflammation of the biliary tract leading to intestinal metaplasia of the mucosa and increase in enterochromaffin (Kulchitsky) cells is a possible basis for development of carcinoid tumors of the biliary tree. The epithelial surface of the duct is generally flat except for tiny pits in the mucosa known as sacculi of Beale, which are luminal openings for the intramural mucous glands. As the duct penetrates the wall of the duodenum, the mucosa appears to become thickened and the surface roughened by longitudinal folds of mucosa, or valvules, particularly at the terminal end of the duct. The valvules were first described in the Fabrica of Vesalius in 1543, followed later by a more detailed description by Santorini in 1724. Brown and Echenberg described a more frequent occurrence of transverse oriented flaps or valvules that face the duodenal lumen and probably function to prevent reflux of duodenal contents into the biliary tree and pancreatic ducts ( Fig. 48.2 ). Baggenstoss also reported that free folds of ductal epithelia in the form of papillary processes may extend 2 to 3 mm beyond the ampullary orifice. Microscopically, a definite transition exists between the mucosa of the duct within the ampulla and the surrounding duodenal mucosa. The ductal mucosa of the ampulla exhibits numerous papillary processes much larger than the adjacent duodenal villi.
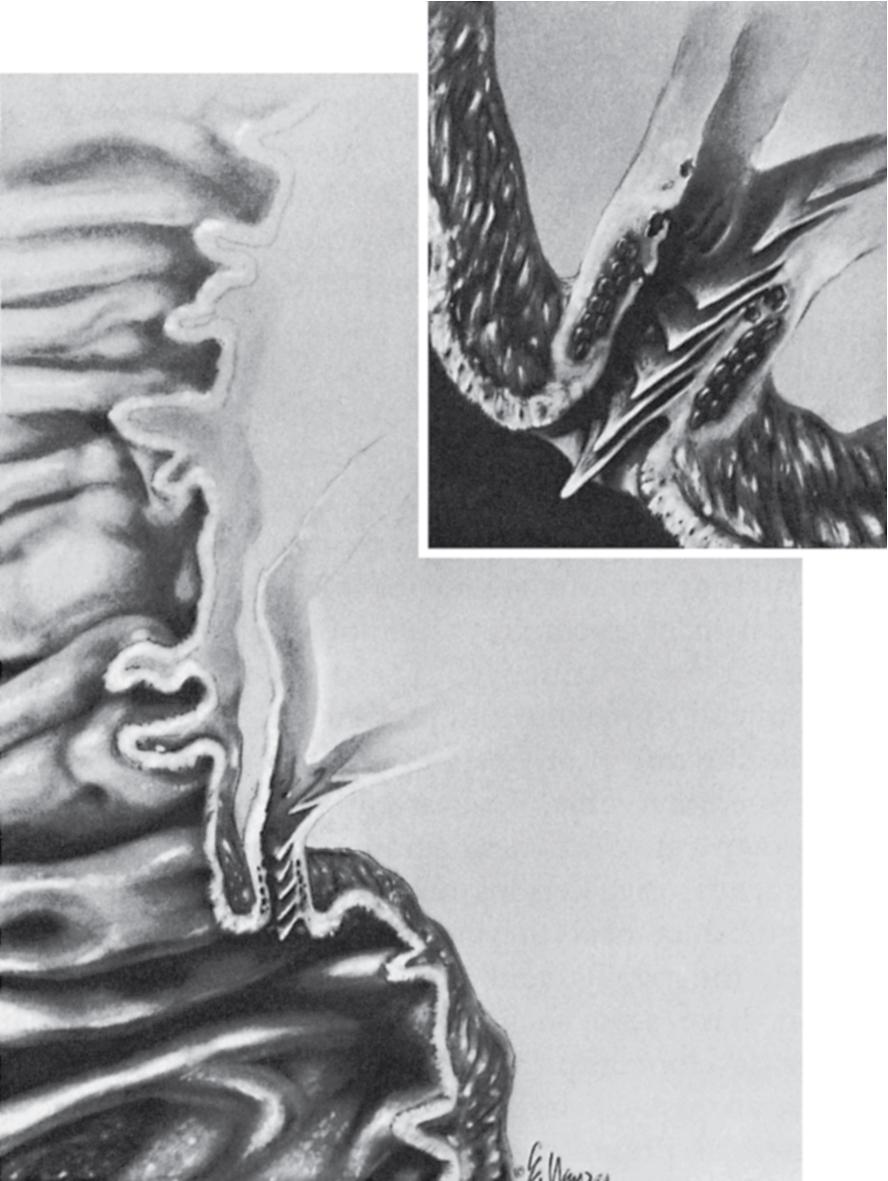
The gallbladder wall is composed of a columnar epithelial mucosa with an underlying basement membrane and lamina propria, irregular smooth muscle, perimuscular connective tissue, and serosa. The perimuscular connective tissue is composed of nerves, blood vessels, lymphatics, and paraganglia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here