Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Heart development begins as the primary germ layers—ectoderm, mesoderm, and endoderm—are induced and progressively changed to various cell types during the morphogenetic process of gastrulation. Combinatorial networks of intercellular signaling events cooperate with massive tissue migrations and internalizations to lay out the basic body plan of the vertebrate embryo. Mesoderm-derived cardiac precursors are among the first cell populations to internalize, coalescing into 2 bilateral populations toward the anterior end of the embryo between 13 and 15 days of human development. The identity of these progenitor pools as cardiac precursors is defined and maintained by expression of a core cohort of developmental gene regulators or transcription factors. These cardiac transcription factors function cooperatively and hierarchically to induce expression of appropriate structural proteins, including components of the specialized cardiomyocyte contractile apparatus and ion channels. Many cardiac transcription factors function not only in the initial specification of cardiac precursors, but also in later aspects of heart morphogenesis, such as establishing chamber identity, chamber-vessel alignment, and conduction system development. Therefore proper spatial and temporal functions of cardiac transcription factors dictate development of a healthy and functional heart. This requirement of correct genetic regulation is exemplified by the numerous congenital heart defects associated with or caused by mutations in cardiac transcription factors.
Even at such early stages of embryonic development, the cardiac precursor pools have been subdivided into two distinct sources of progenitors according to expression of different subsets of cardiac transcription factors. The first, designated as the first heart field, will form the primitive linear heart tube, which will give rise to the left ventricle and most of the atrial tissues. The second heart field, incorporated into the primitive embryonic heart at various stages of development, contributes to the right ventricle and outflow tract. The developing heart receives further contributions from the cardiac neural crest and the mesothelium. The cardiac neural crest is made of ectodermal cells arising outside the heart fields at the lateral borders of the neural plate and because of neural induction from the midline ectoderm. The cardiac neural crest migrates to the heart-forming region, where it contributes to septation of the outflow tract into the arterial and pulmonary vessels. The mesothelium is the embryonic cell source that gives rise to the epicardium, an epithelium that covers the surface of the heart and that plays a role in a number of processes, such as the development of the coronary system and the formation of the annulus fibrosis.
Even before gastrulation has completed, the internalized bilateral cardiac precursor pools continue to migrate in response to signaling cues from neighboring tissues. Remaining as cohesive epithelia, the heart fields move anteriorly and ventrally between 15 and 20 days of development, fusing at the embryonic midline to form the transient cardiac crescent ( Fig. 1.1 ). Proper midline fusion of the bilateral cardiac primordia is essential for development of the heart. Several cardiac transcription factors are required for this process, and loss of function of any one of them causes extensive defects in further morphogenesis, including cardia bifida in severe cases.
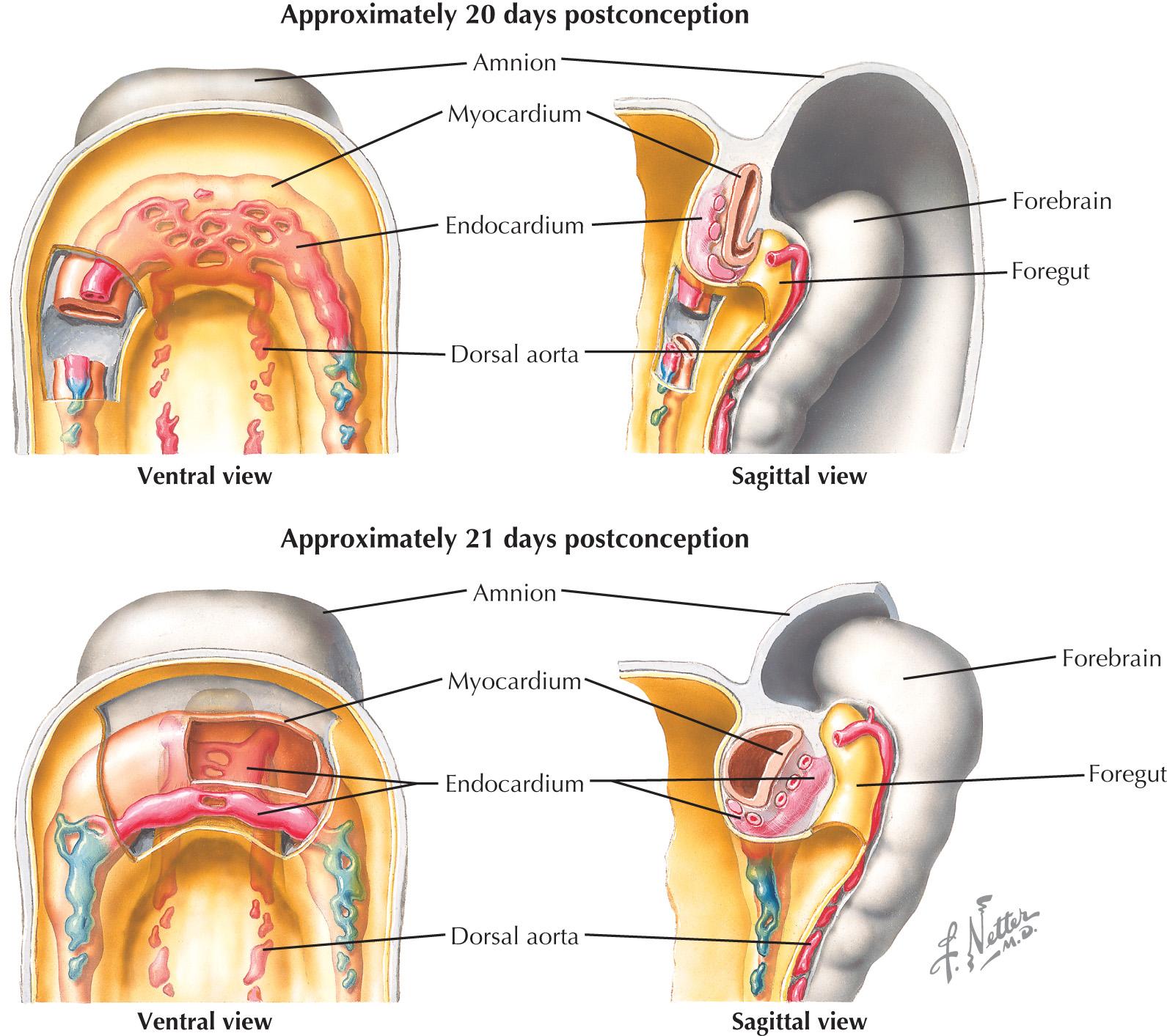
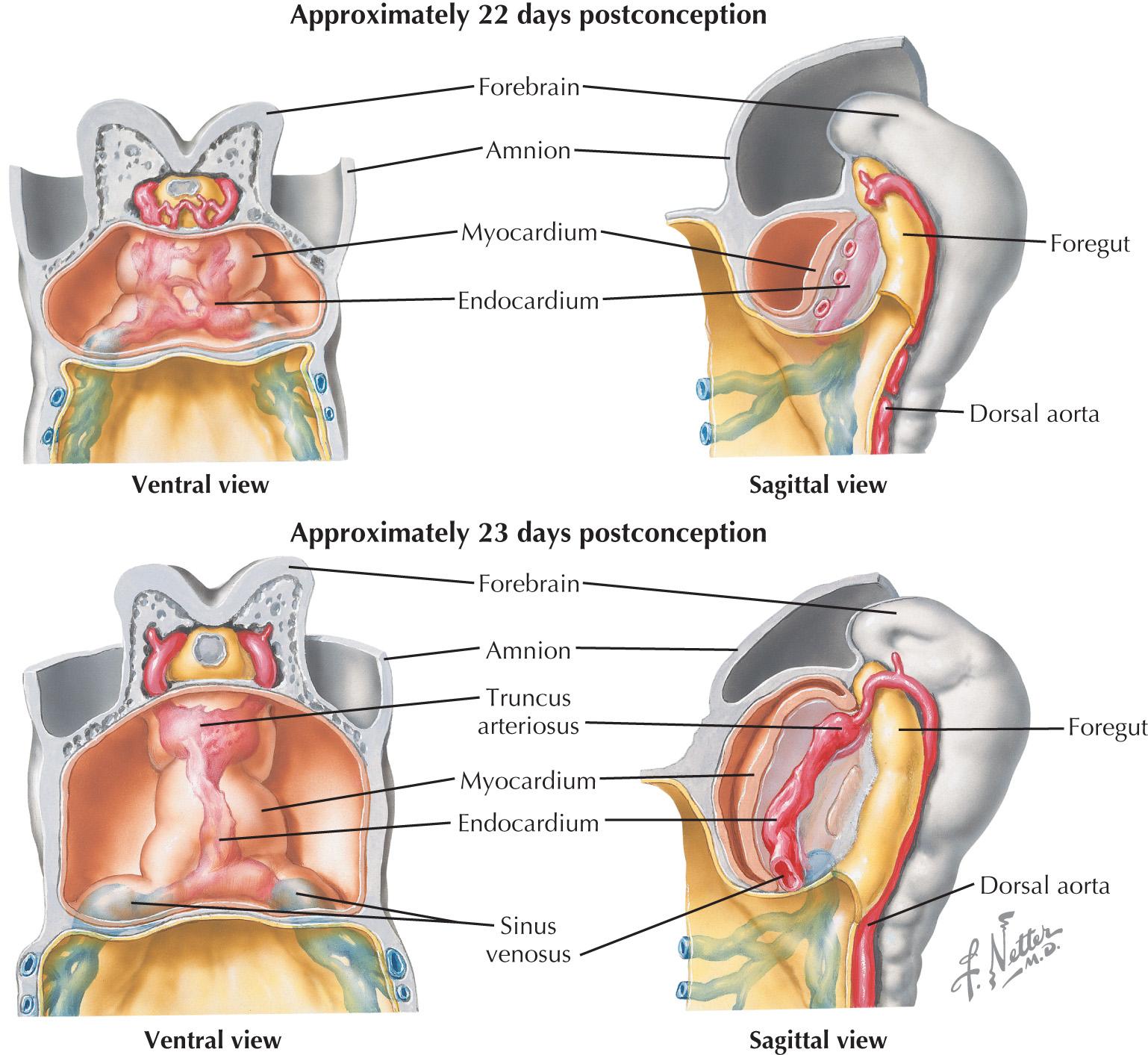
Newly united as the cardiac crescent, the multipotent cardiac progenitors coalesce further to form a linear tube by 3 weeks of development, segregating into the future endocardial lining and myocardial walls ( Fig. 1.2 ). The linear heart tube consists exclusively of differentiated first heart field cells; the second heart field persists as a mesenchymal population, which is a loose association of rapidly dividing precursor cells adjacent to the heart tube. Although no specialized electrical conduction system has yet arisen, the myocardium of the linear heart tube already exhibits autonomous contractions. Compared with those of a mature heart, these contractions are slow and weak, driven only by the intrinsic depolarizing activity and conductivity of the still-maturing cardiomyocytes. Once the conduction system develops and connects to the mature working myocardium, it will serve as an extrinsic regulator of the electrical impulses within the myocardium. Sufficient contractile force will, in turn, allow the heart to beat at the strength required to circulate blood throughout the body.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here