Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Zoonotic viruses ( Chapter 303 ) in animal populations infect humans. An extremely large reservoir of known and unknown viruses represents important causes of described and emerging human infectious diseases. Modes of transmission include human contact with animal bodily fluids or excrement, bite wounds, and direct exposure to an animal carcass. However, many arthropods have evolved to utilize animal blood as a food source, and viruses have exploited this adaptation as a reliable method of transmission. Infectious viruses that are transmitted through arthropod vectors are termed arthropod-borne viruses (arboviruses).
Arboviruses are transmitted to humans most commonly from the bite of a mosquito or tick species. However, other vectors include sand flies ( Phlebotomus spp) and biting midges ( Culicoides spp). Arboviruses infect new vectors most commonly by feeding on an intermediate host, such as small rodents, avian species, or nonhuman primates. The animal host acts as an amplifying host for the virus and can transmit to uninfected vectors. Transovarial transmission from a female to eggs and cofeeding between infected and uninfected insect vectors can contribute to the population of infectious vectors. Many arboviruses also cause persistent infection in the vector, thereby allowing for survival over the winter and transmission over multiple seasons. In general, arboviruses often exist in an enzootic (animal) or sylvatic (jungle-based) cycle of transmission between the vector and an animal host. At times, the vectors may feed on a susceptible population, such as domestic animals (epizootic cycle), or feed on humans to cause an urban cycle or epidemic. If the amount of virus in the blood of infected domestic animals or humans is not high enough or sustained enough over time to support transmission to uninfected vectors, then the host acts as a dead-end or incidental host for the virus. The endemic cycle of an arbovirus between invertebrate and vertebrate hosts determines the geographic range and epidemiology of exposure for humans. As human populations continue to expand into new regions, sylvatic arboviruses that have not yet been identified will have opportunities to develop new epidemic cycles in human populations. Thus, arboviruses represent an important source of future emerging infectious diseases in human populations.
Arboviruses are RNA viruses that are largely grouped into three families of viruses ( Flaviviridae, Togaviridae , and Reoviridae) and one virus order (Bunyavirales).
The Flaviviridae are icosahedral, enveloped viruses about 50 nm in diameter with 180 membrane and 180 envelope proteins arranged in antiparallel dimers on the surface of the viral particle. The capsid protein encloses a positive-sense, single-stranded RNA genome (about 11 kb) with a single open reading frame that has an upstream 5ʹ untranslated region, a 5ʹ m7GpppRNA cap structure, and a 3ʹ untranslated region with conserved RNA structure and no poly-A tail. Within the Flaviviridae family, the genus Flavivirus is divided into mosquito-borne, tick-borne, arthropod-specific, and unknown vector groups. The flaviviruses circulate between mosquito and tick species and specific mammalian hosts that include avian species, small mammals, nonhuman primates, and humans.
The Togaviridae are lipid-enveloped viruses about 60 to 70 nm in diameter with a 5ʹ capped positive-sense RNA genome that is 11 to 12 kb in length with a poly-A tail. The nonstructural proteins are expressed from the first open reading frame, and a subgenomic promoter drives expression of the structural genes. The enveloped virus particle consists of E1 and E2 glycoproteins as heterodimers that then form trimers to make 80 spikes. The Alphavirus genus makes up many of the medically important arboviruses. All Togaviridae are transmitted by mosquito vectors, and individual viruses are generally divided into New World and Old World alphaviruses.
Reoviridae , which include Colorado tick fever virus, are nonenveloped, double-stranded RNA viruses with a segmented genome. Colorado tick fever virus genome is relatively large (~29 kb) compared with other arboviruses and has an icosahedral capsid structure about 80 nm in diameter.
The Bunyavirales order consists of 9 viral families and 13 genera. The arboviruses of human significance are largely represented by the Orthobunyavirus , Phlebovirus , and Orthonairovirus genera. In general, the bunyaviruses are characterized by a spherical, enveloped virus particle that is about 80 to 120 nm in diameter and is decorated with two surface glycoproteins (Gc and Gn). The negative-sense or ambisense RNA genome is divided into three segments (L, M, and S). The L segment encodes the RNA-dependent RNA polymerase, the M segment encodes the viral glycoprotein precursors, and the S segment encodes the nucleocapsid proteins.
Arboviruses are generally found in the salivary glands of the associated vector, where they arrive following replication in the midgut after a blood meal from an infected, amplifying host. In some cases, an invertebrate vector can also become infected from transovarial transmission, in which the female vector transmits virus to eggs, or by cofeeding when an infected vector feeds on a previously uninfected host at the same time as an uninfected vector. Once an infected vector bites a mammalian or avian host, the salivary gland excretion, which precedes feeding, allows the arbovirus to be distributed into the subcutaneous tissue of the host. The virus replicates locally in the dermal tissue, epithelial cells, endothelial cells, or local tissue-resident immune cells such as dendritic cells. Local replication results in viral spread to draining lymph nodes, which are the site of primary viral replication, and subsequent viremia that spreads the virus to target organs, including the spleen and liver. Spread to the spleen or liver can initiate a secondary viremia that can infect the central nervous system, the placenta, or joint tissues.
Arboviruses vary in the rate that they cause disease in humans based on many factors, including age, immune status, comorbid conditions, and prior exposure history. Exposure to an arbovirus can result in a wide range of clinical outcomes, from asymptomatic infection to severe disease. Arboviruses can cause characteristic syndromes broadly defined as a fever/rash/myalgia syndrome or a meningitis encephalitis syndrome. Fever and rash in a returning traveler ( Chapter 265 ) from an endemic area should raise suspicion for an arboviral infection. Some arbovirus members that cause fever and myalgia can also cause hemorrhagic fever syndromes ( Chapter 351 ; Table 352-1 ). Other arboviruses predominantly cause infections of the central nervous system ( Table 352-2 ) but can also cause rashes.
| VIRUS | TRANSMISSION CYCLE | GEOGRAPHIC DISTRIBUTION | CLINICAL SYNDROME | TREATMENT AND PREVENTION |
|---|---|---|---|---|
| TROPIC/SUBTROPICAL GLOBAL DISTRIBUTION | ||||
| Dengue (Flaviviridae) |
Vector: Aedes aegypti Host: primates/humans |
Global tropical distribution | Fever, headache, fatigue, myalgia, arthralgia, weakness | Vaccine |
| Chikungunya (Togaviridae) |
Vector: Aedes aegypti Host: primates/humans |
Global tropical distribution | Fever, myalgias, rash, arthralgias, arthritis, tenosynovitis | None |
| Zika (Flaviviridae) |
Vector: Aedes aegypti Host: primates/humans |
Global tropical distribution | Fever, chills, myalgias, rash, conjunctivitis | None |
| Yellow fever (Flaviviridae) |
Vector: Aedes spp. Host: primates/humans |
South America/Africa | Fever, chills, headache, nausea, vomiting, abdominal pain, jaundice | Vaccine |
| NORTH AMERICA | ||||
| Heartland (Bunyavirales) |
Vector: Amblyomma americanium Host: unknown reservoir |
Midwestern and Southern United States | Fever, headache, fatigue, myalgia, arthralgia, weakness | None |
| CENTRAL AND SOUTH AMERICA | ||||
| Mayaro (Togaviridae) |
Vector: Haemagogus spp. Host: nonhuman primates |
Central and South America | Fever, chills, headache, arthralgias, myocarditis | None |
| Tropical fever (Oropouche virus, Bunyavirales ) |
Vector: Culicoides paranensis biting midge Host: sloths, nonhuman primates |
Central and South America | Mild febrile illness, myalgias, malaise; rare hemorrhagic disease | None |
| Bussuquara (Flaviviridae) |
Vector: Culex melanoconion Host: small rodents |
Central and South America | Fever, arthralgias | None |
| AFRICA | ||||
| O’nyong-nyong (Togaviridae) |
Vector: Anopheles spp. Host: unknown host |
Sub-Saharan Africa | Fever, headache, rash, conjunctivitis, large joint arthralgia, arthritis | None |
| Rift valley fever (Bunyavirales) |
Vector: Aedes spp. and Culex spp. Host: wild ungulates and domesticated livestock |
Sub-Saharan Africa | Fever, headache, myalgias, and severe disease with hepatitis and jaundice, hemorrhage | Vaccines for livestock to prevent epizootic outbreaks |
| EUROPE | ||||
| Sinbis (Togaviridae) |
Vector: Aedes spp., Culex spp., and Culiseta spp. Host: bird species |
Northern Europe, also in Russia, central Africa, Australia | Fever, macular rash, arthralgia of hands and small joints | None |
| Phlebotomus fever (Bunyavirales) |
Vector: Phlebotomus spp. sandfly Host: unknown |
Mediterranean area, western Europe, Turkey, Middle East, western India, Pakistan | Fever, rash, headache, photophobia, myalgia, arthralgia | None |
| ASIA | ||||
| Severe fever with thrombocytopenia syndrome virus (Bunyavirales) |
Vector: Haemaphysalis longicornis tick Host: avian and small mammal species |
Central and eastern Asia | Fever, myalgias, headache, arthralgia; can progress to bleeding diathesis | None |
| Crimean-Congo hemorrhagic fever (Bunyavirales) |
Vector: Hyalomma spp. ticks Host: birds, small and large mammals |
Asia, Africa, Middle East | Febrile illness, myalgia, vomiting and diarrhea; some progress to severe hemorrhagic disease | None |
| Omsk hemorrhagic fever (Flaviviridae) |
Vector: Dermacentor spp. ticks Host: small mammals |
Omsk, Kurgan and regions of western Siberia | Fever, headache, chills, vomiting, myalgia, photophobia, conjunctival suffusion; fatality rate < 3% | Tick-borne encephalitis vaccine may provide cross-protection |
| AUSTRALIA | ||||
| Ross River (Togaviridae) |
Vector: Aedes spp. and multiple mosquito vectors Host: marsupials |
Australia | Fever, rash, arthralgias, arthritis, myalgias | None |
| Edge Hill (Flaviviridae) |
Vector: Aedes spp. and Culex spp. Host: marsupials |
Australia | Fever and proximal joint arthritis | None |
| VIRUS | TRANSMISSION | GEOGRAPHIC DISTRIBUTION | CLINICAL SYNDROME | TREATMENT AND PREVENTION |
|---|---|---|---|---|
| NORTH AMERICA | ||||
| West Nile (Flaviviridae) |
Vector: Culex spp. mosquitoes Host: passerine birds |
North America, South America, Africa, Europe, Middle East, Asia, Australia | Fever, myalgias, rash, nausea, vomiting, diarrhea, meningitis, paralysis, encephalitis | None |
| Powassan (Flaviviridae) |
Vector: Ixodes spp. ticks Host: small mammals and white-tailed deer |
North America | Fever, malaise, myalgia, nausea, rash, meningitis, encephalitis | None |
| Eastern equine encephalitis (Togaviridae) |
Vector: Culiseta melanura mosquito Host: bird species |
East coast and upper Midwest United States | Fever, chills, malaise, myalgias, arthralgias, meningitis, encephalitis | None |
| St. Louis encephalitis (Flaviviridae) |
Vector: Culex spp. Host: avian species |
Continental United States | Fever and myalgias, meningitis, encephalitis | None |
| Western equine encephalitis (Togaviridae) |
Vector: Culex tarsalis mosquitoes Host: avian species |
North America, rare cases | Fever, myalgias, meningitis, encephalitis | None |
| California encephalitis group (Bunyavirales) |
Vector: Aedes spp. mosquitoes Host: small mammals |
North America | Fever, chills, nausea, vomiting, myalgias, seizures, encephalitis | None |
| Colorado tick fever (Reoviridae) |
Vector: Dermacentor andersoni tick Host: small mammals and rodents |
North America, mountain west | Biphasic fever, chills, headache, myalgias, abdominal pain, rash, meningitis | None |
| CENTRAL AND SOUTH AMERICA | ||||
| Venezuelan equine encephalitis (Togaviridae) |
Vector: Culex, Aedes, Mansonia, Psorophora spp. mosquitoes Host: rodents/small mammals |
Central and South America | Fever, malaise, chills, myalgia, headache, encephalitis ataxia, seizures, paralysis | No licensed vaccine |
| Rocio (Flaviviridae) |
Vector: Psorophora spp. mosquitoes Host: birds |
Brazil and South America | Fever, headache, malaise, conjunctivitis; 30% of cases develop meningitis, encephalitis; 10% mortality | None |
| AFRICA | ||||
| Usutu (Flaviviridae) |
Vector: Culex pipiens mosquitoes Host: avian species |
Africa and Europe | Fever and myalgias, meningitis, encephalitis | None |
| EUROPE | ||||
| Tick-borne encephalitis (Flaviviridae) |
Vector: Ixodes spp. ticks Host: mammals, livestock |
Eastern Europe and northern Asia | Fever, myalgias, meningitis, encephalitis, poliomyelitis | Four vaccines available in endemic regions |
| Louping ill (Flaviviridae) |
Vector: Ixodes spp. ticks Host: small rodents |
Europe and Asia | Fever, myalgias, fatigue, meningitis, encephalitis | None |
| Toscana (Bunyavirales) | Vector: Phlebotomus spp. sandfly Host: unknown reservoir |
Mediterranean Europe | Fever, headache, myalgias, meningitis, encephalitis | None |
| ASIA | ||||
| Japanese encephalitis (Flaviviridae) |
Vector: Culex tritaeniorhychnus Host: aquatic birds and pigs |
Southeast Asia, India, western Pacific Islands, northern Australia | Fever, headache, nausea, vomiting, meningitis, encephalitis | Live-attenuated vaccine |
| AUSTRALIA | ||||
| Murray Valley encephalitis (Flaviviridae) |
Vector: Culex spp. mosquitoes Host: small mammals and marsupials |
Northern Australia, New Guinea | Fever, chills, myalgias, rash, meningitis, encephalitis | None |
| spp = species. | ||||
For individuals that develop symptoms, the initial presenting symptoms can be protean, including fever, chills, myalgias, arthralgias, and neurologic symptoms. The only differentiating features may be the geography and surrounding epidemiology of the exposure. Even in a specific region, however, clinical presentations may overlap. For example, Chikungunya virus, dengue virus, and Zika virus can co-circulate using the same vector, or coinfections may occur from Aedes agyepti mosquitos infected with more than one pathogen.
Reverse transcription polymerase chain reaction (RT-PCR) is the preferred diagnostic test when patients have viremia or active virus in the cerebrospinal fluid (CSF). Since the onset of symptoms for most patients follows clearance of active viremia, however, most patients will require virus-specific serologic testing for the presence of IgM or IgG antibodies, often with acute and convalescent titers to distinguish new infection from prior infection. Many of the serologic tests used to diagnose arboviruses cross-react with co-circulating arboviruses, thereby making definitive diagnosis clinically challenging in some regions. For definitive diagnosis, virus-specific neutralizing antibody titer assays, often available through government laboratories, can help distinguish among individual arboviruses.
Specific antiviral therapeutic options are rarely available for acutely ill patients. In general, prevention is based on limiting exposure to vectors. Vaccines are available for yellow fever, dengue, tick-borne encephalitis virus, and Japanese encephalitis viruses. In endemic regions, individual prevention measures include DEET-containing repellants, permethrin-embedded clothing, long pants/sleeves, and bed nets. Vector control methods include drainage or removal of stagnant water sources where vectors reproduce.
Yellow fever virus is a mosquito-borne member of the Flaviviridae family with characteristic, small, spherical, enveloped particles with three structural proteins that contain a positive-sense, single-stranded RNA genome. The genus, Flavivirus (flavi = yellow), acquired its name from the characteristic jaundice associated with yellow fever infection.
Yellow fever virus continues to cause major epidemics in tropical and subtropical regions of Africa and South America despite a safe and effective vaccine. Recent estimates report 63,000 to 158,000 severe annual infections in Africa, resulting in 29,000 to 75,000 deaths per year. In South America, severe infections are estimated to be 4000 to 15,000 per year and deaths to be 2000 to 7000 per year. Yellow fever virus exhibits a sylvatic and urban transmission cycle between nonhuman primates and humans, respectively, with the Haemagogus spp and Aedes aegypti mosquitoes. Large outbreaks in densely populated urban areas are likely spillover infections from sylvatic outbreaks where large populations of A. aegypti mosquitoes breed in urban environments. The magnitude and duration of yellow fever viremia are sufficient to infect naïve, uninfected A. aegypti mosquitoes, thereby resulting in amplification of yellow fever in urban cycles of transmission.
Following a bite from a yellow fever virus–infected mosquito, the virus replicates in the dermal tissue epithelial cells and resident dendritic cells prior to spreading to a local draining lymph node. Following viral replication in the innate immune cells of the draining lymph node, yellow fever virus spreads to the liver, spleen, heart, and kidneys. Infection of Kupffer cells and hepatocytes in the liver results in liver inflammation and injury and can often cause significant liver dysfunction.
The majority of individuals infected with yellow fever virus remain asymptomatic or mildly symptomatic. In others, however, a 3- to 6-day incubation period precedes acute yellow fever, which is characterized by the sudden onset of fever, chills, headache, myalgias, fatigue, weakness, nausea, and vomiting. Symptoms usually resolve within 1 week, but fatigue and weakness may persist. Virus is typically detectable in the serum for the first week of symptoms and clears as symptoms resolve. After a brief remission, about 12% of infected patients develop severe disease characterized by the return of fever, abdominal pain, nausea, vomiting, jaundice, and oliguria. The liver disease results in elevated levels of bilirubin and aminotransferases, with decreased hepatic synthesis of clotting factors. Some patients develop hemorrhagic manifestations of diffuse intravascular coagulation ( Chapter 161 ), which is characterized by ecchymoses as well as bleeding from mucosal surfaces and needle puncture sites. Associated acute renal failure and hemorrhagic shock ( Chapter 92 ) can occur.
The differential diagnosis is broad and includes acute viral hepatitis ( Chapter 134 ), other arboviruses ( Chapter 351 ), and malaria ( Chapter 316 ). Since virus is detectable in human serum during the first 7 to 10 days following the onset of symptoms, yellow fever virus nucleic acid testing using an RT-PCR test may provide a diagnosis prior to serology. Since patients usually present later in the course of disease, laboratory diagnosis of yellow fever is generally accomplished by testing serum for virus-specific IgM antibodies and neutralizing antibodies. An elevated antibody level is diagnostic in unvaccinated patients, but a single antibody test cannot differentiate between the vaccine and natural infection, so serial serology showing a greater than four-fold increase in anti–yellow fever virus IgM or IgG between acute-phase and convalescent-phase samples is required for diagnosis in vaccinated persons. Antibodies to one flavivirus can cross-react with other circulating flaviviruses, such as dengue virus, so confirmatory testing for neutralizing antibodies using plaque-reduction neutralization titers may be needed.
Therapeutic options include supportive care with fluid resuscitation, critical care management, and renal replacement therapy when indicated ( Chapter 351 ). Live-attenuated vaccine produces robust, long-lived neutralizing antibody and cellular immune responses, with a single lifetime dose recommended for travelers to endemic regions. Severe adverse events occur at a rate of <1 case per 100,000 doses. For severe yellow fever, the case-fatality rate is about 50% in unvaccinated individuals.
Dengue virus, which is a member of the Flaviviridae family (genus Flavivirus ), causes infection by one of four serotypes. As with other flaviviruses, the lipid envelope is decorated with viral structural proteins, but its envelope protein varies by as much as 30 to 40% among the different serotypes. This large divergence is the basis for cross-reactive antibody reactions that are non-neutralizing but potentially enhance the severity of the clinical manifestations.
Dengue virus causes an estimated 390 million cases each year and continues to expand into the tropics and subtropics across the world ( E-Fig. 352-1 ), where about 40% of the global population is at risk. Dengue virus circulates in urban cycles largely owing to the success of A. aegypti , its principal vector, to spread and reproduce among human dwellings in small containers of fresh water found in discarded tires, containers, jars, and other vessels. Aedes albopictus can also act as a vector.
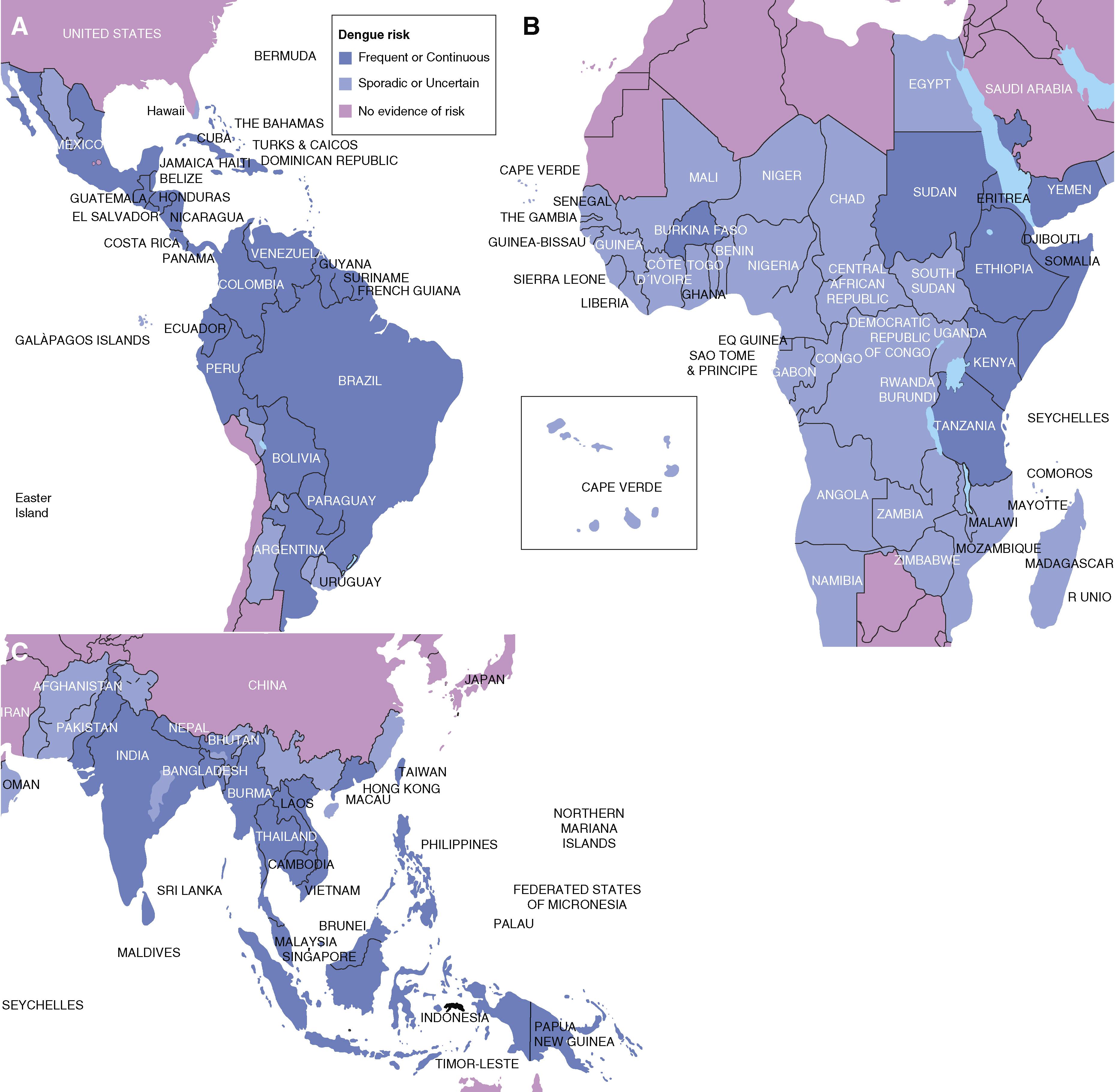
All four serotypes of dengue virus cause dengue fever, and more severe dengue is typically associated with dengue virus 2 infection. The risk for developing severe dengue significantly increases in individuals who have preexisting immunity to a dengue virus serotype and then develop an acute infection with a different dengue virus serotype. The mechanism, which is referred to as antibody-mediated enhancement, occurs when poorly neutralizing or low titer cross-reacting antibodies from the first dengue virus infection are enhanced by a second dengue virus infection from a different serotype or following infection with Zika virus, which is closely related to dengue virus. Human autopsy studies following severe dengue virus infection show antigen and nucleic acid in the cells of the skin, liver, spleen, lymph nodes, kidney, lung, thymus, and brain.
Following an incubation period of 2 to 7 days, patients can exhibit asymptomatic infection (40 to 80%) or dengue fever, which is characterized by sudden onset of high fever (40° C/104° F) accompanied by two or more of the following symptoms during the febrile phase: severe headache, retroorbital pain, myalgias/arthralgias, nausea, vomiting, adenopathy, and macular rash. Petechiae and epistaxis may also be present. The initial illness persists for about 7 days, whereas viremia persists for approximately 4 to 5 days, thereby allowing for transmission from humans to mosquitoes during the typical febrile period of disease. Following the initial febrile phase, most patients defervesce and begin to recover, but 1 to 5% of patients develop severe dengue, which is characterized by plasma leakage, shock, coagulation derangement, and end-organ disease resulting in hepatitis, encephalopathy, myocarditis, or severe bleeding ( Chapter 351 ). Warning signs for severe dengue include abdominal pain, persistent vomiting, fluid accumulation, mucosal bleeding, lethargy, and liver enlargement >2 cm.
Laboratory testing may reveal thrombocytopenia, leukopenia, and mildly elevated liver aminotransferase levels. As patients defervesce, an increasing hematocrit, in part due to plasma leakage, and a rapid decrease in platelet count can indicate risk for development of severe dengue.
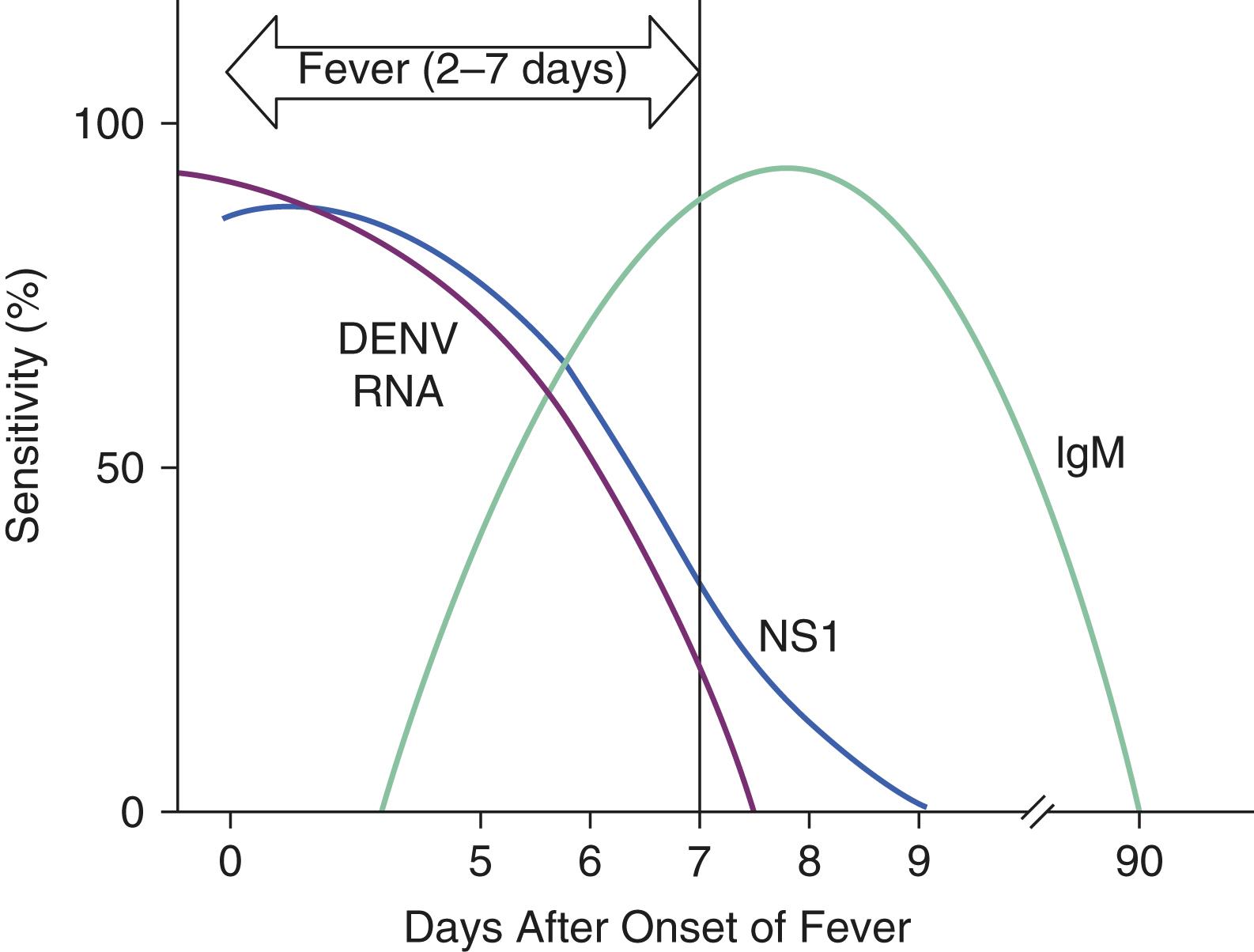
The diagnosis is best made during acute infection by RT-PCR for dengue virus nucleic acid in serum, alone or in combination with antigen testing for dengue nonstructural protein 1 by immunoassay. After about 4 days following the onset of fever, the most useful test is an IgM enzyme-linked immunoassay (ELISA), and a single serum sample strongly suggests recent dengue virus infection in regions where cross-reactive flaviviruses are not likely ( E-Fig. 352-2 ). Plaque reduction neutralization antibody titers can be used to verify a positive dengue IgM antibody test result. The presence of IgG is indicative of past infection and not necessarily current infection. Moreover, cross-reactivity between antibodies to the individual dengue virus serotypes, other circulating flavivirus infections, or vaccines such as yellow fever virus vaccine may provide false-positive serology results.
No specific therapy is effective for dengue virus. Ivermectin can accelerate the clearance of viral protein but has not been shown to be of clinical benefit. Supportive care includes fever reduction with acetaminophen while avoiding nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and aspirin, which can potentiate the risk for bleeding. For patients diagnosed with dengue with warning signs or severe dengue, hospitalization and close monitoring may be required with supportive care, including fluid resuscitation with infusion of crystalloid solutions and oxygen administration.
Vaccination against dengue has shown promising results. , A quadrivalent dengue virus vaccine has an 80% efficacy against infection in children ages 4 to 16 years and provides 90% protection against hospitalization. , Infecting A. aegypti mosquitoes with Wolbachia pipientis makes them less susceptible to dengue virus and can provide about a 75% reduction in dengue infection in endemic areas.
Severe dengue has a case-fatality rate that ranges from <1 to 5%. Appropriate supportive care in a center with experience treating severe dengue reduces overall mortality to close to 1%.
Zika virus is a mosquito-borne flavivirus that is closely related to dengue virus. Previously confined to Africa and Southeast Asia, Zika virus emerged in 2008 to cause large outbreaks of febrile disease with myalgias in adults in tropical and subtropical regions throughout the world. During the global outbreak, newly described Zika virus complications included postinfectious Guillain-Barré syndrome, sexual transmission, and vertical transmission of Zika virus in pregnant women, and congenital Zika virus syndrome became appreciated.
Zika virus, which was first recognized in a febrile macaque and Aedes africanus mosquitoes at the edge of the Zika forest in Uganda, is now an important cause of human disease throughout the world. Two distinct lineages have been identified in Africa and Asia. In 2007, a large outbreak of Zika virus on Yap Island was followed by outbreaks across the Pacific Ocean and subsequently in Brazil, other countries in South America, and Central America. By 2019, 87 countries reported Zika cases. Zika is a cause of Guillain-Barré syndrome ( Chapter 388 ), and infection of pregnant females results in significantly increased risk of fetal abnormalities, including microcephaly (termed congenital Zika syndrome). The largest recorded outbreak was a reported 2,751 cases in Brazil between 2015 and 2016, though this number is likely an undercount.
Zika virus is spread principally by A. aegypti mosquitoes, although A. albopictus can also be a potential vector. The virus replicates in the dermal tissue and subsequently spreads to draining lymph nodes and the reticuloendothelial system, where it precipitates a prolonged viremia that can persist for 7 to 10 days and spread infection to end organs. The viremia can result in transmission to an uninfected mosquito that is feeding on a viremic individual in regions where competent mosquito vectors and susceptible individuals coexist. Infectious virions and viral RNA can be found in genital secretions, and sexual transmission occurs especially in nonendemic regions. In pregnant women, the virus can infect the placenta and the fetus, thereby causing placental inflammation, injury, and insufficiency, with increasingly severe fetal outcomes associated with infection earlier in pregnancy. During fetal infection, Zika virus can infect epithelial cells, thereby resulting in diffuse congenital malformations, but it also infects astrocytes and neuroprogenitor cells in fetal brain tissue, thereby resulting in developmental defects in the central nervous system and microcephaly. A significant number of infants also develop ocular disease characterized by retinal and choroid disease that results in long-term visual disturbances and blindness.
In adults, Zika virus is asymptomatic in about 80% of cases. In symptomatic Zika virus infection, which follows an incubation period of about 7 days, patients develop a mild febrile syndrome characterized by fever, myalgias, arthralgias, a maculopapular rash, and conjunctivitis. In pregnant women, 20% of fetuses develop the severe neurologic abnormalities of congenital Zika syndrome.
The diagnosis is suggested by the acute clinical symptoms and is made by a positive Zika virus nucleic acid amplification testing of serum or urine. Patients generally should be tested for dengue virus IgM antibody as well because of the overlap in symptoms and epidemiology. If a symptomatic pregnant woman is diagnosed with acute Zika virus infection, prenatal ultrasound is often recommended. If amniocentesis is completed as part of clinical care, amniotic fluid should be analyzed for Zika virus nucleic acid amplification testing.
No approved therapy or vaccine is currently available for Zika virus, although a candidate has been shown to be safe and to produce durable immune responses. In immunocompetent adults, symptoms almost always resolve in 7 to 10 days without significant long-term complications. Less than 1% of cases develop Guillain-Barré syndrome ( Chapter 388 ) and typically 7 to 10 days after the onset of symptoms and after the resolution of fever. Children who were exposed to Zika virus in utero are at risk for a wide range of disabilities, including long-term learning disabilities, neurocognitive defects, and abnormal visual and auditory function resulting in long-term disability.
Chikungunya virus, which is in the Alphavirus genus, is an important cause of large epidemics of fever, arthralgia, and arthritis in tropical and subtropical regions throughout the world. Similar to other alphaviruses, Chikungunya virus is an enveloped, plus-strand RNA virus with icosahedral symmetry. The virions are 60 to 70 nm in diameter, and the lipid envelope is decorated with viral glycoproteins, which form heterodimers that are grouped into trimers that form 80 projections on the virion surface.
Chikungunya virus is a mosquito-borne virus that was first characterized in Tanzania but is now transmitted globally, especially where the primary vector, A. aegypti , is active. In addition to occasional outbreaks in Africa and Asia, a large outbreak on Reunion Island infected about 25% of the population in 2005. Since then, Chikungunya virus has produced epidemic fever, arthralgia, and arthritic disease in 114 countries and territories in India, Southeast Asia, Africa, Oceania/Pacific islands, South America, and Central America ( E-Fig. 352-3 ).
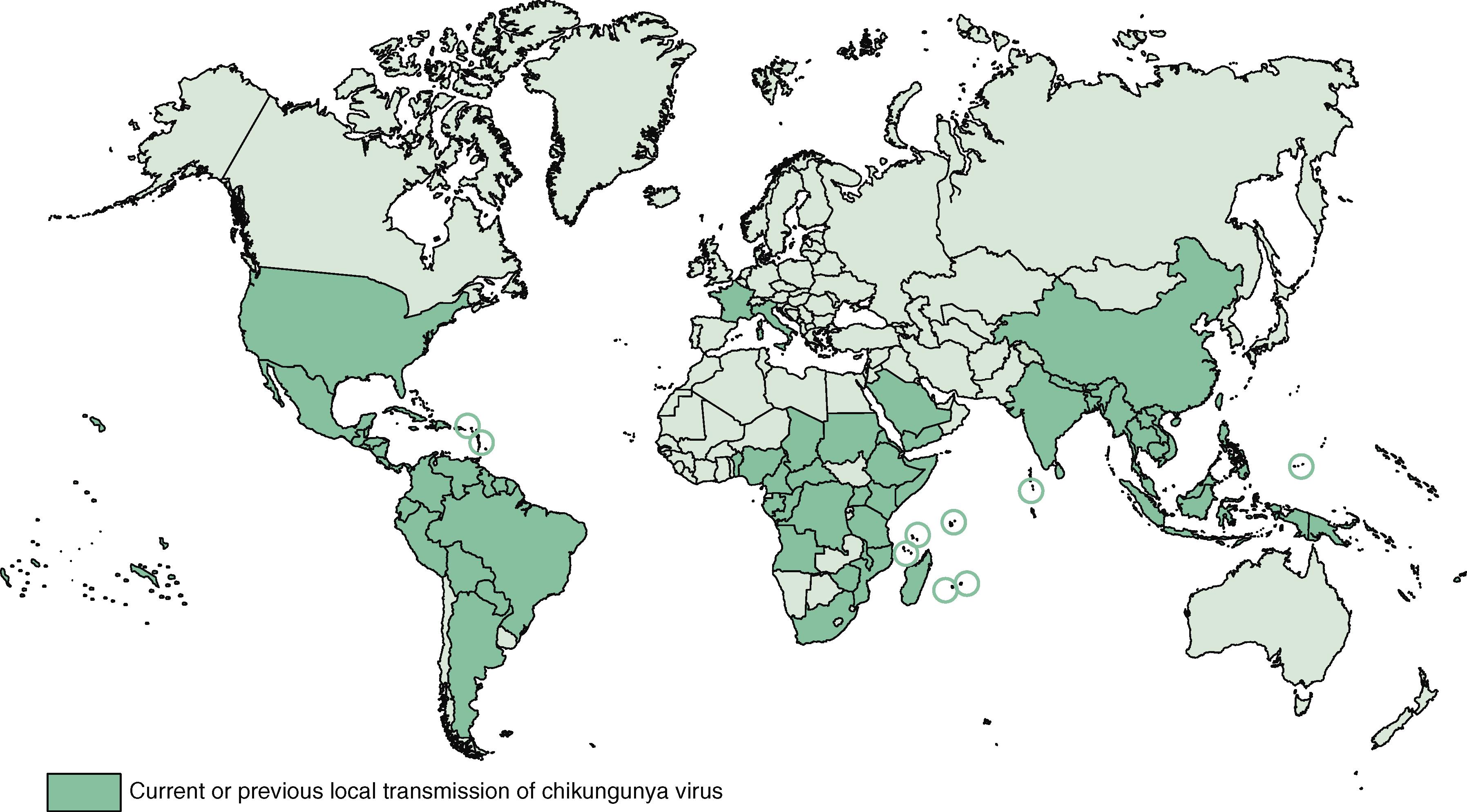
Transmission of Chikungunya virus is maintained in a sylvatic cycle between nonhuman primates and Aedes species of mosquitoes. Chikungunya virus can also maintain large urban epidemics with transmission between primarily A. aegypti or A. albopictus mosquitoes and humans. Following a bite from an infected mosquito, Chikungunya virus replicates in the dermal tissue and spreads to draining lymph nodes and the spleen, thereby resulting in viremia that may precede the onset of symptoms by 2 to 3 days. During acute viremia, direct infection of joint tissue, muscles, skin, and the central nervous system occurs, typically after 6 to 8 days of infection. The duration and magnitude of viremia are sufficient to transmit Chikungunya virus from humans to uninfected A. aegypti mosquitoes, thereby resulting in large urban outbreaks in tropical and subtropical regions.
Approximately 3 to 28% of infected individuals remain asymptomatic, but the remainder develop symptomatic illness characterized by fever (>102° F/39° C) and arthralgias. A maculopapular rash, which develops 4 to 8 days after the onset of fever, involves the trunk, extremities, palms, soles of feet, and face. Other symptoms may include headache, myalgias, arthritis, conjunctivitis, nausea, and vomiting. Joint symptoms, which can be debilitating, are typically manifested as a polyarthritis involving the hands, wrists, and ankles, although more proximal joints also can be involved. Patients may also develop myocarditis, hepatitis, retinitis, acute renal disease, and meningoencephalitis.
The differential diagnosis for Chikungunya virus includes other circulating arboviruses, such as dengue virus or Zika virus, as well as other causes of fever and arthralgias, including malaria ( Chapter 316 ), measles ( Chapter 338 ), enteroviruses ( Chapter 349 ), leptospirosis ( Chapter 298 ), and other endemic infections. Preliminary diagnosis is based on a compatible syndrome associated with exposure to a geographic region with Chikungunya virus transmission. During acute infection, diagnosis can be made using Chikungunya virus nucleic acid amplification testing of the serum. Detection of CHIKV IgM early in the course of disease or a four-fold increase in CHIKV IgG would support a diagnosis of acute CHIKV infection.
Similar to other arboviruses, treatment is supportive with oral or intravenous fluids and with acetaminophen to reduce fever and pain. NSAIDs ( Table 26-4 ) can be used to treat symptoms once dengue is ruled out. A safe and immunogenic vaccine is now approved for use in the U.S. Acute symptoms resolve in 7 to 10 days, and fatalities are rare. Some patients (5 to 80%) have chronic rheumatologic symptoms (e.g., polyarthritis, tenosynovitis, chronic arthralgias, Raynaud syndrome) and persistent and prolonged fatigue for several months after the acute infection. In some cases, the arthritis, pain, and swelling become chronic.
O’nyong-nyong virus is an alphavirus that causes a fever-myalgia syndrome similar to Chikungunya virus, is endemic to sub-Saharan Africa, and was first identified following an outbreak of fever and arthralgia syndrome in Uganda in 1959. O’nyong-nyong virus quickly spread throughout eastern and western Africa causing outbreaks of disease with infection rates ranging from 3 to 45%. Current seroprevalence rates vary from 0.8% in inland Kenya to as high as 56% in a coastal community in Kenya.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here