Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Ancient Egyptians were the first to recognize aortic aneurysms as early as 1550 bce, and Antyllus was the first to attempt treatment of aneurysms through ligation of the vessels, but this was met with poor results. , Through time our understanding of the anatomy, and pathophysiology of abdominal aortic aneurysms (AAA) has progressed and great advances have been made in the management of this complex disease. The 20th century marked a period of rapid advances in the open surgical management of aortic aneurysms by proximal ligation or obliteration (Cooper, 1817; Matas, 1888), extra-arterial wrapping (Poppe, 1946), and direct reconstruction with autologous (Carrel, 1948; Dubost, 1951) or synthetic (Voorhees, 1952) material. Vast improvements in the management and outcomes of AAA came with the work performed by DeBakey and Cooley in the 1950s. However, operative techniques remained relatively stagnant until 1990 when Juan Parodi performed the first endovascular stent of an infrarenal AAA. , Since the US Food and Drug Administration (FDA) approved the first stent grafts for use in repair of infrarenal aortic aneurysms in 1999, endovascular techniques have overtaken traditional open repair as the most common approach to the elective and emergent management of this condition (see Ch. 75 , Aortoiliac Aneurysms: Endovascular Treatment).
Arterial aneurysms are defined as an increase in arterial diameter by 1.5 times the normal diameter with involvement of all three anatomic layers. For the infrarenal aorta, an aneurysm is defined as greater than 3 cm or 50% larger than a normal proximal segment measured in either the anteroposterior or transverse dimension in a plane perpendicular to the longitudinal axis of the aorta. The original definition of a median aortic size of 2.2 to 2.3 cm is based from analysis of 70-year-old Caucasian subjects, but it is now known that there are variations in mean aortic diameter based on sex and race. However, the Aneurysm Detection and Management (ADAM) Veterans Affairs Cooperative Study Group concluded that these differences in average aortic size are not significant enough to change the previous definition of an aneurysm. , Pseudoaneurysms and penetrating aortic ulcers can result in focal increases in arterial diameter, but the pathophysiology of these diseases differs from that of aneurysms.
Aortic aneurysms can form anywhere along the aorta, but approximately 30% of aneurysms are found in the infrarenal aorta. Inferences regarding the affected patient cohorts, incidence, and prevalence of AAAs are drawn from autopsy and screening studies designed to target groups thought to be at increased risk for aneurysm, primarily based on age or sex ( Table 72.1 ). In the United States, the prevalence of AAA as defined by an aortic diameter >3 cm was found to be 1.4% among those between the ages of 50 and 84 years. Age has been determined to be the most significant risk factor for the development of AAA. This risk is increased in the age ranges of 65 to 69, with an odds ratio of 5.4, and 75–79 with an odds ratio of 14.5. Caucasian race and male sex are also additional risk factors for development with aneurysm incidence in women peaking later than in men.
| Study Location | Lederle, USA | Chichester, UK | Viborg, Denmark | Western Australia | MASS UK | Rotterdam, Netherlands | Tromso, Norway |
| N | 73,451 | 15,775 | 12,629 | 41,000 | 67,800 | 5419 | 6386 |
| Gender | Men and women | Men and women | Men | Men | Men | Men and women | Men and women |
| Age (years) | 50–79 | 65–80 | 65–73 | 65–79 | 65–74 | >55 | 55–74 |
| Sampling dates | 1992–1995 | 1988–1990 | 1994–1998 | 1996–1998 | 1997–1999 | 1994–1995 | 1994–1995 |
| Date published | 1997 | 1995 | 2005 | 2004 | 2002 | 1995 | 2001 |
| Aneurysm prevalence | 1.4% (1.4% in men, 0.2% in women) | 4.0% (7.6% in men, 1.3% in women) | 4.0% | 7.2% | 4.9% | 4.1% men, 0.7% women | 8.9% men, 2.2% women |
Smoking has been shown to be the single biggest modifiable risk factor in the development of AAA with a direct correlation to smoking duration. In fact, a smoking history of <0.5 pack per day for 10 years increases the risk of AAA. This has been shown to be dose-dependent with risk increasing by 12-fold when smoking more than one pack a day for over 35 years. Interestingly as smoking prevalence in the United States has decreased, so too has aneurysm prevalence.
Family history is not commonly reported in those with AAA, but when present it increases the risk of AAA development by 20%. , Additional risk factors with less strong associations include increased salt intake, atherosclerotic disease, history of myocardial infarction, peripheral vascular disease, cerebrovascular disease and hypertension. , Negative risk factors include female sex, African American race, and diabetes. , Between 7% and 11% of patients with chronic obstructive pulmonary disease (COPD) have an AAA. Overall, risk is diminished with smoking cessation, well-controlled diabetes mellitus, eating fruits and vegetables more than three times a week, and exercise more than once a week.
Adjacent arterial segments are often involved in aneurysmal dilation of the infrarenal aorta, with 5%–15% extending to the juxta- or suprarenal aorta and 10%–25% involving the iliac arteries. Synchronous thoracic aneurysms are present in 12% of cases, while femoral or popliteal aneurysms are present in 14% of male patients. However, AAAs have been reported in 62% of patients with popliteal artery aneurysms and in 85% of those with femoral artery aneurysms (see Ch. 85 , Lower Extremity Aneurysms). ,
Iliac artery aneurysms (IAAs) most commonly occur in the common iliac artery (CIA) (70%), typically in conjunction with AAA. The largest series of CIA aneurysms noted that 86% presented with AAA or had been treated for one in the past. Only 6.4% of patients will have isolated CIA aneurysms, and one-quarter of those will have bilateral CIA aneurysms. Internal iliac artery aneurysms (IIAAs) are quite rare, accounting for 0.04% of all aortoiliac aneurysms. Again, aneurysms of these vessels are defined as 1.5 times the normal vessel diameter (>1.85 cm in males or 1.5 cm in females). The prevalence of IAAs in patients with clinically relevant AAAs varies from 15% to 40%. Although most IAAs are degenerative in nature, injury from penetrating trauma or iatrogenic injury from operative intervention in the area of the iliac arteries may also play a role in their development. There is also an association of IAAs with vasculitides, such as Behçet disease, fibromuscular dysplasia, Takayasu arteritis and connective tissue disorders. There have been reports of mycotic IAAs, but these are very rare , (see Ch. 141 , Aneurysms Caused by Connective Tissue Abnormalities).
The natural history of IAA, particularly those occurring in isolation, is less well understood but deserves special mention. The Mayo Clinic group reported a median expansion rate in CIA aneurysms of 0.29 cm per year and reported no ruptures at less than 3.8 cm. A recent review of the literature on IIAAs noted that these aneurysms are quite large when they initially present, likely due to an anatomic location that precludes detection on physical examination unless the aneurysm impinges on surrounding anatomic structures sufficiently to produce symptoms associated with the compression of nerves or urogenital/colorectal structures. Asymptomatic aneurysms, discovered by physical examination or incidentally through imaging, averaged 5.1 cm in diameter, whereas nonruptured and ruptured symptomatic aneurysms averaged 7.6 and 8.3 cm, respectively (see Ch. 77 , Isolated Iliac Artery Aneurysms and their Management).
Rupture risk for AAA is stratified by the diameter of the aneurysm, with larger aneurysms conferring increased risk ( Table 72.2 ). Studies have shown that the rupture risk is less than 2% when the aneurysm is less than 4 cm and increases significantly when aneurysm diameter increases beyond 5 cm. , The UK Small Aneurysm Study also found that the risk of rupture was independently associated with female sex, larger AAA diameter at the time of initial diagnosis, smoking, lower FEV1, and higher mean arterial pressure. Risk of rupture was not associated with age, BMI, serum cholesterol or ankle/brachial index. Of note, females were shown to have a three-fold increased risk of rupture compared with males with similar aortic diameters. Modifiable risk factors shown to decrease the risk of rupture were control of hypertension and smoking cessation.
| AAA Diameter (cm) | Rupture Risk (%) |
|---|---|
| 3.0–3.9 | 0.3 |
| 4.0–4.9 | 0.5–1.5 |
| 5.0–5.9 | 1–11 |
| 6.0–6.9 | 11–22 |
| >7 | >30 |
Other aneurysm characteristics also appear to predict rupture risk. Saccular aneurysms carry increased risk of rupture when compared to fusiform aneurysms. This is due to the increased wall stress seen in asymmetric aneurysms. , Imaging characteristics that increase rupture risk are the presence of dissection, mural thrombus, or peripheral calcifications of the aneurysm sac. The UK Morphology of Ruptured AAA study reported that the most likely site for rupture is the middle third of the aneurysm at the point of greatest diameter.
Growth rate must be considered when discussing the progression of AAA. Unfortunately, evidence notes a high degree of variation in AAA diameter assessment when followed by ultrasound due to technician differences. A Canadian study showed that this variability decreased significantly when a measured change in diameter was greater than 0.78 cm. Growth rate is reported as 2.2 mm/year and is not different between men or women. Smoking and the presence of a larger aneurysm, however, increased the rate of growth while presence of diabetes decreased growth rate. Currently, intervention is indicated for AAA with a growth rate greater than or equal to 10 mm in a 12-month period.
Overall, death from AAA rupture has declined in estimates, this can likely be attributed to decline in smoking, increased public awareness, improved operative outcomes, and improvement in overall cardiovascular health. This is further supported by the fact that in countries where cigarette usage has not declined, aneurysm-related mortality continues to increase (see Ch. 76 , Ruptured Aortoiliac Aneurysms and their Management).
The descending thoracic and abdominal aortas are formed through embryonic fusion of the paired dorsal aortas, which communicate with the developing heart via the aortic arches. The wall of the mature aorta is composed of three layers: the intima, with its single layer of endothelial cells; the media, made up of smooth muscle cells within a structural protein matrix; and the adventitia, a tough layer of collagen fiber and fibroblasts (see Ch. 2 , Embryology and Developmental Anatomy).
The embryologic process appears to give rise to regional heterogeneity in different parts of the aorta, with variation in the response of neural crest-derived smooth muscle cells of the thoracic aorta versus mesoderm-derived smooth muscle cells in the abdominal aorta. This regional variation clearly affects the secretion of and susceptibility to various cytokines and growth factors thought to be implicated in aneurysm development. This heterogeneity is also reflected in the tissue characteristics of the aortic media within different anatomic segments. In the thoracic aorta, the media contains 55 to 60 lamellar units, with adventitial vasa vasorum penetrating the vascular zone of the outer layers, whereas there are 28 to 32 lamellar units in the abdominal aortic media. This makes the abdominal aorta relatively avascular compared with the more proximal aorta, relying more on the transintimal diffusion of oxygen and nutrients. ,
Inflammation has been implicated as a key component of aneurysmal degeneration of aortic tissue. Analysis of circulating biomarkers has been extensively studied in those with AAA. Specifically, fibrinogen, WBC, fibrinogen, d-dimer, NT-proBNP, and cTnT levels are elevated. There has also been a linear correlation found between levels of C-reactive protein and aortic diameter. Currently, none of these markers have the sensitivity, specificity, or validation to provide diagnostic prognosis for rupture risk. ,
AAA are the result of the media thinning due to destruction of smooth muscle cells and elastin, inflammatory cell infiltration, neovascularization and collagen deposition. Several factors play a role in the inflammatory component of aneurysmal degeneration. The accumulation of intramural thrombus (ILT) is one such component. It is thought that this effect is mediated through a cascade of platelet activation, ultimately leading to increased oxidative stress and proteolytic injury. Mural thrombus recovered from human AAA samples are notable for recruitment of inflammatory cells such as neutrophils and inflammatory cytokines. The thrombus itself is laminated, progressing from an initial luminal layer with fresh thrombus and cross-linked fibrin to an outer layer in contact with the aortic wall, the latter being characterized by advanced fibrinolysis. At this level, plasmin serves to activate matrix metalloproteinases (MMPs) and TGF-β and participates directly in matrix protein degeneration. The predominant MMPS are MMP 9, MMP2 and MMP12. , The importance of the biologic activity of the ILT is underscored by the observation that increased thrombus burden is associated with an increased AAA growth rate. Wall thinning, medial loss of smooth muscle cells, elastin degradation, and adventitial inflammation have all been associated with the presence of ILT ( Fig. 72.1 ). It has been suggested that the impact of thrombus burden may also be associated with aortic wall hypoxia. ,
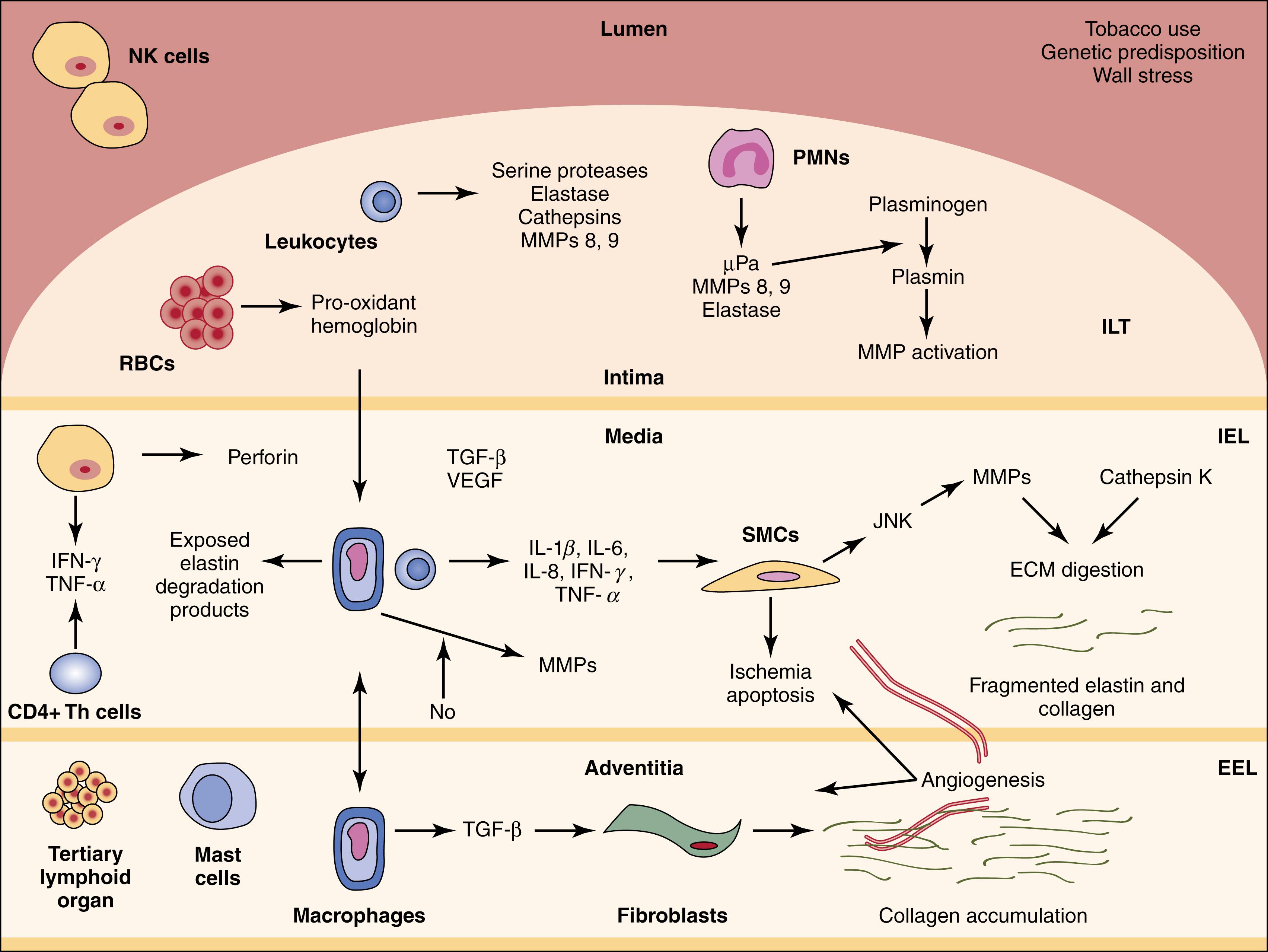
Although much attention has been focused on medial degeneration, marked changes in the adventitia have also been observed in AAAs. Inflammation – in the form of extensive periaortic and adventitial infiltration by lymphocytes, macrophages, and mast cells – is noted histologically. Adventitial degeneration is also characterized by extensive fibrosis. This fibrosis, driven by macrophage/TGF-β–mediated fibroblast proliferation and activation, may provide some degree of protection from rupture, but it may also be associated with pathologic retroperitoneal fibrosis, with adherence to and injury of adjacent retroperitoneal structures. Neoangiogenesis, typically limited to the adventitia and outer media, is also observed in AAAs.
There is also a clear genetic role in the development of AAA, with heritability estimated as high as 70%. If a patient has a first-degree relative with an AAA, their chance of developing an AAA is approximately doubled irrespective of the presence of a connective tissue disorder. , Further evidence is emerging that there are genetic risks for AAA development that do not fall into the spectrum of named connective tissue disorders. Large genome-wide association studies have recognized several novel gene loci that confer increased risk of AAA development regardless of family history. In addition to the novel genetic loci, several named connective tissue disorders confer increased risk for AAA development and subsequent rupture, and these are listed in Table 72.3 .
| Syndrome | Gene | Defect | Inheritance Pattern |
|---|---|---|---|
| Marfan syndrome | FBN1 | Defect in Fibrillin-1 | Autosomal dominant pattern |
| Loeys–Dietz syndrome | TGFBR1 gene mutations cause type I, TGFBR2 gene mutations cause type II, SMAD3 gene mutations cause type III, TGFB2 gene mutations cause type IV, and TGFB3 gene mutations cause type V | Overactive TGF-β pathway | Autosomal dominant pattern |
| Ehlers–Danlos vascular type (formerly type IV) | COL3A1 | Defect in collagen type III | Autosomal dominant pattern |
| Familial abdominal aortic aneurysm | Not specifically identified | ECM Defect in DAB2 interacting protein (DAB2IP) Defect in low-density receptor-related protein (LRP1) |
Unknown |
Frank bacterial infection of the aortic wall is also clearly associated with the development of mycotic aortic aneurysms (MAA). MAA are also known to be rapidly expanding and associated with high mortality. Fortunately, MAA only make up 0.6%–2% of all AAA. It has been proposed that bacterial colonization of either the aortic wall or ILT may contribute to AAA formation and progression either directly or indirectly through alteration of the immunologic milieu. Published literature indicates that the most commonly isolated species are Salmonella, followed by Staphylococcus species , Streptococcus species and E. coli .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here