Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Aortic valve replacement remains the gold standard for the treatment of severe aortic valve disease. However, valve repair is emerging as a feasible and attractive alternative to valve replacement in selected patients. Valve repair can reduce or eliminate the risks of prosthesis-related complications including thromboembolism, endocarditis, anticoagulant-related hemorrhage, and reoperation owing to structural valve deterioration among others. Analogous to the mitral valve, a reconstructive approach to the aortic valve requires a thorough and detailed understanding of three-dimensional valve anatomy, valve function, assessment and classification of pathologic lesions, and treatment of all affected components of the valve. In this chapter, we review the key features of aortic valve insufficiency, aortic valve and root anatomy, an approach to valve assessment and lesion classification, and a demonstration of commonly used reparative techniques for aortic valve repair. Furthermore, we review the outcomes of aortic valve repair in unselected cohorts and in distinct subsets of patients undergoing aortic valve preservation and repair.
The causes of aortic regurgitation (AR) are numerous and can be attributed to a disturbance in any of the components of the functional unit of the aortic valve (e.g., cusps, sinuses of Valsalva, sinotubular junction, ventriculoaortic junction). In general, the causes can be divided into those that affect the valve cusps (e.g., calcific degeneration, congenitally bicuspid valve, infective endocarditis, rheumatic disease, myxomatous degeneration) and those that affect the aortic root (e.g., aortic dissection, aortitis of various etiologies such as syphilis, connective tissue disorders such as Marfan syndrome).
The pathophysiology of AR is dependent on the acuity of onset and duration of the disease process. In acute AR, typically caused by aortic dissection, infective endocarditis, trauma, or valve prosthesis failure, there is a sudden increase in left ventricular end-diastolic volume because of the regurgitation. Since the left ventricle has limited compliance and does not have time to adapt, the left ventricular end-diastolic pressure (LVEDP) rises rapidly.
In chronic AR, there is a slow and insidious progression of left ventricular (LV) dilation and eccentric hypertrophy because of an increase in left ventricular end-diastolic volume, LVEDP, and wall stress. Dilation of the LV, while maintaining normal systolic function, increases total stroke volume and maintains forward flow. This increase in stroke volume coupled with an increase in LVEDP is associated with the wide pulse pressure typical of chronic AR. Eventually, the hypertrophic response is exhausted, and LVEF drops as afterload increases, leading to heart failure and its associated clinical presentation.
Patients with acute AR usually exhibit sudden or rapidly progressive cardiovascular collapse, which can be life threatening if not addressed promptly. They often exhibit ischemic symptoms because of the decrease in coronary blood flow and increased myocardial oxygen demand. In contrast, patients with chronic AR are asymptomatic for a long period of time because of the compensatory LV changes discussed earlier. Once the compensatory response is exhausted, the patients start having heart failure symptoms such as exertional dyspnea, orthopnea, and paroxysmal nocturnal dyspnea. Patients may also feel palpitations and angina.
The classic murmur of AR is an early diastolic, blowing, decrescendo murmur heard best at the level of the diaphragm at the left sternal border while the patient is sitting, leaning forward, and in deep exhalation. It may become holodiastolic in severe AR, and isometric exercises tend to accentuate the murmur. Classic signs of widened pulse pressure may also be found, including Corrigan or water-hammer pulse, De Musset sign (bobbing of the head with heart beats), Quincke pulse (pulsations of the lip and fingers), Traube sign (pistol shot sounds over the femoral artery), and Müller sign (pulsations of the uvula).
Transthoracic echocardiography with Doppler color-flow is the most useful tool for the diagnosis of AR. The jet width and vena contracta width on Doppler color-flow are used to qualitatively assess the severity of AR, whereas the regurgitant volume, regurgitant fraction, and regurgitant orifice area are used for the quantitative assessment.
Decision making for surgical intervention for aortic valve surgery needs to incorporate the natural history of medically managed disease, the risks associated with surgical intervention, and longer-term risks that might accrue related to prosthetic valve implantation. The class I recommendations for aortic valve intervention in patients with AR according to the 2014 American College of Cardiology and the American Heart Association are the following: symptomatic patients with chronic severe AR, asymptomatic patients with chronic severe AR and LV dysfunction (ejection fraction < 50%) at rest, and patients with chronic severe AR who are undergoing concomitant coronary artery bypass grafting, aortic surgery, or other heart valve surgery.
The class IIa recommendation is for patients with asymptomatic AR and normal LV systolic function (ejection fraction > 50%) but with severe LV dilation (end-systolic diameter > 50 mm). The class IIb recommendation is for patients with moderate AR who are undergoing coronary artery bypass grafting, aortic surgery, or other heart valve surgery. Aortic valve intervention may also be reasonable in asymptomatic patients with chronic severe AR, normal LV systolic function, and severe LV dilation (end-diastolic diameter > 65 mm) if the operative risk is low. Other considerations can include evidence of progressive LV dilation, declining exercise tolerance, or abnormal hemodynamic response to exercise.
However, aortic valve repair carries a similar, if not lower, risk of perioperative complication with a low risk of valve-related events over time. Similar to mitral valve repair for mitral regurgitation, there is some suggestion that aortic valve intervention should be considered earlier in patients in whom aortic valve repair is likely.
Another broad category of patients who undergo aortic valve preservation and repair are those with primary aortic pathology involving the aortic root and/or the ascending aorta and varying degrees of associated aortic valvular disease. In these patients, the primary indication for intervention is driven by aortic size, discussed in the American, European, and Canadian Guidelines.
From a technical perspective, all patients with primary aortic insufficiency are potentially candidates for repair. However, the success of aortic valve repair is determined largely by the quality of cusp tissue available. Thus, patients with significant leaflet calcification, destruction owing to active endocarditis, or rheumatic involvement are least likely to undergo successful and durable aortic valve repair. In contrast, repair has been shown to have good results in patients with bicuspid (and in smaller series, unicuspid, and quadricuspid ) aortic valves, despite the abnormalities in cusp anatomy. An important limitation to the universal application of aortic valve repair techniques is the lack of surgical expertise and experience in this field; however, this is changing rapidly with increasing interest in aortic valve repair. Patients who are candidates for repair should be referred to centers with appropriate expertise.
The anatomy of the aortic valve and root is familiar to cardiac surgeons. However, there are some features outlined here that are particularly relevant to aortic valve preserving and repair surgery. Like the mitral valve, aortic valve function involves an important interaction between the valve annulus and leaflets. Importantly, however, the annulus of the aortic valve is not a single structure but rather consists of three different components—namely, the sinotubular junction, the ventriculoaortic junction, and the anatomic crown-shaped annulus, which serves as the insertion point of the aortic valve cusps ( Fig. 78-1 ). These components work together to facilitate normal valve function and together are termed the functional aortic annulus . The aortic valve leaflets insert into the aortic annulus proximally at the ventriculo-aortic junction (VAJ) and distally at the sinotubular junction (STJ). In a normal aortic valve, the cusps coapt at the center of the aortic valve orifice with a coaptation height that is approximately at the mid-level between the VAJ and the STJ. The height of the sinuses of Valsalva (from the VAJ to the STJ) corresponds to the external diameter of the STJ, which can be useful to size prostheses for aortic root replacement and to assess cusp geometry after aortic valve repair.
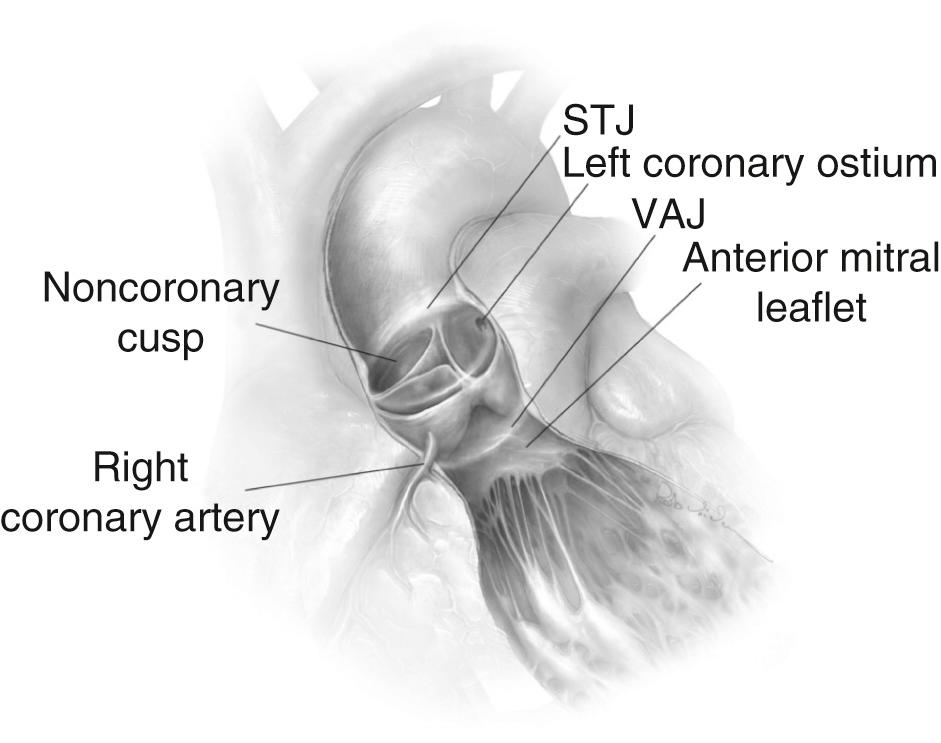
As a functional entity, the aortic valve consists of the functional aortic annulus (FAA), and the valve cusps. The integrity of these two functional components is the basis for good valvular function, and alteration in one of these components is frequently associated with alteration in the other. Thus, a fundamental principle in aortic valve repair is that lesions of the cusps and the FAA should both be addressed at the time of valve repair.
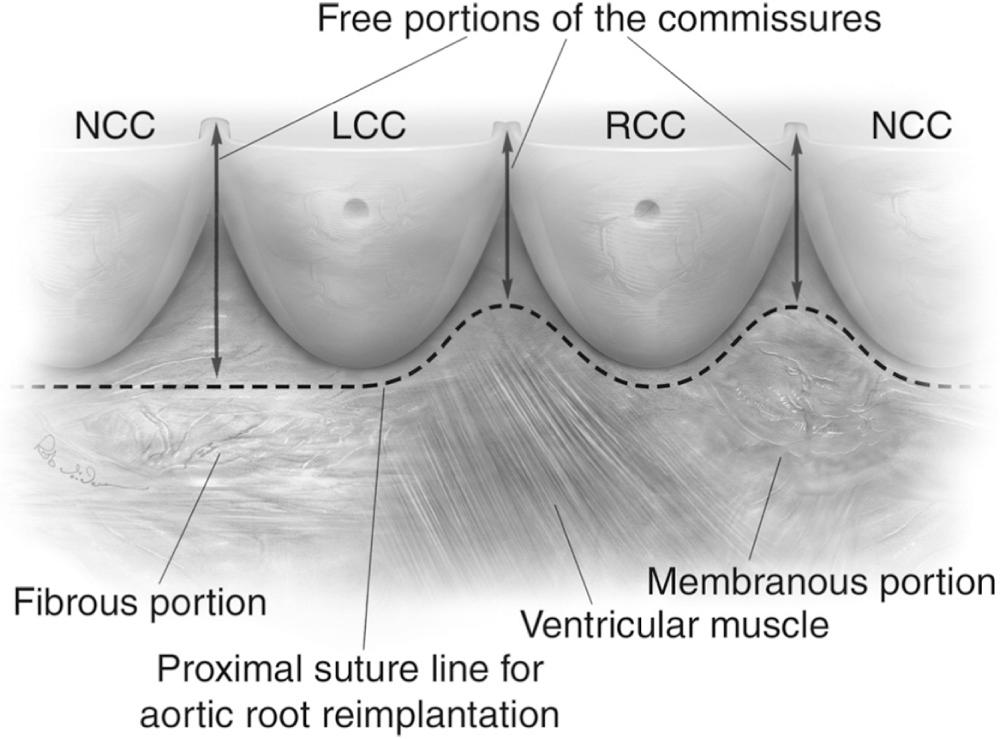
The anatomy of the subvalvular region of the aortic valve and its surrounding structures also has important implications for aortic valve repair ( Fig. 78-2 ). An important observation is that external dissection of the aortic root from its surrounding structures is limited by the membranous septum (at the junction of the noncoronary [NC] and right coronary [RC] cusps) and by ventricular muscle (at the junction of the left coronary [LC] and RC cusps), whereas at all other points, external dissection down to the level of the anatomic valve annulus is possible and necessary when valve-sparing root replacement is performed using the reimplantation technique. Thus, the proximal suture line for the aortic valve reimplantation procedure follows these external limitations in a curvilinear fashion.
Until recently, a major limitation to the more generalized application of aortic valve repair techniques was the absence of a common framework for valve assessment to help guide the approach to valve repair. Important lessons in this regard can be learned from the development of mitral valve repair. The Carpentier classification of mitral valve insufficiency was responsible, in large part, for the development and generalized dissemination of repair techniques for the mitral valve because it provided a common language for cardiologists, anesthesiologists, and surgeons to communicate about disease mechanisms and pathology. Key characteristics of that classification system were that it encompassed the entire spectrum of disease, it clarified and provided insight into the mechanism of insufficiency, it could be consistently applied using different assessment modalities (i.e., echocardiography and surgical assessment), it guided the repair techniques, and it provided a framework for the assessment of long-term outcome for differing mitral valve pathologies.
Over the past decade, a similar classification of aortic valve insufficiency has been developed with the above characteristics in mind ( Fig. 78-3 ). This classification centers on the idea that the aortic valve, much like the mitral valve, consists of two major components: the aortic annulus and the valve leaflets. Contrary to the mitral valve, however, the annulus of the aortic valve is not a single anatomic structure. The functional aortic annulus consists of two separate components, namely the ventriculoaortic junction and the sinotubular junction. As in Carpentier's classification of mitral valve disease, regurgitation associated with normal leaflet motion is designated as type I. This is largely due to lesions of the functional aortic annulus with type 1a aortic insufficiency (AI) owing to sinotubular junction enlargement and dilation of the ascending aorta, type Ib owing to dilation of the sinuses of Valsalva and the sinotubular junction, type Ic owing to dilation of the ventriculoaortic junction, and lastly type 1d owing to cusp perforation without a primary functional aortic annulus lesion. Type II AI is due to leaflet prolapse secondary to excessive cusp tissue or due to commissural disruption. Type III AI is due to leaflet restriction, which can be found in bicuspid, degenerative, or rheumatic valvular disease because of calcification, thickening, and fibrosis of the aortic valve leaflets.
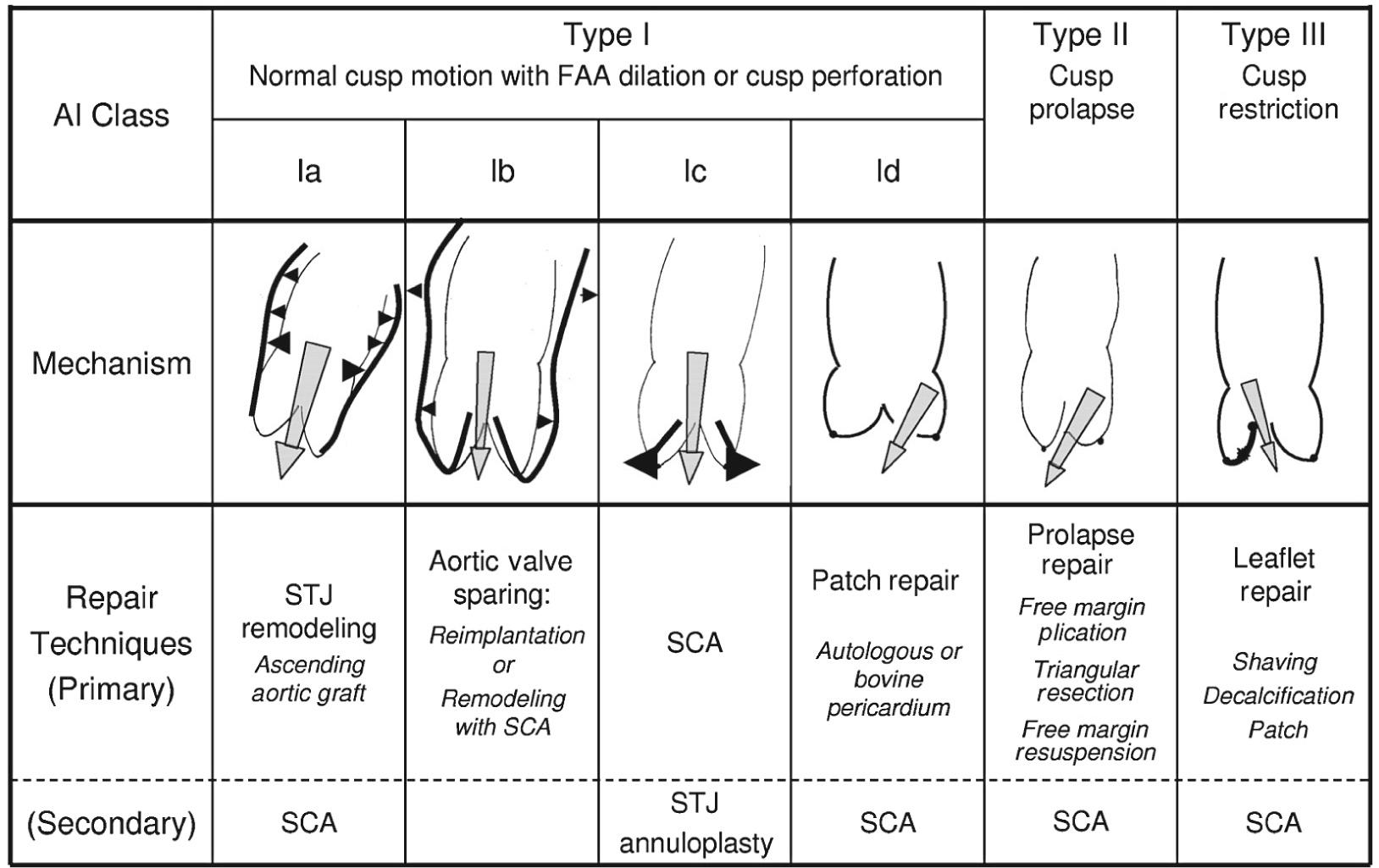
Patients can exhibit either single or multiple lesions contributing to their aortic insufficiency. For example, patients with isolated type Ib AI (owing to dilation of the sinuses of Valsalva) are expected to have a central regurgitant jet. Thus, the presence of a sinus of Valsalva aneurysm with an eccentric AI jet suggests concomitant leaflet prolapse (type II) or restriction (type III). Further assessment of leaflet anatomy can help to better delineate the different mechanisms contributing to AI. Once the mechanism of AI is well understood, the classification system can help to guide the surgeon in the choice of surgical techniques for correction of the pathology.
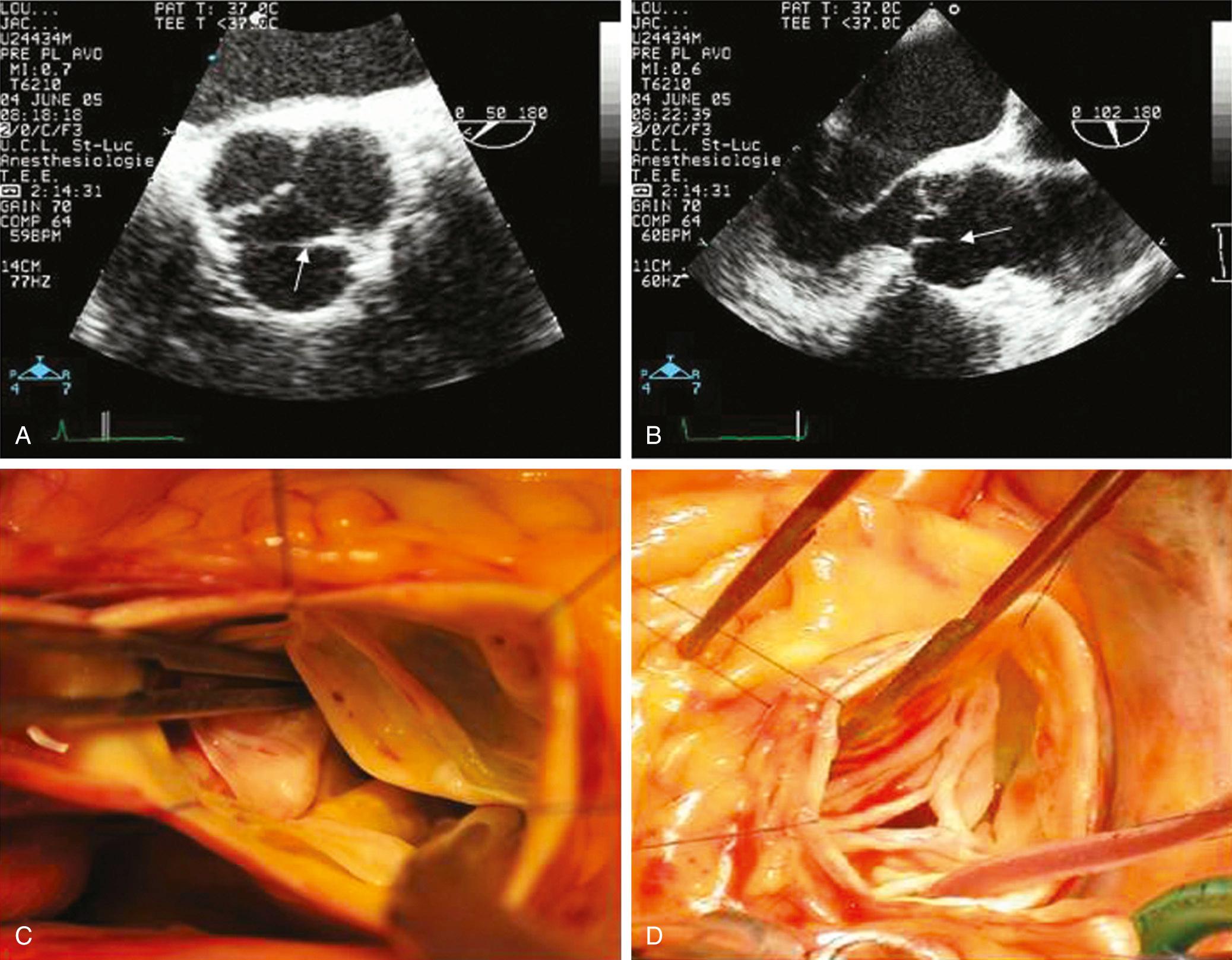
Aortic valve repair procedures are generally performed through a median sternotomy. Arterial cannulation is performed distal to any diseased aortic segments, typically in the distal ascending aorta or aortic arch. Alternatively, axillary artery cannulation may be performed in the setting of aortic arch pathology. A single, two-stage venous cannula is inserted through the right atrial appendage unless alternative cannulation strategies are required to treat coexisting pathology. After cardioplegia, a transverse aortotomy is performed approximately 1 cm above the sinotubular junction starting above the NC sinus and the posterior 2 to 3 cm of aortic wall is left intact. The distal aorta is retracted cephalad. Full-thickness 4-0 polypropylene traction sutures are placed at the three commissures and retracted using clamps, but not tied, to permit a dynamic assessment of valve anatomy. Axial traction is applied (perpendicular to the level of the annular plane) on the commissural traction sutures. This maneuver demonstrates physiologic aortic valve closure position, and the area and height of coaptation is observed. Leaflets are inspected to assess mobility, restriction, calcification, and prolapse. A prolapsing cusp will exhibit a transverse fibrous band at this time, which is also visible on echocardiography ( Fig. 78-4 ).
Type 1 lesions are most frequently due to dilation of the various components of the functional aortic annulus; they can occur in isolation or in association with cusp disease. A type 1a lesion occurs because of a supracoronary ascending aortic aneurysm with concomitant dilation of the STJ. This is corrected by replacing the ascending aorta and remodeling the STJ using a Dacron tube graft. When significant associated AI is present, subcommissural annuloplasty is also performed. Aneurysms of the aortic root (type 1b) are frequently associated with dilation of the STJ and the VAJ. These aneurysms are treated using valve-sparing root replacement, preferentially using the reimplantation technique because it provides better stabilization of the VAJ. Aortic root remodeling can also be used to treat aneurysms of the aortic root, and it is particularly useful when only one or two sinuses are involved or in patients in whom there is no risk of ongoing VAJ dilation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here