Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Anterior segment developmental anomalies (ASDA) are rare but potentially visually devastating. It is important for the pediatric ophthalmologist to know about certain important features when faced with a child with ASDA. It is worth remembering that if there is a developmental anomaly in the eye, there could be one in another organ system. A review by a pediatrician is often fruitful. Specifically, it is important for the pediatric ophthalmologist to be aware of the causes, evaluation, and treatment of these conditions. This chapter discusses the phenotype, genotype, and management options for children with these rare conditions. For ASDA affecting the cornea imaging is essential since it helps to assess the anatomical structure, which can affect prognosis.
Most disease-causing genes proven to play critical roles in ASDA encode transcription factors ( Table 31.1 ), which act by regulating the transcription of their downstream target genes. The explosion of molecular testing and investigation for ASDA would need a chapter dedicated purely to the genes now identified if it were to be comprehensive. Many of these genes have been identified in single case reports or one or two pedigrees. Next generation sequencing and panels are being developed all the time. Examples of the commonest transcription factors involved in the etiology of ASDA are PAX6 , PITX2 , FOXC1 , FOXE3 , and MAF . In this chapter, additionally, those genes that are known to play an important role in the etiology of specific ASDA will also be discussed.
| Human gene | Type | Chromosome location | Human disease | OMIM number |
|---|---|---|---|---|
| CYP1B1 | Enzyme | 2p22 | Congenital glaucoma | 601771 |
| EYA1 | Transcription factor | 8q13 |
|
601653 |
| FOXC1 | Transcription factor | 6p25 |
|
601090 |
| FOXE3 | Transcription factor | 1p23 |
|
601094 |
| LMX1B | Transcription factor | 9q34 | Nail–patella syndrome | 602575 |
| MAF | Transcription factor | 16q23 | ASDA and cataracts | 177075 |
| PAX6 | Transcription factor | 11p13 |
|
106210 |
| PITX2 | Transcription factor | 4q25 |
|
601542 |
| PITX3 | Transcription factor | 10q25 | ASDA and cataracts | 602669 |
| LTBP2 | Latent TGFβ-binding protein 2 | 14q24.3 | Megalocornea, microspherophakia, congenital glaucoma | 602091 |
| COL4A1 | Collagen, type IV, alpha-1 | 13q34 | Cataract/ASDA | 120130 |
| B3GLCT | UDP-Gal:beta-GlcNAc beta-1,3-galactosyltransferase-like | 13q12.3 | Peters-plus syndrome | 610308 |
| KERA | Keratan sulfate proteoglycan | 12q21.33 | Corneal plana | 603228 |
| PXDN | Extracellular matrix-associated protein, peroxidasin | 2q25.3 | ASDA cataracts microcornea | 605158 |
| SLC4A11 | Sodium borate cotransporter | 20p13–p12 | Corneal dystrophy, Fuchs endothelial, 4 | 610206 |
| JAG1 | Ligand of the Notch Receptor | 20p12 | Alagille syndrome with posterior embryotoxon, iris hypoplasia, small corneal diameter, iridocorneal synechiae, and corectopia | 601920 |
| LAMB2 | Extracellular matrix protein, laminin | 3p21 | Pierson syndrome with microcoria or ASDA | 15032 |
| BMP4 | Regulatory molecule | 14q22–q23 | ASDA with or without anophthalmia/microphthalmia | 112262 |
The patterns of expression of genes whose mutation causes ASDAs can be considered as three types: those expressed within migrating periocular neural crest cells ( FOXC1 , PITX2 ); those expressed only within the developing lens ( FOXE3 , MAF ); and those expressed within the whole eye including the lens, of which PAX6 is the only example.
The lens plays an essential role in the induction of anterior segment differentiation. Two genes are implicated in causing ASDA by affecting the inductive properties of the lens. MAF and FOXE3 mutations both cause ASDA with cataracts. Recent work has shown that homozygous mutations in FOXE3 result in primary congenital aphakia.
ASDAs may be considered in terms of their embryologic origin. Therefore they may be:
of neural crest cell origin
of ectodermal origin
of global origin.
This prominent, anteriorly displaced Schwalbe's line, seen in 8%–15% of the normal population appears as a whitish, irregular ridge up to several millimeters from the limbus and is often incomplete ( Fig. 31.1 ). It may be inherited in an autosomal dominant fashion.
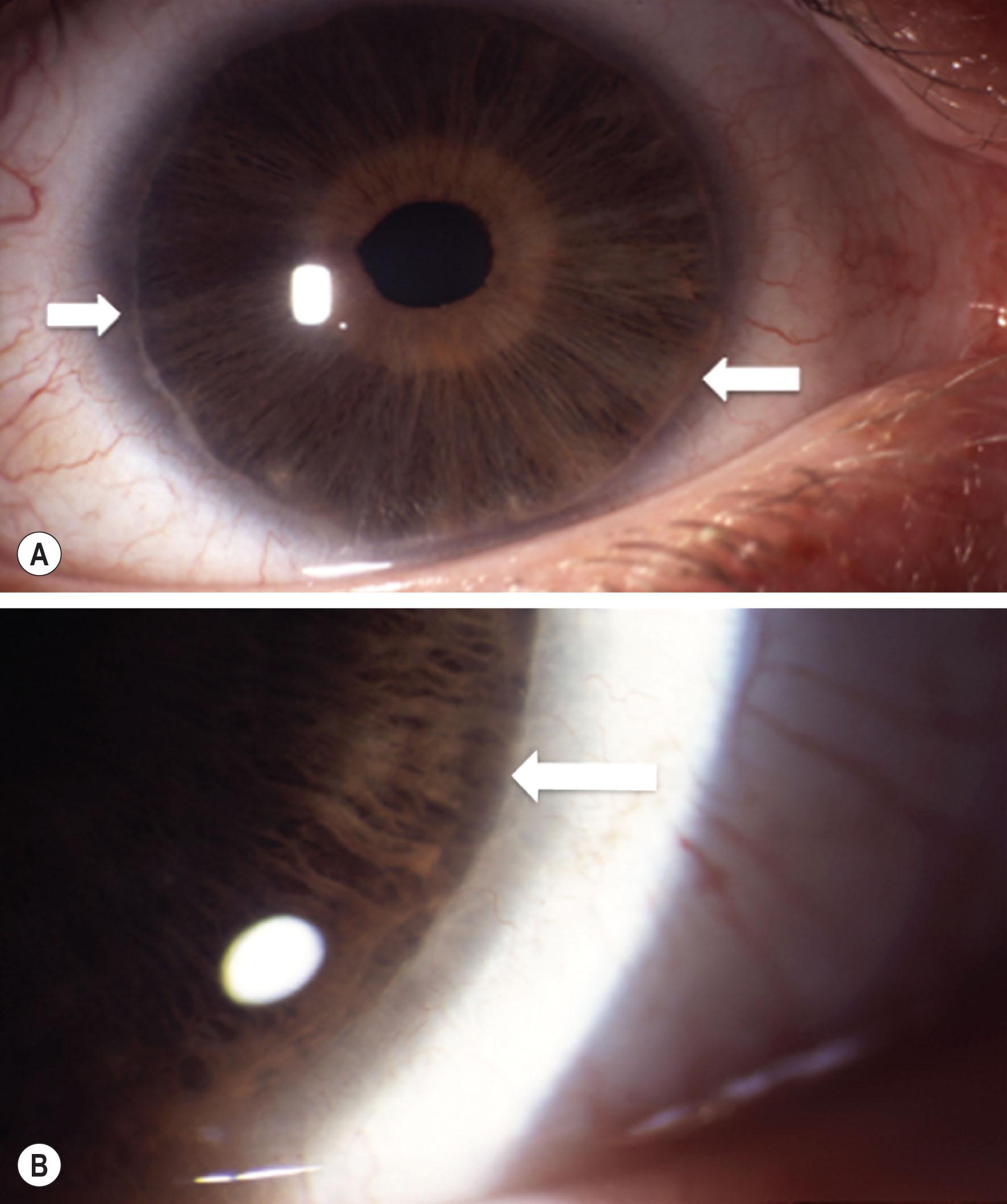
Ocular associations may include iris adhesions with or without iris changes such as hypoplasia, pseudopolycoria, or corectopia, in which case it forms part of the spectrum of the Axenfeld–Rieger anomaly or syndrome (see Fig. 31.2 ).
The presence of posterior embryotoxon in a jaundiced neonate is suggestive of the autosomal dominant condition Alagille syndrome (arteriohepatic dysplasia), which is characterized by intrahepatic cholestasis, peripheral pulmonary artery stenosis, peculiar facies, and butterfly vertebral arch defects. Posterior embryotoxon is seen in 90% of all cases and 77% of cases also have iris strands.
Since isolated posterior embryotoxon is not associated with glaucoma, there is no need for regular intraocular pressure checks unless other members of the family have features of Axenfeld–Rieger anomaly/syndrome.
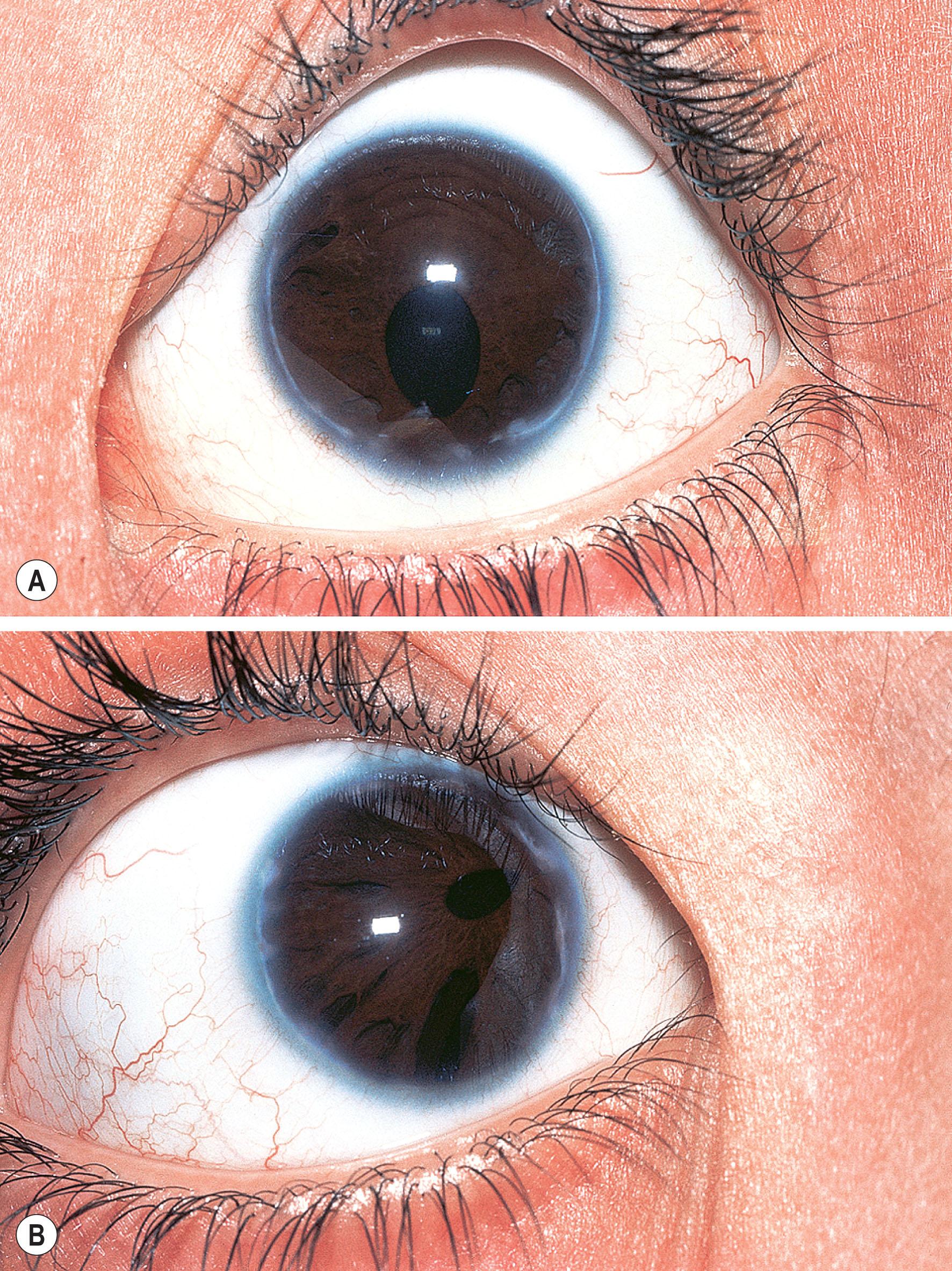
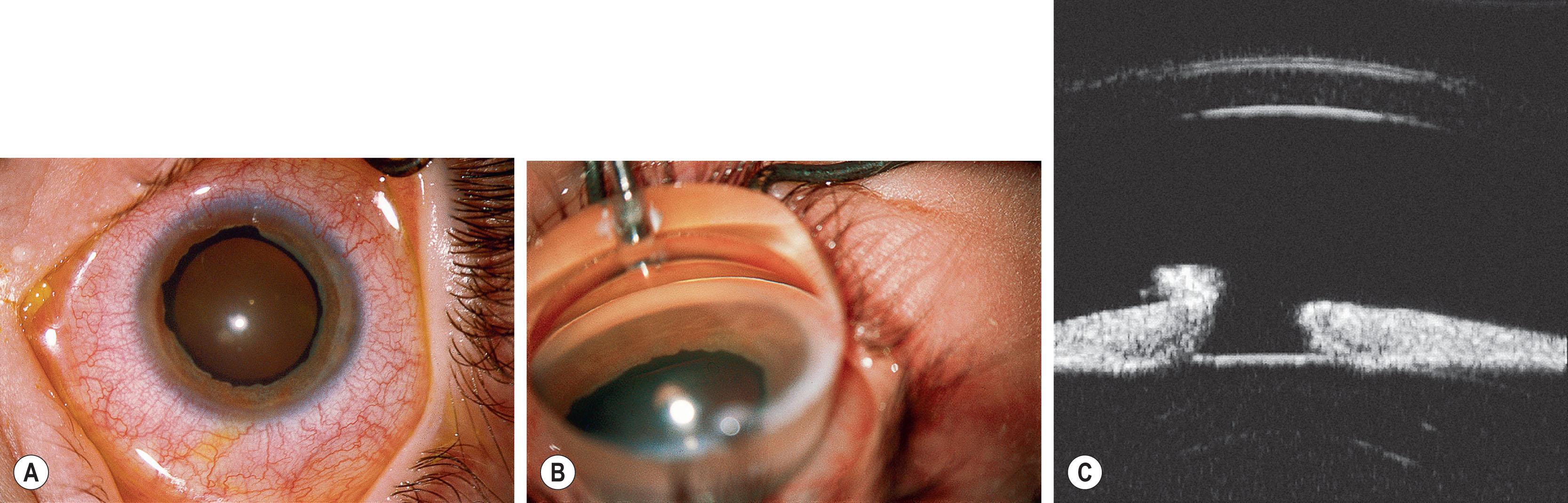
Axenfeld–Rieger syndrome (ARS) has ocular and non-ocular features. The ocular features consist of iris strands attached to posterior embryotoxon. Some of the strands may be very broad and thick and others thread-like. Historically, if these were the only findings, the term Axenfeld anomaly was used. If, in addition, iris defects were present, then this was termed Rieger anomaly. Axenfeld–Rieger anomaly (ARA) now encompasses both. Iris findings range from stromal hypoplasia, pseudopolycoria, corectopia (displaced anomalous pupil; Figs. 31.2–31.4 ), and ectropion uveae. Occasionally the iris may be almost completely absent – a severe form of hypoplasia and is usually seen in cases of FOXC1 deletion ( Fig. 31.5 ). It is important to differentiate this from PAX6 -related aniridia; the lack of foveal hypoplasia, polar lens opacities, nystagmus, and sometimes corneal pannus gives the clue to the diagnosis of ARA. The anterior chamber angle is usually open though there may be a high insertion of the iris into the posterior portion of the trabecular meshwork. Occasionally, the pupil corectopia is severe enough to warrant surgical pupilloplasty. The pupil may still become progressively more eccentric over years despite surgery.
Angle and iris changes are usually stable. Glaucoma develops in 50%−60% of patients with ARS, usually manifesting itself in childhood or young adulthood. Incomplete development of the trabecular meshwork and Schlemm's canal is thought to result from development arrest occurring during the third trimester, causing obstruction to aqueous outflow. Other ocular features may include strabismus, cataracts, limbal dermoids, retinal detachment, macular degeneration, chorioretinal colobomas, and choroidal and optic nerve head hypoplasia.
Familial glaucoma iridogoniodysplasia has been described in one pedigree and entails marked iris hypoplasia, iridocorneal angle anomalies, and, frequently, glaucoma. Iridogoniodysgenesis anomaly (IGDA) has been described as iridocorneal angle anomalies, iris stromal hypoplasia, and glaucoma in 50% of cases. Iridogoniodysgenesis syndrome (IGDS) is a rare condition in which iris hypoplasia and iridocorneal angle anomalies are associated with non-ocular features such as jaw and dental abnormalities. Both IGDA and IGDS are caused by mutations in PITX2 and FOXC1 .
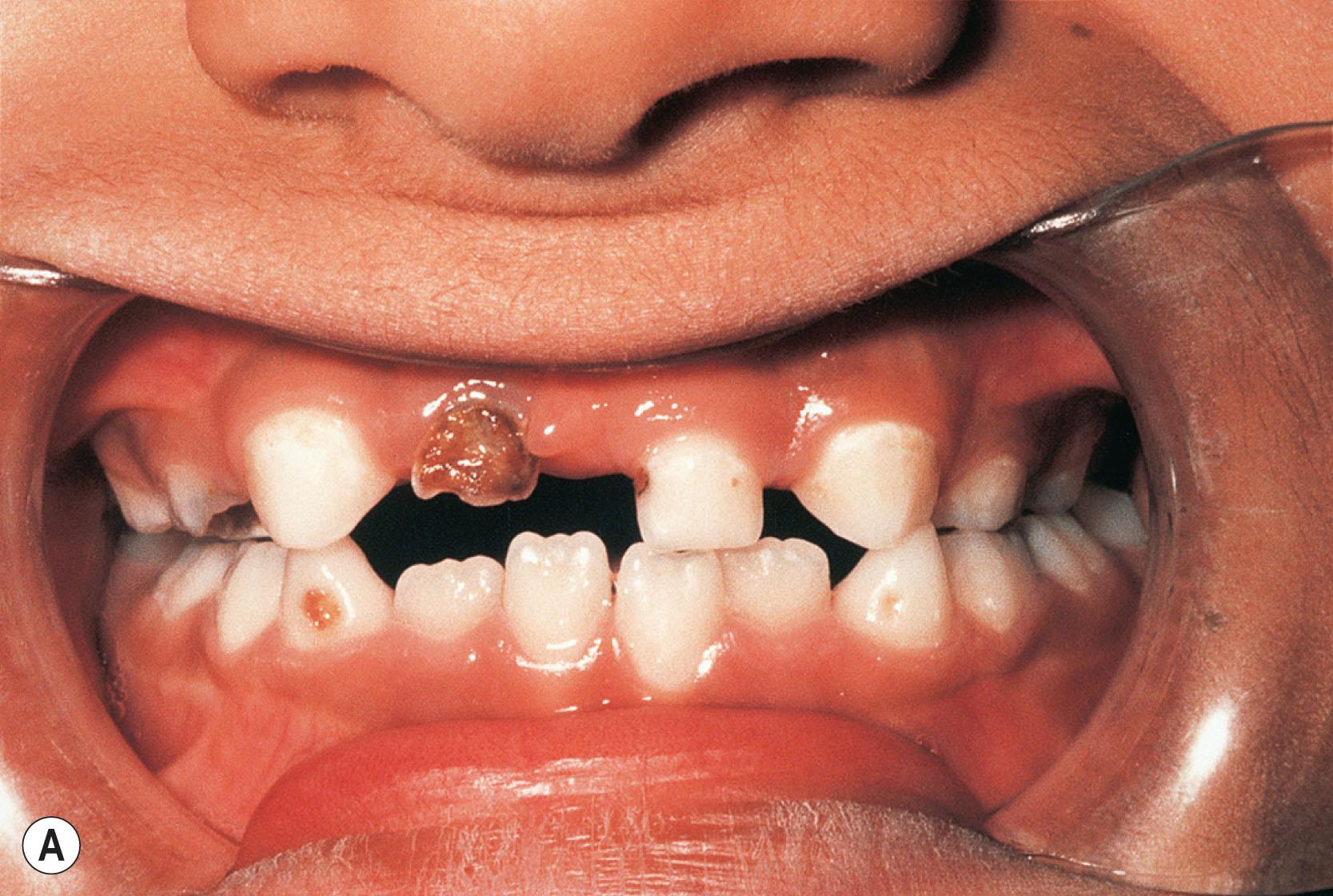
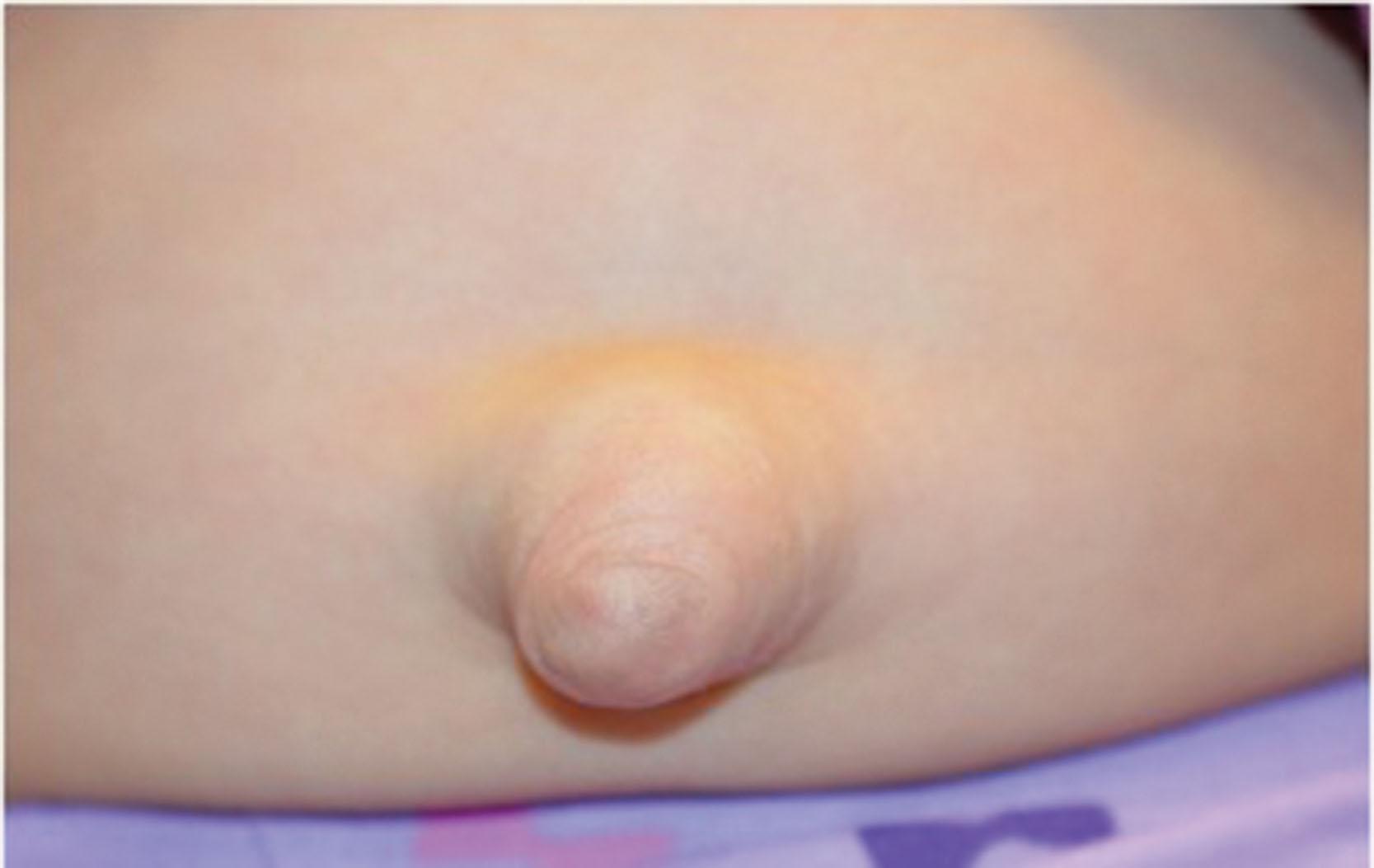
The characteristic non-ocular features of ARS are maxillary hypoplasia, mild prognathism, hypodontia (decreased but evenly spaced teeth), anodontia/oligodontia (focal absence of teeth), microdontia (reduction in crown size), cone-shaped teeth ( Fig. 31.6 ), and excess periumbilical skin ( Fig. 31.7 ) with or without hernia. Hypertelorism, telecanthus, and a broad flat nose have also been described. Other systemic features may include growth hormone deficiency and short stature ( PITX2 usually), heart defects, middle ear deafness, mental deficiency, cerebellar anomalies ( FOXC1 mutations), oculocutaneous albinism, hypospadias, abnormal ears, and, in one pedigree, myotonic dystrophy and Peters anomaly.
Management depends on the presenting complication of the structural anomalies. Some authors suggest that every child with ARS should get a magnetic resonance imaging (MRI) scan given the increased incidence of intracranial anomalies found in PITX2 - or FOXC1 -positive ARS.
Occasionally there is severe pupillary stenosis for which a large pupilloplasty is required, which may be complicated by lens damage and/or late contraction. Severe corectopia without severe stenosis can be adequately treated with occlusion therapy of the less affected eye. Sometimes the pupil is very large, irregular, and distorted (see Fig. 31.3 ).
Medical therapy should be used for glaucoma prior to surgical intervention with the exception of infantile cases. Here goniotomy/trabeculotomy should be considered carefully because angle surgery is less successful than filtration surgery. Miotics should be used with caution, since they may cause trabecular meshwork collapse, with a reduction in aqueous outflow.
Trabeculectomy with antimetabolite augmentation appears to be the procedure of choice for most patients with glaucoma secondary to ARS, especially in older children. The use of cycloablation should be considered for children with limited cooperation where a drainage procedure would be unsuitable. Draining procedures other than trabeculectomy include the use of drainage tubes with or without antimetabolite.
This rare condition is due to homozygous or compound heterozygous mutations affecting CYP1BI gene. While congenital glaucoma is seen in these cases there is also corneal clouding that is not alleviated completely by intraocular pressure control. Unlike the persistent corneal opacity seen in refractory or persistent congenital glaucoma, in these cases the horizontal corneal diameter is seldom more than 11 mm and the corneas lack Haab striae. Histologically the central cornea lacks Descemet membrane and endothelium posteriorly and has a thin or absent Bowman layer anteriorly. Peripherally all these structures are present. While penetrating keratoplasty is effective the glaucoma control is crucial to ensure successful outcome .
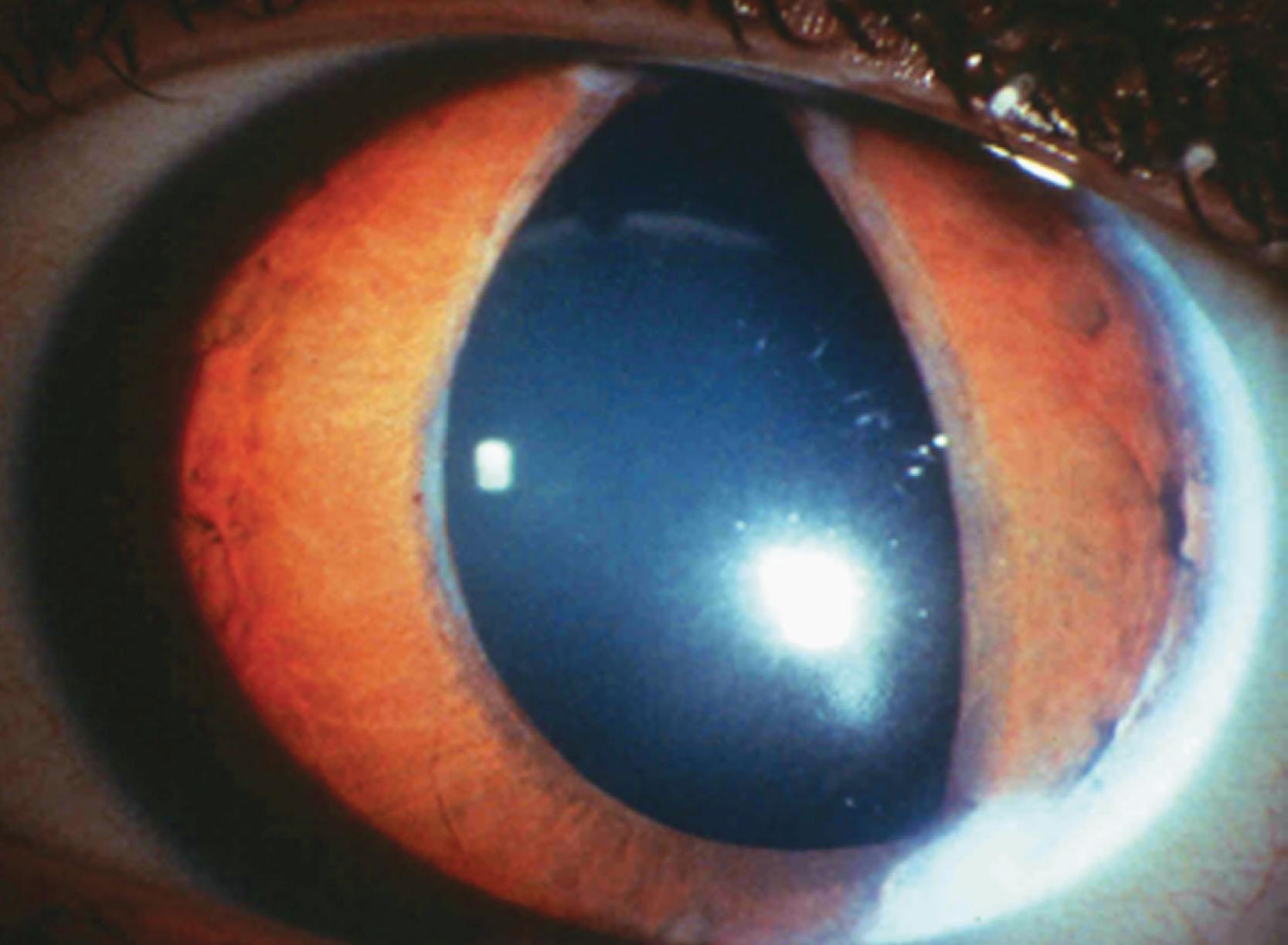
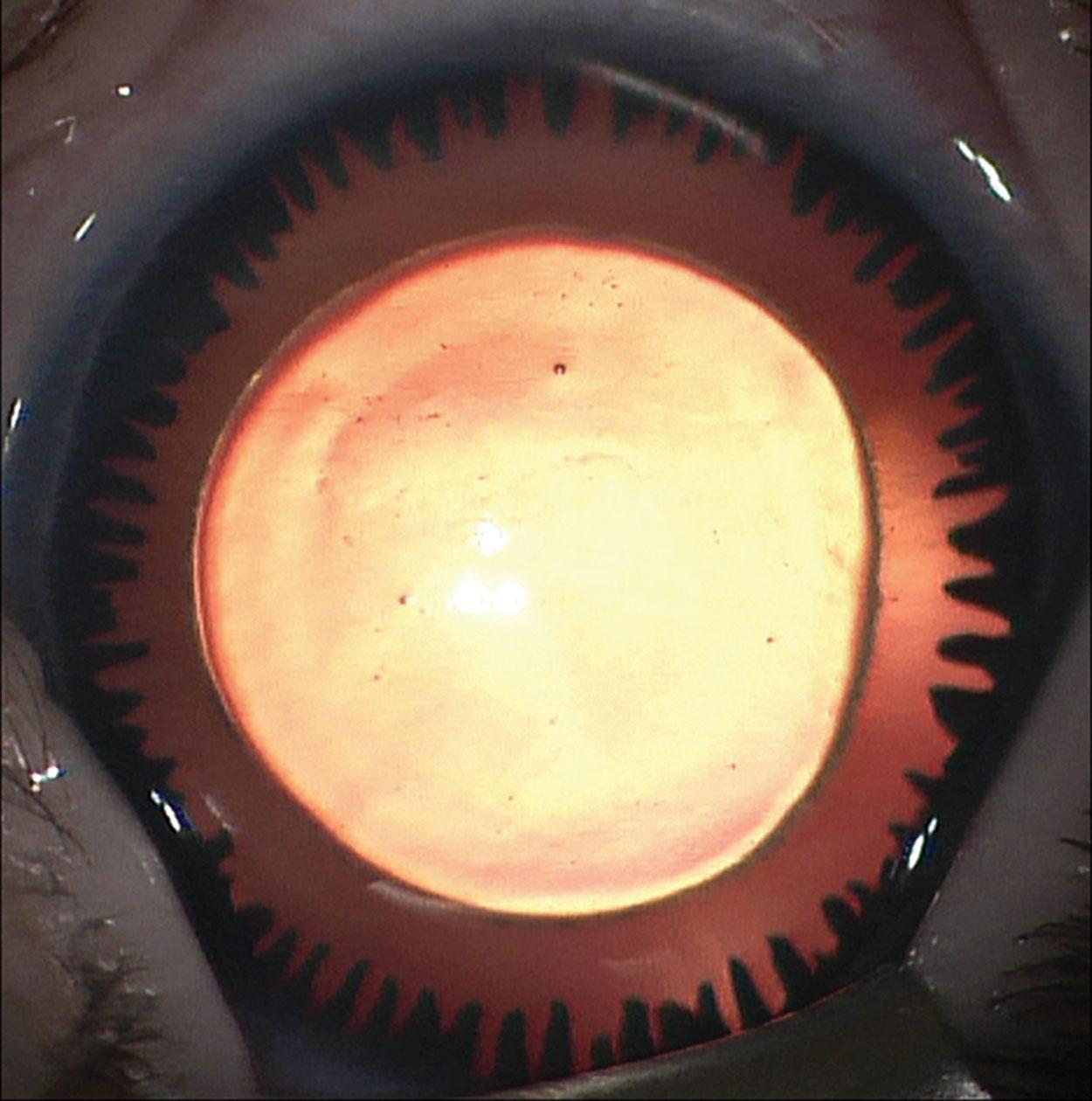
This is a rare, usually unilateral, condition in which there is a congenital, non-progressive, non-tractional hyperplasia of the posterior pigment iris epithelium onto the anterior surface of the iris (ectropion). The child is often thought to have mydriasis and anisocoria because of the dark color of the posterior pigment epithelium. The ectropion may be circumferential ( Fig. 31.8 ) or sectorial. Other ocular features include iris stromal hypoplasia, a high iris insertion into the trabeculum with goniodysgenesis, and secondary glaucoma, including angle closure glaucoma.
The clinical features are thought to result from an arrest in development with abnormal retention of primordial endothelium, which explains the central iris and angle changes. Although the affected pupil reacts to light and accommodation, it may not do so at the same speed as the unaffected eye. Glaucoma occurs in the majority of these patients, usually between early childhood and puberty. Neurofibromatosis type 1 and Prader–Willi syndrome may be associated.
Management of the glaucoma can be difficult; medical therapy is often unsuccessful, as is goniotomy, so augmented trabeculectomy is often the surgical operation of choice. If trabeculectomy fails, a drainage tube is often needed. Some authors prefer to use the drainage tube as the first line of treatment if medical management fails. Recently success with trabeculotomy has been reported in this condition.
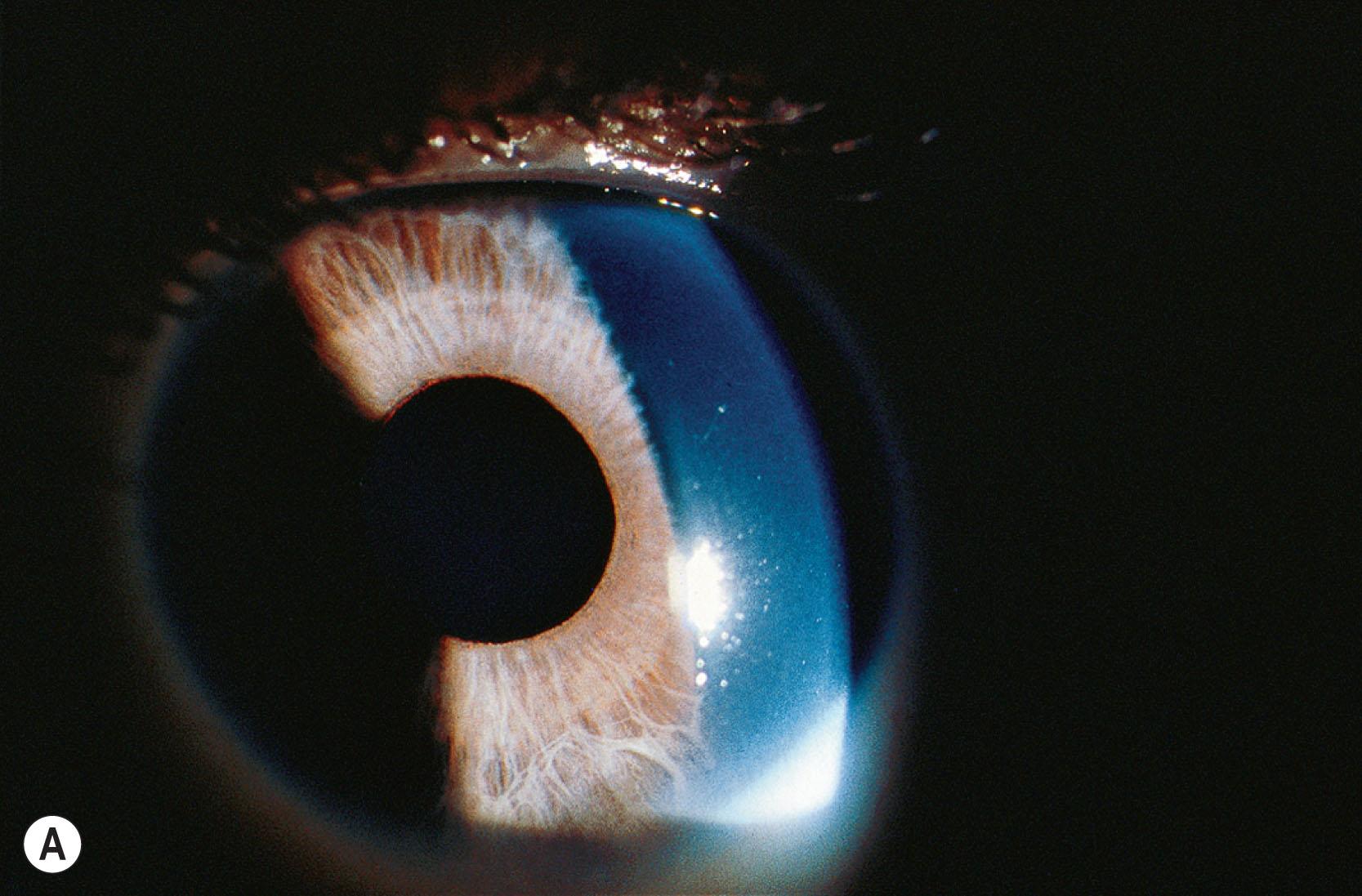
Congenital fixed dilated pupils (congenital mydriasis) is due to hypoplasia of the iris muscles, with absence of iris between the collarette and pupillary border resulting in a scalloped pupillary margin ( Fig. 31.9 ). Often there are wisps of persistent pupillary membrane. This condition has been shown to be part of a multisystem smooth muscle cell dysfunction syndrome with associated moyamoya-like cerebrovascular disease, patent ductus arteriosus, thoracic aortic aneurysm, and other dysfunction of smooth muscle cells throughout the body. As of now, every affected individual has been shown to have a p.R179H heterozygous mutation in the ACTA2 gene (see Chapter 37 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here