Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The focus of this chapter is limited to describing the embryology, anatomy, and physiology of the mesenteric circulation. The terms mesenteric circulation and splanchnic circulation are sometimes used interchangeably; however, they have distinct meanings. The mesenteric circulation refers specifically to the vasculature of the intestines, whereas the splanchnic circulation provides blood flow to the entire abdominal portion of the digestive system that includes the hepatobiliary system, spleen, and pancreas. The primitive gut comprises the foregut, midgut, and hindgut. The foregut is further divided into upper foregut (embryonic pharynx) and lower foregut, which includes the esophagus, stomach, and the descending portion of the duodenum together with the hepatobiliary derivatives. By convention, the boundaries of the specific segments of the gut are determined by the three unpaired abdominal aortic trunks. The celiac axis supplies the abdominal foregut and its accessory organs, which include the abdominal esophagus, stomach, and proximal duodenum, along with the hepatobiliary, pancreatic structures and spleen. The superior mesenteric artery (SMA) supplies the midgut, which begins just distal to the ampulla of Vater and extends to the proximal two-thirds of the transverse colon. The inferior mesenteric artery (IMA) supplies the hindgut, which includes the distal third of the transverse colon, the descending colon, and the rectum, and extends to the upper part of the anal canal. However, studies have shown that demarcation of these specific segments of the gut occurs well before the development of these vessels and depends on specific gene expression within the gut.
The mesenchymal angioblastic tissue covering the yolk sac within the connecting stalk and in the chorionic sac wall give rise to the rudimentary embryonic vessels. By necessity, the cardiovascular arcade and organs are the first system to function in the embryo. Vitelline arteries arising in the yolk sac wall give rise to the arterial supply of the gastrointestinal tract ( Fig. 86.1 ). They connect in the form of plexuses with the ventral surface of the aorta. By the end of week 4, they lose connection with the yolk sac and reduce in number to five cranial and three caudal to the diaphragm, to supply specific regions of the developing abdominal gut.
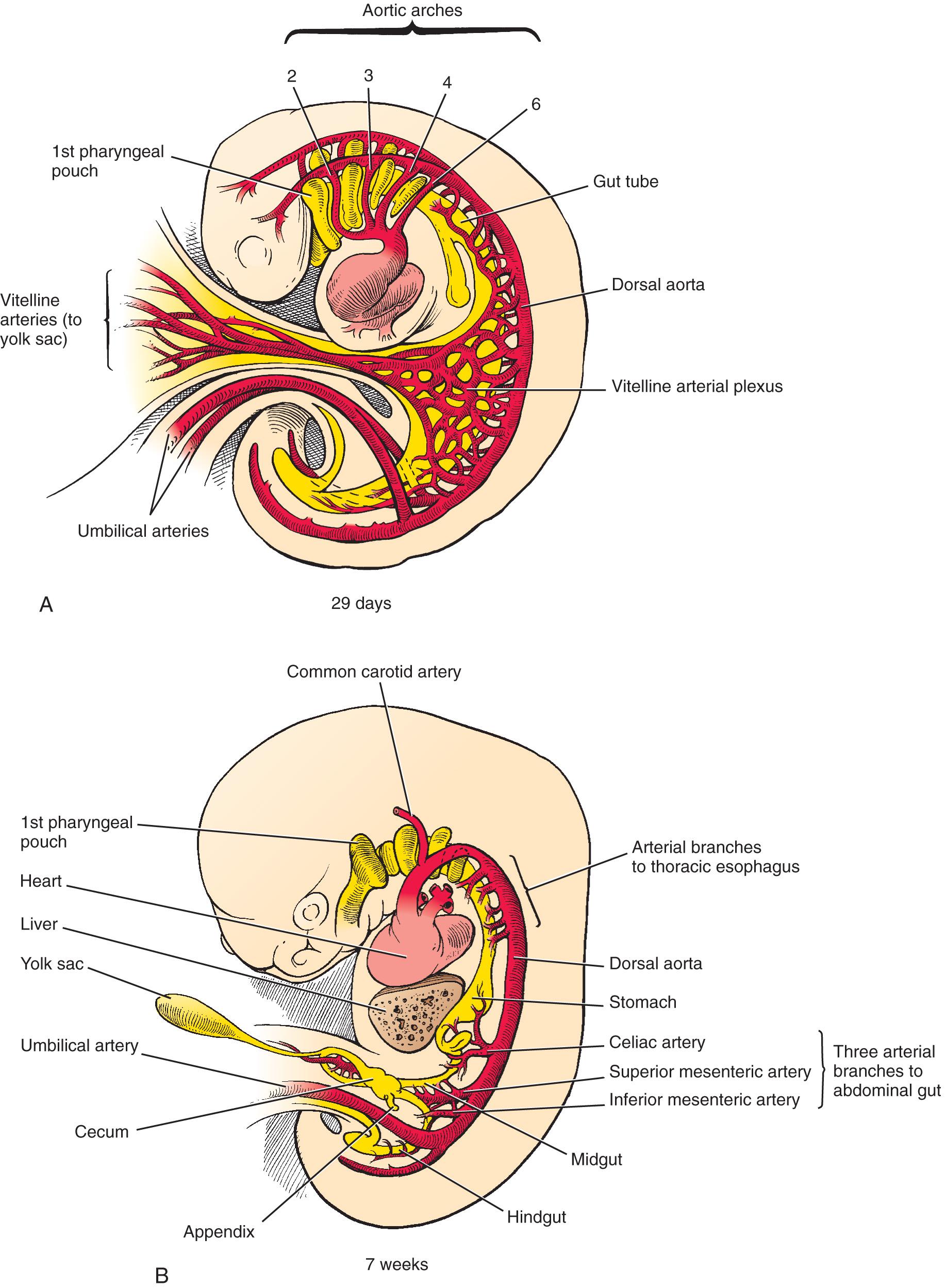
The celiac axis, which is the most superior of the three abdominal vitelline arteries, initially joins the aorta at the C7 level but with the developing gut descends to the T12 level. In addition to the foregut, it supplies branches to the embryologic outgrowths of the foregut, including the liver, gallbladder, pancreas, and spleen.
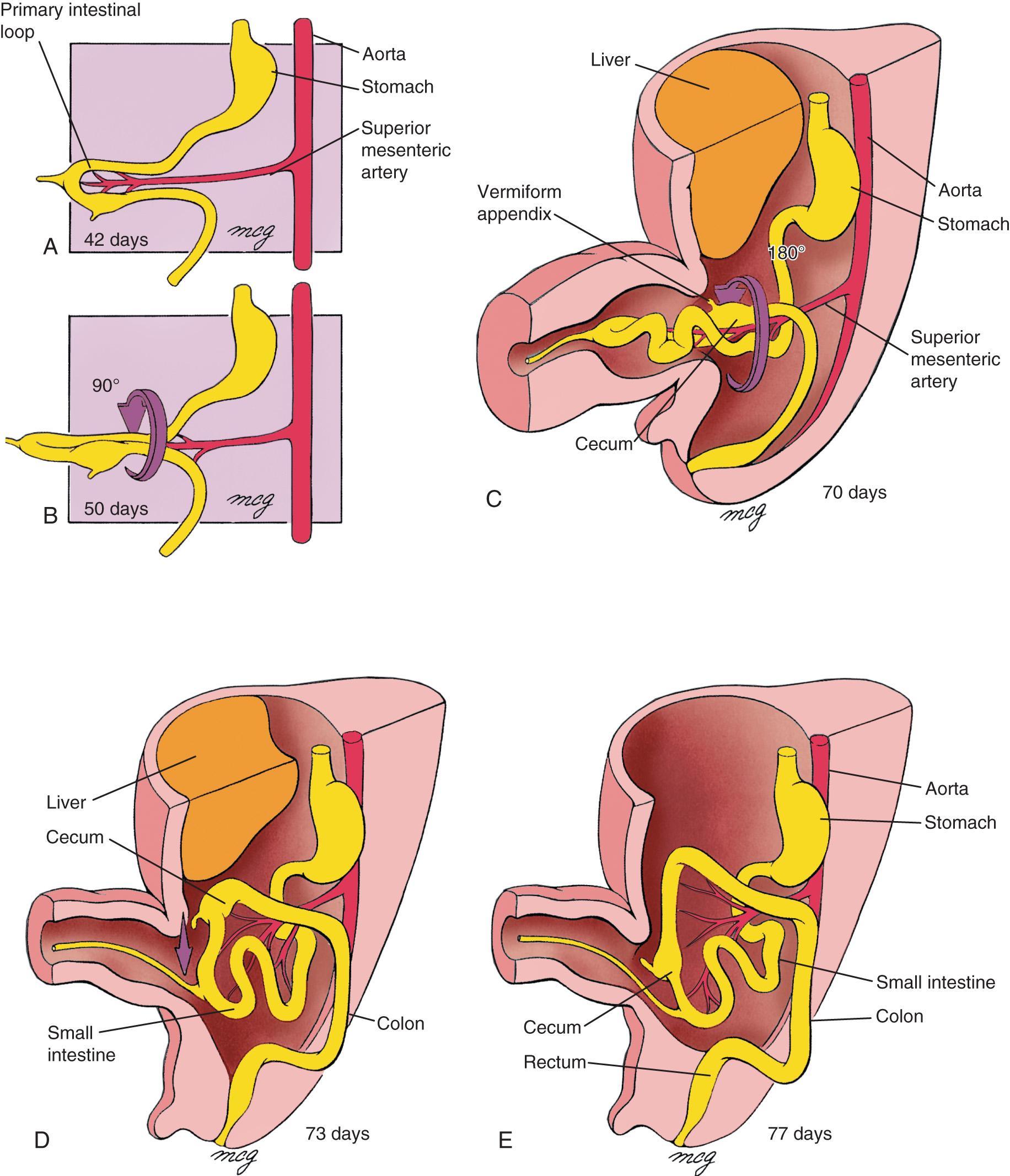
The midgut is supplied by the second abdominal vitelline artery, the SMA. This vessel initially joins the aorta at the T2 level but later migrates to the L1 level. At approximately week 5, the midgut, with its mesentery, elongates more rapidly than does the abdominal cavity, leading to formation of a primary intestinal loop with the SMA running down its long axis. By approximately week 10, the midgut completes a 270-degree counterclockwise rotation around the SMA, as shown in Fig. 86.2 . As a consequence, the proximal duodenal branches arise on the right side of the SMA, branches to the small bowel arise on the left side, and those to the colon arise from the right side of the SMA.
The blood supply to the hindgut is via the third and final abdominal vitelline artery, the IMA. The cloaca, which is an expansion of the primitive gut just superior to the cloacal membrane, is divided into an anterior portion (urogenital sinus) and a posterior portion (anorectal canal) by the urorectal septum. The distal one-third of the anorectal canal develops from an ectodermal invagination called the anal pit. Therefore, the upper two-thirds of the anorectal canal (endodermal origin) is supplied by the IMA and the lower one-third (ectodermal origin) by the systemic circulation.
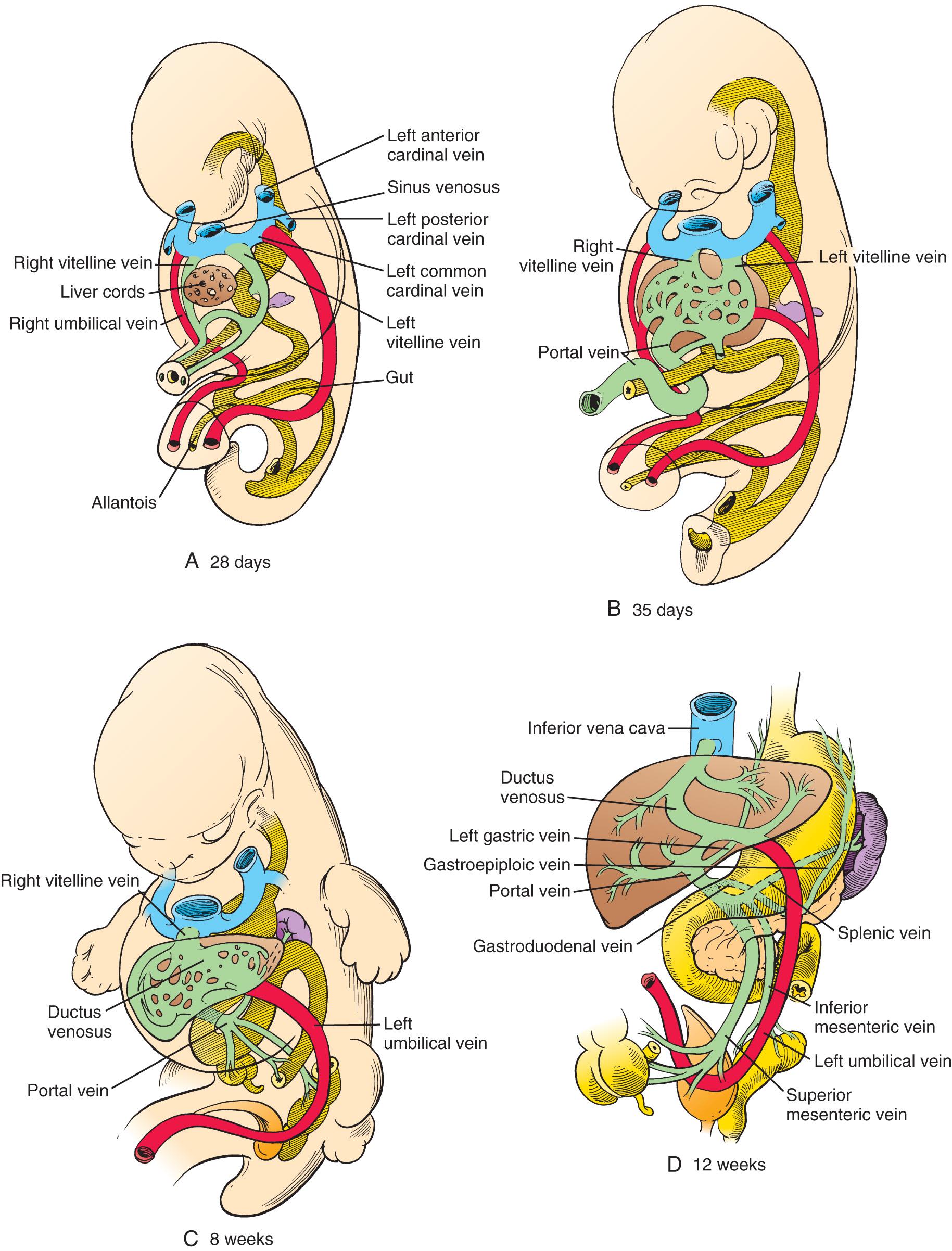
The vitelline veins, like the vitelline arteries, arise from the vascular plexuses of the yolk sac. The vitelline venous system initially starts as paired veins that drain into the sinus horns of the heart, as shown in Fig. 86.3A , and anastomose. The veins subsequently develop plexuses in the septum transversum, within which the liver cords grow to form the liver sinusoids, as shown in Fig. 86.3B . The left vitelline vein regresses and disappears by the third month, except for a few transverse anastomoses between the liver and the abdominal foregut (see Fig. 86.3C ). The blood from the vitelline system now drains to the heart by the right vitelline vein, which enlarges and becomes the hepatocardiac channel, the cranial portion of which becomes the proximal inferior vena cava (IVC). The right vitelline vein, caudal to the liver, also regresses, except for the portion just caudal to the liver together with a few proximal left to right vitelline anastomoses (see Fig. 86.3B ). This segment close to the liver becomes the portal vein (PV) and the superior mesenteric vein (SMV; see Fig. 86.3C and D ). The left to right vitelline anastomoses differentiate into the splenic vein (SV) and the inferior mesenteric vein (IMV). Many of the commonly encountered anomalies of the mesenteric circulation can be attributed to incomplete regression (or persistence) of the early vitelline vessels.
The arterial and venous circulation of the abdominal viscera form an intricate vascular network marked by a large number of collateral pathways offering ample protection against ischemic events. As noted, the three major aortic branches are responsible for the arterial supply to the intestines and include the celiac axis, SMA, and IMA. The mesenteric venous drainage mirrors the arterial system to a certain extent and includes the SMV and IMV.
The celiac axis, one of the largest branches of the aorta, originates at the level of the T12 or L1 vertebral body at a relatively acute angle, traveling for 1 to 2 cm before trifurcating into the left gastric, common hepatic, and splenic arteries. The left gastric artery supplies the distal portion of the esophagus and superior portion of the stomach, then follows the lesser curvature to anastomose with the right gastric artery, which emanates from the common hepatic artery (CHA).
The CHA gives off the right gastric artery and the gastroduodenal artery before becoming the proper hepatic artery. The proper hepatic artery branches into the right and left hepatic arteries. As mentioned earlier, the right gastric artery follows the lesser curvature of the stomach to anastomose with the left gastric artery to supply the lesser curve of the stomach. The gastroduodenal artery descends posterior to the first portion of the duodenum, anterior to the head of the pancreas, and to the left of the common bile duct. It gives off the posterior superior pancreaticoduodenal artery (also called the retroduodenal artery) that supplies the common bile duct, then divides into the right gastroepiploic artery and the anterior superior pancreaticoduodenal artery to supply the antrum of the stomach, the duodenum, and pancreas. The right gastroepiploic artery runs along the greater curvature of the stomach in the greater omentum and eventually communicates with the left gastroepiploic artery, a branch of the splenic artery. The anterior superior pancreaticoduodenal artery divides into duodenal and pancreatic branches.
The splenic artery is the largest branch of the celiac axis and follows a tortuous course along the superior aspect of the pancreas. During its initial course, the splenic artery is a retroperitoneal structure. It then enters the lienorenal ligament before reaching the spleen. The short gastric and left gastroepiploic arteries supplying the greater curvature of the stomach arise from the splenic artery before it enters the hilum. The left gastroepiploic artery continues to join with the right gastroepiploic artery along the inferior aspect of the greater curvature. In addition, the splenic artery gives rise to the dorsal pancreatic artery, which lies posterior to the confluence of the splenic and SMVs.
The classic celiac axis anatomy as described previously is seen in only 55% of patients. A common aortic trunk from which both the celiac axis and the SMA originate, the celiomesenteric artery, occurs in only 1% of patients. Although the potential for individual anatomic variations is countless, common patterns with important clinical relevance have been described. An anomalous right or left hepatic artery has been reported in almost 50% of patients. The right hepatic artery may arise from the SMA in 20% of these cases, the gastroduodenal artery in 2%, and directly from the celiac axis in 1% (also called a replaced right hepatic artery). In addition, the left hepatic artery has been reported to arise from the left gastric artery in 17%, the gastroduodenal artery in 1%, and the celiac trunk in 2% (also called a replaced left hepatic artery).
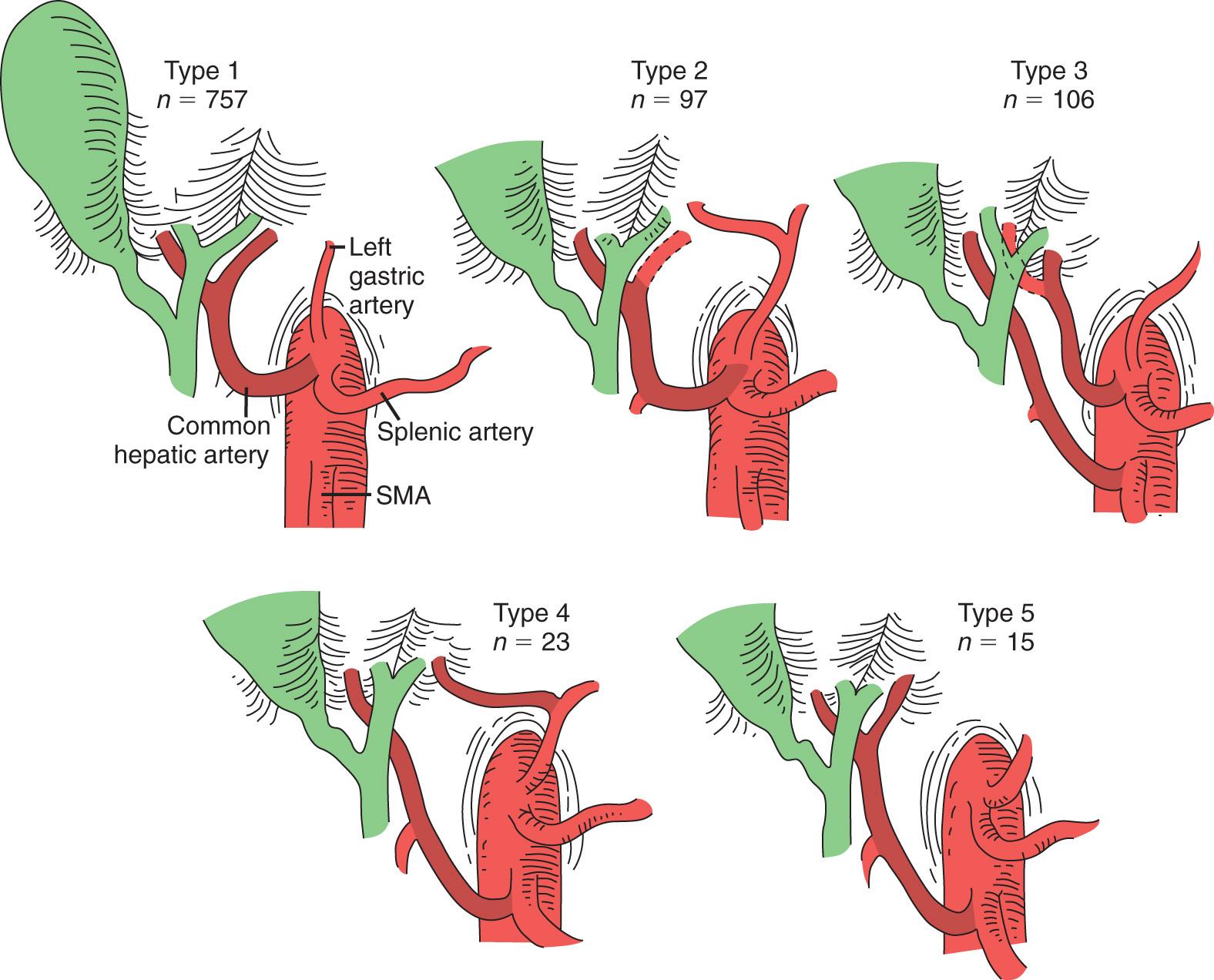
Hiatt et al., in the largest report to date on the surgical anatomy of the hepatic arteries, observed six arterial patterns as follows: type 1, normal anatomy; type 2, a replaced or accessory left hepatic artery arising from the left gastric artery; type 3, a replaced or accessory right hepatic artery originating from the SMA; type 4, both the right and left hepatic artery arising from the superior mesenteric and left gastric arteries, respectively; type 5, the entire CHA arising as a branch of the SMA; and type 6, the CHA originating directly from the aorta ( Fig. 86.4 ). The presence of a replaced right hepatic artery is particularly significant when pancreaticoduodenectomy or liver transplantation is being considered. In addition to anomalous origins, hepatic arteries have been shown to follow anomalous courses. For example, a replaced or accessory right hepatic artery originating from the SMA may follow a retropancreatic path or remain anterior to the pancreatic neck.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here