Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The conventional approach to teaching about inflammatory disorders of the gastrointestinal (GI) tract is to describe the clinical and histological features of individual disease entities. Unfortunately, rendering a specific diagnosis is not always possible when evaluating mucosal biopsies of the GI tract. Therefore it is incumbent on the pathologist to evaluate all of the clinical, endoscopic, and histological features in each case to help establish a specific diagnosis. One of the biggest challenges in mucosal biopsy pathology is recognizing a specific morphological pattern of injury. Use of standardized terminology is critical because each discrete morphological pattern of injury has well-defined etiological associations and therapeutic implications. For instance, a diagnosis of Barrett’s esophagus (BE) or inflammatory bowel disease (IBD) leads to lifelong endoscopic surveillance of the patient and imposes a significant burden on patients and on the health care system. Use of nonstandardized terms such as nonspecific esophagitis, nonspecific colitis, or colonic mucosa with acute and chronic inflammation is misleading and therefore not helpful for patient management.
The purpose of this chapter is to outline a simple algorithmic approach to the diagnosis of common inflammatory disorders of the GI tract in mucosal biopsies. The process begins by dividing biopsies into one of several broad categories (e.g., normal, inflammatory, neoplastic) and is followed by a systematic evaluation of morphological features to determine a specific morphological pattern of injury. This approach often implies a fairly standard etiological differential diagnosis.
Esophageal biopsies can be broadly categorized as normal, inflammatory, or neoplastic ( Fig. 13.1 ). Normal esophageal mucosa is composed of nonkeratinizing squamous epithelium with a basal zone that is typically one to three cell layers thick, papillae that are confined to the lower half of the mucosa, and lamina propria that is composed of loose connective tissue without inflammatory cells ( Fig. 13.2 ). The presence of surface keratinization (hyperkeratosis), parakeratosis, spongiosis, or an abnormal number of inflammatory cells in the epithelium is a sign of mucosal injury and should lead to further evaluation of the biopsy specimen for a specific morphological pattern of injury.

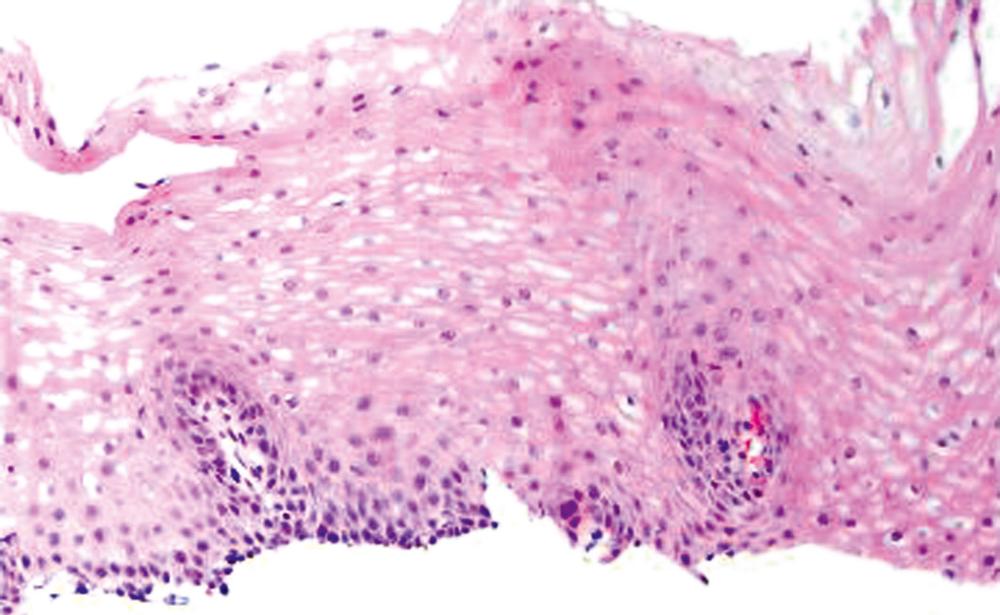
If a biopsy is considered “inflammatory,” the next step is to evaluate the dominant inflammatory cell type. Inflammation in the esophagus may be dominated by eosinophils, neutrophils, lymphocytes, or a combination of these cells, and each of these categories has well-defined etiological associations (see Fig. 13.1 ). Disorders with prominent eosinophilic and/or neutrophilic patterns of inflammation are classified as active esophagitis. As illustrated in Fig. 13.1 , gastroesophageal reflux disease (GERD) and infections may be associated with more than one pattern of inflammation; for this reason, they are always in the differential diagnosis of inflamed (or even near-normal) esophageal biopsies. However, endoscopic or histological evidence of esophagitis is present in only approximately 40% of patients with well-established GERD.
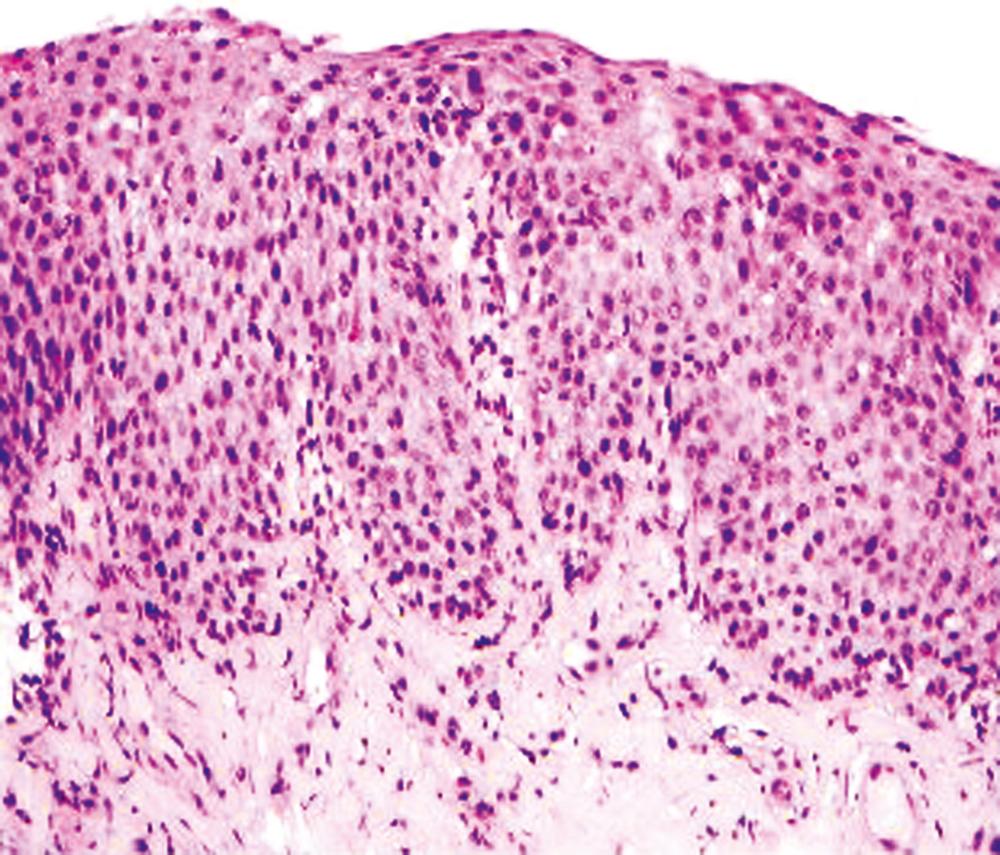
Intraepithelial eosinophilic infiltration is one of the most common patterns of inflammatory injury in esophageal biopsies. GERD and eosinophilic esophagitis (EoE) are the diseases most commonly associated with this pattern of injury. The presence of eosinophils in esophageal mucosa is not necessarily diagnostic of primary EoE (a well-defined clinicopathological disorder that is discussed in detail in Chapter 14 ). No single clinical, endoscopic, histological, or treatment parameter allows for absolute distinction of GERD from EoE. However, there are a variety of histological features and combinations of features that can certainly help point a pathologist toward a diagnosis of EoE. EoE is a clinicopathological diagnosis and requires correlation with patient symptoms; endoscopic features; the density, pattern, and gradient of eosinophilic infiltration in mucosal biopsies; and treatment response. Some of the salient features that help distinguish GERD from EoE are summarized in Table 13.1 and illustrated in Fig. 13.3 . Eosinophilic infiltration in esophageal mucosa may also be present in disorders other than GERD and EoE. A list of these disorders and the clinical or morphological features that are helpful in making these diagnoses is presented in Table 13.2 .
| Features | Reflux Esophagitis | Eosinophilic Esophagitis |
|---|---|---|
| Clinical History | ||
| Allergy/asthma | Low incidence | High incidence |
| Food impaction | Uncommon | Common |
| Symptoms | ||
| GERD symptoms | Typical | May be present |
| Dysphagia | May be present | Common |
| Endoscopy | ||
| Rings | Uncommon | Typical |
| Linear furrows | Uncommon | Typical |
| Erosions/ulcers | Typical | Uncommon |
| pH/impedance | Abnormal | Normal |
| Pathology | ||
| Inflammation gradient | Distal >> Mid/upper | Mid/upper >> Distal |
| Eosinophilic Infiltrate | ||
|
|
|
|
|
|
|
|
|
|
|
|
| BCH/spongiosis | Mild | Severe |
| Therapy | ||
| Response to PPIs | Yes | No |
| Response to steroids | No | Yes |
| Diagnosis | Diagnostic Features |
|---|---|
| Infections | Viral inclusions/parasites on biopsy |
| Eosinophilic gastroenteritis | Gastric/SI involvement |
| Achalasia | Motility studies |
| Hypereosinophilic syndrome | Peripheral blood hypereosinophilia |
| Crohn’s disease | Granulomas, colon/SI involvement |
| GVHD | Apoptosis, history of BMT |
| Drug hypersensitivity | Clinical history |
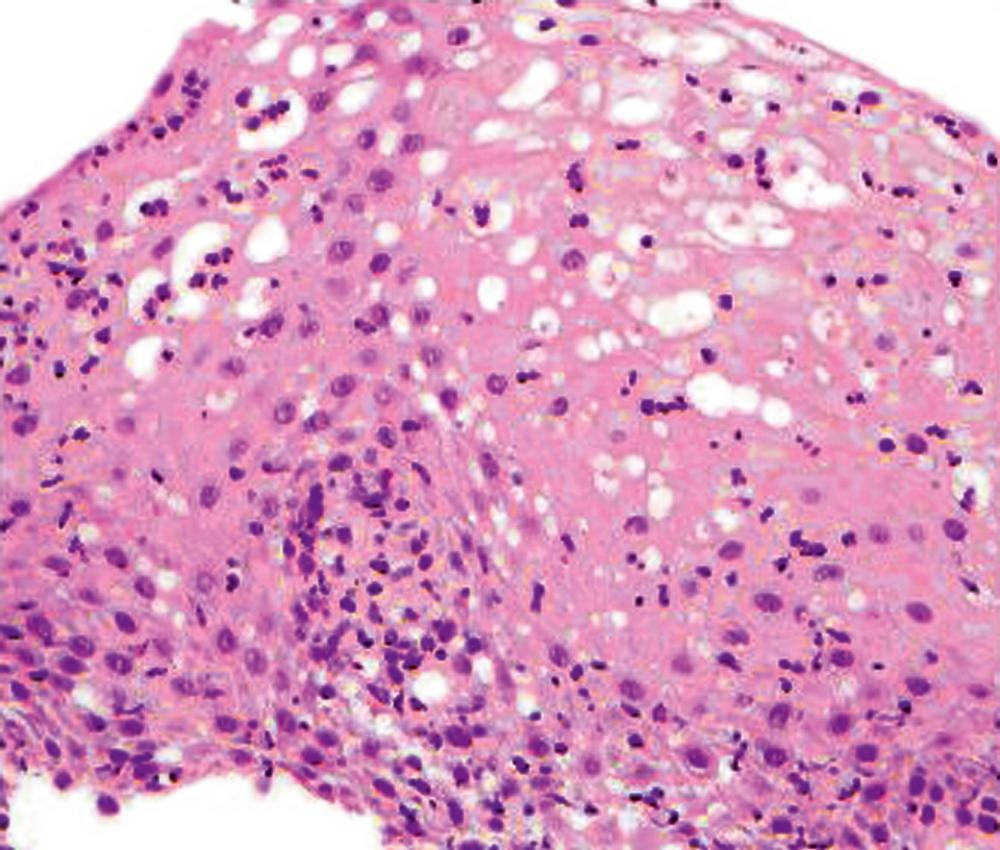
A neutrophil-predominant inflammatory pattern of injury may be associated with infections, GERD, pill esophagitis, or corrosive mucosal injury ( Fig. 13.4 ), among others. The presence of surface hyperkeratosis in association with a neutrophilic infiltrate should always prompt a search for fungi, such as Candida . Yeast and pseudohyphae may, at times, be confined to surface keratinous debris and may be associated with either minimal or no inflammation in the epithelium and lamina propria. Cytomegalovirus (CMV) and herpes esophagitis may also be associated with a neutrophil-predominant pattern of inflammation, and these infections may be diagnosed when typical viral inclusions are demonstrated in the biopsy. A diagnosis of pill esophagitis should be considered when the inflammation, or ulcer, is present in the upper and middle levels of the esophagus. The biopsy may show refractile, crystalline particles within necroinflammatory debris that represent pill fragments. This diagnosis requires correlation with the patient’s medication history. Similarly, the presence of active esophagitis containing scattered atypical cells with large, hyperchromatic epithelial and/or stromal nuclei may be indicative of radiation esophagitis. Corrosive mucosal injury often leads to necrosis of the upper half of the squamous epithelium, and a band of neutrophils may be present between the superficial necrotic and deeper viable mucosa.
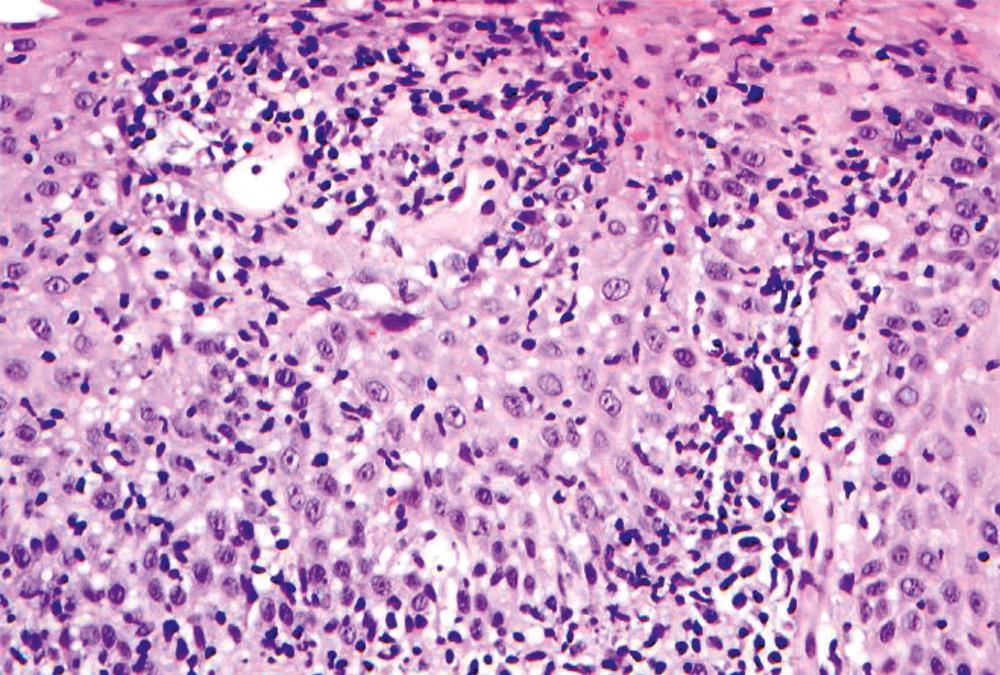
Intraepithelial lymphocytosis of variable degrees is not an uncommon finding in esophageal biopsies. When it is associated with basal cell hyperplasia and scattered eosinophils and/or neutrophils, it is most often a result of GERD. The presence of hyperkeratosis and scattered neutrophils should always raise suspicion for Candida esophagitis. The presence of a pure lymphocytic infiltrate (usually ≥ 20 lymphocytes per high-power field) associated with mucosal injury in the form of edema and basal cell hyperplasia, particularly in the peripapillary epithelium, is consistent with lymphocytic esophagitis ( Fig. 13.5 ). Lymphocytic esophagitis is a poorly defined entity with a myriad of etiological associations; the most notable are motility disorders and systemic immune-mediated diseases such as Crohn’s disease and connective tissue disorders. Crohn’s disease manifests in the esophagus with a lymphocytic esophagitis pattern of injury more often in pediatric patients than in adults. Other disorders that cause intraepithelial lymphocytosis in the esophagus are listed in Box 13.1 . When the lymphocytic infiltrate is distributed predominantly at the epithelial lamina propria interface and scattered necrotic keratinocytes along with vacuolization of basal cells are present, the possibility of a dermatological disorder such as lichen planus should be considered.
Crohn’s disease (particularly in pediatric age group)
GERD
Infections
Motility disorders
Allergy/asthma
Autoimmune diseases
Immunodeficiency (HIV, CVID)
Celiac disease
Esophageal involvement in dermatological disorders
Granulomatous esophagitis ( Fig. 13.6 ) is associated with Crohn’s disease or sarcoidosis in almost all cases. Rare causes of granulomatous esophagitis include mycobacterial or fungal infection, vasculitis, and drug-induced mucosal injury.
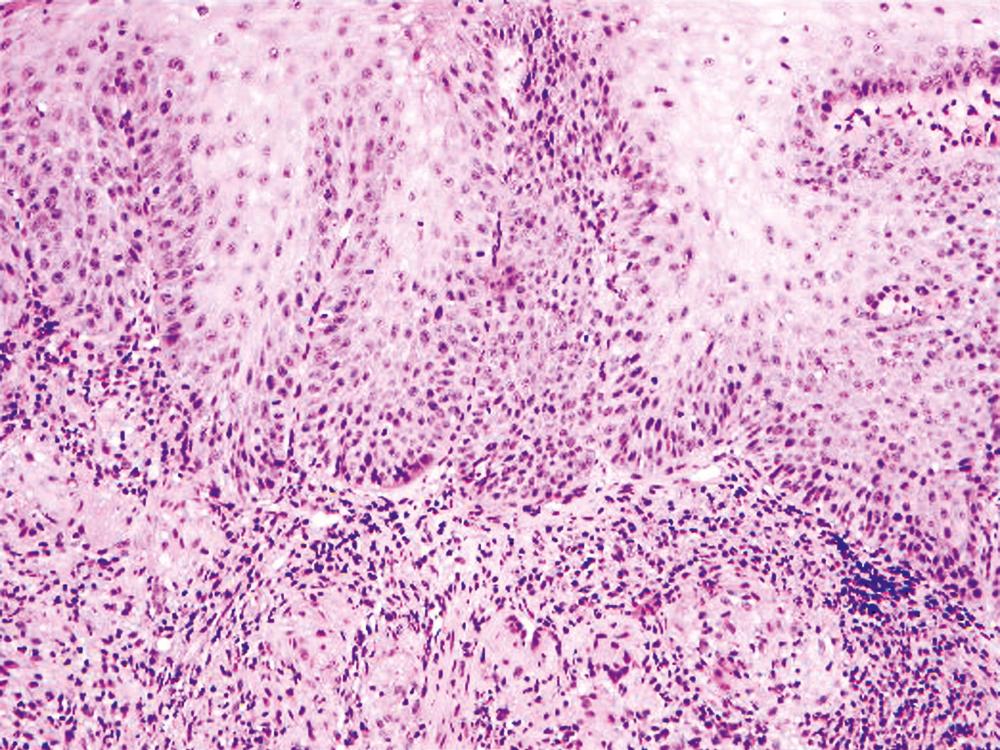
A list of pauciinflammatory disorders is shown in Table 13.3 . Graft-versus-host disease (GVHD) is the disorder that most frequently manifests with this pattern of mucosal injury ( Fig. 13.7 ). Apoptosis and necrosis in the absence of inflammation, characteristic of GVHD, overlaps with mycophenolate-induced mucosal injury. Infections in immunocompromised patients also may lack an inflammatory response. Amyloidosis is another disorder that may be easily missed because the mucosa is typically completely normal, and the diagnostic changes are subtle, usually confined to the blood vessels in the lamina propria and superficial submucosa.
| Diagnosis | Features |
|---|---|
| CMV, HSV | Viral inclusions |
| GVHD, mycophenolate | Increased apoptosis |
| Corrosive injury | Superficial necrosis |
| Taxol, colchicine toxicity | Mitotic arrest |
| Skin disorders (pemphigus) | Vesicles, bullae |
| Scleroderma | Submucosal fibrosis |
| Amyloidosis | Perivascular deposits |
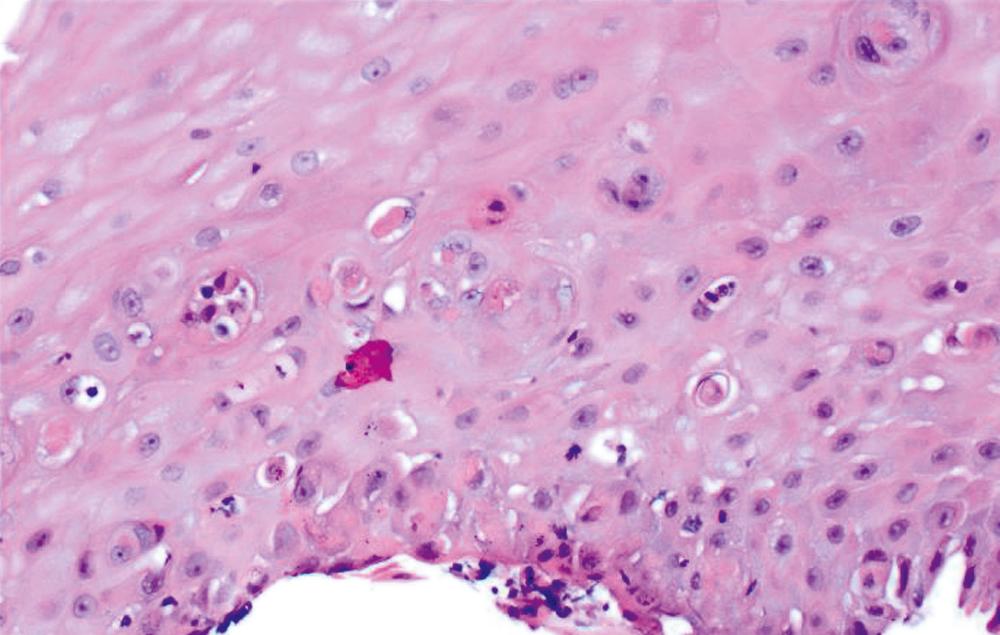
In some instances, one may see ulcerated granulation tissue, without viable mucosa, in a biopsy of the esophagus. A specific diagnosis cannot be rendered in most of these cases. However, biopsies with this pattern of injury should always be evaluated for viral inclusions, pill fragments, and neoplastic cells before they are dismissed as nonspecific .
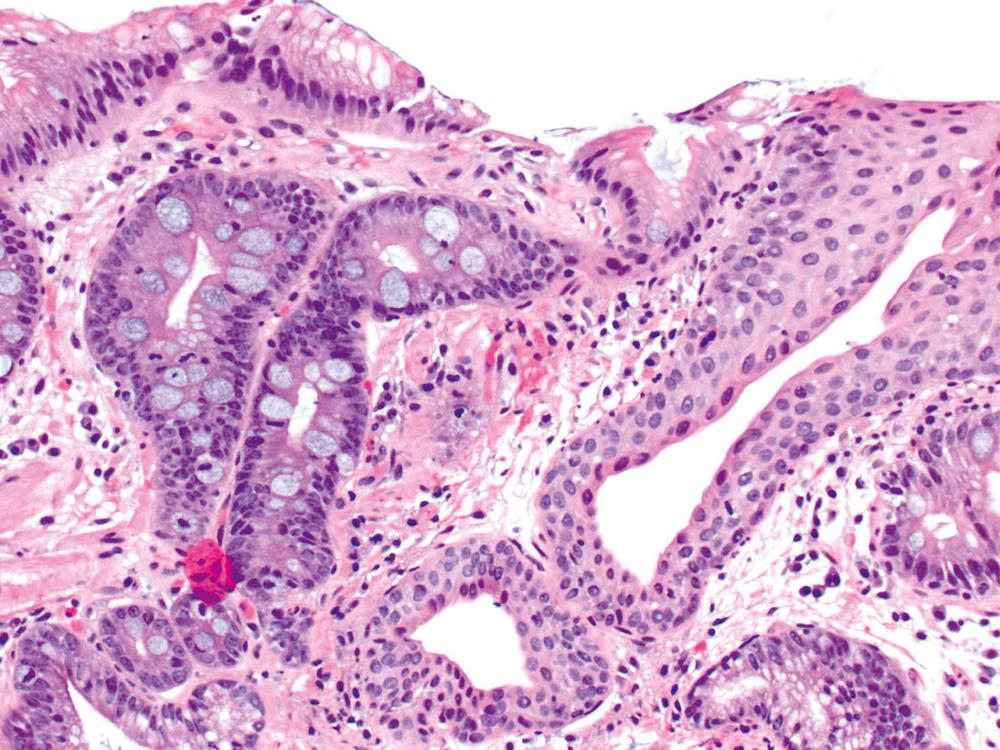
The anatomic gastroesophageal junction (GEJ) is endoscopically defined as the proximal limit of the gastric rugal folds. The squamocolumnar junction (SCJ) represents the point of transition from pearly-gray squamous mucosa of the esophagus to salmon-red columnar mucosa of the proximal stomach (or distal esophagus in cases of BE). In healthy individuals, the SCJ is located at the GEJ, whereas in patients with BE, the SCJ is located proximal to the anatomic GEJ. When an endoscopic biopsy is submitted with a question of BE, it is imperative for the pathologist to know the precise location of the biopsy, even before histological evaluation is performed. Biopsies labeled as GEJ, SCJ, or Z-line cannot be definitely labeled as BE, even when goblet cells are identified in the biopsy, unless there are morphological features in the biopsy specimen that help determine whether the columnar epithelium was taken from the anatomic esophagus. These features include multilayered epithelium, mucosal or submucosal glands or ducts ( Fig. 13.8 ), subsquamous (“buried”) glands, or hybrid epithelium. Additionally, based on current American College of Gastroenterology guidelines, a definitive diagnosis of BE can be rendered only if the biopsies have been obtained at a distance of 1 cm above the GEJ (from and confined to the distal esophagus) and in which goblet cells are identified in the columnar epithelium.
Gastric mucosal biopsies may be broadly categorized on morphological evaluation as either normal, inflammatory, or neoplastic. Another category consists of lamina propria infiltrates or pigment depositions in the absence of significant lymphoplasmacytic, neutrophilic, or eosinophilic inflammation ( Fig. 13.9 ). Normal gastric corpus mucosa is composed of closely packed oxyntic glands, whereas the antrum is composed of pure mucus glands arranged in a vague lobular configuration ( Fig. 13.10 ). In the proximal corpus, basally located compact mucosal (mucinous) glands, similar in appearance to those at the SCJ and in the gastric cardia of patients with BE, have recently been described ( Fig. 13.11 ). These glands have been proposed to represent a potential source of stem cells, but this needs further research to define properly. In older children and young adults, both gastric compartments (antrum and corpus) show minimal intervening stroma and either few or no plasma cells in the lamina propria. With advancing patient age, up to 5 to 10 mononuclear cells per high-power field are considered normal, particularly in the antrum. A few lymphocytes or plasma cells in the lamina propria in the absence of epithelial injury or regeneration should not be considered indicative of active or chronic disease. Primary lymphoid follicles without active germinal centers are normally present at the base of the gastric corpus mucosa as well, and these should not be considered a manifestation of chronic gastritis. However, the presence of multiple clusters of plasma cells (at least 3 to 4 clusters, composed of 6 to 8 plasma cells each), lymphoid aggregates in the antrum, lymphoid aggregates with active germinal centers, and a bandlike lymphoplasmacytic infiltrate beneath the surface epithelium are all considered features of chronic gastritis.


Determination of the predominant inflammatory cell type in the lamina propria is the first step in diagnosing gastritis (see Fig. 13.9 ). A lymphoplasmacytic infiltrate is by far the most common type of inflammatory reaction seen in gastric biopsies in routine practice. This pattern (also broadly referred to as chronic gastritis ) includes several well-defined clinicopathological entities. This pattern of inflammation should be evaluated for pattern of injury (focal vs. diffuse), region of the stomach predominantly involved (antrum vs. corpus, or both), location in the mucosa (superficial vs. deep), presence or absence of active inflammation, presence or absence of microorganisms, atrophy of oxyntic glands, endocrine cell hyperplasia, intraepithelial lymphocytes (IELs), and quality of the subepithelial collagen layer, among others ( Table 13.4 ).
| Feature | Helicobacter | Other Infectious | Autoimmune | Lymphocytic | Collagenous | Eosinophilic |
|---|---|---|---|---|---|---|
| Lymphoplasmacytic infiltrate | Prominent | Present | Present | Present | Present | May be present |
| Antral predominance | Yes | No | No | May be seen | May be seen | Absent |
| Superficial bandlike infiltrate | Prominent | May be present | Absent | May be present | May be present | Absent |
| Microorganisms or viral inclusions | Present | Present | Absent | Absent | Absent | Absent |
| Oxyntic gland atrophy | May be present | May be present | Prominent | Absent | Absent | Absent |
| Endocrine cell hyperplasia | Absent | Absent | Prominent | Absent | Absent | Absent |
| Increased IELs | May be present | Absent | Absent | Prominent | May be present | Absent |
| Thick subepithelial collagen layer | Absent | Absent | Absent | Absent | Present | Absent |
| Eosinophil clusters, aggregates, and epithelial damage | Absent | May be present | Absent | Absent | May be present | Prominent |
Focal active gastritis may be a gastric manifestation of IBD, particularly in pediatric patients ( Fig. 13.12 ). The etiological associations of focal active gastritis in adults are diverse, and the association with IBD is weak. Infections such as CMV and adenovirus, Helicobacter heilmannii, and drug reactions, especially immune checkpoint inhibitor–induced gastritis, can also cause focal active gastritis.

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here