Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Immune defense against microbes is mediated by sequential and coordinated responses, termed innate and adaptive immunity . Innate immunity represents early response mechanisms that facilitate rapid reaction to invading pathogens in the first few hours or days after infection (see Chapter 4 ). Adaptive immunity, in contrast, provides stronger and more specialized responses, as well as memory of the response, leading to more rapid and effective control upon future exposures to the same pathogen. In health, immune defenses are not directed at self-antigens or tissues, whereas a breakdown in this “tolerance” is thought to underlie the mechanisms that lead to autoimmunity. In many inflammatory rheumatologic conditions, both innate and adaptive mechanisms have been shown to play a role in pathogenesis.
Adaptive immunity can be divided mechanistically into cellular immunity and humoral immunity. Cellular immunity is mediated by T lymphocytes bearing antigen-specific T-cell receptors (TCRs). When stimulated by antigen, T cells are able to produce cytokines and kill microbe-infected or malignant cells. Humoral immunity is mediated by antigen-specific antibodies produced by B lymphocytes bearing antigen-specific B-cell receptors (BCRs). TCRs recognize peptide fragments bound to major histocompatibility complex (MHC) molecules on antigen-presenting cells (APCs), whereas BCRs recognize intact proteins, carbohydrates, or other macromolecules.
T cells and B cells play central roles in mediating adaptive inflammatory responses, both in health and in disease. The fundamental properties of the adaptive immune response include the following:
Specificity and diversity. During early development, a large number of naïve T- and B-cell clones with unique antigen receptors are produced and circulate throughout the body. TCR and BCR recognition of antigen results in the activation and subsequent proliferation and differentiation of T and B cells, producing clonal populations reactive with the stimulating antigen. Naïve T cells develop into mature helper, effector (including regulatory/immunosuppressive), and memory populations directed to the same antigen initially recognized by the naïve T cell. One role of helper T cells is to interact with B cells that have encountered the same antigen and induce their maturation into antibody-producing cells. It is estimated that the human immune system can discriminate up to 10 9 distinct antigenic determinants. This diversity of antigen reactivity is essential to defend the individual against the many potential pathogens in the environment.
Memory. A central feature of the adaptive immune system is an enhanced response to antigens that have been encountered previously. The first or primary response results in clonal expansion of T cells and B cells specific for that antigen; some of these activated cells remain as long-lived memory cells after the pathogen is cleared. On a second or subsequent exposure to the same antigen, these memory cells support a secondary immune response that is more rapid, greater in magnitude, and often qualitatively different from the primary immune response. Memory enables the immune system to respond more quickly and effectively and likely evolved to combat recurrent infections.
Self-tolerance. A fundamental property of the adaptive immune system is the ability to recognize and respond to an enormous variety of foreign (nonself) antigens while maintaining tolerance, or nonreactivity, to a similarly large variety of self-antigens. Importantly, the processes that generate antigen-specific receptors inevitably produce self-reactive TCR and BCR, such that self-tolerance cannot be genetically hardwired but must be acquired during lymphocyte development. Immune tolerance is created and maintained by several mechanisms, including elimination of T cells or B cells with antigen receptors that recognize self during development in the thymus or bone marrow, respectively ( central tolerance ), and/or via inhibition of mature lymphocyte functions. Mechanisms of the latter, together known as peripheral tolerance , include induction of anergy, deletion of autoreactive cells, and/or development of antigen-specific regulatory T and B cells. Mistakes in regulation of self-tolerance are thought to underlie many autoimmune disorders.
T cells are key mediators of the adaptive immune system, contributing to the control of infection, the promotion of inflammation via production of cytokines, and the capacity to kill infected or abnormal cells. Progress in understanding how T cells develop and are regulated has contributed significantly to insights into the functions of T cells in both normal and aberrant immune responses.
T-cell progenitors develop from bone marrow stem cells, which migrate to the thymus, where they undergo a process of maturation, T-cell receptor gene rearrangement and selection, giving rise to a large population of naïve T cells. The molecular mechanisms that regulate T-cell development and the somatic gene rearrangement mechanisms that result in production of antigen-specific TCR are well described in immunology textbooks. Naïve T cells circulate between blood and secondary lymphoid tissues (bone marrow, spleen, and peripheral lymph nodes). Upon activation through their TCRs, T cells proliferate and differentiate into distinct phenotypic and functional subpopulations ( Table 5.1 ), including effector cells that mount or coordinate attacks on the pathogen that produced the stimulating antigen, memory cells, and regulatory T cells.
| Subset | Functions | Antigen Receptor and Antigen Specificity | Phenotype |
|---|---|---|---|
| CD4 + helper T lymphocytes | B-cell activation (humoral immunity) Macrophage activation (cell-mediated immunity) Stimulation of inflammation |
αβ TCR heterodimers Peptide–class II MHC complexes |
CD3 + , CD4 + , CD8 − |
| CD8 + cytotoxic T lymphocytes | Killing of cells infected with intracellular microbes, tumor cells | αβ TCR heterodimers Peptide–class I MHC complexes |
CD3 + , CD4 − , CD8 + |
| Regulatory T cells | Suppress function of other T cells (regulation of immune responses, maintenance of self-tolerance) | αβ TCR heterodimers Specific for self and some foreign antigens (peptide-class II MHC complexes) |
CD3 + , CD4 + , CD25 + , FOXP3 + |
| Natural killer T (NKT) cells | Suppress or activate innate and adaptive immune responses | αβ TCR heterodimers Limited specificity for glycolipid-CD1 complexes |
CD56 + , CD16 + , CD3 + |
| γδ T lymphocytes | Helper and cytotoxic functions (innate immunity) | γδ TCR heterodimers Limited specificities for peptide and nonpeptide antigens |
CD3 + , CD4 and CD8 variable |
| Mucosa-associated invariant T (MAIT) cells | Helper and cytotoxic functions in the gut | αβ TCR heterodimers Limited specificity for bacterial metabolites |
CD3 + , CD8 + (majority) |
Effector T cells include cluster of differentiation (CD)4-expressing helper T cells (Th) and CD8-expressing cytotoxic T cells (Tc). Th cells express surface molecules and cytokines that activate and promote the function of B cells, macrophages, and dendritic cells (DCs) and recruit neutrophils, particularly at mucosal barriers. Tc cells express proteins in cytoplasmic granules that, when released, kill target cells (e.g., virus-infected or tumor cells). Both Th and Tc cells express variable cell surface proteins, receptors, and cytokines depending on their state of activation, as well as adhesion molecules that direct migration to specific tissues (e.g., lymph nodes, skin, gut).
Memory T cells are generated concurrent with effector T cells after antigen activation, and they survive in a functionally quiescent state after antigen is eliminated. Some memory T cells remain in the recirculating lymphocyte pool and lymphoid organs. Others express adhesion molecules that promote their migration to the tissue(s) where antigen was previously encountered, and these can remain in situ for a very long time (potentially the lifetime of the host). Memory T cells accumulate with age in the skin, gut, and mucosal tissues, forming a library of rapid response cells specific for previously encountered antigens.
Regulatory T cells (Treg) are key mediators of peripheral tolerance and dampen or restrict specific adaptive immune responses. Treg cells are characterized by expression of the transcription factor FOXP3 and can either be directly derived from the thymus (natural Treg cells) or induced in the periphery (adaptive Treg cells). , Treg cells control immune reactions to self and to some foreign antigens (e.g., proteins derived from food). In humans, mutations in FOXP3 lead to the IPEX syndrome (immunodysregulation polyendocrinopathy enteropathy X-linked), a fatal multisystem autoimmune disease with onset early in life, underscoring that Treg cells are crucial for maintaining immune tolerance. , On the other hand, in most human autoimmune diseases there seems to be no quantitative deficiency of Treg cells. Instead, an increased number of Treg cells is often found at sites of inflammation, such as the synovial fluid in patients with juvenile idiopathic arthritis (JIA). Whether the Treg cells at these sites are deficient in function remains unclear. A subset of Treg cells termed Tr1 cells are characterized by interleukin (IL)-10 production and the expression of particular surface markers (e.g., CD49b and LAG-3) and are thought to be crucial for maintaining peripheral tolerance. , To date, little is known about the role of Tr1 in JIA or other pediatric autoimmune diseases.
T-cell proliferation and differentiation require antigen recognition, a costimulatory signal, and growth factor cytokines. TCR recognition of peptide/MHC complexes expressed on APC is required for T-cell activation (signal 1). Th cells are activated by TCR recognition of peptide/MHC class II complexes, whereas Tc cells are activated by TCR recognition of peptide MHC class I complexes (see section titled Antigen Presentation). The lineage, developmental stage, and activation history of the T cell, as well as the nature and activation state of the APC presenting the peptide/MHC complex, are critical to the outcome. At the T-cell/APC interface, peptide/MHC, TCR, and other ligand/receptor pairs assemble in a complex, ordered arrangement called the immunological synapse ( Fig. 5.1 ). Within this interface, APCs deliver costimulatory signals via cell surface molecules (signal 2), as well as via soluble factors (signal 3). Naïve T cells that only receive signal 1 without other signals are anergic, contributing to self-tolerance.
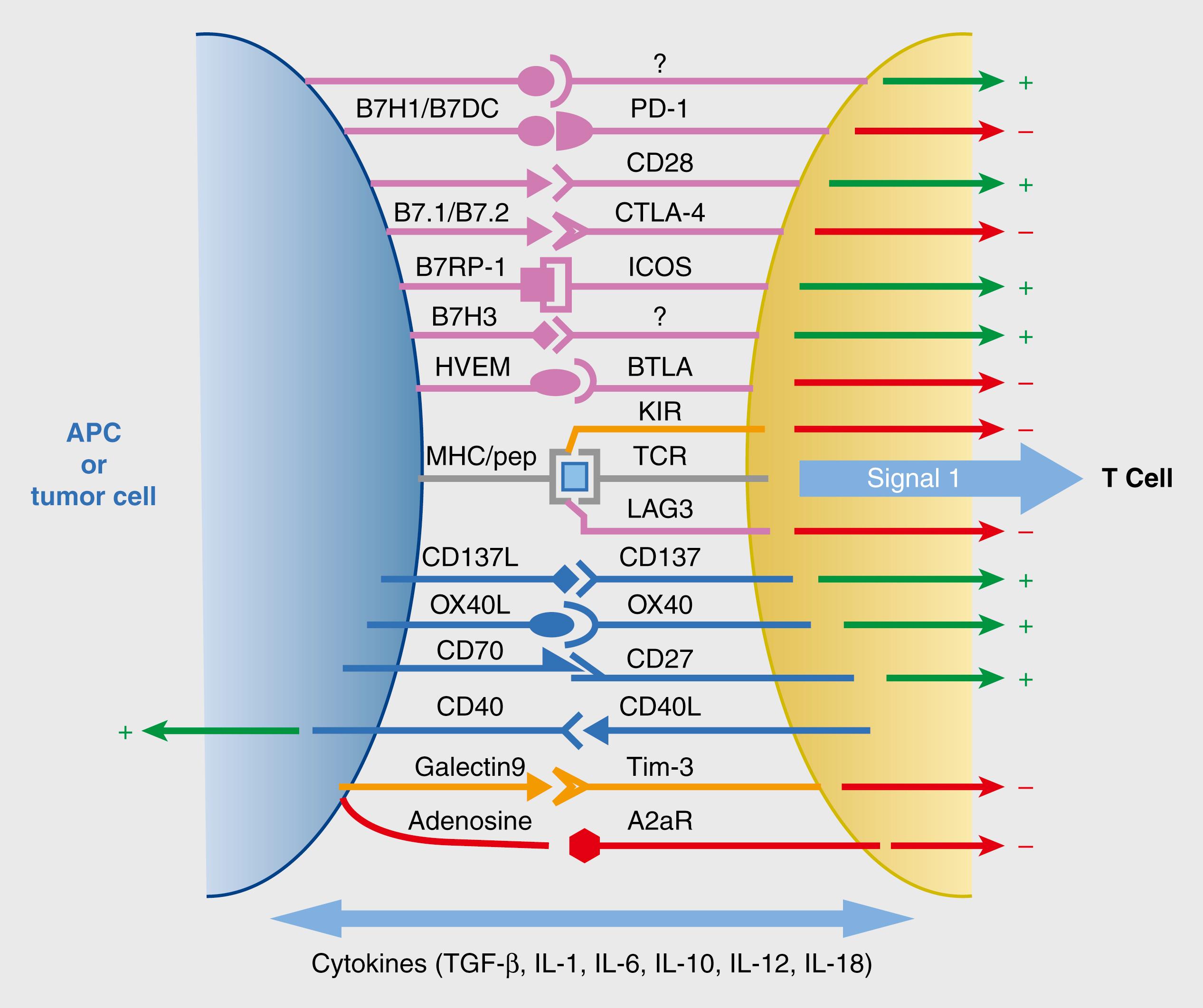
Naïve T-lymphocytes are activated primarily in peripheral (secondary) lymphoid tissues (lymph nodes and spleen), where these circulating cells encounter antigens presented by DCs. Naïve T cells are highly motile within lymphoid organs, interacting with many DCs and stopping when they recognize antigen to allow the immunological synapse to form. Costimulatory signals are expressed at high levels by DCs. The best characterized costimulatory pathway involves the T-cell surface receptor CD28, which binds the costimulatory molecules B7-1 (CD80) and B7-2 (CD86) expressed on the DC surface. Microbial products and cytokines produced by innate immune cells increase the expression of CD80 and CD86 to facilitate T-cell activation during infection. Effector and memory T cells do not need as much costimulation for activation and thus can respond to antigens presented by APCs other than DCs.
The earliest responses of antigen-stimulated, activated T cells consist of cytokine secretion and changes in the expression of various surface molecules, including cytokine receptors. Proliferation occurs next, driven in part by autocrine functions of the secreted cytokines (e.g., IL-2 for Th), along with differentiation of activated cells into effector and memory cells. Characteristic changes in the expression of surface molecules after antigen activation include upregulation of CD69 and CD25 (part of the IL-2 receptor) within the first few hours, followed by increased expression of CD40-ligand and CTLA-4 both ligands for costimulatory molecules, over the next 24 to 48 hours. Adhesion molecules and chemokine receptors are also altered in expression, directing the migration of cells to or from lymphoid organs and/or peripheral tissues.
Resting APCs in normal tissues are capable of presenting self-antigen/MHC complexes to T cells, but as they express only low levels of costimulatory molecules, potentially self-reactive naïve T cells are not activated. Treg cells also depend on CD28-mediated costimulation for their generation and maintenance. It is thought that the low levels of CD80/CD86, constitutively expressed by resting APCs, function together with self-antigens displayed by these APC to maintain Treg cells. Other receptors homologous to CD28 and their ligands (homologous to CD80/CD86) provide positive or negative signals to effector T cells.
Three major subsets of CD4 + effector T cells, termed Th1 , Th2 , and Th17 , function in host defense against distinct types of infectious pathogens and are involved in different types of tissue injury in rheumatic diseases. A fourth subset, T follicular helper cells , direct the formation and function of germinal centers in lymph nodes (see section titled The Germinal Center) and trigger B-cell production of IL-21 and IL-4. The defining characteristics of differentiated CD4 + T effector subsets are the cytokines they produce. The signature cytokines are interferon (IFN)-γ for Th1 cells; IL-4, IL-5, and IL-13 for Th2 cells; and IL-17, IL-22, and granulocyte-macrophage colony-stimulating factor (GM-CSF) for Th17 cells. The cytokines determine T-cell subset functions in health and disease. The production of distinct cytokines is initiated by expression of subset-specific transcription factors and sustained by epigenetic modifications of cytokine gene loci, a process called polarization . Th1, Th2, and Th17 cells also display distinct patterns of tissue homing, mediated by combinations of chemokine receptors and adhesion molecules, which direct them to specific tissues or sites of inflammation. Th1, but not Th2, cells express high levels of the chemokine receptors CXCR3 and CCR5 , which bind to chemokines produced in tissues during innate immune responses. Th1 cells also express high levels of ligands for E-selectin and P-selectin, which assist in migration to sites of inflammation, where these selectins are expressed on the endothelium. In contrast, Th2 cells express the chemokine receptors CCR3, CCR4, and CCR8, which recognize chemokines that are highly expressed at sites of helminthic infection or allergic reactions, particularly in mucosal tissues. Th17 cells express CCR6 which binds the chemokine CCL20, a chemokine produced by various tissue cells and macrophages in certain bacterial and fungal infections.
Th1 differentiation is driven mainly by IL-12 and IFN-γ, produced primarily by DCs and macrophages in the lymphoid organ where the response is initiated. These cytokines induce and activate the transcription factors T-bet, STAT1, and STAT4. The ability of IFN-γ to stimulate T-bet expression and the ability of T-bet to enhance IFN-γ transcription sets up a positive amplification loop for Th1 differentiation. IFN-γ from Th1 cells activates macrophages to ingest and destroy microbes and increases expression of proteins involved in antigen processing and presentation in many cell types. IFN-γ also inhibits development of Th2 and Th17 cells, thus reinforcing Th1 polarization.
The primary cytokine that initiates Th2 development is IL-4, though this may be enhanced by others, including IL-25, IL-33, and thymic stromal lymphopoietin produced by damaged epithelia and other cells. IL-4, which is produced both by mast cells and Th2 cells, functions as both an inducer and an effector cytokine of the Th2 cell subset. IL-4 receptor signaling activates STAT6, which induces transcription of genes that account for many of the actions of IL-4, including promotion of Th2 differentiation and proliferation. IL-4–driven Th2 cells augment phagocyte-independent defense, in which eosinophils and mast cells play central roles. Other functions of Th2 cells are mediated by IL-5, which activates eosinophils, and IL-13, which has diverse actions on macrophages and mucosal epithelial cells of the airway and intestine. IL-4 also stimulates B cells to produce immunoglobulin E (IgE) antibodies, which are involved in many Th2-mediated defense reactions and allergic responses.
Th17 development is stimulated by proinflammatory cytokines produced in response to bacteria and fungi, including IL-6, IL-1, and IL-23. Notably, transforming growth factor-beta (TGF-β), which is produced by many cell types and is an antiinflammatory cytokine, promotes the development of proinflammatory Th17 cells when other mediators of inflammation, such as IL-6 or IL-1, are present. Th17 differentiation is inhibited by IFN-γ and IL-4; therefore strong Th1 and Th2 responses tend to suppress Th17 development. The development of Th17 cells is dependent on the transcription factors RORγt and STAT3 . In addition to IL-17, Th17 cells produce IL-22 and IL-21, which help maintain the integrity of epithelial barriers and promote the development of germinal centers, respectively. Th17 cells at inflammatory sites may also produce other cytokines, including tumor necrosis factor (TNF) and GM-CSF. Th17 cells are especially abundant in mucosal tissues, particularly of the gastrointestinal tract. Their development and activity at these sites appear, at least in mice, to depend on the presence of local commensal bacteria. This observation suggests that Th17 cells may be especially important in regulating intestinal homeostasis and indicates potential mechanisms for their observed role in the development of inflammatory bowel disease. Th17 cells play a key role in recruiting neutrophils and, to a lesser extent, monocytes to sites of infection and inflammation. These reactions contribute significantly to tissue damage in inflammatory diseases. Th17 cells also induce production of innate antimicrobial substances, including defensins, by many cell types.
CD8 + Tc are characterized by the capacity to kill target cells infected with intracellular pathogens (e.g., viruses or intracellular bacteria). Tc also play critical roles in eradication of tumors and the acute rejection of organ allografts. The activation of naïve Tc depends on specialized subsets of DCs and in some cases requires help from CD4 + Th cells. Th cells promote Tc activation by producing cytokines that stimulate Tc differentiation directly and also express CD40 ligand ( CD154 ), which binds to CD40 on antigen-loaded DCs to upregulate costimulatory molecules, making DCs more efficient drivers of Tc differentiation.
Mature Tc develops cytoplasmic granules containing proteins, including perforin and granzymes, that are capable of disrupting the target cell membrane and triggering apoptosis. In addition, activated Tc produce cytokines, primarily IFN-γ, that activate phagocytes and enhance their functions. Inherited mutations that interfere with Tc function, such as perforin mutations, are associated with familial and acquired hemophagocytic lymphohistiocytosis and macrophage activation syndrome (see Chapter 42 ).
In addition to CD4 + and CD8 + T cells, there are smaller populations of T cells that have distinct features and probably serve specialized functions in host defense. The best defined of these subsets are γδ T cells, natural killer T (NKT) cells, and mucosa-associated invariant T (MAIT) cells. , These cell types are abundant in epithelial tissues, such as the gastrointestinal tract, and each recognizes a limited variety of antigens, many of which are not peptides and do not require presentation by MHC molecules. Their antigen receptors have limited diversity, suggesting that each of the three cell types may have evolved to recognize a specific subgroup of microbial antigens. It is also possible that these cells mainly respond to cytokines produced by other cells at sites of infection and tissue damage. Because of these features, these T-cell populations are often said to bridge innate and adaptive immunity.
Whereas the phenotypes of Th1, Th2, and Th17 cells are readily recognized and were initially thought to be mutually exclusive, it is apparent that many effector CD4 + T cells at inflammatory sites produce combinations of cytokines from different subsets or only some of the cytokines characteristic of a particular subset and are not readily classifiable into distinct populations. For example, at sites of inflammation, including joints of JIA patients, there are frequently T cells that produce both IFN-γ (characteristic of Th1 cells) and IL-17 (typical of Th17 cells). These “polyfunctional” T cells appear to be highly pathogenic in several models of arthritis. Conversely, some cells may produce cytokines that are not characteristic of any of the three major subsets (e.g., IL-9). The observation of cells with alternative or restricted cytokine profiles has led to an expanding nomenclature describing these populations (e.g., Th9, Th22). It is also clear that effector T cells may convert from one cytokine profile to another when subjected to changes in activation conditions. The extent and significance of plasticity or stability of differentiated effector T cells remain topics of active research. Maintaining a healthy immune balance between vigorous host defense and control of damage to self involves a highly interactive system of T cells that adapt quickly to a changing environment. However, it will be vital to fully understand what regulates such T-cell plasticity if T cells are to be used in cell therapy approaches (e.g., to ensure that infused Tregs do not become effectors in vivo).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here