Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The ABO blood group antigens and the human leukocyte antigens (HLA) encoded by the major histocompatibility gene complex represent substantial barriers to transplantation of tissues and organs between individuals. The antigens of these two systems are expressed on most cells in the body, and antibodies directed against either ABO or HLA antigens of the donor can cause severe damage to the graft. The allogeneic donor HLA antigens themselves are major targets of rejection reactions. The important roles of ABO, tissue typing, and crossmatch incompatibilities in transplantation were recognized early in the experience with kidney transplants. Most ABO-incompatible kidney transplants reported between 1955 and 1964 were rapidly and irreversibly rejected because of anti–blood group antibodies in the recipient’s circulation. As a result of those early experiences, all organs are now transplanted to ABO-identical or ABO-compatible recipients with few exceptions. By the mid-1960s it was also clear that patients who had circulating anti–donor HLA antibodies at the time of transplantation experienced irreversible hyperacute rejection of their kidney graft. Screening tests to identify those sensitized patients are now routinely performed before kidney transplantation to determine whether circulating anti-HLA antibodies directed against donor HLA antigens are present and have nearly eliminated hyperacute rejection of transplanted kidneys. Thus circulating antibodies to graft ABO or HLA antigens caused rapid kidney graft loss, characterized by renal artery thrombosis, neutrophil infiltrates, intimal fibrin deposition, and cortical necrosis. Similar observations of hyperacute rejections were subsequently reported in recipients of hearts, lungs, and livers.
Despite the general acceptance of the roles of ABO and HLA antigens and antibodies in organ and tissue transplantation based on these early observations, the perception has been that liver transplants are exceptional with regard to the rules of ABO and HLA. The liver is a massive organ compared with the kidney or heart, and with its dual blood supply and capacity for regeneration, it appears to be less susceptible to irreparable immune damage than other organs. After more than 35 years of experience and more than 200,000 liver transplants worldwide, controversy remains regarding the importance of the ABO and HLA antigen systems in liver transplants. Clearly the effects of ABO, HLA, and crossmatch incompatibilities are more complex as they apply to liver transplantation than to other organ transplants; however, emerging data suggest that these incompatibilities affect the outcome of liver transplants and strategies can be developed to reduce their impact. In this chapter we summarize the current status of histocompatibility for ABO and HLA in liver transplantation.
The blood group ABH antigens expressed on almost all cell types in the body show subtle differences in structure causing major differences in antigenicity. The ABH antigens in the liver are expressed on the hepatic artery, portal vein, capillary, sinusoidal lining cells, and bile duct epithelium but not on the bile ductile or hepatocytes. Three genes, A, B, and O located on chromosome 9, transcribe for ABH antigens, respectively. The ABH antigens contain a common precursor structure, the H antigen. Expression of the A and B gene-encoded glycosyltransferases catalyzes the addition of specific carbohydrate determinants to the H antigen, producing the A and B blood groups, respectively. Expression of the O gene induces no functional glycosyltransferase for the H antigen. Because of the occurrence of two different A glycosyltransferases with either high or low glycosylation efficiency, two A blood subgroups, A 1 and A 2 , are distinguished that differ in the amount of A antigen expressed.
Immunocompetent individuals produce “natural antibodies” against the A and B structures that differ from their own. Those who are blood type O have antibodies against A and B antigens; those who are blood type A have antibodies against B and vice versa; and those who are blood type AB express both antigens but no antibodies. Interestingly, the natural antibodies are not present in newborns but develop during the first year of life, presumably in response to food and environmental antigens. The presence of preformed natural antibodies and the expression of blood group antigens on blood vessel endothelia are the basis for the initial paradigm that organ transplantation across blood group barriers is impossible. Without preconditioning to lower the levels of circulating natural antibodies, transplantation across the ABO barrier can lead to hyperacute rejection within minutes. The recipient’s preformed natural antibodies react with the AB carbohydrate antigens expressed on the vascular endothelial cells in the graft. Antibody binding leads to fixation and activation of complement, which induces endothelial cell activation and damage and finally results in the formation of microthrombi and microhemorrhages.
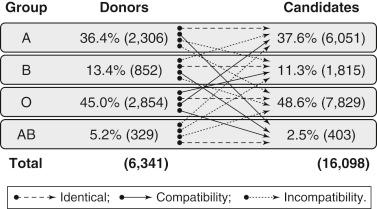
The blood groups are evenly distributed among liver donors and liver transplant candidates ( Fig. 89-1 ). The less common blood groups, B and AB, constitute only 13.4% and 5.2% of donors and 11.3% and 2.5% of candidates, respectively. However, the donor pool is considerably larger for patients with these less common types, and finding an ABO-compatible liver donor to avoid damage from natural blood group antibodies is readily achievable.
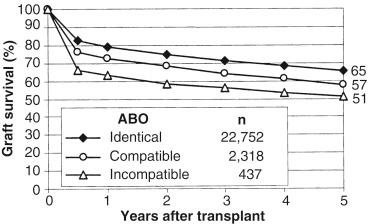
The liver was originally regarded as a “tolerogenic organ,” and it was therefore believed that it would be more easily transplanted from ABO-incompatible donors, compared with hearts or kidneys. However, trials of ABO-incompatible liver transplantation in the 1980s and in the early 1990s demonstrated that the postoperative course was extremely poor, with severe rejection crises, hepatic artery thrombosis, and intractable intrahepatic bile duct injury occurring commonly. Therefore ABO-incompatible liver transplantation in the United States is generally performed only in emergency situations when given no choice but to proceed with the first available organ. Since its introduction by Thomas Starzl et al, ABO-incompatible liver transplantation has evolved through an era of controversy. An early retrospective study of 671 liver transplants performed in Pittsburgh showed organ loss and patient death with graft failure rates from 30% to more than 50%, and nearly half of the ABO-incompatible transplant patients ultimately required retransplantation. The ABO-incompatible liver transplantations performed between 1986 and 2000 and reported in 24 different studies were reviewed by Rydberg, who concluded that ABO-incompatible liver transplantations were substantially less successful than ABO-compatible liver transplants. A recently performed systematic review and meta-analysis of 701 ABO-incompatible (116 pediatric and 585 adult) and 2103 ABO-compatible (348 pediatric and 1755 adult) liver transplants reported in 14 different publications revealed ABO-incompatible liver transplantations have markedly inferior results, with an increased incidence of vascular and biliary complications and rejection, when compared to ABO-compatible grafts. The recipient age plays an important role in the development of allograft failure, with younger recipients having better outcomes after ABO-incompatible transplant. Egawa et al reported that the patient survival rate after ABO-incompatible transplant gradually decreased with increasing recipient age; the 5-year patient survival rate was 85% in infants and only 52% in adults. Several reasons may account for this phenomenon: first, anti-A and anti-B antibody titers remain at low levels in infants because of incomplete maturity of the immune system. Second, the complement system in the very young is not as sensitive compared to that in adults. Data from the United Network for Organ Sharing (UNOS) Organ Procurement and Transplantation Network (OPTN) presented in Figure 89-2 show that recipients of ABO-compatible or ABO-incompatible livers transplanted between 1995 and 2000 had poorer outcomes than recipients of ABO-identical organs. The graft survival rates at 6 months ranged from 83% for ABO-identical to 76% for compatible and 66% for ABO-incompatible livers. Five years after transplantation the differences in graft survival rates remained essentially the same, suggesting that the deleterious effect of ABO differences manifests early within the first posttransplant year and that there is no additional long-term disadvantage associated with ABO-nonidentical transplants. Thus liver graft survival rates for recent ABO-incompatible transplants performed at centers throughout the United States were about 15% lower at 5 years than those from ABO-compatible donors overall. It is noteworthy that among these 25,507 deceased-donor liver transplants, ABO-nonidentical transplants were uncommon. Only 9% of liver transplants were performed from ABO-nonidentical but compatible donors and fewer than 2% were from ABO-incompatible donors.
The often urgent nature of ABO-compatible and ABO-incompatible transplants could explain the poorer outcomes in these combinations. However, two early studies compared ABO-identical, ABO-compatible, and ABO-incompatible transplants done in urgent situations and found that the 35% to 45% lower 2-year survival rate of ABO-incompatible grafts was not due to the emergency conditions under which the transplant was performed. In fact, the incompatible grafts in each case were lost early, and immunoglobulin and complement components were readily identified on sinusoidal cells and arterial endothelium, indicating a clear humoral component to the graft failures. This conclusion was supported by a subsequent analysis comparing 31 ABO-incompatible transplants with 199 ABO-compatible emergency transplants at UCLA, which revealed a significantly increased incidence of rejection, thrombosis, and biliary stricture among incompatible graft recipients, resulting in a 20% lower 1-year graft survival rate. Most recently a study of 229 highly urgent liver transplantations in Scandinavia noted that patients who received an ABO-identical graft had significantly higher patient survival rates than those who received ABO-compatible (n = 76) or ABO-incompatible livers (n = 10). In this study, which included transplants performed in five countries during 1990-2001, the authors noted that the outcomes for highly urgent transplants had improved during the course of the study; however, ABO compatibility produced superior results throughout.
Liver allocation algorithms often provide for mandatory sharing of livers for highly urgent patients, allowing ABO-compatible and even ABO-incompatible offers. However, such prioritizations may disadvantage other less urgent patients, particularly blood group O patients. Eurotransplant has reported a simulation study that suggests a restricted ABO matching policy provides the optimum balance for both urgent and elective patients from the allocation point of view. Under this system, blood group O livers are offered to blood group O or B patients and blood group A livers are offered to blood group A or AB patients. UNOS in the United States employs a similar policy. Blood group O livers are allocated first to blood group O patients then to blood group B patients ranked according to their medical condition. A point system is used to promote ABO-identical combinations over ABO-compatible and ABO-incompatible transplants.
Despite the poorer survival rates for ABO-incompatible transplants, many survive and function well. This observation contrasts sharply with the results for inadvertent ABO kidney transplants, nearly all of which have failed very early after the transplant. When ABO-incompatible transplants are performed inadvertently or in urgent situations, there is no time to assess the suitability of the recipient to receive an incompatible organ. However, the results of ABO-incompatible transplants might be improved when the incompatibility is anticipated and the recipient can be conditioned through antibody reduction therapy such as plasmapheresis in advance.
During the 1980s a small number of deliberate ABO-incompatible kidney transplants were undertaken when a willing a medically suitable living donor was available, but ABO-incompatibility presented a barrier. The largest experience with deliberate ABO-incompatible liver transplants may be in Asia and specifically in Japan, where the lack of deceased donor livers limits the availability of liver transplantation to those with a living donor. According to the Japanese registry of living donor liver transplantation (LDLT) across ABO blood type barrier, 97 ABO-incompatible LDLTs were performed in Japan before 2005, and the 5-year survival rate of the patients was 38% before 2001 and improved to 63% among patients who underwent transplantation after 2002.
LDLTs in ABO-compatible and ABO-incompatible combinations have been frequently performed after conditioning the patient to reduce the likelihood of early antibody-mediated damage. The goal of conditioning is to reduce the titer of anti-A or anti-B antibody in the patient before transplantation and to prevent its rapid rebound in the early posttransplant period. This has been accomplished by plasmapheresis often accompanied by splenectomy, immunosuppressant drugs, and other approaches, with monitoring of antibody titers. Most studies agree that an antibody titer of less than 1:8 is sufficiently low to avoid most problems.
Despite these preparations, however, there remains a high risk for failure because several of these studies have reported a 40% to 60% early graft loss rate among recipients of ABO-incompatible LDLTs. The most encouraging results were obtained by Hanto et al, who reported no immunological graft losses in 14 patients after a combination of pretransplant and posttransplant total plasma exchange and splenectomy with quadruple immunosuppression. In 16 pediatric ABO-incompatible pediatric liver transplantations, Heffron et al reported 1-year actuarial graft survival of 92% using standard immunosuppression with selective postoperative plasmapheresis and without splenectomy. Plasmapheresis may be useful by reducing the recipients’ antibody titers before and after transplantation.
Interestingly, anti–ABO blood group antibodies may return to high levels after the transplant without causing apparent damage to the graft. Alexandre et al first described this phenomenon, called “accommodation,” in recipients of deliberate ABO-incompatible kidneys. The mechanism underlying graft accommodation remains unclear. Platt et al proposed three possible mechanisms for this phenomenon, postulating that either the antibody or the antigen was somehow modulated on the graft or, alternatively, that the graft itself becomes resistant to the damaging effects of antibody. There is some evidence now to support the latter possibility—that cells exposed to low levels of antibody may express antiapoptotic genes early after exposure and that accommodated grafts acquire a distinct phenotype characterized by expression of tumor necrosis factor (TNF)-α, transforming growth factor (TGF)-β 1 , SMAD5, protein kinase GFRA1, and MUC1 3 months or more after successful transplantation.
Promising results with ABO-incompatible liver transplantation using A 2 donors (with a subgroup of A that is less reactive and occurs in approximately 20% of group A individuals) have been reported. Transplants of A 2 livers to O recipients appear to be safe without augmented immunosuppression or restrictions with regard to anti-A antibody titers, according to a study of six such transplants at Mount Sinai Medical Center in New York. In a Swedish study of 10 adult blood group O recipients who received A 2 cadaveric grafts, patient and graft survival were 10 of 10 and 8 of 10, respectively, at 8.5 months’ median follow-up with a tacrolimus-based protocol and initial immunosuppression with antithymocyte globulin, interleukin-2-receptor antagonists, or anti-CD20 antibody.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here