Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Chronic diarrhea is defined as stool volume of more than 10 g/kg/day in toddlers/infants and greater than 200 g/day in older children that lasts for 4 wk or more. Persistent diarrhea began acutely but lasts longer than 14 days. In practice, this usually means having loose or watery stools more than 3 times a day . Awakening at night to pass stool is often a sign of an organic cause of diarrhea. The epidemiology has 2 distinct patterns. In developing countries, chronic diarrhea is, in many cases, the result of an intestinal infection that persists longer than expected. This syndrome is often defined as protracted (persistent) diarrhea , but there is no clear distinction between protracted (persistent) and chronic diarrhea. In countries with higher socioeconomic conditions, chronic diarrhea is less frequent, and the etiology often varies with age. The outcome of diarrhea depends on the cause and ranges from benign, self-limited conditions, such as toddler's diarrhea, to severe congenital diseases, such as microvillus inclusion disease, that may lead to progressive intestinal failure.
The mechanisms of diarrhea are generally divided into secretory and osmotic , but often diarrhea is a combination of both mechanisms . In addition, inflammation and motility disorders may contribute to diarrhea. Secretory diarrhea is usually associated with large volumes of watery stools and persists when oral feeding is withdrawn. Osmotic diarrhea is dependent on oral feeding, and stool volumes are usually not as massive as in secretory diarrhea ( Fig. 367.1 ).
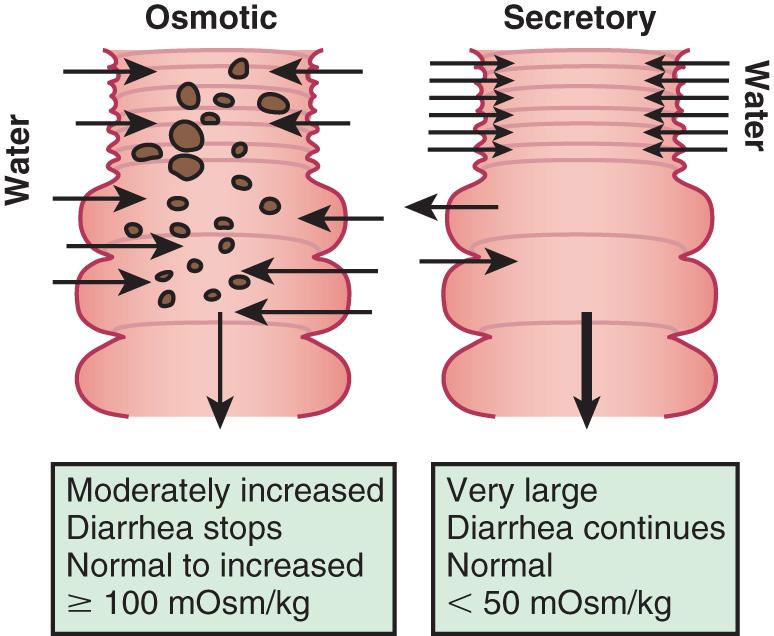
Secretory diarrhea is characterized by active electrolyte and water fluxes toward the intestinal lumen, resulting from either the inhibition of neutral NaCl absorption in villous enterocytes or an increase in electrogenic chloride secretion in secretory crypt cells as a result of the opening of the cystic fibrosis transmembrane regulator (CFTR) chloride channel or both. The result is more secretion from the crypts than absorption in the villous that persists during fasting. The other components of the enterocyte ion secretory machinery are (1) the Na-K 2Cl cotransporter for the electroneutral chloride entrance into the enterocyte; (2) the Na-K pump, which decreases the intracellular Na + concentration, determining the driving gradient for further Na + influx; and (3) the K + selective channel, that enables K + , once it has entered the cell together with Na + , to return to the extracellular fluid.
Electrogenic secretion is induced by an increase of intracellular concentration of cyclic adenosine monophosphate, cyclic guanosine monophosphate, or calcium in response to microbial enterotoxins, or to endogenous endocrine or nonendocrine molecules, including inflammatory cytokines. Another mechanism of secretory diarrhea is the inhibition of the electroneutral NaCl-coupled pathway that involves the Na + /H + and the Cl − /HCO 3 − exchangers. Defects in the genes of the Na + /H + and the Cl − /HCO 3 − exchangers are responsible for congenital Na + and Cl − diarrhea, respectively.
Osmotic diarrhea is caused by nonabsorbed nutrients in the intestinal lumen as a result of one or more of the following mechanisms: (1) intestinal damage (e.g., enteric infection); (2) reduced absorptive surface area (e.g., active celiac disease); (3) defective digestive enzyme or nutrient carrier (e.g., lactase deficiency); (4) decreased intestinal transit time (e.g., functional diarrhea); and (5) nutrient overload, exceeding the digestive capacity (e.g., overfeeding, sorbitol in fruit juice). Whatever the mechanism, the osmotic force generated by nonabsorbed solutes drives water into the intestinal lumen. A very common example of osmotic diarrhea is lactose intolerance. Lactose, if not absorbed in the small intestine, reaches the colon, where it is fermented to short-chain organic acids, releasing hydrogen that is detected in the lactose breath test, and generating an osmotic overload. Another risk for chronic osmotic diarrhea often noted in patients with diarrhea-associated irritable bowel syndrome are foods containing FODMAPs (fermentable oligo-di-monosaccharides and polyols).
In many children chronic diarrhea may be caused by the combination of multiple mechanisms.
Table 367.1 summarizes the main etiologies of chronic diarrhea in infants and children.
| ETIOLOGY | YOUNGER THAN 2 YR | OLDER THAN 2 YR |
|---|---|---|
| Infections | +++ | +++ |
| Postenteritis syndrome | +++ | +++ |
| Immune deficiency | ++ | Rare |
| Celiac disease | +++ (after gluten introduction) | +++ |
| Food allergy | +++ | + |
| Inflammatory bowel disease | + (rare) | +++ |
| Pancreatic insufficiency | ++ | ++ |
| Cholestasis and insufficient bile acids | ++ | ++ |
| Cystic fibrosis | ++ | + |
| Lactose intolerance | ++ (mostly postinfectious) | +++ |
| Intestinal lymphangiectasia | + | + |
| Motility disorders | ++ | Rare |
| Short bowel syndrome | +++ | + |
| Toddler's and functional diarrhea | ++ | ++ |
| Excessive intake of fruit juices and fluids | ++ | ++ |
| Congenital diarrheal disorders, including structural enterocyte defects and enzymatic or transport malabsorption syndromes | ++ | Unlikely |
Enteric infections are by far the most frequent cause of persistent or chronic diarrhea, both in developing and industrialized countries, however outcomes are often very different. In the former, comorbid conditions, such as HIV/AIDS, malaria, or tuberculosis, result in malnutrition that impairs the child's immune response, thereby potentiating the likelihood of prolonging diarrhea or acquiring another enteric infection. In children with HIV/AIDS, the viral infection itself impairs immune function and may trigger a vicious circle with malnutrition. Sequential infections with the same or different pathogens may also be responsible for chronic diarrhea. In developing countries, enteroadherent Escherichia coli and Giardia lamblia have been implicated in chronic diarrhea, whereas, in developed countries, chronic infectious diarrhea usually runs a more benign course and the etiology is often viral, with a major role of rotavirus and norovirus ( Table 367.2 ).
| AGENT/DISEASE | |
|---|---|
| INDUSTRIALIZED COUNTRIES | DEVELOPING COUNTRIES |
|
|
| Astrovirus Norovirus Rotavirus * Small intestinal bacterial overgrowth (SIBO) Postenteritis diarrhea syndrome |
|
* More frequent in industrialized than in developing countries as agent of chronic diarrhea.
Chronic diarrhea in travelers to or expatriates from developing countries may depend on the country of origin. Nonetheless, common pathogens include giardia, E. coli , shigella, campylobacter, salmonella, and enteric viruses. Less common pathogens include amebiasis, strongyloides, and tropical spruce.
Opportunistic microorganisms induce diarrhea exclusively, more severely, or for more prolonged periods, in specific populations, such as immunocompromised children. Specific agents cause chronic diarrhea or exacerbate diarrhea in many chronic diseases. Clostridium difficile or cytomegalovirus act as opportunistic agents in oncologic patients as well as in patients with inflammatory bowel diseases. Cryptosporidium may induce severe and protracted diarrhea in AIDS patients.
Small intestinal bacterial overgrowth results in chronic diarrhea by either a direct interaction between the microorganism and the enterocyte, or the consequence of deconjugation and dihydroxylation of bile salts and hydroxylation of fatty acids due to an increased proliferation of bacteria in the proximal intestine.
Postenteritis diarrhea syndrome ( Chapter 364.4 ) is a clinicopathologic condition in which small intestinal mucosal damage persists after acute gastroenteritis. Sensitization to food antigens, secondary disaccharidase deficiency, persistent infections, reinfection with an enteric pathogen, or side effects of medication may be responsible for causing postenteritis diarrhea syndrome, thought to be related to dysregulation of the intestinal microbiota. Functional diarrhea which may be related to the pathogenesis of irritable bowel syndrome may be caused by complications of an acute gastroenteritis.
Celiac disease ( Chapter 364.2 ) is a genetically determined permanent gluten intolerance that affects about 1 in 100 individuals, depending on geographic origin. In the genetically susceptible host, gliadin, the major protein of gluten, reacts with the immune system to cause villous atrophy. A reduction of intestinal absorptive surface is responsible for the diarrhea in celiac disease, which is reversible upon restriction of gluten from the diet.
Food allergy (mainly cow milk protein allergy Chapter 176 ) may present during infancy with chronic diarrhea. An abnormal immune response to food proteins can cause a proctitis/colitis or an enteropathy. Eosinophilic gastroenteritis is characterized by eosinophilic infiltration of the intestinal wall and is strongly associated with atopy. However, whereas diarrhea in food allergy responds to withdrawal of the responsible food, this does not always occur in eosinophilic gastroenteritis, in which immune suppression may be needed.
Inflammatory bowel diseases, including Crohn disease, ulcerative colitis, and inflammatory bowel disease–undetermined, cause chronic diarrhea that is often associated with abdominal pain, elevated inflammatory markers, and increased concentrations of fecal calprotectin or lactoferrin (see Chapter 362 ). The age of onset of inflammatory bowel disease is broad, with rare cases described in the 1st few mo of life, but the peak incidence in childhood occurs in adolescence. The severity of the symptoms is highly variable with a pattern characterized by long periods of well-being followed by exacerbations.
Autoimmune processes may target the intestinal epithelium, alone or in association with extraintestinal symptoms. Autoimmune enteropathy is associated with the production of antienterocyte and antigoblet cell antibodies, primarily immunoglobulin A, but also immunoglobulin G, directed against components of the enterocyte brush-border or cytoplasm and by a cell-mediated autoimmune response with mucosal T-cell activation. An X-linked immune-dysregulation, polyendocrinopathy, and enteropathy ( IPEX syndrome ) is associated with variable gene mutations and phenotypes of chronic diarrhea (more on autoimmune enteropathy and IPEX syndrome is available on Chapter 364.3 ).
Immune deficiency can present as chronic diarrhea in children. In these cases (for example, severe combined immunodeficiency or AIDS) the child can be infected by an opportunistic pathogen; can exhibit a persistent diarrhea due to a pathogen usually causing an acute gastroenteritis; or be infected by multiple and recurrent different pathogens causing mucosal damage to the intestines. Other immunoregulatory defects, found in patients with agammaglobulinemia, isolated immunoglobulin A deficiency, and common variable immunodeficiency disorder, may result in mild persistent infectious diarrhea.
Chronic diarrhea may be the manifestation of maldigestion caused by exocrine pancreatic disorders (see Chapters 376 and 378.2 ). In most patients with cystic fibrosis , exocrine pancreatic insufficiency results in steatorrhea and protein malabsorption. In Shwachman-Diamond syndrome , exocrine pancreatic hypoplasia may be associated with neutropenia, bone changes, and intestinal protein-losing enteropathy. Specific isolated pancreatic enzyme defects, such as lipase deficiency, result in fat and/or protein malabsorption. Familial pancreatitis, associated with a mutation in the trypsinogen gene, may be associated with exocrine pancreatic insufficiency and chronic diarrhea. Mutations in CFTR , CTRC , PRSS1 , PRSS2 , SPINK 1 , and SPINK 5 are associated with hereditary pancreatitis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here