Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Jaundice, the yellow discoloration of skin and sclerae, results when the serum level of bilirubin, a pigmented compound, is elevated. Jaundice is not evident until the total serum bilirubin is at least 2–2.5 mg/dL in children out of the neonatal period.
Bilirubin is formed from the degradation of heme-containing compounds, particularly hemoglobin ( Fig. 18.1 ). Microsomal heme oxygenase, located principally in the reticuloendothelial system, catabolizes heme to biliverdin, which is then reduced to bilirubin by biliverdin reductase. This unconjugated bilirubin (UCB) is lipophilic and cannot be easily eliminated via the kidney because of its insolubility in water. It can easily cross cell membranes and the blood-brain barrier. UCB is transported bound primarily to albumin. A receptor on the hepatocyte surface facilitates bilirubin uptake. Bilirubin is then conjugated with glucuronic acid by bilirubin uridine diphosphate glucuronosyltransferase (UDPGT). UDPGT can be induced by a variety of drugs (e.g., narcotics, anticonvulsants, and oral contraceptives) and by bilirubin itself. Enzyme activity is decreased by restriction of calorie and protein intake.
Conjugated bilirubin (CB) is a polar, water-soluble compound. It is excreted from the hepatocyte to the canaliculi, through the biliary tree, and into the duodenum. Once CB reaches the colon, bacterial hydrolysis converts CB to urobilinogen. A small amount of urobilinogen is reabsorbed and returned to the liver via enterohepatic circulation or excreted by the kidneys. The remainder is converted to stercobilin and excreted in feces. In neonates, β-glucuronidase in the intestinal lumen hydrolyzes CB to UCB, which is then reabsorbed and returned to the liver via the enterohepatic circulation.
Hyperbilirubinemia can result from alteration of any step in this process. Hyperbilirubinemia can be classified as conjugated (direct) or unconjugated (indirect), depending on the concentration of CB in the serum. Conjugated and unconjugated are more accurate terms, because “direct” and “indirect” refer to the van den Bergh reaction, historically used for measuring bilirubin. In this assay, the unconjugated fraction is determined by subtracting the direct fraction from the total and, therefore, is an indirect measurement. The direct fraction includes both conjugated bilirubin and Δ-bilirubin, an albumin-bound fraction. Conjugated hyperbilirubinemia exists when more than 20% of the total bilirubin or more than 2 mg/dL is conjugated. If neither criterion is met, the hyperbilirubinemia is classified as unconjugated.
Unconjugated hyperbilirubinemia can be caused by any process that results in increased production, decreased delivery to the liver, decreased hepatic uptake, decreased conjugation, or increased enterohepatic circulation of bilirubin. The primary concern in patients with high levels of unconjugated bilirubin is kernicterus, resulting from the neurotoxicity of UCB across the blood-brain barrier mostly in the basal ganglia, pons, or cerebellum. This is a concern primarily in neonates.
Conjugated hyperbilirubinemia can occur due to hepatocellular dysfunction, biliary obstruction, and abnormal excretion of bile acids or bilirubin.
The causes of jaundice in the neonate and older infant are not the same as the causes of jaundice in the older child or adolescent ( Figs. 18.2 and 18.3 ). The approach to the problem varies with age.
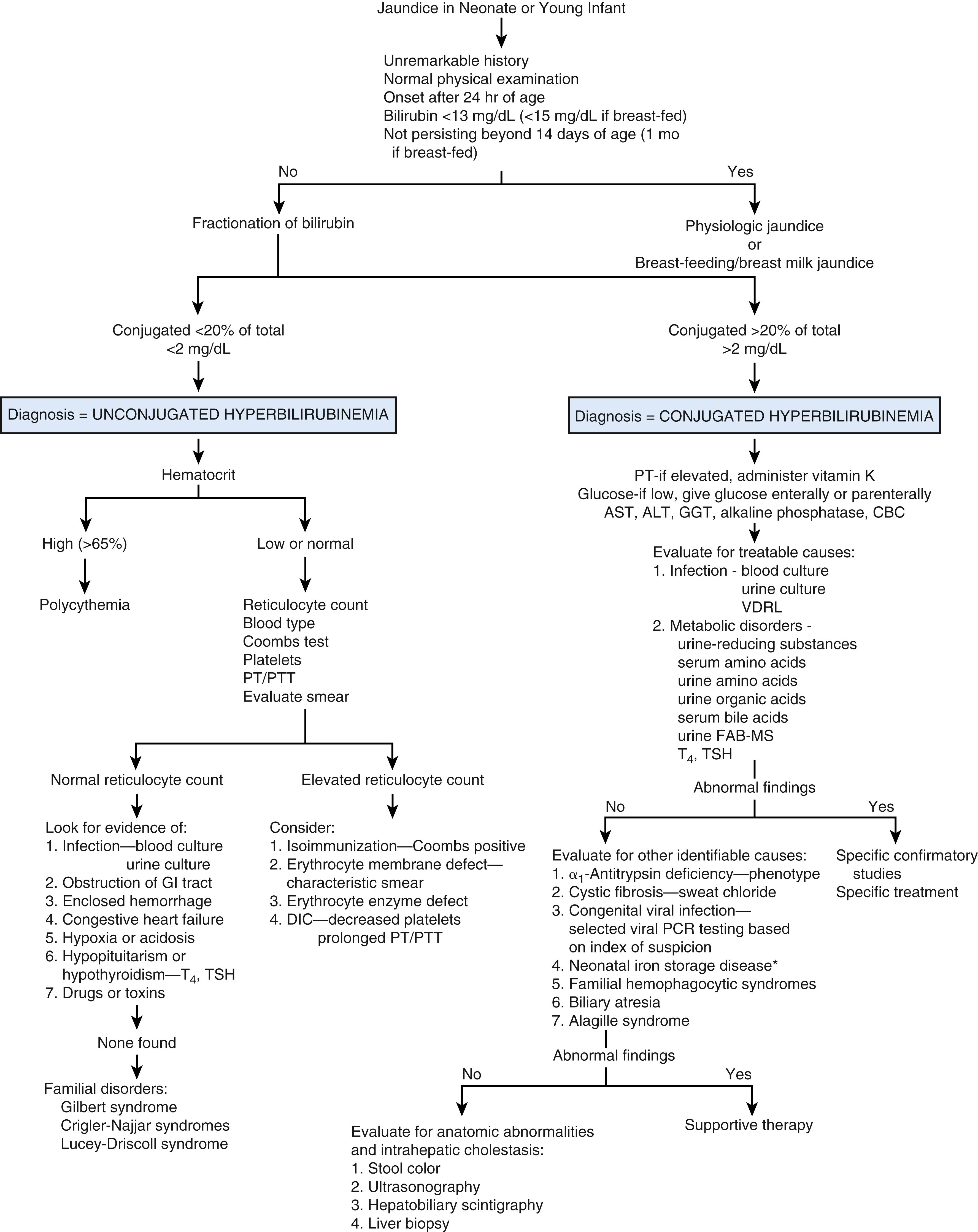
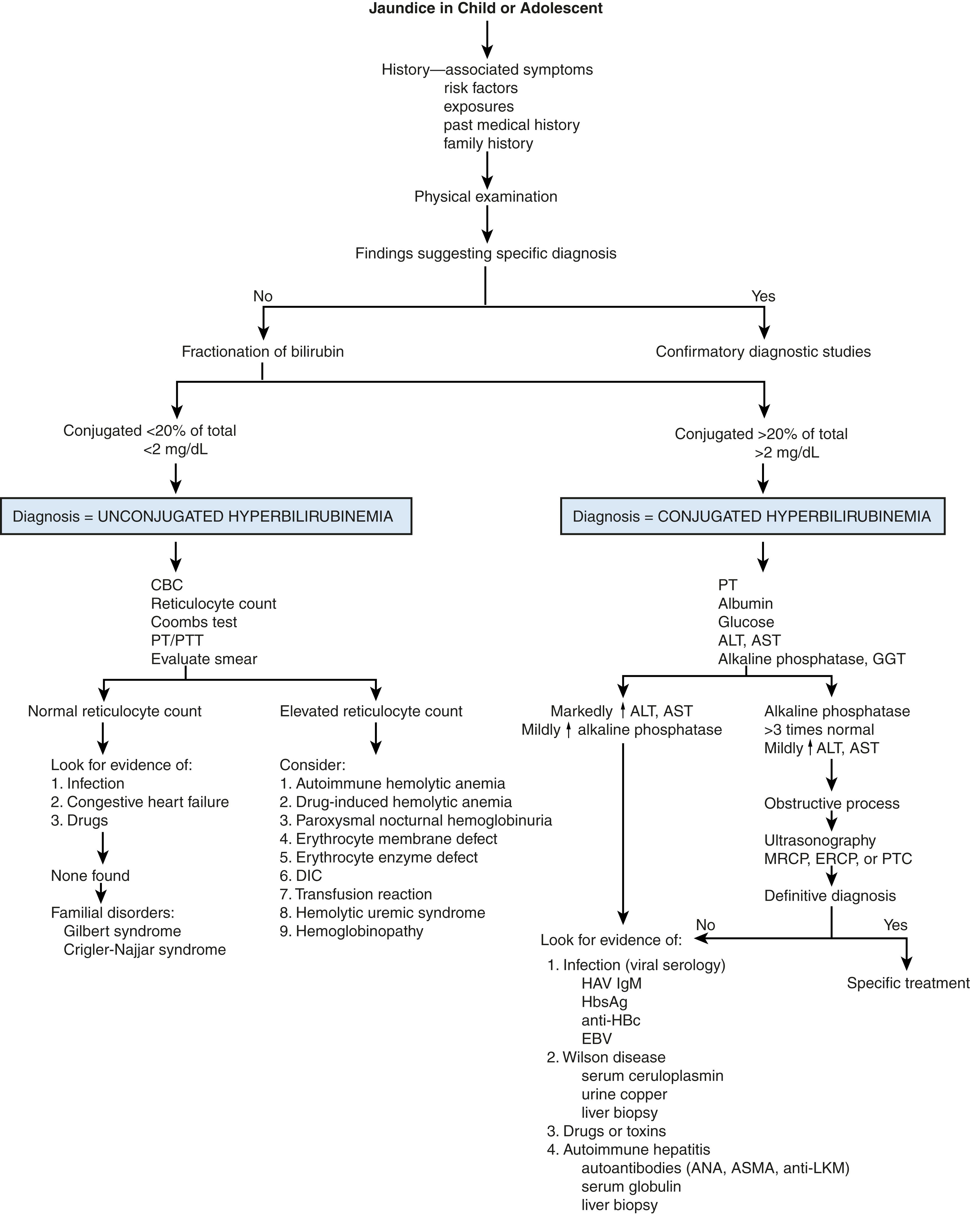
In any patient with jaundice, the total serum bilirubin should be fractionated, as the differential diagnosis of unconjugated hyperbilirubinemia is distinct from that of conjugated hyperbilirubinemia (see Figs. 18.2 and 18.3 ). On occasion, hemolysis interferes with some assays and may result in a falsely elevated conjugated fraction. This can be problematic with specimens obtained by heelstick or fingerstick. If the clinical picture is consistent with unconjugated hyperbilirubinemia, the assay should be repeated with a venous sample.
Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are frequently used as markers of hepatocellular injury. AST is expressed in mitochondria of the liver and cytosol of red blood cells and muscles; thus, it is not specific for liver injury. Since ALT is less abundant outside of the liver, an increased ALT level is more suggestive of liver disease. Levels of both are markedly elevated (>5- to 10-fold normal) with hepatocellular injury caused by hepatitis, hepatotoxicity, ischemia, and genetic or metabolic liver disorders. Elevation of AST in excess of ALT suggests an extrahepatic source of injury. With acute biliary obstruction, there are initial sharp increases in ALT and AST levels and a rapid decline in 12–72 hours as obstruction is relieved. In chronic cholestasis, aminotransferases are usually only mildly elevated. With hepatocellular injury, ALT and AST levels tend to remain more significantly elevated longer. In acute liver failure a rapid decline in ALT and AST levels with worsening coagulopathy is a poor prognostic factor.
It is important to remember that aminotransferases reflect cell injury, not liver function. There is no correlation between the severity of the liver dysfunction and the degree of elevation of ALT and AST levels. Temporal trends in serum aminotransferase levels are useful in monitoring disease activity in chronic viral and autoimmune hepatitis.
Alkaline phosphatase is an enzyme found in bile ducts, bone, intestine, placenta, and tumors. Elevations in the serum alkaline phosphatase level occur with hepatobiliary disease but also normal growth, healing fractures, vitamin D deficiency, bone disease, pregnancy, and malignancy. Fractionation of the alkaline phosphatase isoenzymes can help to determine its site of origin. A mild increase can be seen transiently in normal individuals. In the evaluation of conjugated hyperbilirubinemia, an alkaline phosphatase level of >3 times normal indicates cholestasis; a milder elevation is more consistent with hepatocellular disease.
The γ -glutamyltransferase (GGT) level is more specific for biliary tract disease than are ALT and AST levels. GGT elevations are inducible by alcohol and certain drugs, including phenytoin and phenobarbital. GGT is found in a variety of tissues and can be elevated in chronic pulmonary disease, renal failure, and diabetes mellitus. The GGT concentration is most helpful in confirming that an elevated alkaline phosphatase level is a result of liver disease rather than bone disease and in differentiating familial cholestatic syndromes.
Serum bile acids are a very sensitive measure of cholestatic disease. Bile acid levels may be elevated before an increase in bilirubin. Levels are generally very high in primary cholestasis and biliary obstruction but only mildly increased (more than twice normal) in hepatocellular disease. Bile acids should be measured while fasting.
Albumin is produced in the liver, and levels can reflect hepatic synthetic function. Serum albumin levels can be useful in monitoring progression of chronic liver disease and in discriminating an acute illness from a previously unrecognized chronic disorder. Hypoalbuminemia can also be secondary to nephrotic syndrome or a protein-losing enteropathy. Due to a long half-life (20 days), albumin is of limited use in assessing synthetic dysfunction in acute liver failure.
Prothrombin time (PT) is the best marker of hepatic synthetic function, as most clotting factors are produced in the liver. It is important not only to measure the PT but also to document the response to parenteral administration of vitamin K because vitamin K deficiency may be an alternative explanation of the elevation of the PT. With severe hepatocellular injury, there is little improvement in the PT. Disseminated intravascular coagulation and thrombosis of a major blood vessel should not be overlooked as the cause of a prolonged PT.
Ultrasound studies are useful, noninvasive, relatively inexpensive diagnostic tools for the evaluation of liver disease. Ultrasonography provides information on the size and consistency of the liver and spleen and anatomic abnormalities of the biliary tree, gallstones, and hepatic masses such as cysts, tumors, or abscesses. Dilated intrahepatic ducts may indicate extrahepatic obstruction; however, the absence of dilatation on ultrasonography cannot exclude obstruction, and further studies are required for definitive diagnosis. The utility of ultrasonography is limited in obese patients and in patients with excessive bowel gas. Doppler ultrasonography also demonstrates dynamic flow in hepatic blood vessels and the portal vein; it can identify vascular anomalies of the liver and suggest presence of portal hypertension. Ultrasound elastography can provide an indirect measure of liver fibrosis.
Hepatobiliary scintigraphy can aid in the diagnosis of biliary atresia. In a healthy individual, hepatic uptake and excretion of the radionuclide via the biliary system are prompt. When there is an injury to the hepatocyte, the uptake of radionuclide by the liver is diminished; however, the tracer should eventually be visualized in the intestinal tract. With obstructive processes, such as biliary atresia, uptake should be relatively normal unless the problem has been present long enough to have caused hepatocellular injury; however, there is no excretion into the intestinal tract. Administration of phenobarbital (5 mg/kg/day) for 5 days before the study may increase bile flow and thus can increase the diagnostic accuracy. Unfortunately, a significant percentage of patients with intrahepatic cholestasis and neonatal hepatitis do not demonstrate biliary excretion, and further evaluation is needed; thus, final diagnosis is delayed. In patients with high level of suspicion for biliary atresia (acholic stools, high GGT), liver biopsy and percutaneous cholangiogram provide a faster and more direct way to reach the diagnosis.
CT is useful for identifying mass lesions within the liver and when there are technical problems with ultrasonography. CT with contrast can be used to define the nature of liver tumors. CT angiography can define the anatomy of portal and hepatic circulation. CT has limited value in the evaluation of biliary anatomy.
Magnetic resonance (MR) studies provide valuable information regarding the anatomy of the liver. Since many imaging protocols can be used depending on the purpose of the study, contacting a radiologist prior to ordering the study is recommended. Cost and frequent need for sedation make MR evaluation the tool for secondary evaluation after screening imaging with ultrasound leaves diagnostic questions. MRI can demonstrate storage of heavy metals, such as iron in neonatal iron storage disease. MR with contrast can define the nature of liver tumors. MR angiography is useful in studying the vascular system, including the vascular supply of tumors. MR cholangiopancreatography (MRCP) visualizes abnormalities of the intrahepatic and extrahepatic biliary tree and is also quite useful in evaluating the pancreatic duct system. At this point resolution of MRCP is inadequate to diagnose biliary atresia. Unlike endoscopic retrograde cholangiopancreatography (ERCP) or percutaneous transhepatic cholangiography (PTC), MRCP is noninvasive.
ERCP is performed for the evaluation of biliary anatomy in situations when endoscopic intervention is likely. Unlike MRCP, ERCP is both diagnostic and potentially therapeutic for common duct stones and for strictures. Complications of the procedure include cholangitis and pancreatitis. ERCP is recommended for evaluation of the biliary tree when therapeutic intervention is likely.
PTC can be used as an alternative to ERCP as a diagnostic and therapeutic tool in evaluating the biliary tree. Under ultrasound guidance, a needle is passed through the liver and into the biliary tree, and contrast material is injected. If obstruction is identified, biliary drainage, if required, can be performed at the same time. PTC is contraindicated if there are marked ascites or irreversible coagulopathy. The complications of PTC include bleeding, pneumothorax, infection, and bile leakage.
Percutaneous liver biopsy is often necessary to determine the cause of conjugated hyperbilirubinemia. In some instances, a specific pattern of injury, such as paucity of bile ducts or bile duct proliferation, may be evident. In other cases, specific markers of disease may be identified (the distinctive inclusions in α 1 -antitrypsin deficiency) or measured (metabolic enzyme activity). Ultrasound-guided biopsy is useful when a specific lesion needs to be evaluated or if there is abnormal anatomy of the liver. An open biopsy may be necessary when a large sample of tissue is needed or when there are contraindications to the percutaneous approach, such as ascites or severe coagulopathy. Transjugular liver biopsy can reduce the risk of bleeding in patients with coagulopathy. The complications of liver biopsy are the same as those for PTC.
Evaluation of the infant with jaundice starts with a thorough history, including age at onset and duration of jaundice (see Fig. 18.2 ). In the neonate, the causes of jaundice range from a benign, self-limited process associated with immaturity of bilirubin excretion (physiologic jaundice) to life-threatening biliary atresia or metabolic disorders (galactosemia, fructosemia, tyrosinemia). In older infants, there are fewer benign explanations for jaundice. For example, physiologic jaundice generally resolves by 1–2 weeks of age, and jaundice associated with breast milk usually resolves by the time the infant is 1 month old.
Acholic stools usually indicate obstruction of the biliary tree; however, nonpigmented stools can be seen with severe hepatocellular injury. The clinician should document the presence or absence of acholic stool in every infant evaluated for jaundice. The center of the stool should be examined because the outside may be lightly pigmented from sloughed jaundiced cells of the intestinal tract. Delayed passage of meconium may be secondary to cystic fibrosis or Hirschsprung disease. Delayed passage of stools, by itself, can lead to increased enterohepatic circulation of bilirubin.
Clues to the diagnosis of hyperbilirubinemia are often found in the prenatal and perinatal history ( Table 18.1 ). Maternal infections that can be transmitted to the fetus or neonate, such as syphilis, toxoplasmosis, cytomegalovirus (CMV), hepatitis B, enterovirus, herpes simplex, and HIV, are rare causes of cholestatic liver disease in the neonate. Prenatal growth pattern should be carefully evaluated. Perinatal infections such as CMV, rubella, and toxoplasmosis can present with intrauterine growth restriction. Premature infants are prone to higher bilirubin levels and more prolonged hyperbilirubinemia; they are also more likely to have risk factors for hyperbilirubinemia such as delayed enteral feedings, require parenteral nutrition, and have perinatal insults with hypoxia and acidosis.
| Symptom | Possible Diagnosis |
|---|---|
| Prenatal/Perinatal Findings | |
| Polyhydramnios | Intestinal atresia |
| In utero growth restriction | Cytomegalovirus; rubella; toxoplasmosis |
| Vomiting/poor feeding | Metabolic disorders |
| Delayed passage of meconium | Cystic fibrosis; Hirschsprung disease |
| Constipation, hypotonia, hypothermia | Hypothyroidism |
| Maternal preeclampsia | HELLP: fatty acid oxidation disorders |
| Microphallus | Hypopituitarism associated with SOD |
| Intrahepatic cholestasis of pregnancy | PFIC type 2 and 3 |
| Repeated affected neonates | Alloimmune hepatitis |
| Characteristic Facies | |
| Narrow cranium, prominent forehead, hypertelorism, epicanthal folds, large fontanel | Zellweger syndrome |
| Triangular face with broad forehead, hypertelorism, deep-set eyes, long nose, pointed mandible | Alagille syndrome |
| Microcephaly | Congenital viral infections |
| Ophthalmologic Findings | |
| Cataracts | Galactosemia; rubella |
| Chorioretinitis | Congenital infections |
| Nystagmus with hypoplasia of optic nerve | Hypopituitarism with SOD |
| Posterior embryotoxon | Alagille syndrome |
| Perinatal Infections | |
| Syphilis | Syphilis |
| Toxoplasmosis | Toxoplasmosis |
| Cytomegalovirus | Cytomegalovirus |
| Hepatitis B | Hepatitis B |
| Herpes simplex | Herpes simplex |
| Enterovirus | Enterovirus |
| HIV | HIV infection |
| Renal Disease | |
| RTA | Tyrosinemia |
| RTA | Galactosemia |
| Congenital hepatic fibrosis | ARPKD |
| Alagille syndrome | Alagille syndrome |
| Arthrogryposis | RTA–cholestasis syndrome |
| Fibrocystic disease | Congenital hepatic fibrosis |
Delay of feeding can contribute to both conjugated and unconjugated hyperbilirubinemia; this effect is usually transient and should not be overinterpreted. Breast-feeding is associated with higher levels of unconjugated bilirubin and a longer duration of jaundice than in formula-feeding. Even when diagnosis of breast milk jaundice is likely, conjugated bilirubin should be checked because it provides an easy screening tool for liver disorders, including biliary atresia. Galactosemia does not manifest in the infant who receives a lactose-free formula. Hereditary fructose intolerance is not clinically apparent until the infant ingests fluids or solids containing fructose or sucrose. Infants with metabolic disorders often present with a history of vomiting, lethargy, and poor feeding. Vomiting may also be a symptom of intestinal obstruction including malrotation/volvulus.
The family history can often provide direction to the evaluation, particularly with some of the less common hereditary disorders. This can include most of the metabolic disorders, hemolytic diseases, and disorders associated with intrahepatic cholestasis ( Tables 18.2 and 18.3 ). Diseases that lead to severe liver dysfunction resulting in neonatal hepatic failure have some characteristic features ( Table 18.4 ).
| Physiologic Jaundice |
| Breast-Feeding/Breast Milk Jaundice |
| Polycythemia |
|
|
|
|
|
|
| Hemolysis |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Infection |
| Intestinal Obstruction |
|
|
|
|
| Enclosed Hematoma (Cephalohematoma, Ecchymoses) |
| Congestive Heart Failure |
| Hypoxia |
| Acidosis |
| Hypothyroidism or Hypopituitarism |
| Drugs/Toxins |
|
|
|
|
|
| Familial Disorders of Bilirubin Metabolism |
|
|
|
|
|
| GALD | HLH | Mitochondrial | Viral | Ischemic | |
|---|---|---|---|---|---|
| Transaminase levels (IU/L) | Normal/mild increase <100 | Moderate/significant increase (>1,000) | Moderate increase (100–500) | Significant increase (>1,000) | Significant increase (>1,000–6,000) |
| INR | Significant increase | Moderate/significant increase | Moderate/significant increase | Moderate/significant increase | Moderate/significant increase |
| Ferritin level (ng/mL) | 800–7,000 | Significant increase (>20,000) | Variable | Significant increase (>20,000) | Variable depending on underlying cause of ischemia |
| Triglyceride levels | Normal | Increased | Normal | Normal | Normal |
| Hypoglycemia | Yes | Often | Yes | Often | Variable |
| Lactic acidosis | Normal | Normal | Increased | Normal unless shock | Often |
| α-Fetoprotein level (for age) | Increased | Normal | Normal/increased | Normal | Normal |
| Cholestasis | Progressive after birth | Moderate/significant | Moderate | None/mild at presentation | Mild/moderate |
With increasing levels of bilirubin, neonatal icterus becomes more extensive, spreading in a cephalopedal direction. Pallor may indicate hemolytic disease. Petechiae alert the clinician to thrombocytopenia, possible sepsis, congenital infections, or severe hemolytic disease.
Dysmorphic face can be present in Zellweger syndrome or Alagille syndrome (see Table 18.1 ). The characteristic facies of Alagille syndrome may not be recognizable until later in childhood. Microcephaly that accompanies jaundice is associated with congenital viral infections.
An ophthalmologic examination can demonstrate a variety of abnormalities. Cataracts are seen in galactosemia and rubella. Chorioretinitis accompanies congenital infections (toxoplasmosis, syphilis, rubella, CMV, herpes simplex virus). Nystagmus with hypoplasia of the optic nerve suggests hypopituitarism associated with septo-optic dysplasia. Posterior embryotoxon is found in Alagille syndrome.
A heart murmur may be caused by an underlying congenital heart disease, which may be associated with Alagille syndrome, one of the trisomies, and syndromic forms of biliary atresia (polysplenia syndrome). Heart disease that results in hepatic ischemia or congestion can be a cause of conjugated or unconjugated hyperbilirubinemia.
Hepatomegaly, splenomegaly, and ascites may be caused by both hepatic and nonhepatic etiologies, but they always require evaluation as they are not associated with physiologic or breast milk jaundice.
Microphallus can be associated with septo-optic dysplasia and hypopituitarism.
When a neonate has jaundice, a thorough history, including the obstetric history, and physical examination should provide most of the information necessary to determine whether the condition represents physiologic jaundice (see Fig. 18.2 ). A total and fractionated bilirubin measurement should be performed if there is any question about the diagnosis of physiologic jaundice.
In neonates, increased bilirubin production is caused by the normally increased neonatal red blood cell mass and the decreased life span of the red blood cells (80 vs 120 days). Albumin binding is decreased because of lower albumin concentrations and diminished binding capacity, which results in decreased transport of UCB to the liver with increased deposition in tissues. Uptake of bilirubin by the hepatocytes during the first weeks of life is defective. Low levels of glutathione S-transferase B decrease intracellular binding, which may impede the transport of UCB to the endoplasmic reticulum. Conjugation is impaired by decreased activity of UDPGT. Secretion into the canaliculi is impaired. There is increased enterohepatic circulation of unconjugated bilirubin as a result of increased activity of β-glucuronidase in the intestinal lumen and as a result of intestinal bacterial flora with lower capacity for urobilinogen formation (see Fig. 18.1 ).
These features contribute in varying degrees to physiologic jaundice , characterized by a peak bilirubin level of <13 mg/dL on postnatal days 3–5, a decrease to normal by 2 weeks of age, and a conjugated fraction of <20%. In premature, breast-fed infants, infants of diabetic mothers, and Asian and Native American infants, the peak is higher and lasts longer. Conjugated bilirubin should be checked if there is any question of the nature of jaundice.
Breast-feeding has been associated with an increased incidence of unconjugated hyperbilirubinemia outside the expected range (>13 mg/dL). Jaundice of this level may occur in 10–25% of breast-fed infants, in contrast to 4–7% of formula-fed infants. It can occur within the first 5 days of life and is referred to as “early” or “breast-feeding” jaundice. Breast-feeding jaundice is seen in infants who are not feeding adequately and may be dehydrated or malnourished. In a second group of breast-fed infants, the jaundice develops slowly, occurring after the first week of life, and peaks between the second and third weeks of life at 10–20 mg/dL. This is referred to as “late” or “breast milk” jaundice. The precise cause of increased bilirubin levels in this latter setting has not been established; alternative theories include inhibition of glucuronosyltransferase activity and increased enterohepatic circulation of UCB. Kernicterus appears to be very rare but has been reported in association with breast-feeding. No treatment is necessary for physiologic jaundice. Practices that support breast-feeding, such as rooming-in on the maternity ward and frequent feedings, decrease the risk for breast-feeding jaundice. If the bilirubin exceeds 20 mg/dL in the breast-fed infant, discontinuing breast-feeding for 24 hours and supplementing with formula feeds results in a decreased bilirubin level. Phototherapy or rarely exchange transfusion may also be needed. Diagnosis of breast milk jaundice should include a blood test for conjugated bilirubin and can be diagnosed only with appropriate clinical history in the absence of conjugated hyperbilirubinemia.
If there are any red flags or uncertainty about the diagnosis ( Table 18.5 ), or if treatment is being considered, the hyperbilirubinemia should be investigated further. Any abnormality identified by history, physical examination, or laboratory findings is a matter of concern.
| Onset |
|
|
| Bilirubin |
| Conjugated |
|
| Total |
|
|
| Course |
|
|
| Prenatal History |
|
|
|
|
|
| Delivery |
|
|
|
| Feeding |
|
|
|
|
| Stools |
|
|
| Family History |
|
|
|
|
|
| Physical Examination |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The differential diagnosis of unconjugated hyperbilirubinemia in the neonate and infant is presented in Table 18.2 . Unless abnormalities in the history and physical examination direct the evaluation more specifically, hematologic evaluation, which may identify causes of increased bilirubin production, should be performed. This includes a CBC with examination of the smear, a reticulocyte count, a direct Coombs test, and blood typing (mother and infant).
Neonatal polycythemia, defined as a hematocrit >65% by venipuncture, can be caused by maternal diabetes, twin-twin transfusion, intrauterine hypoxemia, endocrine disorders, and delayed cord clamping (see Table 18.2 ). Polycythemia results in increased bilirubin production because of the increased red blood cell mass.
Reticulocytosis, unconjugated hyperbilirubinemia, and an increased nucleated red blood cell count, with either a low or normal hematocrit, suggest hemolysis. This can result from isoimmunization; erythrocyte membrane, hemoglobin, or enzyme defects; or sepsis with disseminated intravascular coagulation. Some causes of isoimmunization have low reticulocyte counts because the antibody binds to these precursor cells. Rarer causes of hemolysis include hemangiomas and congenital erythropoietic porphyria.
In this group of disorders, maternal antibodies (immunoglobulin G) to the infant’s erythrocytes cross the placenta, resulting in red blood cell destruction. The administration of anti-D gamma globulin (Rh O [D] immune globulin [RhoGAM]) after delivery to women who are Rh-negative has reduced the incidence of Rh sensitization and erythroblastosis fetalis. If a woman has been sensitized, the fetus can be monitored with serial amniocenteses. If necessary, intrauterine transfusion can then be performed to prevent the sequelae of severe hemolysis, which include fetal and neonatal anemia, edema, hepatosplenomegaly, and circulatory collapse or stillbirth of an infant with hydrops fetalis. If the problem has not been recognized prenatally, the infant with Rh incompatibility presents with pallor, hepatosplenomegaly, and rapidly developing jaundice.
The diagnosis is confirmed by demonstrating that the infant is Rh-positive, that the direct Coombs test result is positive, and that maternal antibody is coating the infant’s red blood cells. These test results are modified with in utero transfusions with Rh-negative cells. Depending on the degree of hemolysis, postnatal phototherapy and/or exchange transfusion may be required.
ABO blood type incompatibility causes a less severe form of isoimmune hemolytic disease with a less rapid development of jaundice. It is more common in infants with blood type A or B who are born to mothers with blood type O. Hemolysis develops in 50% of sensitized infants; of these infants, 50% have a bilirubin level >10 mg/dL. In addition to showing anemia, reticulocytosis, and spherocytes on the smear, the direct Coombs test result is weakly positive, and the indirect Coombs test result is positive. In rare cases, other minor blood group antibodies can also cause hemolysis.
Red blood cell membrane defects are relatively uncommon causes of unconjugated hyperbilirubinemia. There is often a family history of hemolysis, transfusions, cholecystectomy for bilirubin stones, or splenectomy. Hemolysis results from fragility of the red blood cell membrane. When the defect is present in infancy, there are anemia, jaundice, and splenomegaly, and the smear is often characteristic (e.g., spherocytosis or elliptocytosis). Spherocytes are also seen with ABO incompatibility. All membrane defects yield negative results of the Coombs tests.
Glucose-6-phosphate dehydrogenase (G6PD) deficiency is common. Jaundice is seen more frequently in persons with a Mediterranean or Far Eastern ancestry who have a complete absence of the enzyme. In these individuals, hemolysis can occur without a precipitant. In African-American patients, the disease is generally less severe, and hemolysis is rare without exposure to a drug, a toxin, or an infection that causes an oxidant stress. G6PD deficiency can manifest as neonatal jaundice on day 2 or 3 after birth; alternatively, it may not manifest until later in childhood, when jaundice is associated with an acute hemolytic crisis. The diagnosis of G6PD deficiency is confirmed by documenting deficiency of the enzyme in red blood cells.
Numerous deficiencies of enzymes in the glycolytic pathway have been identified. Pyruvate kinase deficiency is the most common of these rare disorders. Most of these disorders are thought to have an autosomal recessive mode of transmission and have been identified in only a small number of individuals. They all result in hemolysis. The time of manifestation depends on the degree of hemolysis.
If the hematocrit is normal and there is no evidence of hemolysis or a consumptive process, other explanations for unconjugated hyperbilirubinemia should be sought. Blood and urine cultures rarely identify infectious etiologic agents if the patient is otherwise clinically normal. Vomiting, abdominal distention, and delayed passage of meconium suggest obstruction of the gastrointestinal tract and should be further investigated by imaging. Clinical examination should also identify cephalohematoma, ecchymoses, heart failure, hypoxia, and acidosis. Thyroxine and thyroid-stimulating hormone levels should be obtained or checked from the state neonatal screening program to look for evidence of hypothyroidism or hypopituitarism.
Drugs, administered to either mother or neonate, and toxins should be identified by careful record review. Examples include oxytocin, excess vitamin K in premature infants, some antibiotics, and phenol disinfectants used in nurseries. Use of herbal remedies should also be investigated.
In the evaluation process, it is important to remember that the division of causes into hemolytic and nonhemolytic is an arbitrary one. Drugs, infection, and G6PD deficiency can contribute to both hemolytic and nonhemolytic neonatal jaundice. In addition, the cause of jaundice can be multifactorial.
Gilbert syndrome is a benign condition that occurs in up to 8% of the population. A familial incidence is reported in 15–40% of cases. Gilbert syndrome is a heterogeneous group of disorders that have in common at least a 50% decrease in UDPGT activity as a result of a defect in the gene responsible for this enzyme. In 20–30% of individuals with Gilbert syndrome, there is also a decrease in hepatocyte bilirubin uptake. Affected individuals are generally asymptomatic and may not present with jaundice until the second or third decade of life. Gilbert syndrome may be responsible for some cases of neonatal jaundice. Mild jaundice with a bilirubin level up to 7 mg/dL can occur transiently with fatigue, exercise, fasting, febrile illness, and alcohol ingestion in older patients. Except for showing an increased indirect bilirubin level, all laboratory studies are normal. The diagnosis is generally a clinical one but can be confirmed by documenting a twofold to threefold rise in unconjugated bilirubin during a 24-hour fast.
Crigler-Najjar syndrome types I and II (also known as Arias syndrome) are rare autosomal recessive conditions caused by variants (different alleles from those of Gilbert syndrome) in the gene coding for UDPGT. Crigler-Najjar syndrome type I is characterized by marked hyperbilirubinemia (20–40 mg/dL) in the neonatal period in an otherwise healthy infant. Among untreated infants, kernicterus is universal, and affected individuals usually die with severe neurologic problems. Because UDPGT is undetectable, there is no conjugated bilirubin in the bile or serum, and the bile is colorless. There is no decrease in serum UCB levels during phenobarbital administration. The only therapies are exchange transfusion, intensive phototherapy, and liver transplantation.
The onset of Crigler-Najjar syndrome type II is usually at birth, although it can be in late childhood. There is <5% of the normal UDPGT activity. Bile contains bilirubin monoglucuronides. Bilirubin levels, generally 8–25 mg/dL, respond to phenobarbital administration with a significant decrease. Neurologic disease is rare.
Lucey-Driscoll syndrome is a transient familial neonatal hyperbilirubinemia that appears in the first few days of life and resolves by 2–3 weeks of age. This results from inhibition of UDPGT by a substance that has been found in both maternal and infant serum. The bilirubin level can rise to >60 mg/dL in untreated infants, resulting in severe neurotoxicity. The condition is treated with exchange transfusion.
Treatment of unconjugated hyperbilirubinemia depends on the degree of elevation of bilirubin. Considerable controversy exists over which level is toxic and when treatment should be initiated. Because it is lipid soluble, unconjugated bilirubin can diffuse into the central nervous system, which results in neurologic toxicity. Most authorities agree that kernicterus does not occur below a bilirubin level of 20–30 mg/dL in the healthy, full-term infant without evidence of hemolysis. Kernicterus may occur at lower bilirubin levels in premature or sick neonates.
Treatment options include phototherapy and exchange transfusion. Phototherapy produces a reduction of bilirubin by 1–2 mg/dL in 4–6 hours by causing the photoisomerization and photodegradation of unconjugated bilirubin to more water-soluble forms that are more readily excreted in bile and urine. Potential complications include retinal damage, diarrhea, and dehydration. Phototherapy is begun at levels below that for exchange transfusion (∼5 mg/dL less) or during preparations for an exchange transfusion.
Exchange transfusion with blood cross-matched against that of the mother is indicated for severe hyperbilirubinemia. This decision must be based not only on the bilirubin level but also, as important, on the infant’s age and clinical condition. In full- or near-term infants (>2,000 g in weight) with evidence of hemolysis, exchange transfusion is indicated if the serum unconjugated bilirubin level is higher than 25–30 mg/dL or if the bilirubin level does not rapidly respond to phototherapy. Signs of kernicterus (i.e., a high-pitched cry, gaze paralysis, fever, lethargy, and opisthotonic posture) warrant exchange transfusion, no matter what the bilirubin level.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here