Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
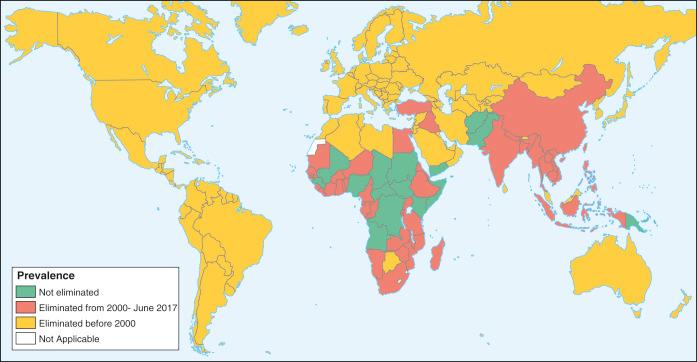
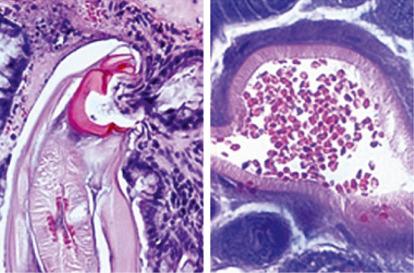
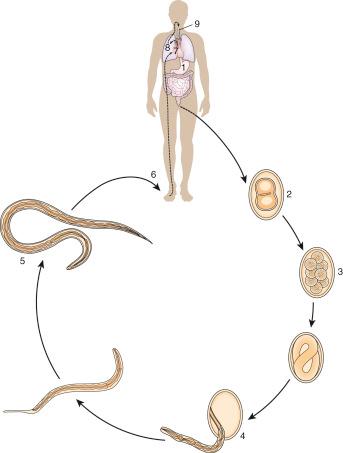
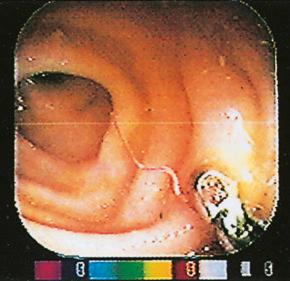
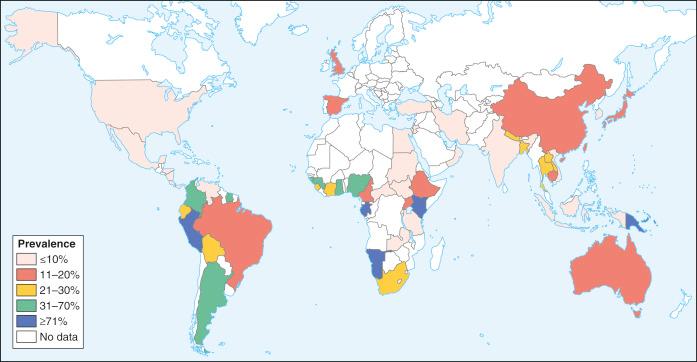
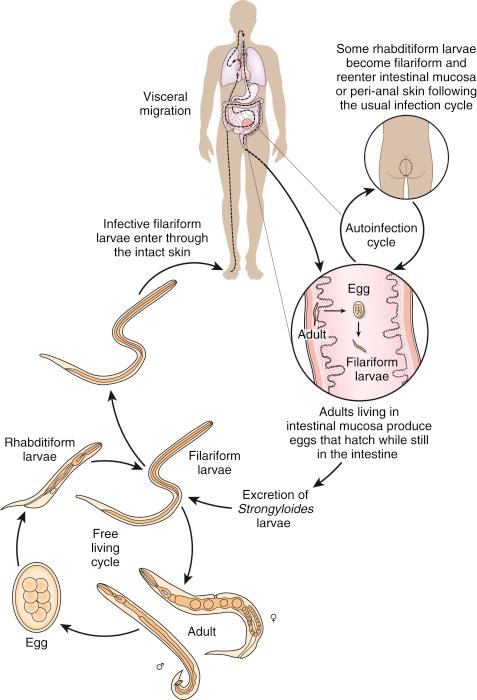
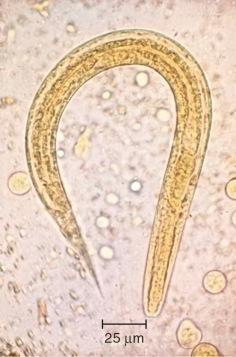
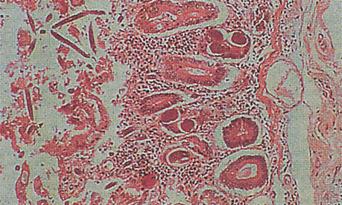
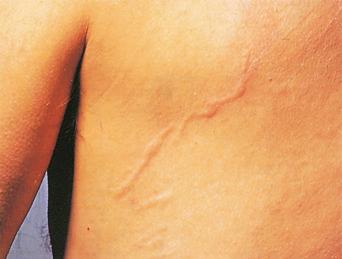
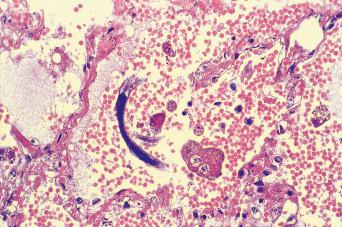
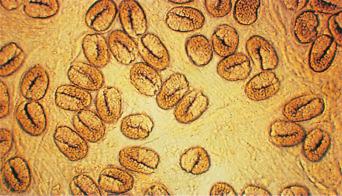
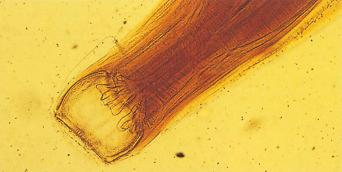
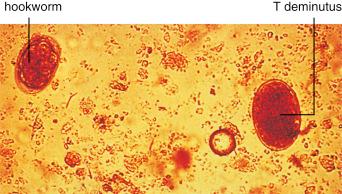
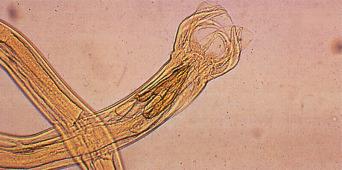
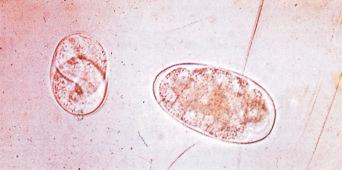
There are estimated to be billions of microorganisms in a handful of soil, so it might be considered surprising that it does not serve as a source for more pathogens. Indeed, given that soil cannot be decontaminated globally, and thus soil-acquired infections like tetanus can never be eradicated, this is fortunate. ‘Soil-transmitted helminths’ (STHs) is the collective term (interchangeable with ‘geohelminths’) applied principally to three intestinal nematode infections for which egg-contaminated soil plays an important lifecycle role: roundworm ( Ascaris lumbricoides ), whipworm ( Trichuris trichiura ) and hookworms ( Ancylostoma duodenale and Necator americanus ). However, the term can be confusing, as the former two are acquired through ingestion of food or water contaminated by helminth eggs from the soil (and therefore described in Chapter 3 ), whereas hookworm larvae enter the human body through the skin by direct contact. Though Strongyloides stercoralis is acquired in a similar fashion to hookworm and is thus strictly speaking an STH, it is rarely considered as part of this group because of lower prevalence and lower associated morbidity. It is also not included in mass drug administration programmes for STH control.
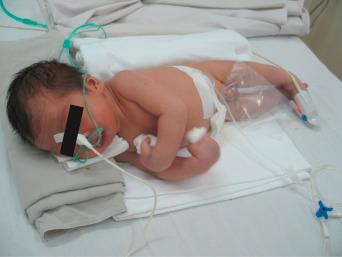
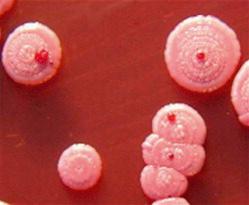
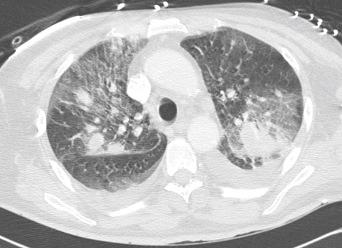
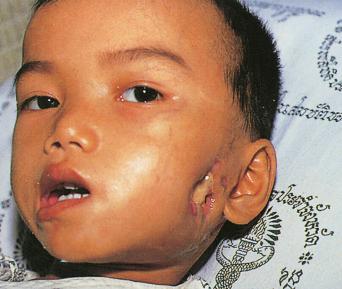
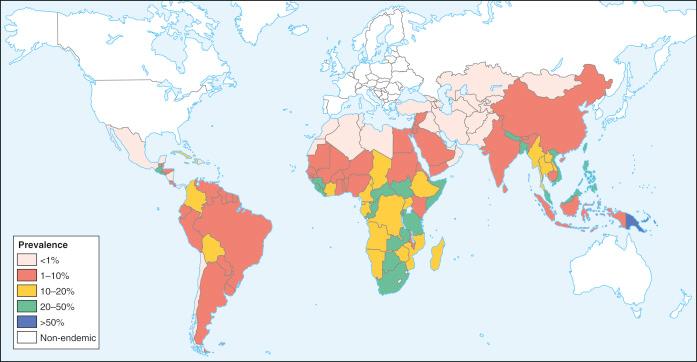

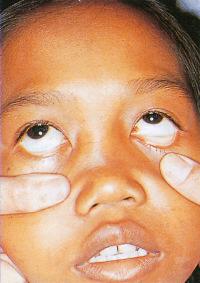
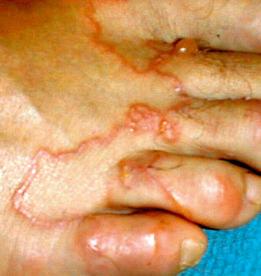
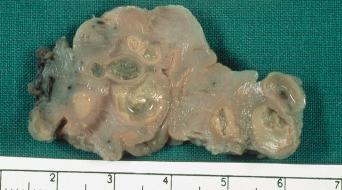
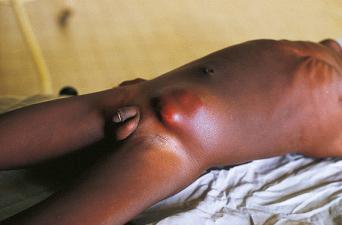
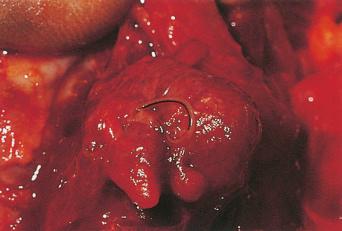
Humans have a daily need for water for multiple purposes. In addition to the danger posed by ingestion of unsafe water ( Chapter 3 ), cutaneous and mucocutaneous contact with contaminated water can also expose individuals to a significant number of infectious pathogens.
| Category | Agent | Clinical syndrome | Distribution | Atlas reference |
|---|---|---|---|---|
| Viral infections | CMV | Retinitis, colitis, oesophagitis | Global | 2.103 , 2.104 |
| HSV | Mucocutaneous ulceration of increased severity | Global | 2.72 | |
| VZV | Increased risk of varicella zoster reactivation (‘shingles’) and dissemination | Global | 4.16 , 4.17 | |
| JC virus | Progressive multifocal leucoencephalopathy | Global | 3.12 | |
| Molluscum contagiosum virus | Molluscum contagiosum (more extensive and persistent in HIV) | Global | 2.105 | |
| EBV | Oral hairy leukoplakia, lymphoma | Global | 2.102 | |
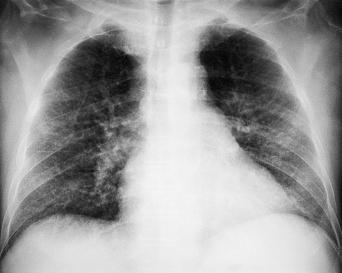
| Bacterial infections | Mycobacterium tuberculosis | Pulmonary and extrapulmonary TB | Global | 4.34 - 4.70 |
| Mycobacterium avium intracellulare | Fever, reticulo-endothelial system involvement | Global | - | |
| Streptococcus pneumoniae | Pneumonia and sepsis | Global | 4.18 - 4.20 | |
| Non-typhoidal salmonellae | Sepsis syndrome | Global | 1.137 | |
| Mycobacterium leprae | Modified disease manifestations | Leprosy-endemic regions | 4.71 - 4.93 | |
| Bartonella henselae | Bacillary angiomatosis | Global | 2.135 | |
| Fungal infections | Cryptococcus sp. | Meningitis, sepsis | Global | 4.111 - 4.113 |
| Pneumocystis jirovecii | Pneumonia | Global | 4.94 , 4.95 | |
| Candida sp. | Oral and oesophageal candidiasis | Global | 2.160 , 2.161 | |
| Talaromyces (formerly Penicillium ) marneffei | Sepsis syndrome, rash | South-east Asia | 4.97 - 4.99 | |
| Histoplasma capsulatum | Rash, pneumonia, bone-marrow involvement | Global | 4.100 - 4.102 | |
| Paracoccidioidomycosis | Cutaneous, mucosal, pulmonary, reticulo-endothelial system involvement | Central and South America | 2.175 , 4.103 - 4.107 | |
| Protozoal infections | Plasmodium sp. | Malaria | Malaria endemic regions | 1.102 - 1.166 |
| Toxoplasma gondii | Cerebral abscess | Global | 3.53 , 3.57 | |
| Trypanosoma cruzi | Meningoencephalitis, myocarditis | Central and South America | 1.197 - 1.220 | |
| Leishmania sp. | Visceral leishmaniasis 1 | Leishmania endemic regions | 1.221 - 1.282 | |
| Cryptosporidium sp. | Diarrhoea | Global | 3.38 , 3.52 | |
| Isospora sp. | Diarrhoea | Global | 3.38 , 3.51 | |
| Microsporidia ( Encephalitozoon hellem, Enterocytozoon bineusi ) | Diarrhoea | Global | 3.59 | |
| Malignancies | Kaposi sarcoma | Cutaneous, pulmonary and gastrointestinal involvement | Global – aetiological agent is KSHV (HHV-8) | 8.5 |
| Non-Hodgkins lymphoma | Visceral and lymph node lymphoma | Global – associated with EBV infection | - | |
| Primary cerebral lymphoma | Cerebral space-occupying lesion | Global – associated with EBV infection | - | |
| Cervical carcinoma | Cervix mass lesion | Global – associated with HPV infection | 2.76 | |
| Anal squamous cell carcinoma | Anal mass lesion | Global – associated with HPV infection | - | |
| Multicentric Castleman’s disease | Lymph node enlargement | Global – associated with HHV-8 infection | - |
1 Lack of evidence of increased risk of cutaneous leishmaniasis in HIV.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here