Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Over the past century, clinicians have been challenged by the more frequent identification of primary pancreatic cystic neoplasms (PCNs). Increased use of cross-sectional imaging studies, such as computed tomography and magnetic resonance imaging (MRI), have led to a sustained increased “incidence” of these lesions. Management decisions are extremely difficult due to the uncertain biologic behavior of these lesions and the lack of prognostic biomarkers to assist with counseling patients on treatment. Reports of cystic neoplasms in the pancreas are documented in the literature as early as 1908. However, the distinction between serous and mucinous cysts was not made until 1978 by Compagno and Oertel. This proved to be an important differentiation due to the risk of malignant transformation associated with mucinous cysts. Only a few years later, in 1982 Ohashi et al. published the first report of a mucinous secreting cancer of the pancreas, which was later redefined as an intraductal papillary mucinous neoplasm (IPMN) in 1996. Despite histopathologic advancements in diagnosis, PCNs remain a diagnostic dilemma for even the most experienced clinician to accurately predict the malignant potential of these lesions. This chapter will focus on the classifications and characteristics of PCNs, the diagnostic modalities, the therapies, and future ways of treating this disease.
Most pancreatic cysts are nonneoplastic, originating from injury, inflammation, or congenital abnormalities of the pancreas. A much smaller percentage of cysts are neoplastic. In spite of previously being defined as a rare entity, PCNs have increased in diagnostic prevalence due to increased use of cross-sectional imaging. Based on a retrospective review in 2010, the overall prevalence of incidental pancreatic cysts discovered by MRI is 13.5% and increases with age. An autopsy study identified PCNs in 378 of 1374 cases (27.5%) with an increased prevalence correlated with age. Thus, as imaging techniques continue to improve, more incidental PCNs will inevitably become discovered.
The three most common types of PCNs are serous cystic neoplasms (SCNs), mucinous cystic neoplasms (MCNs), and IPMNs, representing approximately 90% of all PCNs. MCNs and IPMNs are the most common and more importantly have the highest potential for malignant transformation. SCNs occur much less frequently and are almost always benign.
SCNs are most commonly observed in women (3 : 1 female-to-male ratio), with an average age of diagnosis of 62 years. There are four subtypes of SCNs: serous microcytic adenoma, serous macrocytic (oligocystic) adenoma, von Hippel-Lindau (VHL)-associated pancreatic cysts, and serous cystadenocarcinoma.
SCNs are characterized by serous fluid–filled cysts lined by a single layer of cuboidal epithelial cells with uniform, round, darkly stained nuclei and a glycogen-rich cytoplasm. Notably, there is a lack of atypia, necrosis, and mitotic features in SCNs. On gross examination, microcystic SCNs have a characteristic honeycomb appearance with multiple thin-walled cysts around a central scar ( Fig. 98.1 ). Commonly, calcifications can be found in the proximity of the central scar. The cystic spaces do not communicate with the pancreatic ductal system. The surrounding stroma usually contains nerves, islets, lymphoid aggregates, and vascular channels.
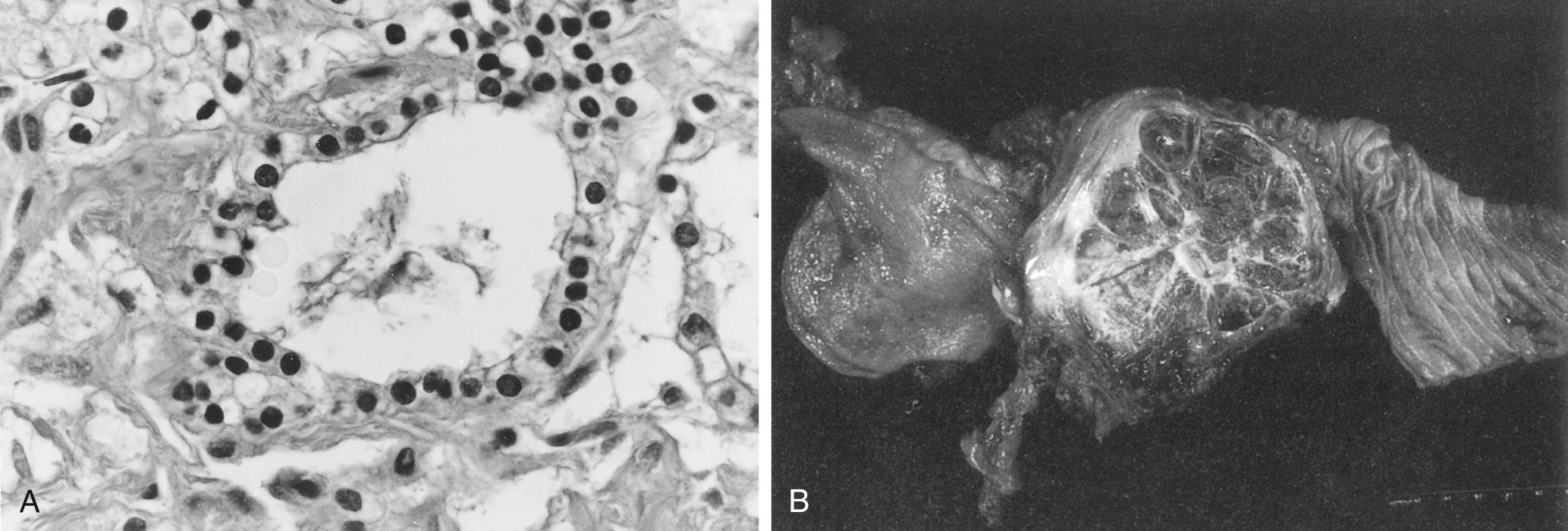
Serous macrocystic or oligocystic adenomas are rare and usually have larger and fewer cystic structures. The oligocystic variant does not have the central scar that is seen with the microcystic subtype. These cysts are found equally in men and women and usually occur earlier in life, with an average age of 50 years. They are predominately found in the pancreatic head, which can lead to symptoms related to biliary duct obstruction. Microscopically the cellular architecture of macrocystic and microcystic serous adenomas are similar.
VHL disease is an autosomal dominant mutation of chromosome 3p25.3 causing multiorgan pathology. VHL-associated pancreatic cysts show a loss of heterozygosity on chromosome 3p25 and mutations in the VHL gene. A 2012 systemic review of the literature by Charlesworth identified 252 out of 420 (60%) patients with VHL and a pancreatic lesion. These lesions are indistinguishable from sporadic serous cystadenomas. In contrast, VHL patients tend to have multifocal disease rather than a single lesion. These lesions behave similarly to non-VHL serous cystadenomas with minimal malignant potential.
Malignant SCNs are exceedingly rare, with a few case reports in the literature. George et al. first described a patient with a serous cystadenocarcinoma in 1989, and since that time, fewer than 40 cases have been published. These malignant cysts are nearly identical to benign serous cystadenomas and are distinguished only by the presence of metastases. Literature suggests the possibility that serous cystadenocarcinomas are misdiagnosed as a malignancy due to the presence of vascular impingement on imaging. Histologic review of these reported serous cystadenocarcinoma specimens failed to demonstrate any malignant attributes.
MCNs are cystic tumors of the pancreas that have a lower incidence than serous cysts or IPMNs. The average age range of presentation is 40 to 50 years old, and they are found predominantly in perimenopausal women (20 : 1 female-to-male ratio).
The vast majority of MCNs are found in the body and tail of the pancreas. Most MCNs are approximately 6 to 10 cm at the time of diagnosis but range from 1.5 to 35 cm in diameter. On gross examination, they are spherical and infrequently encapsulated by a calcified fibrous wall (approximately 15%). Almost all MCNs are multiloculated. The cyst can be filled with mucin, blood, or a watery fluid. This fluid tends to be thicker and more viscous than in SCN, due to the presence of mucus. MCNs do not communicate with the pancreatic ductal system except when there is erosion into the duct or the formation of a fistula. Any evidence of solid mural nodules should be thoroughly investigated to rule out stromal invasion.
MCNs are lined by tall mucin-producing columnar cells ( Fig. 98.2 ). These epithelial cells that line the cyst may be papillary or flat and can show a tendency toward gastric or intestinal differentiation. The stroma resembles an ovarian corpora albicantia due to luteinized cells and foci of hyalinization. The ovarian-like stroma that underlies that epithelium is pathognomonic of MCN and thus is required for diagnosis. In difficult to diagnose cases the presence of the ovarian-like stroma can help to differentiate between MCN and IPMN. Areas of denudation in the epithelial lining can occasionally be seen.
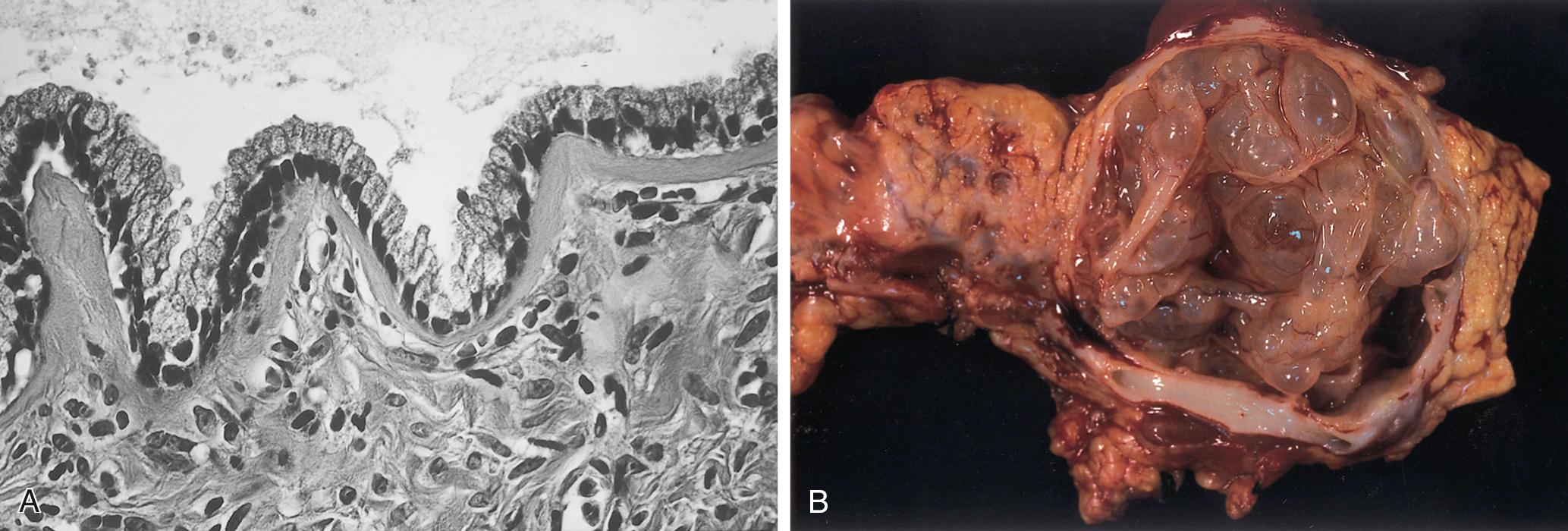
The epithelium of MCN demonstrates immunoreactivity for cytokeratins 7, 8, 18, and 19, epithelial membrane antigen, carcinoembryonic antigen (CEA), MUC5AC, and carbohydrate antigen (CA) 19-9. The stroma cells stain for estrogen (25% to 63%), progesterone (50% to 80%), and alpha-inhibin (50% to 70%).
In 2010 the World Health Organization (WHO) classification divided noninvasive MCNs into three distinct categories based on the degree of cellular atypia: low-, intermediate-, and high-grade. The classification is based on the greatest degree, and not the average, of the epithelial dysplasia ( Fig. 98.3 ). Less than 20% of MCNs are associated with invasive carcinoma and thus should be considered a potential precursor to pancreatic cancer. In these cases the invasive component is histologically consistent with the classic pancreatic ductal adenocarcinoma, although colloid carcinoma, undifferentiated carcinoma, osteoclast-like giant cells, adenosquamous carcinoma, and sarcomas may infrequently occur. Extensive sampling of the cyst is recommended, given the relatively small volume of the invasive component. It appears that the incidence of malignant transformation is directly correlated to the overall size of the cyst and the complexity of the cyst.
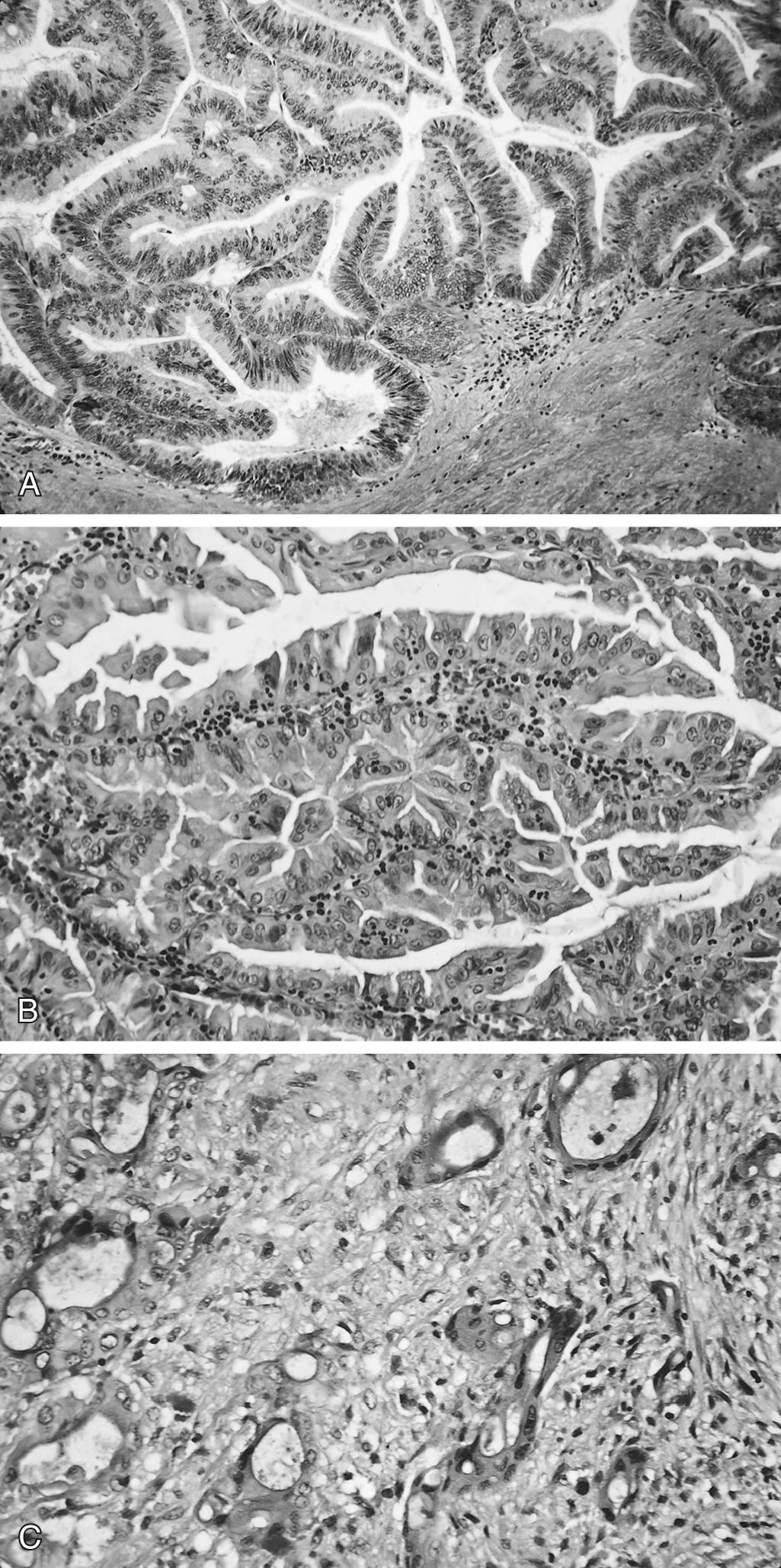
The majority of patients with an MCN present with nonspecific symptoms, including weight loss, nausea, vomiting, diarrhea, and abdominal pain. Approximately 30% of patients with an MCN have a palpable abdominal mass.
IPMNs were first recognized in 1982 by Ohashi, but the term IPMN was not officially used until 1993. IPMNs are defined in the WHO Classification of Tumors of the Digestive System as an intraductal, grossly visible epithelial neoplasm of mucin-producing cells. Using imaging and histology, IPMNs can be classified into three types based on duct involvement :
Main-duct IPMN (approximately 25% of IPMNs): Segmental or diffuse dilation of the main pancreatic duct (>5 mm) in the absence of other causes of ductal obstruction.
Branch-duct IPMN (approximately 57% of IPMNs): Pancreatic cysts (>5 mm) that communicate with the main pancreatic duct.
Mixed type IPMN (approximately 18% of IPMNs): Meets criteria for both main and branch duct.
Due to the asymptomatic nature of the disease, the overall incidence of IPMNs is difficult to define but is thought to account for approximately 3% to 5% of all pancreatic tumors. Most IPMNs are discovered as incidental lesions from the workup of an unrelated process by imaging or endoscopy. IPMNs are slightly more prevalent in males than in females, with a peak incidence of 60 to 70 years of age. Branch-duct IPMNs tend to occur in a slightly younger population and are less associated with malignancy compared with main-duct or mixed variants.
In the main-duct variant of IPMN the main pancreatic duct is usually dilated (≥5 mm), mucin filled, and tortuous. The ampulla of Vater on endoscopy tends to have a characteristic “fish-mouth” appearance. These tumors are generally found near the head of the pancreas and grow along the main duct. In patients, when the major or minor papilla is involved, evidence of mucin in the duodenum can be visualized when performing endoscopy. Adjacent pancreas can be fibrotic and firm due to chronic pancreatitis. Branch-duct IPMNs are most frequently found in the uncinate process. On gross examination, branch-duct IPMNs are grapelike structures that are multicystic, containing mucin-filled ducts. The adjacent pancreas is usually normal due to noninvolvement of the main pancreatic duct.
Studies have categorized IPMNs based on their epithelial lining of the papillary component ( Fig. 98.4 ). These include gastric, intestinal, pancreatobiliary, and oncocytic. Furthermore, IPMNs contain highly glycosylated proteins called mucins (MUC) that have different gene expression patterns and can help to distinguish between the histopathologic IPMN subtypes. The gastric-subtype IPMNs contain basal small nuclei with apical cytoplasmic mucin, similar in appearance to gastric epithelium. These lesions are almost always low-grade and express MUC5AC and MUC6. The majority of branch-duct IPMNs are of the gastric variant. The intestinal-type IPMNs contain fingerlike projections lined by mucin-containing cells with large cigar-like nuclei. These tumors, which express MUCH5AC, MUC2, and CDX2, are strongly associated with colloid carcinomas. A large proportion of main-duct IPMNs are of the intestinal type. Pancreatobiliary-type IPMNs contain cuboidal cells with atypical nuclei and prominent nucleoli. These lesions tend to be a higher grade and are associated with invasive carcinomas that tend to be tubular and aggressive. Their expression pattern includes MUC1 and MUC5A. Finally, the oncocytic subtype contains cells with abundant eosinophilic cytoplasm secondary to large amounts of intracellular mitochondria. This type tends to be complex with arborizing papillae, cribriform formations, and solid nests growing into a dilated duct. Most of these lesions are classified as high grade with expression of MUC1 and MUC6.
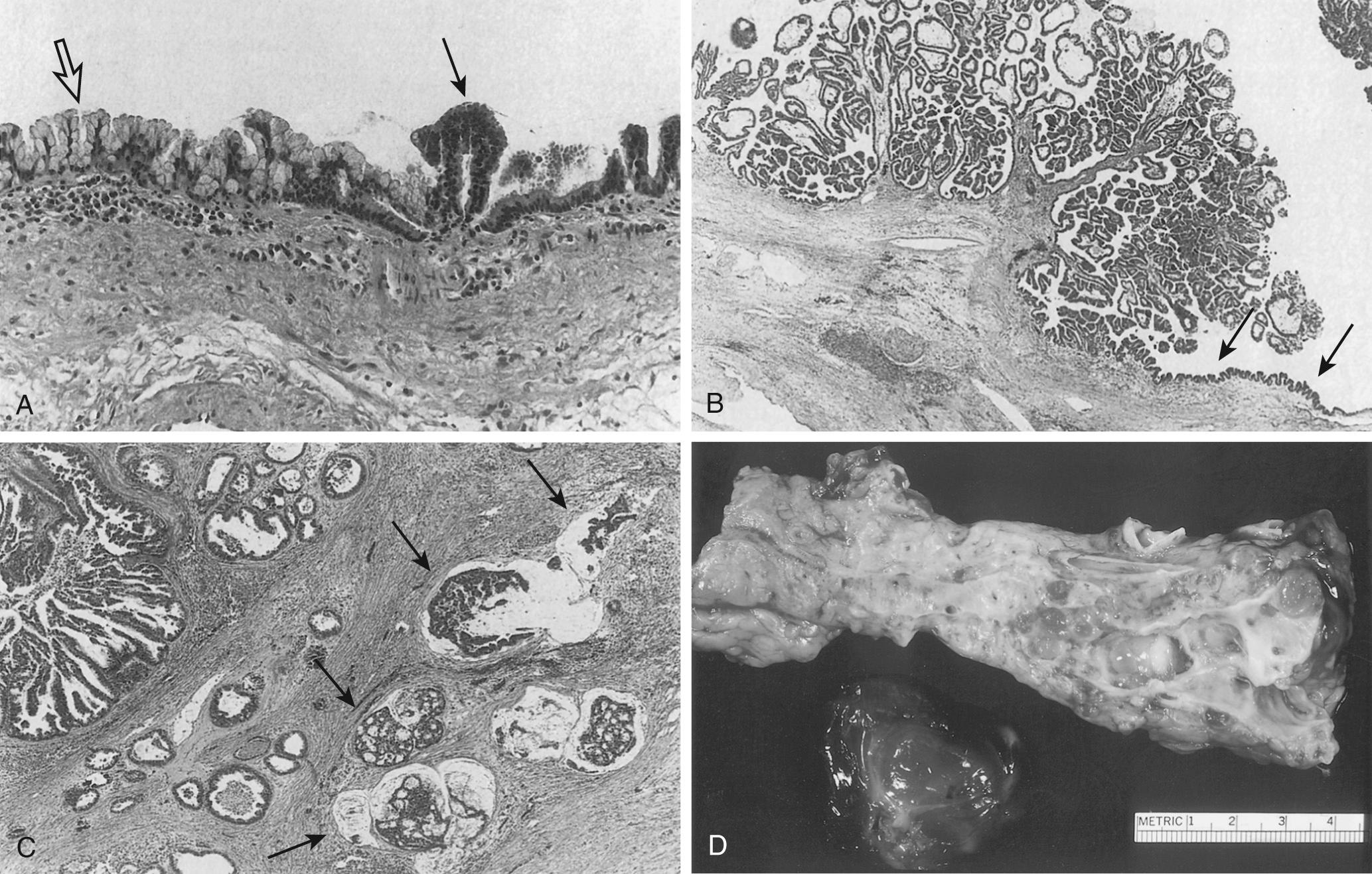
Because a majority of IPMNs are discovered incidentally, most are asymptomatic. When symptoms do occur, they tend to be nonspecific and include unexplained weight loss, anorexia, abdominal pain, and back pain. Jaundice can occur with mucin obstructing the ampulla or with an underlying invasive carcinoma. The obstruction of the pancreatic duct can also lead to pancreatitis.
IPMNs may represent genomic instability of the entire pancreas. This concept, known as a “field defect,” has been described as a theoretical risk of developing a recurrent IPMN or pancreatic adenocarcinoma at a site remote from the original IPMN.
See Table 98.1 for cystic variants of other pancreatic neoplasms.
| Cystic endocrine tumors | Cyst usually contains clear fluid lined by neoplastic endocrine cells Radiographically tumor appears solid with small cystic changes |
| Cystic acinar cell tumors | Includes:
|
| Other pancreatic neoplasms with cystic changes | Includes: ductal adenocarcinoma, adenosquamous carcinoma, undifferentiated carcinoma with osteoclast-like giant cells, and squamous cell carcinoma |
Most primary pancreatic cysts are discovered incidentally by imaging during the evaluation of an unrelated process. Approximately 47% of patients with SCNs are asymptomatic, with the cyst identified only incidentally. The nonspecific symptoms associated with SCNs include abdominal pain (25%), palpable mass (10%), and jaundice (7%). The size of the cyst plays a large role in the manifestation of symptoms with cysts greater than 4 cm compared with less than 4 cm (72% vs. 22%). MCNs and IPMNs are generally more symptomatic (approximately 75%) than SCNs due to their larger size. These lesions are more likely to cause acute pancreatitis due to mass effect on the pancreatic duct (9.2%). IPMNs can also cause acute pancreatitis from mucin production leading to duct obstruction. Other symptoms include abdominal pain (60%), fatigue (10%), and palpable mass (12%). Smaller-size branch-duct IPMNs are more likely to be asymptomatic and diagnosed incidentally.
Each of the PCNs has varying degrees of malignant potential and requires an accurate diagnosis to ensure adequate therapeutic decision making. Clinical presentation is variable due to nonspecific or lack of symptoms in patients. To aid in the diagnosis of PCNs, physicians rely on different imaging techniques. The three modalities that will be discussed in this section include multidetector-row computed tomography (MDCT), MRI, and positron emission tomography–computed tomography (PET–CT).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here