Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The neuromuscular system may be affected at various levels, each of which is characterized by changes in motor function peculiar to the site and extent of involvement. The differential features of various levels of motor function are illustrated in Table 31.1 .
| Type of Disturbance | Spinomuscular | Extrapyramidal | Pyramidal | Cerebellar | ||
|---|---|---|---|---|---|---|
| Muscular | Neural | Spinal | ||||
| Loss of motor power | Focal-segmental Usually proximal and axial muscle groups Complete |
Focal-segmental Usually distal limb musculature Complete |
Focal-segmental Usually distal limb musculature Complete |
Generalized Entire limb and movements Incomplete |
Generalized Entire limb and movements Incomplete |
None Ataxia may simulate loss of power |
| Tone | Flaccid | Flaccid | Flaccid | Rigid | Spastic | Hypotonic (ataxia) |
| Atrophy | Present | Present | Present | Absent | Minimal (caused by disuse and chronic paresis) | Absent |
| Fasciculations | May be present | Absent | May be present | Absent | Absent | Absent |
| Reaction of degeneration | Present | Present | Present | Absent | Absent | Absent |
| Reflexes | ||||||
| Deep | Diminished and preserved until late | Absent early | Absent early | Normal or variable | Hyperactive | Diminished or pendular |
| Superficial | Diminished | Absent | Absent | Normal or increased | Diminished or absent | Normal |
| Sensory deficit | Absent | Usually present | Absent | Absent | May be present | Absent |
| Trophic disturbance | Present | Present | Present | Absent | Usually absent | Absent |
| Ataxia | Absent | Absent | Absent | Absent | Absent | Present |
| Abnormal movements | Absent | Absent | Absent | Present | Absent | May be present (intention tremor and ataxia) |
At the spinomuscular level, motor activity is simple; impulses arising in the anterior horn cells of the spinal cord are transmitted through peripheral nerves to myoneural junctions and then to individual muscles. In disorders at the spinomuscular level, the loss of motor power is focal and segmental, with complete paralysis of the muscles or muscle groups that are supplied by a peripheral nerve or by the anterior horn cells in the spinal cord. Muscular paralysis is flaccid or hypotonic, and degeneration, atrophy, fibrillations, and fasciculations are typical findings. The deep tendon and superficial reflexes are diminished or absent. Pyramidal tract signs, abnormal involuntary movements, and ataxia are absent. Trophic changes may be seen in the skin, nails, and bone.
Pathologic processes at the spinomuscular level may be further classified into various sublevels. When the disease originates in the anterior horn cells, as in poliomyelitis, the spinal level of the motor system is affected. Other examples of diseases at the spinal level are progressive spinal muscular atrophy of the Werdnig-Hoffmann type, progressive bulbar palsy, syringomyelia, and intramedullary neoplasm. Loss of function of the anterior horn cells or the motor nuclei of the brainstem results in clinical findings of flaccid paralysis, atrophy, areflexia, degeneration, and fasciculations.
At the neural level of the motor system, the peripheral nerves and nerve roots are affected, as in obstetric brachial plexus palsy and progressive neuromuscular atrophy (Charcot-Marie-Tooth disease). In processes affecting nerves, the sensory fibers are usually involved, with resultant sensory changes such as anesthesia or hyperesthesia. Otherwise, the clinical findings are similar to those of involvement of the spinal level; that is, flaccid paralysis, atrophy, degeneration, and areflexia develop as a result of loss of conduction of motor impulses. In the absence of sensory changes, it is difficult to distinguish between diseases of the peripheral nerves, anterior roots, and anterior horn cells.
If the pathologic process arises at the myoneural junction, as in myasthenia gravis and familial periodic paralysis, it is a disease at the myoneural level. In diseases of primarily muscular origin, the motor system is involved at the muscular level. The muscular dystrophies are familiar examples of disturbance at the muscular level in diseases with spinomuscular involvement. The paralysis is flaccid, but reflexes persist until the late stages, when marked atrophy has already occurred. Contractility is lost but not excitability; that is, the muscle fibers have degenerated and have been replaced by fibroadipose tissue, but the peripheral nerves and anterior horn cells are normal.
Disorders of the motor system at the extrapyramidal level are characterized by generalized involvement of the muscles of the limbs and trunk. Muscle tone is hypertonic. Atrophy, fasciculations, and degeneration are absent. Motion of the limbs is hyperkinetic, with loss of associated or automatic movements. The deep tendon and superficial reflexes are normal. No pyramidal tract responses or sensory deficits are present. Athetoid cerebral palsy (CP) is a common example of a disease at the extrapyramidal level.
At the pyramidal or corticospinal level, motor deficit arises from involvement of motor nuclei of the cerebral cortex. The paresis is usually generalized and associated with hypertonicity or spasticity of muscles. Pyramidal tract signs and pathologic reflexes are generally present. Usually some atrophy that is not focal is present; it is caused by chronic paralysis and disuse. Fasciculations, trophic disturbances, degeneration, and abnormal movements are absent. The deep tendon reflexes are hyperactive, and the superficial reflexes are diminished or absent. Spastic CP illustrates the pyramidal level of motor involvement.
Lesions at the cerebellar level are characterized by loss of coordination and control, or ataxia. No real loss of motor power occurs. Fasciculations, degeneration, atrophy, and trophic disturbances are absent. The deep tendon reflexes may be diminished, but the superficial reflexes are normal.
![]() For References, see expertconsult.com .
For References, see expertconsult.com .
CP was first described by William Little in 1862. Little correlated the findings seen in young children with CP and associated them with difficult births. The term cerebral palsy originated with Freud. Static encephalopathy has been used interchangeably with cerebral palsy.
A succinct and accurate definition of CP is difficult to construct because of wide variability in the manifestations of CP. In 2008 CP was proposed as “a group of permanent disorders of the development of movement and posture, causing activity limitation, that are attributed to nonprogressive disturbances that occurred in the developing fetal or infant brain.” In all cases, the following must be true:
CP is the result of a brain lesion; therefore, the spinal cord and muscles are structurally and biochemically normal.
The brain lesion must be fixed and nonprogressive. Thus, all the progressive neurodegenerative disorders are excluded from the definition.
The abnormality of the brain results in motor impairment.
The insult to the brain may occur prenatally, perinatally, or during childhood. Although older children with brain damage were traditionally excluded from the definition, this is not clinically relevant from an orthopaedic standpoint. Certainly, any orthopaedist caring for a child who has sustained an anoxic injury after nearly drowning or who is spastic after infectious meningitis would argue that these slightly older children functionally have CP.
The clinical manifestations of CP depend on which part and how much of the brain are involved. The range of manifestations is huge, with both a young child who is intellectually bright but walks on his toes and a noncommunicative wheelchair-bound child with seizures meeting the aforementioned definition of CP.
The orthopaedic surgeon is consulted by a pediatrician or family for management of the musculoskeletal problems that follow from the underlying brain lesion. It is of utmost importance for the orthopaedist to evaluate the child thoroughly to ascertain why the child has CP. If the child was born full-term, if no perinatal medical problems were noted, and especially if the child began to develop normally and then regressed, prompt neurologic consultation must be sought. The neurologist will differentiate CP from such dangerous entities as brain and spinal cord tumors, metabolic encephalopathies, and progressive neurodegenerative diseases, some of which are treatable.
The incidence of CP is increasing slightly. In recent reports the incidence has been estimated to be between 2.4 and 2.7 per 1000 live births. , , The prevalence of CP appears to be increasing secondary to an increase in the number of infants with very low birth weight being born and the increased survival of these tiny neonates, whereas the rate of CP in infants of a given birth weight has remained stable. , This increase in incidence is of concern because the economic impact of CP is considerable, with the cost per case estimated at $503,000 in 1992. The risk for CP in a child born full-term is approximately 1 in 1000. The incidence of CP has been correlated with both gestational age and birth weight. Approximately 50% of children with CP have low birth weight, and 28% weigh less than 1500 g at birth. ,
The incidence of CP is increased with multiple births. In more recent studies of multiple births the incidence of CP is 9 to 12 per 1000 in twins, 31 to 45 per 1000 in triplets, and 111 per 1000 in quadruplets. , , The predisposition to CP in twin pregnancies is present even when controlling for birth weight and gestational age. The risk for CP is high for a surviving twin when the other twin dies in utero.
The brain of the fetus is susceptible to damage from maternal infections and toxins. The TORCHES group of infections (toxoplasmosis, rubella, cytomegalovirus, herpes, and syphilis) is known to cause significant damage to the developing brain of the fetus, and such damage leads to very neurologically involved infants with mental retardation, microcephaly, and seizures. Orthopaedic deformities are noted in 82% of these children.
Fetal exposure to drugs and alcohol through maternal use can also result in injury to the developing brain. Cocaine, heroin, and marijuana can all cross the placental barrier and cause damage to the central nervous system of the fetus.
Congenital malformations of the brain that occur during early pregnancy often result in severe CP. It has been stated that approximately 10% of patients with CP have congenital brain malformations that are apparent on neuroimaging.
Rhesus blood group incompatibility resulting in kernicterus as a cause of CP is decreasing in incidence with improvements in prenatal care. RhoGAM treatment of Rh-negative mothers has led to a decline in kernicterus, which often resulted in the development of such movement disorders as athetoid CP.
Maternal health problems, such as renal failure or infections, can lead to problems with brain development in the fetus. Prenatal chorioamnionitis and maternal infection have been associated with an increased risk for premature onset of labor and CP in the infant. , , Placental abnormalities have been linked with a higher frequency of CP.
Anoxia as a result of perinatal complications may lead to the development of CP. A tight nuchal cord or placental abruption can lead to anoxia and thus result in CP. Fetal hypoxia may be detected by fetal heart rate monitoring, but changes consistent with hypoxia, such as late deceleration of the heart rate with uterine contractions, are common and not specific. The frequency of CP associated with birth asphyxia is estimated to be 1 in 3700 full-term live births. Fetal distress during delivery has been documented in some children with CP. The mode of delivery—vaginal or cesarean—has not been found to influence the incidence of CP.
Sepsis in the neonatal period can predispose to the development of CP in a low–birth weight infant. Bronchopulmonary dysplasia and prolonged ventilation in preterm infants may result in hypoxia, which predisposes the infant to CP. Extracorporeal membrane oxygenation (ECMO) has been used to sustain babies with severe cardiorespiratory failure. CP has been diagnosed in up to 20% of surviving children who were treated with ECMO.
Cardiac surgery for the treatment of severe congenital heart disease has been linked with an increased incidence of CP. Heart surgery before the age of 1 month resulted in CP in 25% of infants. Clearly, these children are quite ill, with an increased risk for hypoxia, sepsis, and prolonged ventilation.
Infections such as meningitis in early childhood can lead to CP. Any episode of hypoxia, such as cardiopulmonary arrest, near-drowning, and suffocation, can also produce brain damage leading to CP. Trauma, such as motor vehicle accidents producing head injury, severe falls, and child abuse, may result in CP as well.
The first classification is physiologic and describes the type of movement disorder present. The most common movement abnormality is spasticity. Spasticity results from damage to the pyramidal system, particularly the motor cortex in the brain. Disinhibition of pathologic reflex arcs leads to increased tone in the extremities. The tone is dependent on velocity, which means that if a muscle is stretched rapidly, tone increases more than if the same muscle group were stretched gradually and gently.
Hypotonia is, as its name implies, abnormally decreased tone. Infants with CP are often described as floppy or hypotonic. Hypotonia is usually a phase and most frequently leads to spasticity as the infant matures.
Dystonia is described as increased tone, which is not dependent on velocity. Whereas tone in spasticity is described as “clasped knife,” tone in dystonic CP is described as “lead pipe,” which means that tone does not decrease with gentle prolonged stretching.
Athetosis is characterized by abnormal writhing movements that the patient cannot control. The movements become more exaggerated as the patient tries to complete a purposeful motion. Athetosis results from damage to the basal ganglia. Speech is often garbled and difficult to understand, yet affected patients may be intelligent. Athetosis has frequently been the result of neonatal kernicterus.
Cerebellar lesions lead to ataxic CP. The disturbed balance of these children results in a wide-based and clumsy gait. Pure ataxic CP is rare.
Patients with CP frequently have a mixed form of movement disorder. It is important to correctly classify the movement disorder of a patient with CP because the results of surgical treatment are unpredictable for all but purely spastic patients.
The second classification system is geographic and describes what part of the body is affected by CP. Hemiplegia is present when only one side of the body is involved, with the upper extremity usually more involved than the lower extremity ( Fig. 31.1 ). Patients with spastic hemiplegia can be further divided by their degree of gait impairment. Winters and colleagues subdivided patients with spastic hemiplegia into four groups: (1) loss of swing-phase ankle dorsiflexion (i.e., footdrop) but stance-phase dorsiflexion present; (2) loss of stance- and swing-phase ankle dorsiflexion (equinus) and possible knee hyperextension in stance phase; (3) ankle involvement plus increased stance-phase knee flexion with limited range of knee motion; and (4) ankle, knee, and hip involvement with increased stance-phase hip flexion and limited range of hip motion.

Diplegia implies involvement of both sides of the body, with both lower extremities being involved (though not always symmetrically) and lesser involvement of the upper extremities ( Fig. 31.2 ). A word of caution is needed. If the patient has abnormal tone in both lower extremities but the upper extremities are completely normal, the examiner should beware. Patients with diplegia will have some abnormality in the upper extremities, such as decreased fine motor control, spasticity, or increased reflexes. If the upper extremities are normal, it is imperative to evaluate the spinal cord. Spinal cord pathology, including tumor, may masquerade as CP.
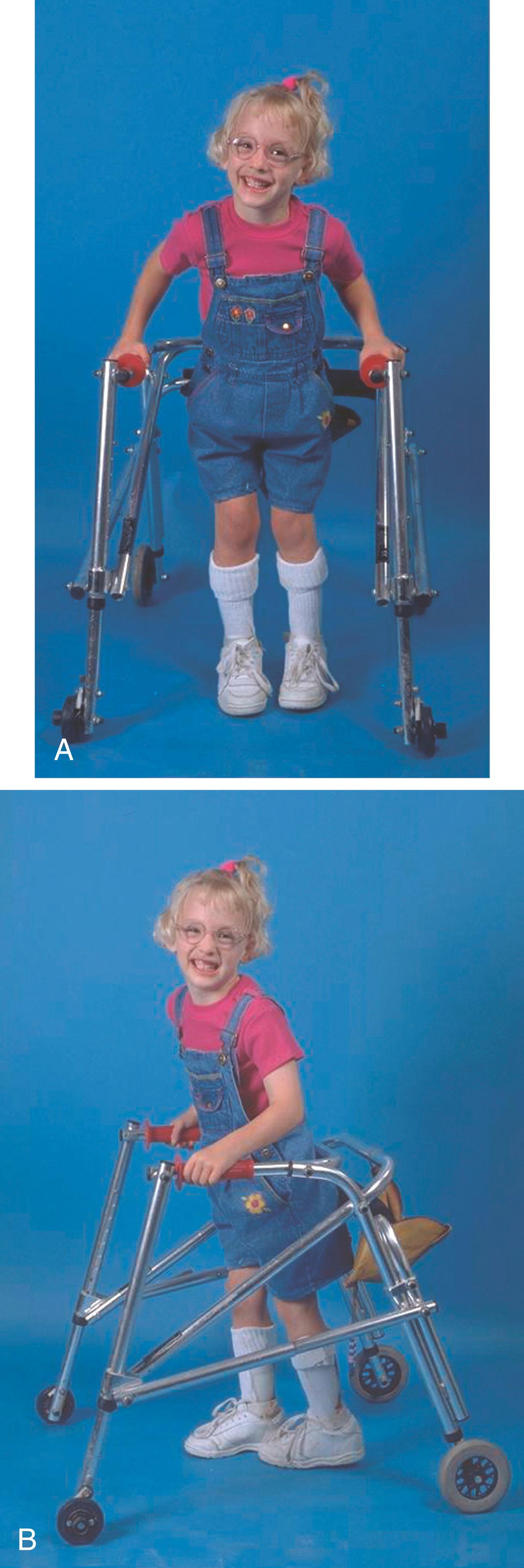
Involvement of both lower extremities and one upper extremity is termed triplegia. Quadriplegia, or total body involvement, is present when all four extremities are severely involved, with poor trunk control as well ( Fig. 31.3 )

Familial spastic paraparesis, a genetic neurologic disease, may resemble CP in that both lower extremities are spastic, yet the upper extremities are normal. Various forms of the disease exist, and a history of other affected family members is helpful.
Current emphasis is on classifying patients with CP by functional level ( Fig. 31.4 ). The Gross Motor and Functional Classification System (GMFCS) is most commonly used to describe the patient’s level of function before and after an intervention. The GMFCS scale has five levels. GMFCS 1 describes a patient who ambulates without aids on all surfaces and keeps up with peers. In GMFCS 2, the patient is fully ambulatory, may use lower extremity orthoses, and does not keep up fully with peers. At GMFCS 3, the patient uses ambulatory aids such as a walker or crutches and may use a wheelchair for longer distances. GMFCS 4 describes nonambulatory patients who are able to propel their own wheelchair, whereas GMFCS 5 indicates an inability to transfer, propel a wheelchair, or support the trunk. A comprehensive review of nine CP registries throughout the world revealed the following proportion of GMFCS levels: level 1, 34.2%; level 2, 25.6%; level 3, 11.5%; level 4, 13.7%, and level 5, 15.6%. Because levels 1 and 2 are considered lesser involvement, most patients are mildly involved, although more severely involved children are more apparent in a pediatric orthopaedic practice.

The first step in the evaluation of a child with CP is to obtain a complete history, especially the birth history. Birth weight, gestational age, complications, and whether the child required ventilator assistance or hospitalization in the neonatal intensive care unit are important data. If the birth history is normal, neurologic consultation should be considered. Evaluation of motor milestones will reveal delayed development. Head control should be present at 3 to 6 months, sitting by 6 to 9 months, crawling by 9 months, standing and cruising by 10 to 12 months, and walking between 12 and 18 months. Adjustments for prematurity should be made; a premature child may not walk by 15 months of age. Preferential use of one hand or leg and early handedness, particularly left-handedness in small infants, are often clues that spastic hemiparesis may be present. Likewise, dragging one leg when crawling or scooting also may be an indication of hemiparesis. Ascertaining whether the child has other problems, such as strabismus, difficulty swallowing, frequent choking, delayed speech development, poor eyesight, and seizures, is important. Some 20% to 40% of children with CP have seizures, most common in hemiplegic and quadriplegic patients. , These observations may all be clues leading to the diagnosis of CP.
Physical examination of a child with CP should include muscle tone in the extremities. With the patient relaxed (even sitting on the lap of a parent), the extremities are brought through a full range of motion. Spasticity feels like tightness in the muscles, which become tighter the quicker the limbs are passively moved. Greater range of motion can be gained by slowly and gently stretching the joints in question. The Tardieu test is a measure of spasticity. For example, if the examiner is assessing hamstring spasticity, the angle at which a “grab” of resistance occurs when quickly extending the knee with the hip in flexion is compared with the amount of extension possible when the knee is stretched slowly.
Fine motor activities should be assessed. Passing the child a toy or a pen often reveals spastic hemiplegia in one extremity. Having the child clap the hands or wiggle the fingers may reveal difficulties in fine motor control.
Deep tendon reflexes are increased in patients with CP. Repetitive tapping of the deep tendons or quick passive dorsiflexion of the ankle may produce clonus, which establishes the presence of an upper motor neuron neurologic abnormality. In hemiparesis, reflexes will be asymmetric.
Infantile reflexes disappear in normal children by 3 to 6 months of age as the motor cortex matures; however, they are retained in children with CP. Bleck’s textbook on CP outlines these reflexes in clear detail. The startle reflex, or the Moro reflex, which should disappear by 4 months of age, is elicited by letting the infant’s head drop back into extension with the infant supine but slightly elevated. This causes the legs and arms to extend abruptly. A sudden loud noise can likewise cause an older child to extend and lurch from a wheelchair. The parachute reflex is tested by holding the child in the air and then lowering him quickly headfirst toward the examining table. Children older than 5 months will reach out with both arms to protect themselves. Children with CP cannot do so, and those with hemiplegia will reach out with only one arm.
The tonic neck reflex is elicited by turning the supine infant’s head to one side. The ipsilateral arm and leg will extend while the contralateral arm and leg flex. This reflex should disappear in infancy; persistence should raise suspicion for CP.
Balance, sitting, and gait are assessed by noting whether the child can sit unsupported without use of hands or get into a sitting position without assistance or whether balance is easily disturbed in the sitting position or as the child walks.
Clinical assessment of gait requires that the child’s joints be readily seen, so the child should be barefoot and in shorts or a short gown. The evaluation should be conducted with the examiner seated on a stool at the level of the child. Enough room should be available for the child to walk naturally. Heel-to-toe walking, hopping on either foot, and running are observed. A patient with mild hemiplegia may walk nearly normally but exhibit abnormal movement patterns while running; the affected upper extremity will draw upward and not have a normal arm swing.
Gait should be observed from the front of the child and then from the side, and the hips, knees, and ankles should be systematically assessed from each perspective. A crouched gait consisting of increased flexion at the hip and knee, toe-walking with genu recurvatum, or a footdrop in the swing phase of gait may all be indicative of CP. Disturbance in clearance of the swing-phase limb may be caused by footdrop or an inability to flex the knee.
Rarely are imaging studies ordered by orthopaedic surgeons when establishing the diagnosis of CP. If questions persist regarding a correct diagnosis, referral to a pediatric neurologist is indicated. At the neurologist’s discretion, imaging studies such as cranial ultrasonography, brain magnetic resonance imaging (MRI), and computed tomography (CT) may be pursued. Similarly, laboratory studies may be necessary to look for evidence of metabolic diseases associated with delays in development and CP-like symptoms, such as congenital hypothyroidism or dopa-responsive dystonia. However, a detailed discussion of these metabolic conditions is beyond the scope of this chapter.
Two findings frequently described on histopathologic examination or imaging studies of the brain in children with CP are periventricular leukomalacia and intraventricular and periventricular hemorrhage. Periventricular leukomalacia is defined as patchy areas of necrosis in the periventricular white matter adjacent to the lateral ventricles. It results from an ischemic insult to the arterial watershed area close to the ventricular walls. Pyramidal tract fibers mapping to the lower extremities pass through this area and are therefore more susceptible to injury than fibers responsible for the upper extremities and face.
The areas of the brain immediately adjacent to the ventricles are also most susceptible to hemorrhage. Hemorrhage may be seen on ultrasound, MRI, or CT. Mild hemorrhages involve the germinal matrix adjacent to the ventricles, whereas more severe hemorrhages extend into the ventricles themselves and into the parenchyma of the brain. Hypoxia is known to predispose to periventricular and intraventricular hemorrhage. Approximately one-half of preterm infants with CP are found to have abnormalities on neuroimaging, such as echolucency in the periventricular white matter or ventricular enlargement on cranial ultrasound. In children with CP born at or near term, approximately two-thirds have abnormalities on neuroimaging, including focal infarction, malformations, and periventricular leukomalacia.
Gait analysis has become popular in the assessment of movement disorders in children with CP. Accurate documentation of dynamic range of motion may help in planning surgical treatment and assessing the results of orthopaedic operations, and complements but does not replace a good clinical examination. When gait analysis graphs are scrutinized together with information from the clinical examination and slow-motion videotape, better understanding of a patient’s gait can be gained. , A detailed discussion of gait analysis can be found in Chapter 5 . The Functional Mobility Scale is a questionnaire that assesses the child’s ambulatory ability at 5 m (within the home), 50 m (short distances), and 500 m (community ambulation) and should be considered in conjunction with gait analysis in surgical decision making. ,
Cadence parameters include walking speed, step length, number of steps per minute, and the proportion of time spent in the stance and swing phases. Patients with CP usually have disturbances in cadence parameters. In good walkers with spastic diplegia, walking velocity is maintained despite decreased step length by increasing cadence (quick, short steps). Good walkers with CP increase their speed by increasing cadence, but spasticity interferes with increasing step length. In those with more severe diplegia and quadriplegia, walking speed is diminished, with decreased cadence and step length. The proportion of time spent in stance phase, particularly double-limb stance, increases because the child has greater difficulty with balance and advancing the limb. Children with hemiplegia show asymmetry in step length and in single- and double-support time.
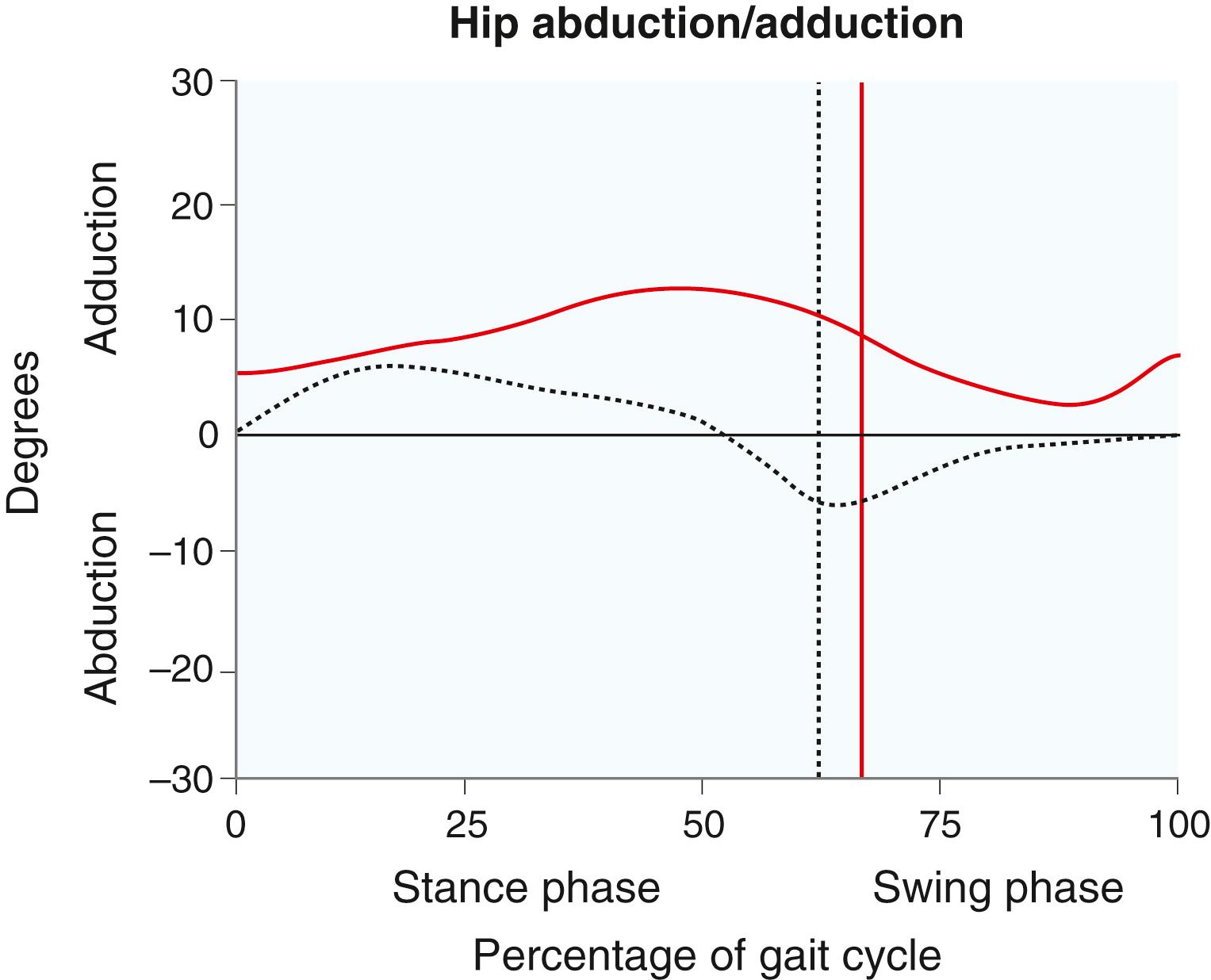
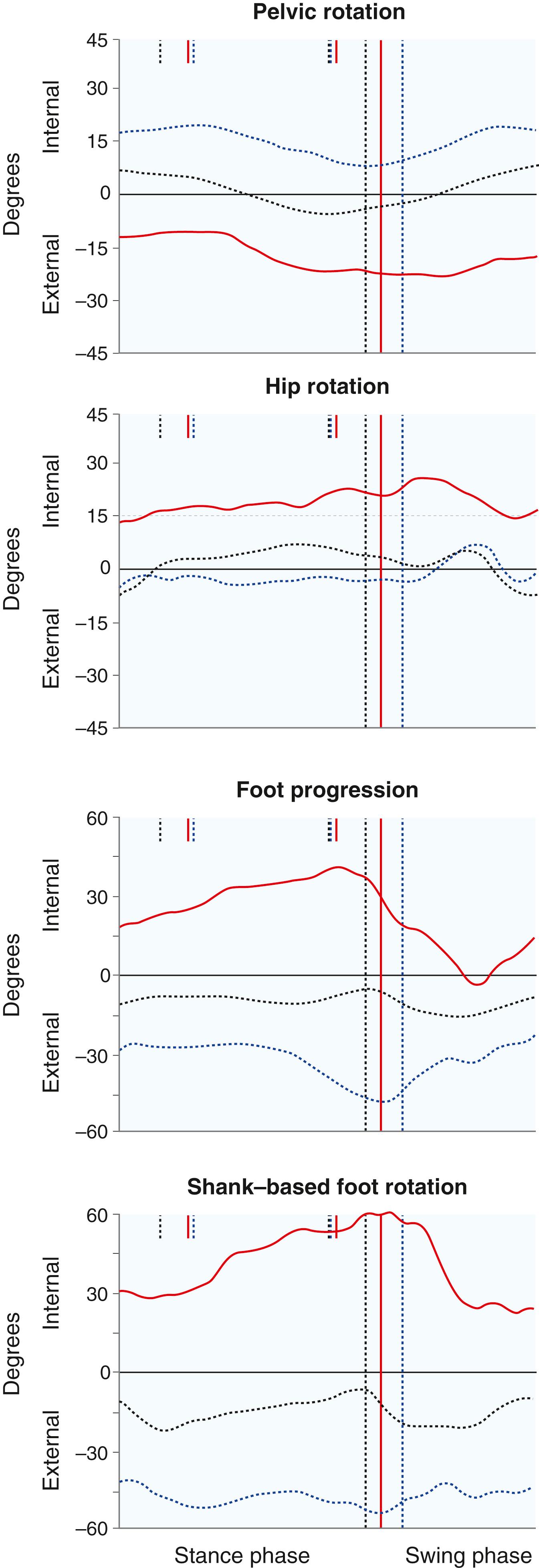
Certain kinematic patterns are seen in patients with CP. a In the hip, scissoring, which is caused by tightness in the adductor musculature and, in part, by weakness of the hip abductors, leads to a narrow base of gait and difficulty advancing the swing-phase limb past the stance-phase limb ( Fig. 31.5 ). In the sagittal plane, increased hip flexion and anterior pelvic tilt may be part of crouch gait as a result of increased tone in the iliopsoas ( Fig. 31.6 ). Increased femoral anteversion may be documented by gait analysis as increased internal rotation of the hips. Asymmetric pelvic rotation may be present, and gait analysis is particularly helpful when the examiner is trying to ascertain whether the abnormal rotation is originating from the pelvis, hips, or tibiae ( Fig. 31.7 ).
References , , , , , .

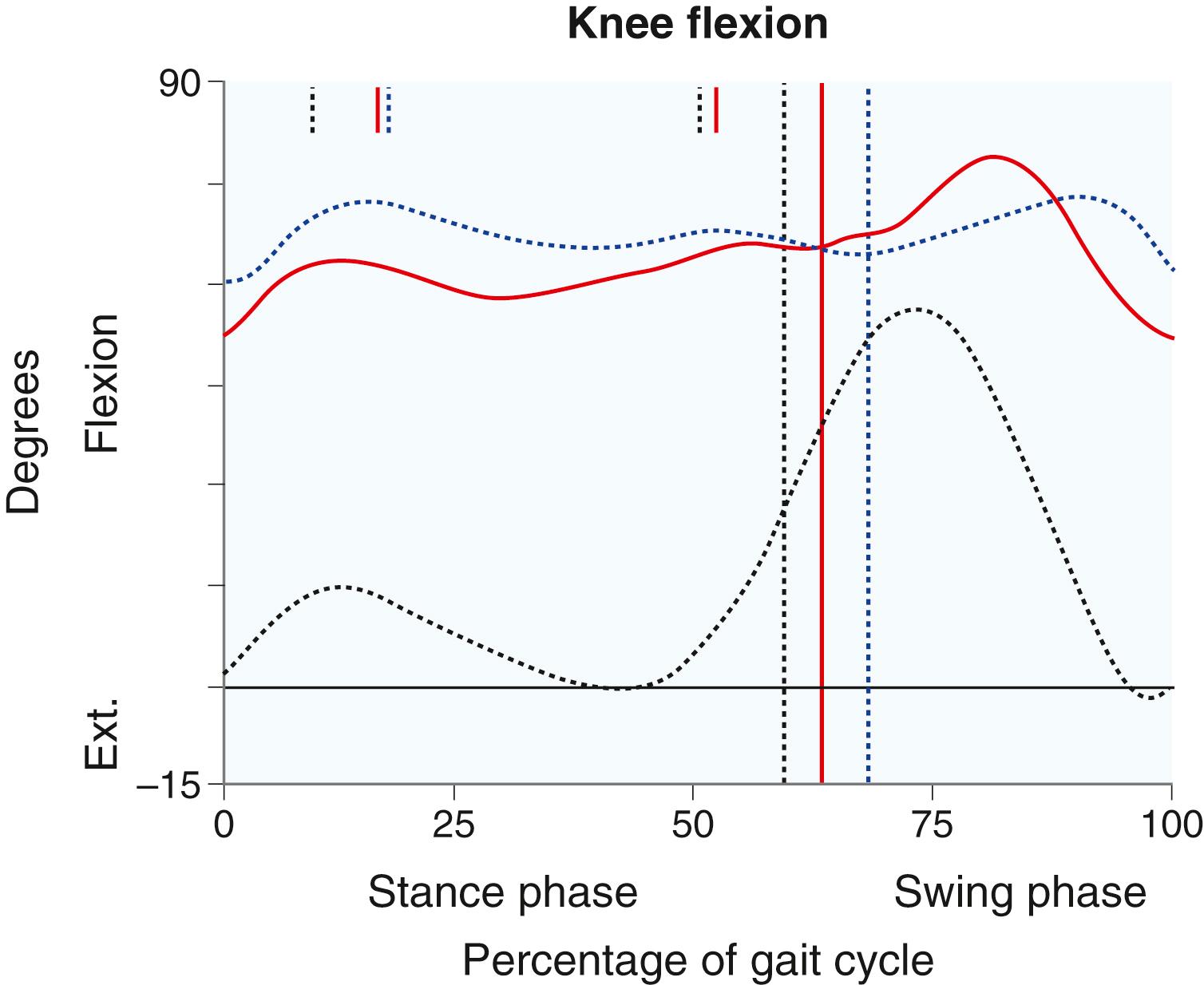
At the knee, sagittal-plane motion is usually abnormal. In patients with tight hamstrings, the knee remains flexed at initial contact and is unable to extend normally during stance phase. In swing phase, spasticity of the rectus femoris may inhibit the patient’s ability to flex the knee and clear the floor ( Fig. 31.8 ). Genu recurvatum during stance phase may be present in response to a tight Achilles tendon, which causes difficulty advancing the tibia forward over the foot.
Ankle kinematics often shows disturbances in plantar flexion, dorsiflexion, and push-off. Plantar flexion on weight acceptance is generally abnormal in patients with equinus contractures. Dorsiflexion during midstance is diminished in the presence of a tight Achilles tendon, and push-off is reduced if the ankle is already plantar-flexed from the equinus. Swing-phase dorsiflexion may be absent as a result of weakness or inactivity of the tibialis anterior and lead to footdrop ( Fig. 31.9 ).

Gait analysis is particularly useful in assessing the cause of toe-walking. It is tempting to attribute all toe-walking to tight Achilles tendons; however, some children walk on their toes in response to crouch above the ankle and have neutral ankle dorsiflexion but increased flexion at the knee and hip. Lengthening of the gastrocnemius or Achilles tendon in these children would result in a calcaneus gait, with persistent crouch at the hip and knee but excessive dorsiflexion at the ankle leading to inefficient push-off.
Spasticity leads to electrical overactivity on electromyography (EMG) during gait, and it seems that the more spastic the child, the greater the EMG signal and the less phasic the muscles in their contraction ( Fig. 31.10 ). Dynamic EMG data collected during gait analysis can be correlated with kinematic and kinetic graphs to gain a fuller understanding of the biomechanics of the child’s gait. For example, normally at initial contact, the ankle plantar-flexes while the anterior tibialis fires on the EMG. Kinetic plots show absorption of power as weight acceptance occurs. In stance phase a child with CP may exhibit early heel rise, seen as plantar flexion on kinematic plots, which correlates with gastrocsoleus overactivity on EMG. We find data from EMG most useful when evaluating a child with a stiff knee in the swing phase for rectus femoris transfer and when assessing a child with an equinovarus foot for tendon transfer (either anterior tibialis or posterior tibialis split transfer).
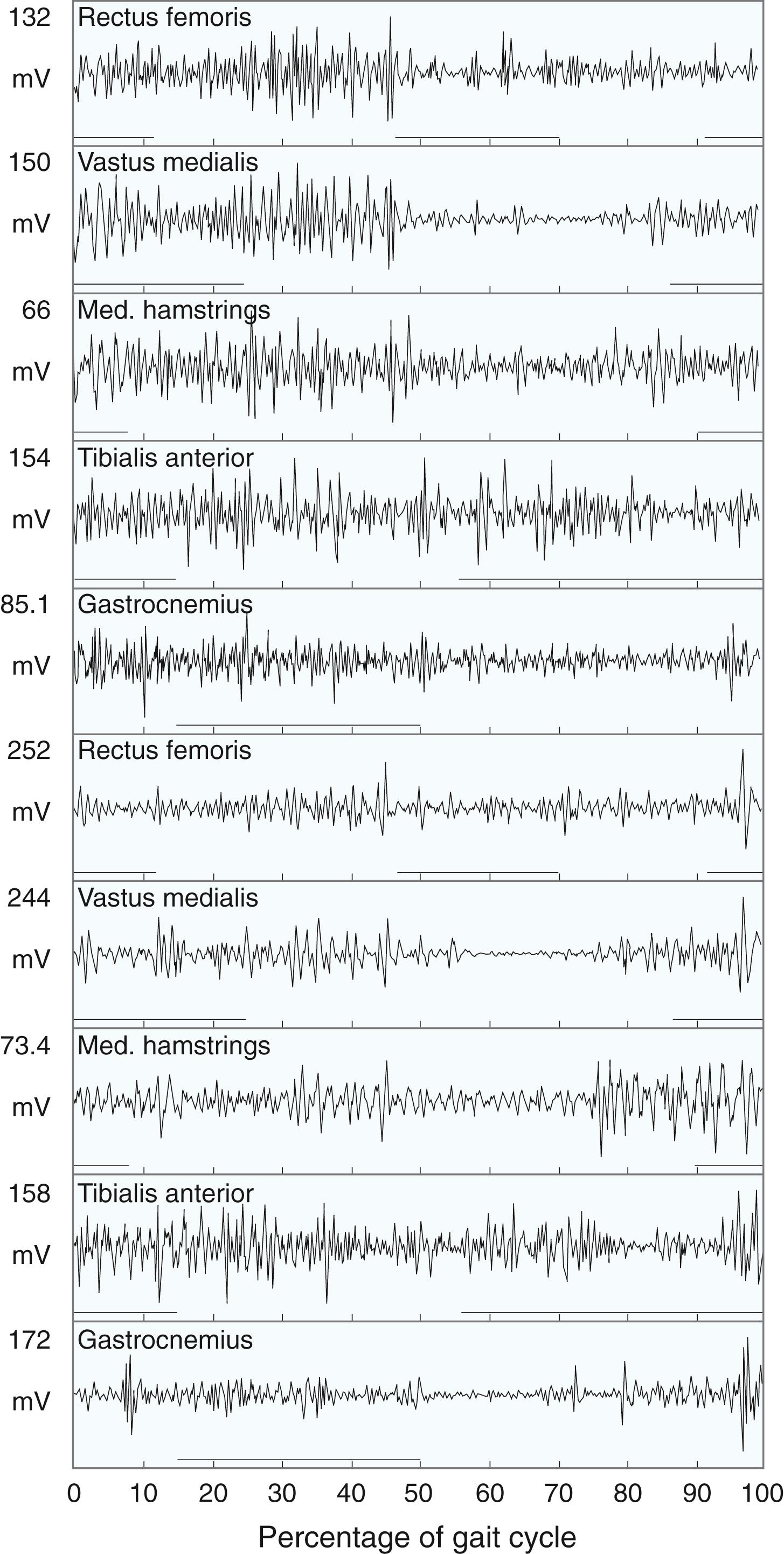
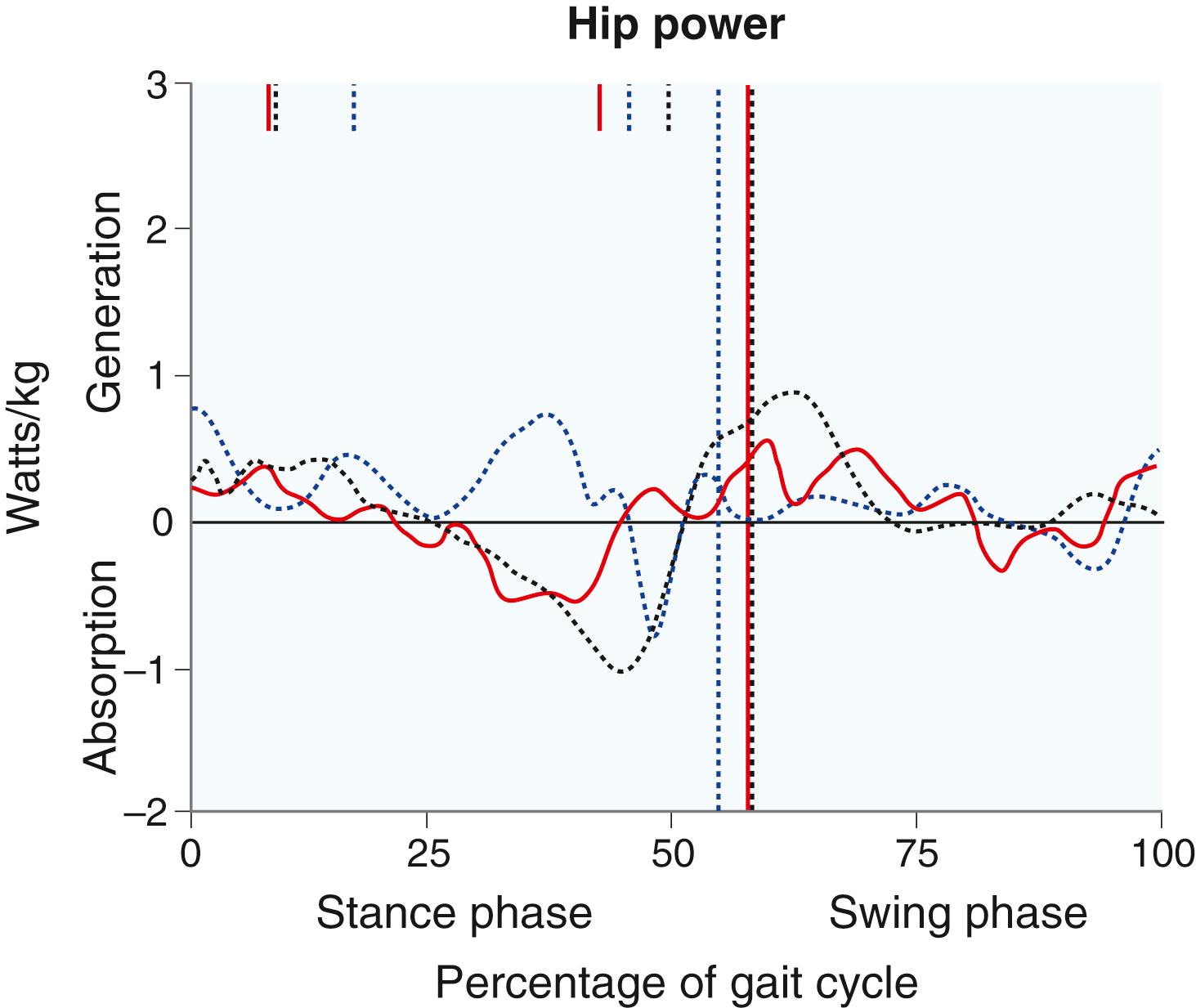
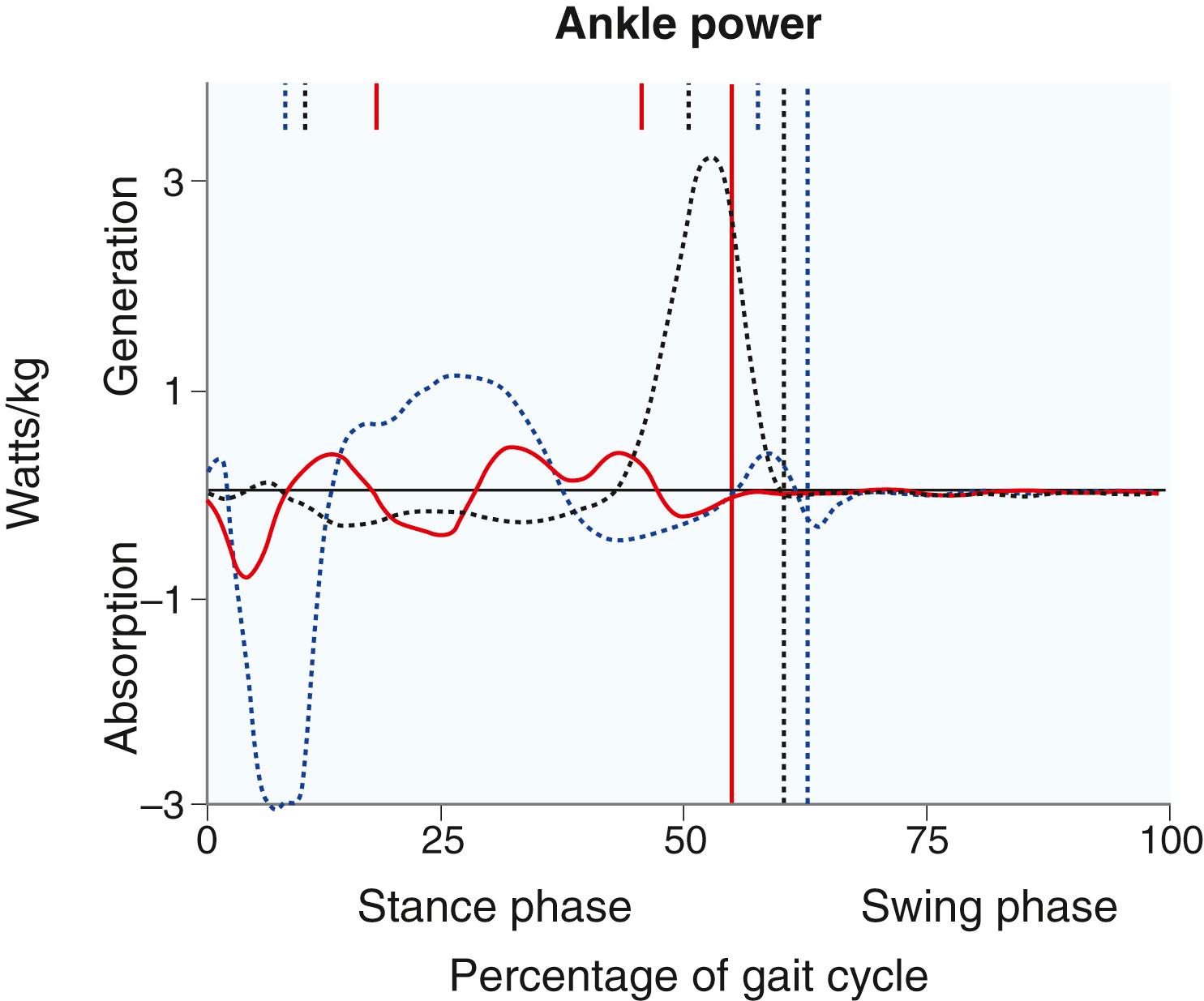
Kinetics are the forces exerted across joints during gait. Each joint has well-described kinetic patterns, and the reader is referred to work by Gage for descriptions. Two particular forces are clinically interesting: hip pull-off power and ankle push-off power. Hip pull-off power, the force exerted by the iliopsoas and other hip flexors to lift the stance-phase limb off the ground and into the swing phase, is often diminished in patients with CP and leads to decreased energy efficiency ( Fig. 31.11 ). Ankle push-off power, the force exerted by the gastrocsoleus at terminal stance to push the stance-phase limb off the ground, is diminished in patients with equinus or calcaneus gait ( Fig. 31.12 ).
The goal of orthopaedic intervention is to improve the quality and efficiency of walking. By comparing oxygen cost and consumption with normal values through collection of preoperative and postoperative data, the efficiency of the child’s gait can be measured objectively. , Heart rate is an indirect measure of oxygen consumption and can easily be measured in the clinic. , Children with CP have been found to have six times higher heart rates when walking than able-bodied peers do, with the highest heart rates seen in children with crouch gait.
Critics of gait analysis in patients with CP have pointed out that patients fitted with markers and electrodes and placed in front of video cameras do not walk as they do at home or school. Considerable variability in how patients walk from clinic visit to clinic visit has been noted. Basing treatment plans on kinematic and kinetic data from a few strides across a gait laboratory may not always be in the patient’s best interest. Gait analysis can be an adjunct to clinical examination, but the data should be scrutinized and the videotape reviewed to judge whether the data are representative of the gait seen in the clinic or described by the parents in the home.
Very active gait analysis laboratories note that their studies change the surgical plan in patients with CP in up to 52% to 70% of instances. , , In many cases, gait analysis reduces the number of individual procedures needed in a single-event multilevel surgery (SEMLS) approach. The decision-making process with the use of gait analysis data is only as good as the astuteness and familiarity of the orthopaedic surgeon reading the graphs.
The Gillette Gait Index (GGI) is a numerical calculation that represents how different the kinematic data are from age-matched normal values. Improvement in the GGI following SEMLS has been documented in studies.
Gait analysis in our center is used to
Clearly document the three-dimensional movement of the lower extremity during gait preoperatively
Document changes in gait over time as the patient grows, because gait may deteriorate in children with CP ,
Allow preoperative and postoperative comparisons of results after tendon and/or bone surgery and to gather research data
Analyze the rotational profile of the patient before surgery to help the surgeon select the correct site and amount of rotational change desired
Confirm a surgical plan when needed
Gait analysis does not tell surgeons whether they should operate, but it may help them fine-tune the operative plan when questions exist. Additionally, gait analysis provides an objective assessment of changes seen after SEMLS, including alterations in joint position, dynamic range of motion, and time and distance parameters.
It has long been known that muscles in children with CP are typically spastic and that motor control of the muscles (i.e., the child’s ability to volitionally contract and relax the muscle) is impaired. Reports have documented that muscle weakness is also a problem in children with CP. Documented muscle weakness is more pronounced as the CP GMFCS level worsens.
Many authors have proposed criteria for predicting the ultimate ability of a child with CP to walk. Inability to sit by 2 years of age, persistence of two or more infantile reflexes beyond 12 to 15 months, and lack of head control by 20 months imply a poor prognosis for ambulation. The presence or absence of mental retardation does not influence the ability to walk.
The geographic type of CP that a child has influences whether the child will walk. All children with spastic hemiplegia develop the ability to ambulate. Eighty-six percent to 91% of children with spastic diplegia become ambulatory and 0% to 72% of patients with spastic quadriplegia learn to walk. The discrepant figures are due to variation in differentiation between spastic diplegia and quadriplegia among studies.
The age at which children with CP begin to walk varies with the severity of their neurologic disease. Patients with spastic hemiplegia generally walk between the ages of 18 and 21 months. Children with spastic diplegia who walk usually do so by 4 years of age. Many agree that the ability to walk plateaus by 7 years of age, thus implying that if a child is nonambulatory at 7 years, the child will probably never become ambulatory.
Once the diagnosis of CP has been made, the physician must select a course of treatment. Treatment choices are now more numerous than ever and include observation, physical therapy, botulinum toxin, intrathecal administration of baclofen, rhizotomy, and orthopaedic surgery. With the increasing popularity of nonorthopaedic management of spasticity, some centers have noted an overall decrease in orthopaedic surgical procedures in this patient group. Treatment, whether surgical or nonsurgical, must be goal oriented. The goals of treatment of children with CP that have been linked to productive lives as adults are communication, education, mobility, and ambulation.
Note that walking ranks below mobility. Although ambulation remains a desirable goal, it should not be pursued so fervently that attention to overall development of the child is ignored.
Bleck quoted Rang as advising orthopaedists to remember that “the child with cerebral palsy becomes the adult with cerebral palsy.” Childhood is the optimal time for intervention to maximize the function of a patient with CP. It is the orthopaedists’ duty to ensure that the musculoskeletal treatment of the child prevents future problems with pain and deformity as an adult. Patients with CP do not usually have severely shortened life spans. In a study of all children with CP born between 1966 and 1984, the 20-year survival rate was 89%. If the patients were ambulatory, had manual dexterity, and were not mentally retarded, the 20-year survival rate was higher than 99%. Survival is clearly linked with the patient’s GMFCS level. In a study from the Swedish database, all GMFCS level 1 and 2 children survived to 19 years of age, whereas only 60% of GMFCS level 5 children were still alive. Patients with gastrostomy tubes were most likely to succumb to early death, which is indicative of their medical fragility. However, survival of these frail patients has increased over the time period from 1983 to 2010. The overall 30-year survival rate is estimated to be 87%.
Frequently, the first treatment rendered to a child with CP is physical therapy. Yet no controlled studies have confirmed that regular physical therapy improves the outcome of a child with CP. One of the first well-designed studies investigating the effect of physical therapy was performed by Wright and Nicholson in 1973. They found no difference in motor skills or reflexes after 12 months in children who had neurodevelopmental training or physical therapy and those who did not. Other studies followed and again showed no discernible improvement after different forms of therapy. , , , In defense of physical therapy, these studies are difficult to carry out because they involve different age groups and children with varying severity of neurologic impairment and usually encompass just a brief period of therapy. However, as Bleck pointed out, “The burden of proof is on the proponents of the treatment. Critics need not prove ineffectiveness but can insist on positive data.” The efficacy of physical therapy can be proved or disproved only with a properly designed, collaborative, multicenter, randomized, controlled trial.
Physical therapy, ranging from the medical model, with the aims of attaining ambulation, range of motion, or transfers, to neurodevelopmental training, sensory integration, and even craniosacral therapy, has been proposed. Electrical stimulation of the muscles has also been used in these patients. , Families like physical therapy and attribute gains in their young child’s ability to interaction with the therapist. However, some of these gains are simply the result of neurologic maturation of the child.
Our approach to physical therapy is to establish therapy for monitoring the developmental milestones of very young children, around the age of 2 or 3 years. Therapy is continued if gains are being made in attaining ambulation. School-based programs are used in elementary school and often include adaptive physical education. Postoperative intensive physical therapy is essential to reestablish range of motion and strength after surgical intervention. Strength training of weak muscles has been successful in improving motor function. , We also draw on the expertise of physical therapists in assessing orthotic needs and wheelchair seating when appropriate. No evidence supports the continued use of physical therapy for range of motion, particularly in a nonambulatory child. Physical therapists play an important consultative role in making treatment decisions for patients whom they treat on a regular basis and whom we examine on a relatively infrequent basis.
It is common for parents of children with CP to resist discontinuing physical therapy. We believe that goals must be set for therapy and, if progress toward these goals is not being made, either the goals need to be reassessed or therapy should be stopped because it is not useful. Setting measurable functional goals has led to greater success after physical therapy.
In an older child, transitioning from physical therapy to therapeutic recreation is desirable and generally met with enthusiasm by the patient. Adaptive sports or swimming allows the child to participate with peers and affords greater enjoyment than exercises in the therapy gym do. Time in school should be spent on education at this age and not on physical therapy.
Inhibitive casting has waxed and waned in popularity as a mode of treatment of spasticity in children with CP. It is based on the presence of areas on the feet that are target centers for increased tone at the ankle and, some believe, throughout the lower extremities in certain patients. Usually, short-leg casts are applied with extended toe plates, careful molding of the heel, and metatarsal head control. This has been used by physical therapists and by some physicians. The time spent in casts varies but is generally a minimum of 6 weeks and is followed by the use of orthoses.
In our experience, casting has a limited role in patients with CP. We have used casting in rare cases of very young children (<4 years) with equinus contractures to delay surgical heel cord lengthening. Because equinus recurs after casting, permanent resolution of the equinus is not a goal. Some studies show short-lived improvements in passive and dynamic ankle range of motion following casting, whereas others have reported longer resolution of equinus contractures in children younger than 6 years.
Orthoses can be helpful in improving gait in ambulatory patients with CP. Bracing is best prescribed when specific treatment goals for gait improvement are identified. Ankle-foot orthoses (AFOs) are helpful in positioning the ankle and foot during gait. In young children, dynamic equinus can be improved, with a plantigrade foot position obtained and the tendency for genu recurvatum decreased.
AFOs come in various designs ( Fig. 31.13 ). Solid ankle AFOs are used when minimal dorsiflexion of the ankle is possible. Of the various types, they afford the easiest fit. Solid ankle AFOs also can be used in patients with excessive dorsiflexion and knee flexion; by stabilizing the ankle, extension of the knee is encouraged. Ground reaction AFOs have an anterior shell of plastic across the proximal end of the tibia and are rear entry. The goal of a ground reaction AFO is to provide push-back on the knee during stance phase and help the knee extend. Patients who walk in mild crouch with knee flexion contractures of less than 10 degrees may benefit from this design. Hinged AFOs permit a more natural gait by allowing some dorsiflexion during stance but blocking plantar flexion and therefore eliminating equinus and footdrop. Greater power generation in preswing is seen with hinged AFOs than with solid ankle designs ; however, hinged AFOs are bulkier and break more frequently, and they offer no benefit if the patient does not have passive dorsiflexion of the ankle. If crouch gait is present, a hinged AFO will worsen knee flexion, thereby leading to ankle calcaneus and a less efficient gait. Posterior leaf spring AFOs are fabricated from more pliable polypropylene and have more severe posterior trim lines at the ankle. They are touted as providing a little push-off at terminal stance. They tend to break and may not provide enough hindfoot control in most patients with CP. Gait analysis has shown that posterior leaf spring AFOs function similar to hinged AFOs; that is, they allow some dorsiflexion in stance phase through deformation of the brace, and they control equinus in the swing phase but do not return energy at push-off.
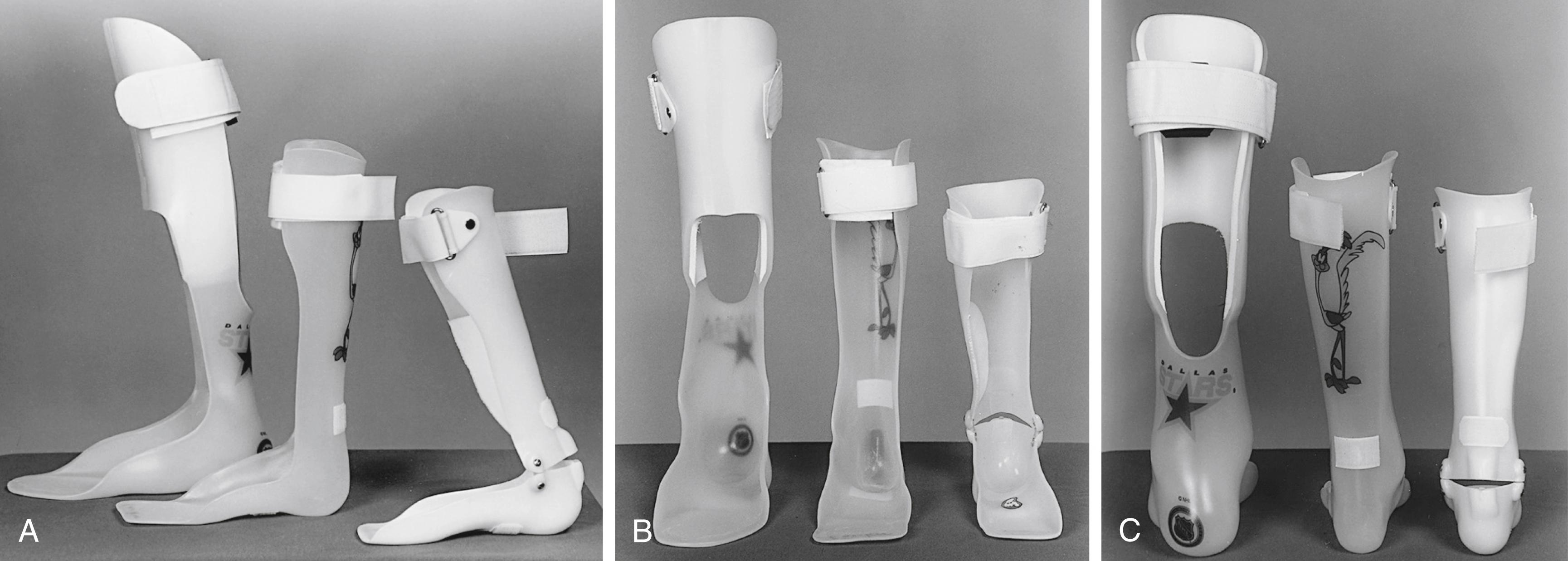
Tone-reducing (or dynamic) AFOs incorporate a molded footplate, termed an inhibitive footplate, and have higher trim lines that extend across the dorsum of the foot. The goal of these orthoses is to apply pressure similar to that of an inhibitive cast and therefore reduce tone throughout the lower extremities. Little scientific evidence supports this design inasmuch as no significant functional changes in gait have been noted with the use of tone-reducing as opposed to standard AFOs. Nonetheless, they remain popular with many families.
Bracing above the knee with knee-ankle-foot orthoses (KAFOs) or hip-knee-ankle-foot orthoses is not generally done in those with CP. The weight and bulk of the braces usually interfere with rather than improve the child’s ability to walk. Short-term use of KAFOs or knee immobilizers after soft tissue surgery for crouch gait can be helpful in retraining children to walk.
Our indications for bracing are
To obtain a plantigrade foot position and reduce genu recurvatum in patients with dynamic equinus
To support the foot in dorsiflexion during the swing phase when footdrop is present
To assist the foot postoperatively while weakness is being treated by physical therapy
To improve mild crouch
We do not advocate the use of AFOs in nonambulatory patients who are able to wear shoes, but we do on occasion prescribe them when footwear is difficult and is improved with orthoses. We also do not use AFOs in a preambulatory young child because they interfere with the child’s ability to crawl and move about the floor.
Some patients walk worse with AFOs than without them. In patients with significant crouch gait, flexion of the knee and hip with the foot supported in an AFO forces the child to either pull up out of the braces or toe-walk despite the orthosis ( Fig. 31.14 ). Toe-walking has not ever been found to be harmful, so it is often better to allow the child to walk on toes brace free or to proceed with surgery when appropriate. Valgus deformities of the hindfeet are often difficult to brace because braces can produce painful calluses and blisters over the prominence of the talar heads. Comfortable footwear without orthoses is preferable for these feet. Finally, as young children with CP become adolescents, they often refuse to wear orthoses for cosmetic reasons. Allowing the patient to make a personal choice in whether to wear a brace is particularly important in this age group.

Oral pharmacotherapy, including diazepam and baclofen, has been used in an attempt to reduce tone in patients with CP. Side effects include somnolence. Oral tizanidine has been used to relieve spasticity with improved results. Patients with movement disorders such as dystonia or choreiform movements have been treated by various medications, with varying success.
The inability to adequately reduce tone with oral baclofen because of the side effects with large dosages led to trials of intrathecal baclofen. When baclofen is injected into the intrathecal space, small doses can effectively reach the target neural tissue of the spinal cord and produce a greater reduction in tone. Baclofen, a γ-aminobutyric acid agonist, acts at the spinal cord level to impede release of the excitatory neurotransmitters that cause spasticity. After proof of efficacy with test injections of the drug, implantable pumps filled with baclofen are surgically inserted into the anterior abdominal wall and the dose of medication is titrated. Continuous infusion of the medication is delivered by the pump, which requires refilling approximately every 3 months. Studies have shown decreased upper and lower extremity tone and improvements in range of motion with continuous intrathecal baclofen infusion.
Complications occur in approximately 20% of patients. The complications are usually related to catheter dislodgment or fracture, but serious infection occurs in 5%. , Overdose of intrathecal baclofen is very rare but serious and can produce somnolence and hypotonia, which leads to respiratory depression requiring mechanical ventilation.
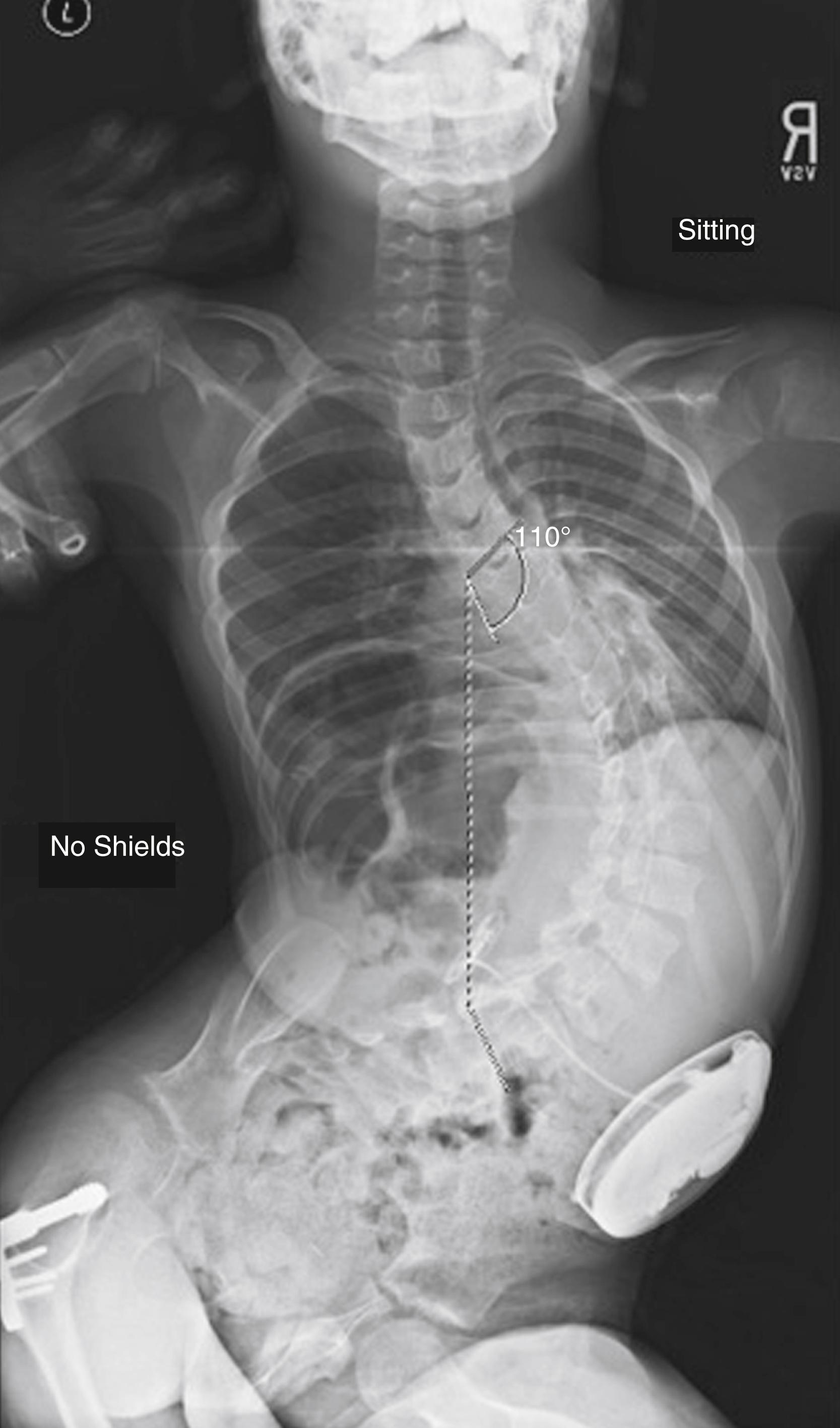
Currently, no strict indications for the use of intrathecal baclofen have been determined, but most pumps are implanted in patients with severe spasticity that interferes with positioning and function of the upper and lower extremities. Pumps are currently being implanted in both nonambulatory and ambulatory patients. Spasticity, as graded by the Ashworth scale, has been shown to decrease 1 year following implantation of intrathecal baclofen pumps regardless of preoperative severity. Caregiver satisfaction in the nonambulatory group is high, with parents noting easier positioning, transfers, and personal care, as well as improved pain control. , Pain relief and improved sleep are typically seen within 6 months of pump implantation. Many of these patients still need orthopaedic surgery for hip subluxation and should be monitored for the development of such. Although progressive hip subluxation can occur, significant deterioration in migration percentage is uncommon. Close monitoring for scoliosis is also warranted because rapid progression of deformity has been described in some studies but refuted in others. , Several studies comparing progression of scoliosis in patients who had intrathecal baclofen pumps implanted versus a well-matched control group of children with severe CP who were not treated with baclofen found no statistical difference in progression of scoliosis in the treated group. Both groups of patients experienced curve progression, which supports the premise that it is the severity of neurologic involvement and functional disability that predisposes the patient to scoliosis and not the medical management of spasticity ( Fig. 31.15 ). , , Additionally, the presence of an intrathecal baclofen pump has not been linked with a higher complication rate following scoliosis surgery.
Botulinum toxin injections have become a popular form of treatment in patients with CP. The toxin is produced by the anaerobic bacterium Clostridium botulinum and works by blocking the release of acetylcholine at the neuromuscular junction. It is injected at known anatomic sites of innervation, often guided by EMG or ultrasound. The targeted muscle becomes weak because of lack of innervation until the neuromuscular junction theoretically sprouts new endings. Muscle histopathology studies following serial botulinum toxin injections has shown worrisome muscle fiber neurogenic atrophy in a recent study, bringing into question the concept of reversability of the effects of the medication. Botulinum toxin is available in two forms, Botox and Dysport, which are not pharmacologically equivalent.
Muscles that are considered to be candidates for injection are those that produce dynamic deformities in the absence of fixed contracture. For example, a child who walks in ankle equinus because of spasticity in the gastrocsoleus but who exhibits dorsiflexion on passive range of motion may be a candidate for injection into this muscle. The drug begins taking effect after 2 to 3 days, and its effect wears off after approximately 3 months. The amount of botulinum toxin that can be injected at any one setting has an upper limit, so it works best in children in whom only one or two muscles are to be injected. It offers less benefit to a child with multiple muscle involvement. From a technical standpoint, superficial large muscles are the easiest to inject. Deep muscles, such as the iliopsoas, can be injected under imaging for accurate localization. ,
Most research that has been published in the orthopaedic literature on the use of botulinum toxin for CP has involved gastrocsoleus injection. Two randomized, double-blind, placebo-controlled trials found improved initial foot contact and gross motor function in botulinum toxin–treated patients. , Gait studies have found improvements in sagittal-plane stance-phase dorsiflexion—in other words, less equinus—after botulinum toxin injection. , Cosgrove, one of the first proponents of this treatment, notes that patients who are younger, who have diplegia rather than hemiplegia, and who have greater dynamic shortening of the muscle with less contracture have the best response. He and others proposed that botulinum toxin may allow tendon surgery to be delayed until a later age, when the risk for recurrence is lower. , Annual injections have been found equally efficacious as injections every 4 months.
Botulinum toxin injections have been compared with serial casting for the nonsurgical treatment of equinus. In one study, the results were similar between children treated with serial casting and those who underwent botulinum toxin injection. Conflicting results were seen in another report, which found Botox alone to be less effective than either casting or combined Botox and casting for ankle equinus. Yet another study reported that casting plus Botox provided improved passive dorsiflexion and motor control when compared with Botox alone. Finally, a meta-analysis of all randomized controlled studies found no benefit of Botox and casting over either casting or Botox alone.
Botox injections into other muscles have been studied. , Hamstring injections may decrease crouch at the knee. Injection of the adductors is reported to lessen scissoring and significantly increase hip abduction. However, adductor injections to decrease hip subluxation is ineffective. A randomized controlled study in Australia compared young children with CP treated with botulinum toxin injections into the adductors and hamstrings every 6 months and daily use of an abduction brace with a similar group of young children who were observed without intervention. Both groups experienced progressive subluxation, although the treated group had a minimal decrease in the rate of progression. The authors concluded that the program of botulinum toxin injection and bracing did not significantly influence the outcome of the hip.
Frequently, parents adopt the philosophy that no harm will be done with injection and surgery can be performed later if necessary. One study found that only 10% of children treated with Botox (and guided by gait analysis) underwent surgery by 7 years of age as opposed to 52% of 7-year-olds who did not receive injections. Although the use of Botox may delay the need for operative treatment, it has not been shown to decrease the need for tendon surgery in the long run. The results wear off, and over time repeated injections become undesirable.
Indications for botulinum toxin injection are
A child with a dynamic equinus deformity and no fixed plantar flexion contracture
A child with equinus gait without multilevel crouch
A child younger than 4 years who cannot tolerate AFOs because of dynamic equinus
Parents’ desire for injections and refusal of tendon-lengthening surgery
Rarely, we recommend botulinum toxin injection before surgery to investigate possible response to a planned surgical intervention.
Botox has been used in a few hospitals to reduce muscle spasm after orthopaedic surgery. Although improved pain relief and reduced medication requirement have been reported in a small series, widespread use of Botox in the perioperative period has not yet been adopted. Similarly, Botulinum toxin has been used in nonambulant children to improve ease of care and comfort. A randomized controlled study comparing injection of botulinum toxin in nonambulators to a sham procedure showed positive results in the treated group.
Some reports of adverse events linked to botulinum toxin injection have led to warnings from regulatory agencies that have tempered the enthusiasm for Botox treatment. , Sixteen deaths from dysphagia and respiratory compromise led to issuing the warning. A follow-up study of 334 patients who received 596 Botox treatments found a 23% incidence of temporary adverse events, which included generalized weakness in one child, worsening of dysphagia, and respiratory infections; however, no deaths occurred in this group. A large Australian series of 1147 children treated with botulinum toxin injections found a 1.3% incidence of hospital admission for respiratory compromise and linked the adverse reactions to higher GMFCS levels. Because many severely involved patients have multiple comorbid conditions as a result of the severity of their medical condition, it is difficult to definitively establish whether the complications described are due to the injection of botulinum toxin or are inherent because of the patients’ underlying fragile health.
In conclusion, the botulinum toxin injection is of use in the treatment of lower extremity spasticity. Recommended dose guidelines have been published. Complications are rare but serious and are seen most frequently in the GMFCS 5 population.
When evaluating a child with CP for surgical intervention, a few general principles must be kept in mind. Speaking frankly with the family about the goals of surgery and the expected postoperative course is important. Frequently, the family will have unrealistic expectations and believe that surgery will “cure” the child and help him walk normally. It must be explained that CP is a brain disease and that orthopaedic surgery for CP will make the child walk differently, hopefully better, but will not make the child walk “normally.”
Experts recommend combining multiple tendon surgeries and osteotomies into a single surgical event (SEMLS). Rang advised avoiding “birthday surgery,” or hospitalizing the child every year for yet another soft tissue surgery or osteotomy. Aside from avoiding repeated hospitalizations as one joint is released at a time, contractures present at one joint affect the position and movement of the rest of the extremity, so correcting all concomitant contractures and bony malalignment simultaneously during one surgery is important to avoid recurrence or overcorrection of deformity. Because gait changes and matures until approximately 7 years of age, when feasible it is wise to avoid surgery until this time. At this age, multiple levels can be treated simultaneously to optimize the ambulatory skills of the patient, followed by a single aggressive course of physical therapy to maximize the benefits from surgery. This surgical approach has been termed “single-event multilevel surgery.” The literature supports the fact that well-executed surgery addressing all contractures can result in stabilization of patients’ GMFCS level, thereby preserving their function or in some cases resulting in an improved GMFCS level at follow-up (i.e., improving their gait closer toward normal).
In some patients, surgery cannot be delayed until the age of 7 years. Young children with hip subluxation should undergo surgery when the problem is first recognized to improve coverage of the hip. At younger ages, hip surgery is often less extensive and may consist of only soft tissue release, whereas in older children, additional osteotomies of the femur and pelvis are required.
For other children who are nearly ambulatory but whose progress has been halted by contractures in the lower extremities, earlier surgery may allow them additional range of motion to make walking less cumbersome. Adductor release for scissoring and gastrocsoleus surgery with or without hamstring lengthening in these younger children may be indicated. Parents should be forewarned about recurrent contractures requiring additional surgery in the future.
Studies have found that the natural history of gait disturbances in patients with CP is gradual deterioration beginning at 12 years of age. Decreased ankle, knee, and hip sagittal-plane motion, decreased cadence, and slower walking speed have been documented over time in patients who did not undergo intervening surgical procedures. It is common for crouch, contractures, and out-toeing to worsen in the early teen years. Surgery may offer some benefit, but realistic goals must be discussed. Surgery to maintain ambulation can be successful in many children who are GMFCS 3; however, those patients who are GMFCS 4 (therefore nonambulatory) prior to surgery are unlikely to achieve maintenance or restoration of mobility, including even standing transfers, with extensive surgery.
A word of warning regarding tone and its effect on postoperative results is warranted. Best results following SEMLS surgery are seen in patients with pure spasticity. Those patients with dystonia, although preoperatively resembling their spastic counterparts, may experience unpredictable results such as early recurrence or worsening of gait in a subset of cases.
Patients with CP experience latex allergy with increased frequency, and for those with suspected latex allergy, a latex-free surgical environment is necessary. Allergy testing is available for latex products. At-risk children are those who have undergone multiple previous operations or who have indwelling latex devices such as gastrostomy tubes or ventriculoperitoneal shunts. ,
Antiseizure medications may produce alterations in clotting. Increases in bleeding times and decreased platelet counts are known side effects of some antiepileptic agents. Preoperative evaluation of clotting parameters is recommended for major operations such as hip reconstruction or spinal fusion. A routine clotting profile consisting of a prothrombin time and partial thromboplastin time will miss bleeding time abnormalities. Platelet function assays can help identify these bleeding disorders.
Postoperative pain control is difficult in patients undergoing hip reconstruction, spinal surgery, and multilevel tendon surgery. We use short-term inpatient continuous epidural infusions at our hospital and find that if the pain is well controlled, problems with muscle spasm are lessened. Oral medication often needs to include both pain control medications and muscle relaxants such as diazepam.
Weakness is a frequent short-term sequela of lower extremity surgery in those with CP. Persistent diminished muscle strength has been documented 1 year following surgery. Plans should be made for frequent postoperative physical therapy beginning shortly after surgery with attention given to muscle strengthening. If at all possible, the patient should be kept ambulatory or weight bearing after the operation. Prolonged time in a wheelchair adds to the overall weakness of the limbs. Patients with osteotomies are the exception to this rule. In these children, weakness may be even more of a problem once casts are discontinued. A few centers have exhibited new enthusiasm for minimally invasive surgery to correct contractures in children with CP. Percutaneous releases with safe intramedullary fixation of rotational osteotomies may result in less loss of strength in the postoperative period. However, because it is imperative that safety be ensured, percutaneous releases are not applicable in all situations.
A trend toward less immobilization after soft tissue surgery has emerged. In our practice, short-leg casts are still used after Achilles tendon lengthening, but knee immobilizers rather than casts are becoming more frequently used after hamstring lengthening, and removable abduction bars are preferable to Petrie casts after adductor release. The goal of soft tissue surgery is greater range of motion, so avoiding overimmobilization and secondary stiffness is logical.
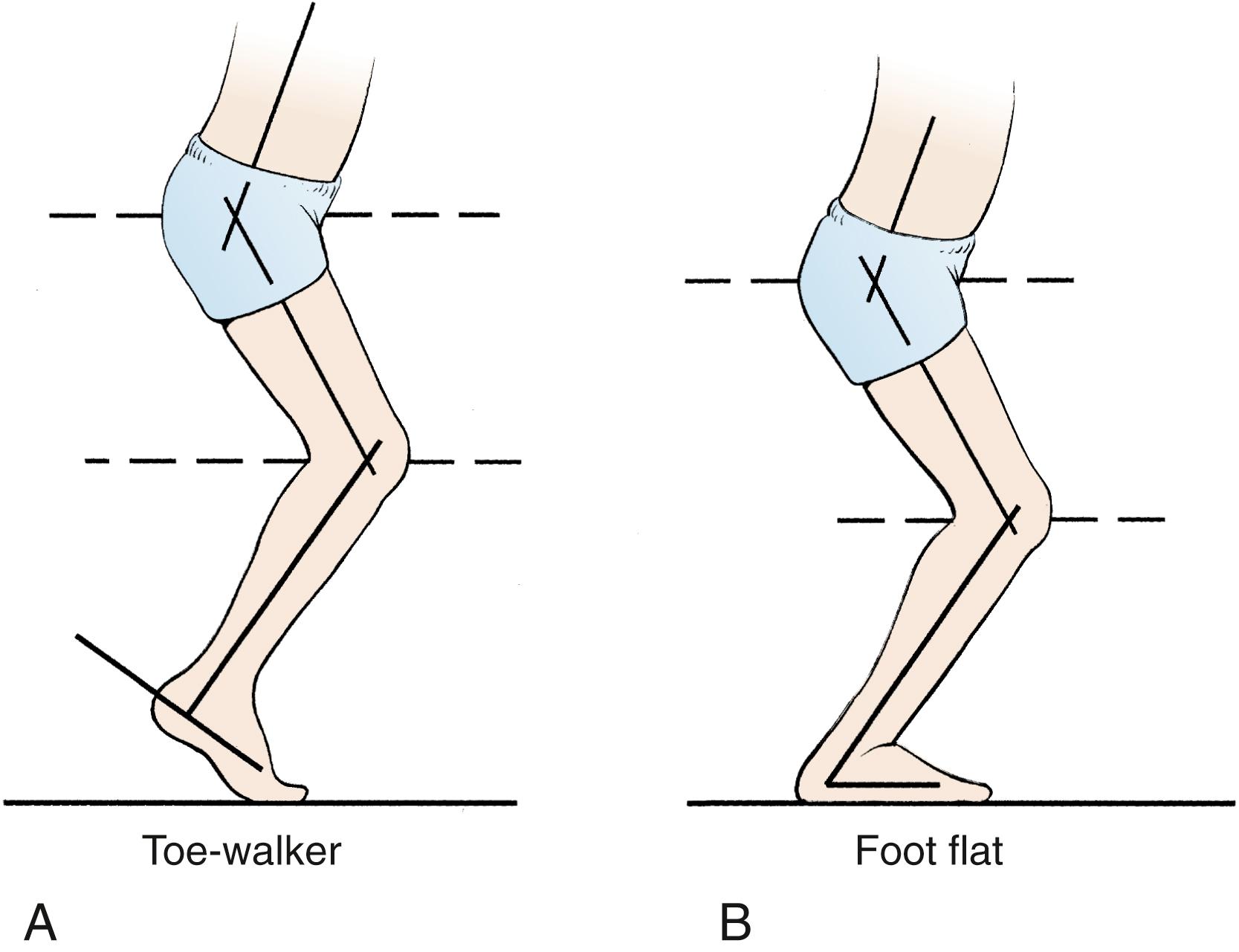
Equinus is defined as increased plantar flexion secondary to a plantar flexion contracture or dynamic plantar flexion secondary to overactivity of the gastrocsoleus during gait ( Fig. 31.16 ). Patients who walk on their toes often have equinus, but some may be on their toes as a consequence of crouch at the hip and knee and may in fact have a neutral ankle position. The physician must differentiate these two groups of children.
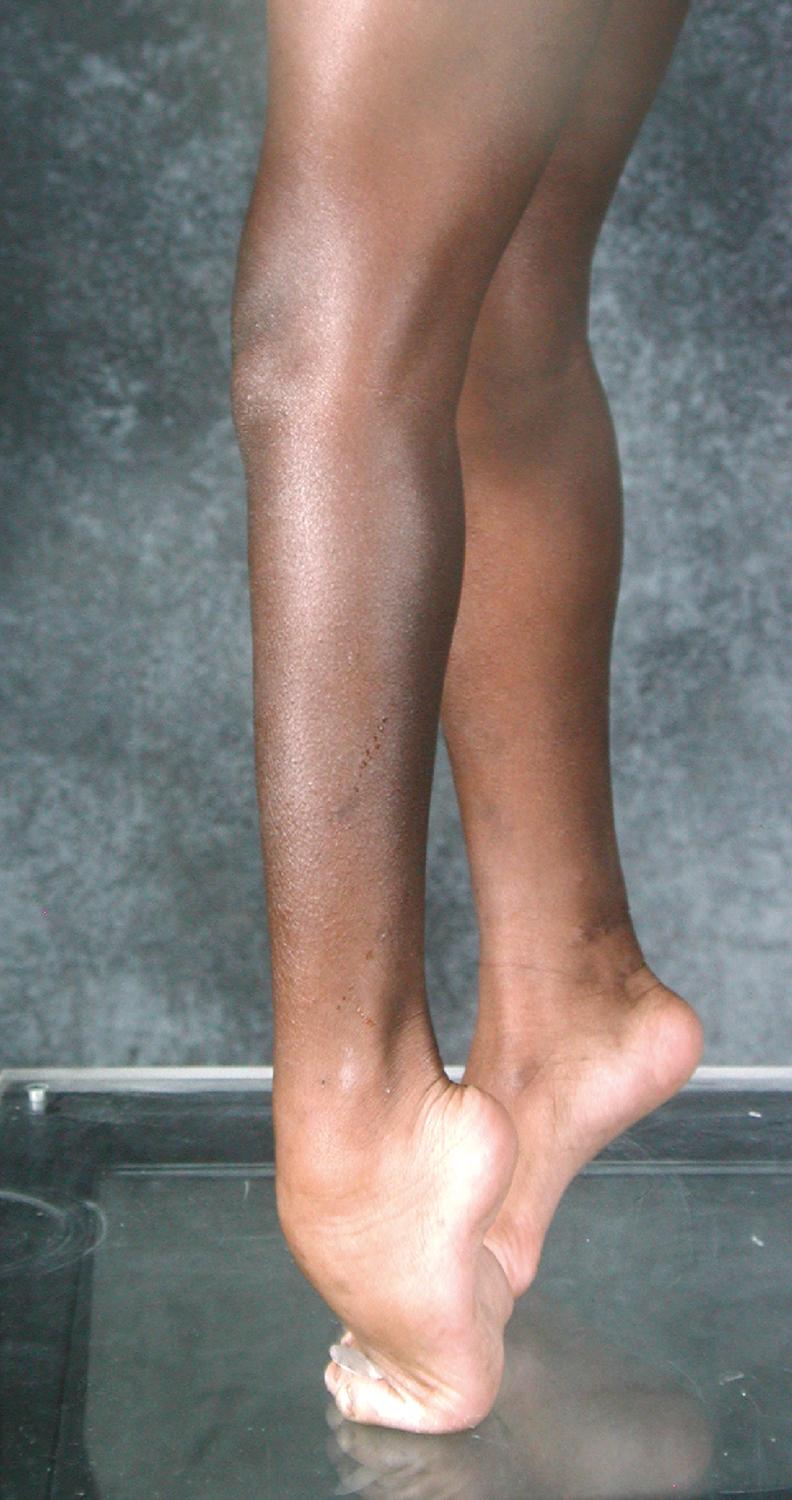
Not all children who walk on their toes have CP. Idiopathic toe-walking, frequently caused by a congenitally short Achilles tendon, is a condition in which otherwise neurologically normal children walk on their toes. Unlike children with CP, these children are not developmentally delayed and learn to walk on time, have normal knee motion during gait, have no neurologic signs of spasticity, and have normal reflexes. Gait studies looking at kinematic and EMG data have attempted to differentiate the two diagnoses on these grounds, , but a thorough history and physical examination by an experienced clinician are really all that is needed in most children.
Another condition that produces toe-walking is muscular dystrophy. The condition should be suspected in any young boy who walks on his toes and has a normal birth history. A delay in the age of walking is frequently seen in patients with Duchenne muscular dystrophy, so the developmental history may not differentiate it from CP. Testing for the Gower sign by having the child rise up rapidly from a sitting position on the floor will accentuate the presence of proximal muscle weakness. If the test for the Gower sign is suspicious, laboratory evaluation for serum muscle enzymes (creatine phosphokinase) is indicated.
Clinical examination of a child with equinus associated with CP reveals an inability to fully dorsiflex the ankle. If the ankle can be passively dorsiflexed with the knee bent to 90 degrees but cannot be dorsiflexed with the knee extended, it is believed that the gastrocnemius is tight but the soleus is not contracted (Silfverskiöld test; Fig. 31.17 ). Many have used this test to determine which type of surgical lengthening to perform. If the ankle has sufficient dorsiflexion on passive range-of-motion examination, the equinus is termed dynamic , and surgical treatment is not usually needed.
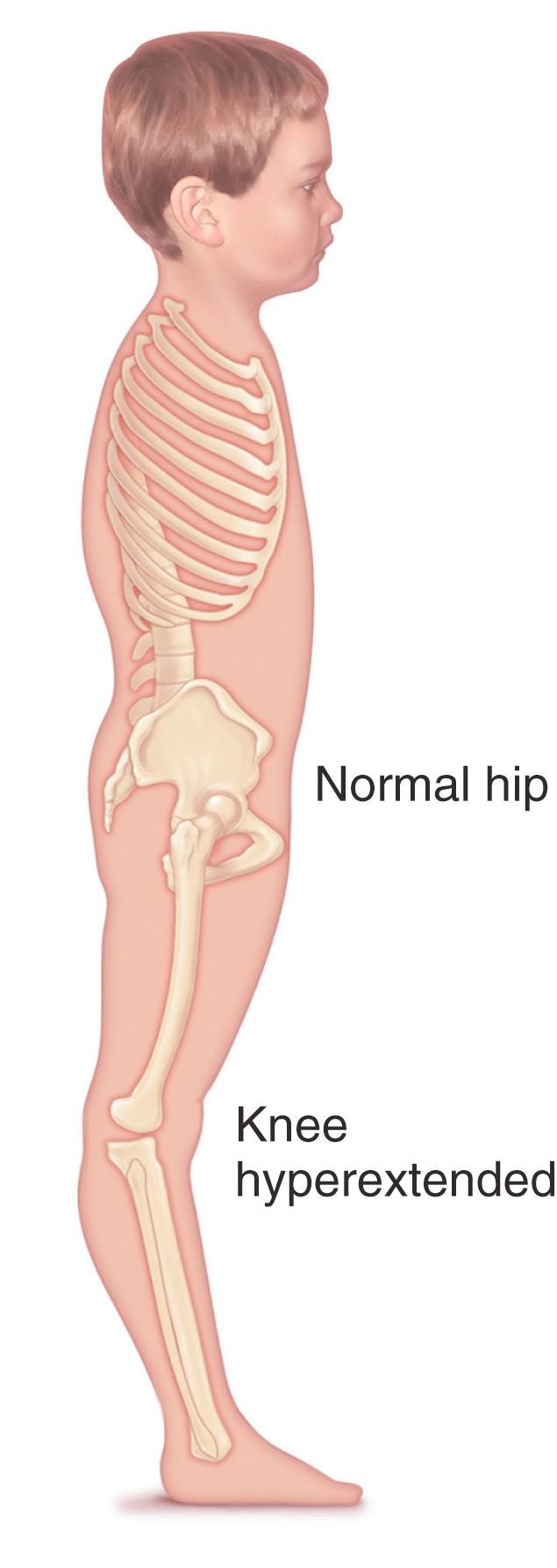
Equinus interferes with forward progression of the tibia over the foot during stance phase and therefore shortens stride length. Because the ankle is already plantar-flexed at terminal stance, little push-off power is generated and the gait is less efficient. If the tibialis anterior is unable to lift the foot to neutral during the swing phase, footdrop results, with possible problems in clearing the foot in the swing phase and tripping. Lack of normal swing-phase ankle dorsiflexion leads to forefoot contact on foot strike at the beginning of stance phase. Knee disturbances also result from ankle equinus. Genu recurvatum is seen when the tibia is unable to move forward over the plantigrade foot because of tightness in the gastrocsoleus, so the knee thrusts backward during midstance ( Fig. 31.18 ). The body and femur continue to move forward over the stationary tibia, and an extensor moment is produced at the knee. This aligns the ground reaction force anterior to the knee, thereby reducing demands on the quadriceps and improving the stability of the knee. Likewise, compensatory knee flexion can be seen during stance phase in patients who walk on their toes.
Over time, ankle equinus leads to valgus positioning of the hindfoot and stretching of the plantar arch (midfoot break) . Although the foot may appear plantigrade, the talar head is very prominent in the longitudinal arch and the calcaneus is actually in equinus. Pain and calluses form over the head of the talus ( Fig. 31.19 ). Hallux valgus can develop in response to the valgus positioning of the foot.

Surgical treatment of equinus is lengthening of the Achilles tendon, the gastrocnemius/soleus aponeurosis, or selective gastrocnemius fascial recession. Advocates of gastrocnemius recession and aponeurotic lengthening state that these operations preserve or even increase push-off power more than Achilles tendon lengthening does by not involving the soleus muscle or tendon. , Immobilization is minimized after gastrocnemius recession, whereas casting is necessary after Achilles tendon lengthening. The risk for overcorrection into a calcaneus gait is negligible. Perry and Hoffer believed that gastrocnemius recession should be performed when a Silfverskiöld test performed under anesthesia is positive and dynamic EMG shows more abnormality of the gastrocnemius than of the soleus during gait.
Those who prefer lengthening of the Achilles tendon recognize a greater rate of recurrence of equinus after gastrocnemius recession, which has been as high as 48% in some studies. They also state that Achilles tendon lengthening may be done percutaneously, unlike gastrocnemius recession. , In one comparative gait analysis study, no significant difference was found between patients who had undergone gastrocnemius recession and those who had undergone lengthening of the Achilles tendon. In another study, if equinus was an isolated gait abnormality (i.e., the knee did not lack extension during stance phase), isolated percutaneous Achilles tendon lengthening to a plantigrade position was very successful in improving ankle kinematics and kinetics without resulting in knee abnormalities or a calcaneus gait. A higher risk for calcaneal overcorrection has been noted after percutaneous Achilles tendon lengthening than after Baker gastrocsoleus aponeurotic lengthening. At 9 years of follow-up, a calcaneus gait had developed in only 10% of 44 spastic diplegics treated by gastrocsoleus aponeurotic recession. In contrast, Dietz and colleagues studied 79 children who had undergone Achilles tendon lengthening. At an average of 7 years’ follow-up, they found that a calcaneus gait did not develop in the hemiplegic patients whereas 41% of the diplegic and 50% of the quadriplegic patients did walk with excessive knee flexion and ankle dorsiflexion. Vuillermin and colleagues side with Dietz. They studied the prevalence of crouch in two periods, the first when Achilles tendon lengthening was commonly performed and the second when gastrocnemius recession replaced Achilles tendon lengthening. They found a marked decrease in the development of crouch gait in the second cohort of patients. Confounding factors were the use of gait analysis for preoperative planning and adoption of the SEMLS approach in the group treated with gastrocnemius recession. In longer-term follow-up, the same group has found that 12.5% of children treated with either a gastrocnemius recession or selective gastrocsoleus lengthening did require repeat surgery for equinus, but only 2.5% went on to develop an overcorrected calcaneus gait, strengthening their recommendation for gastrocnemius selective lengthening. The Melbourne group, strong advocates of selective gastrocsoleus lengthening, found that 35% of patients who had conservative surgical treatment of their equinus rather than tendon achilles lengthening (TAL) did have recurrent equinus at midterm follow-up, but that mild equinus was actually favorable in maintaining the knee in extension. Controversy on this subject continues, with surgeons split between those who believe that the adolescent crouch gait is the result of weakness from previous Achilles tendon–lengthening surgery and those who believe it to be due to growth and untreated hamstring spasticity. Increasing knee flexion (crouch gait) has been documented to develop over time in patients who have not previously undergone orthopaedic procedures. Meta-analysis of the existing literature failed to show a scientific preference for one procedure over another, although it was agreed that recurrence was most likely in younger patients and that overcorrection (or talipes calcaneus) was unlikely in hemiplegics when compared with diplegics.
Gastrocnemius recession may be done with the techniques of Strayer, Baker, or Vulpius. In a Vulpius procedure, the aponeurosis of the gastrocsoleus is divided in chevron fashion and the midline fibrous septum of the soleus is transected, but the soleus muscle fibers are not disturbed ( Fig. 31.20 ). The fascia slides on the underlying soleus. , The Melbourne group advocates for a transverse cut rather than a chevron cut, with emphasis on division of the midline raphe. The cut in the gastrocnemius is transverse and more proximal in the Strayer procedure, which does not lengthen the soleus whatsoever ( Fig. 31.21 ). In the Baker technique, the gastrocsoleus aponeurosis is cut in tongue-in-groove fashion and dissected free from the underlying soleus muscle. The fascia is allowed to slide on the underlying muscle, thereby increasing the overall length of the muscle, and the four corners of the aponeurosis are sutured in the lengthened position ( Fig. 31.22 ). These surgeries can be divided into groups based on the zone in which lengthening occurs. Zone 1 is from the gastrocnemius origin to the distal end of the medial gastrocnemius. Zone 2 is from the distal end of zone 1 to the end of the soleus muscle belly. Zone 3 represents the Achilles tendon. The Strayer lengthening occurs in zone 1 and the Vulpius and Baker lengthenings in zone 2. ,

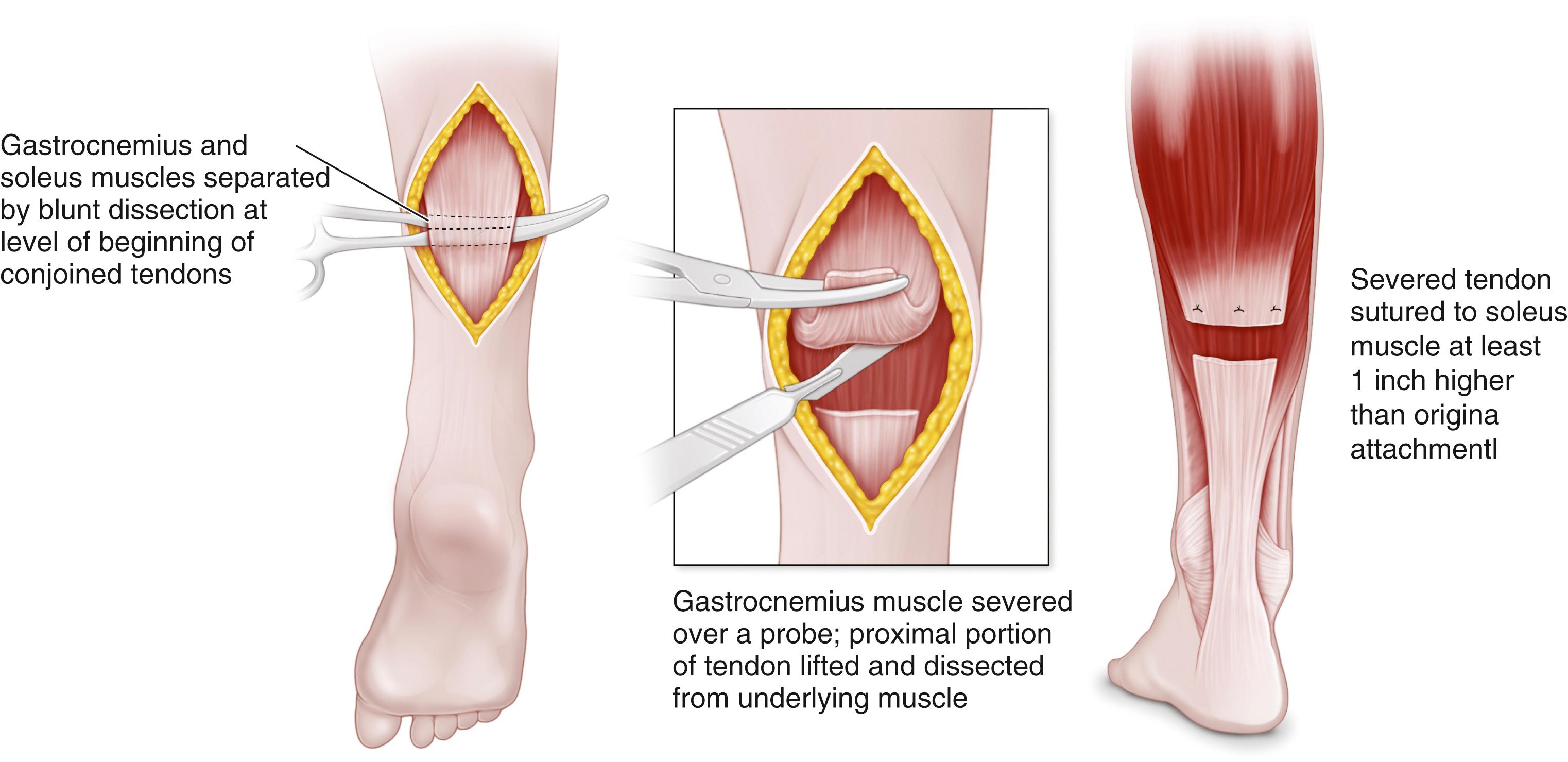
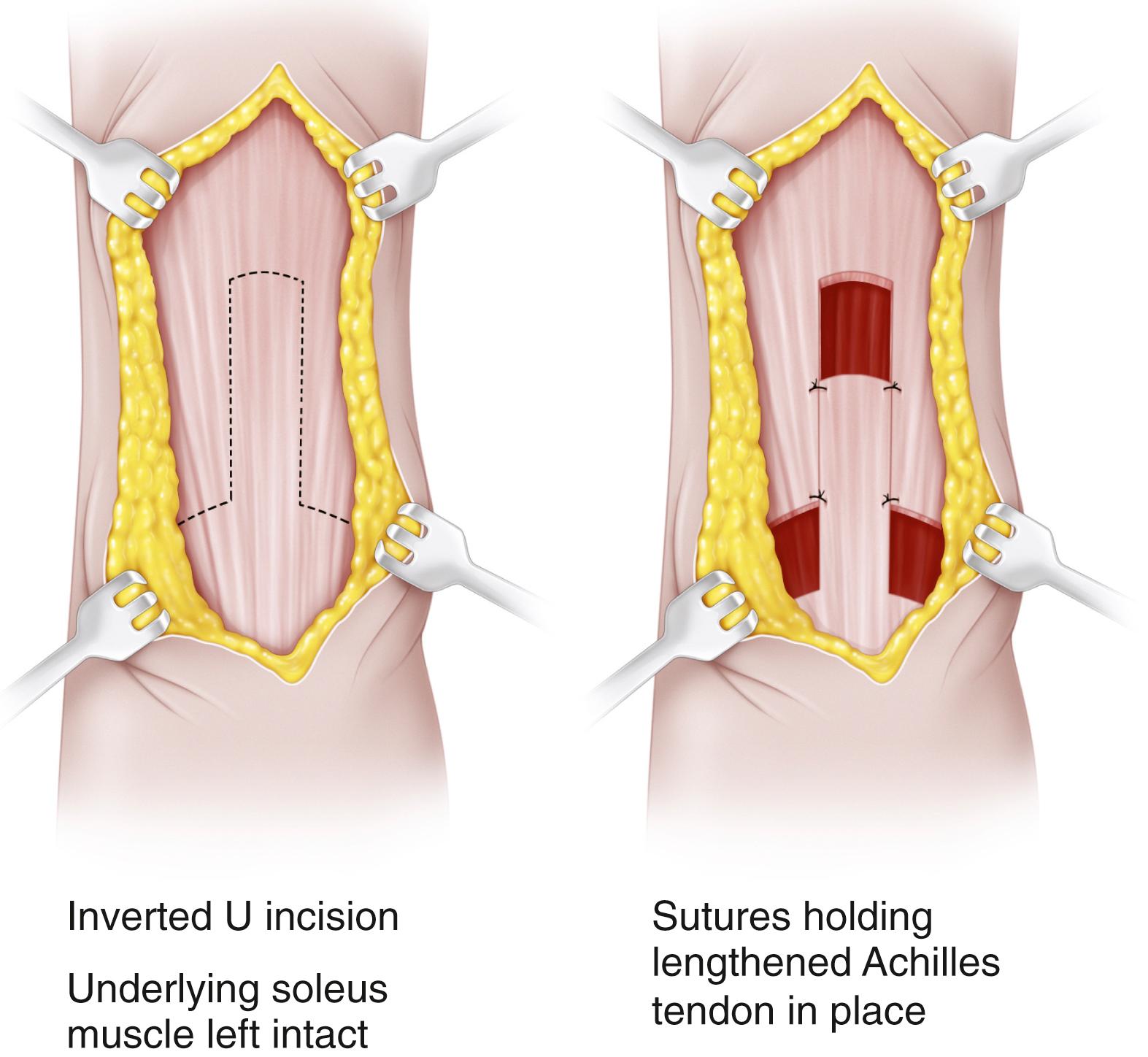
Achilles tendon lengthening may be performed by open or percutaneous techniques. In the open technique, a longitudinal incision is made just lateral to the Achilles tendon. The tendon is lengthened in Z-fashion and repaired with stout nonabsorbable suture ( Fig. 31.23 ). The tendon must be repaired with sufficient tension to avoid postoperative calcaneus gait. , , The ankle is then immobilized in a short-leg cast for 6 weeks. An open sliding lengthening of the Achilles tendon also may be performed; this procedure does not require suturing of the tendon.
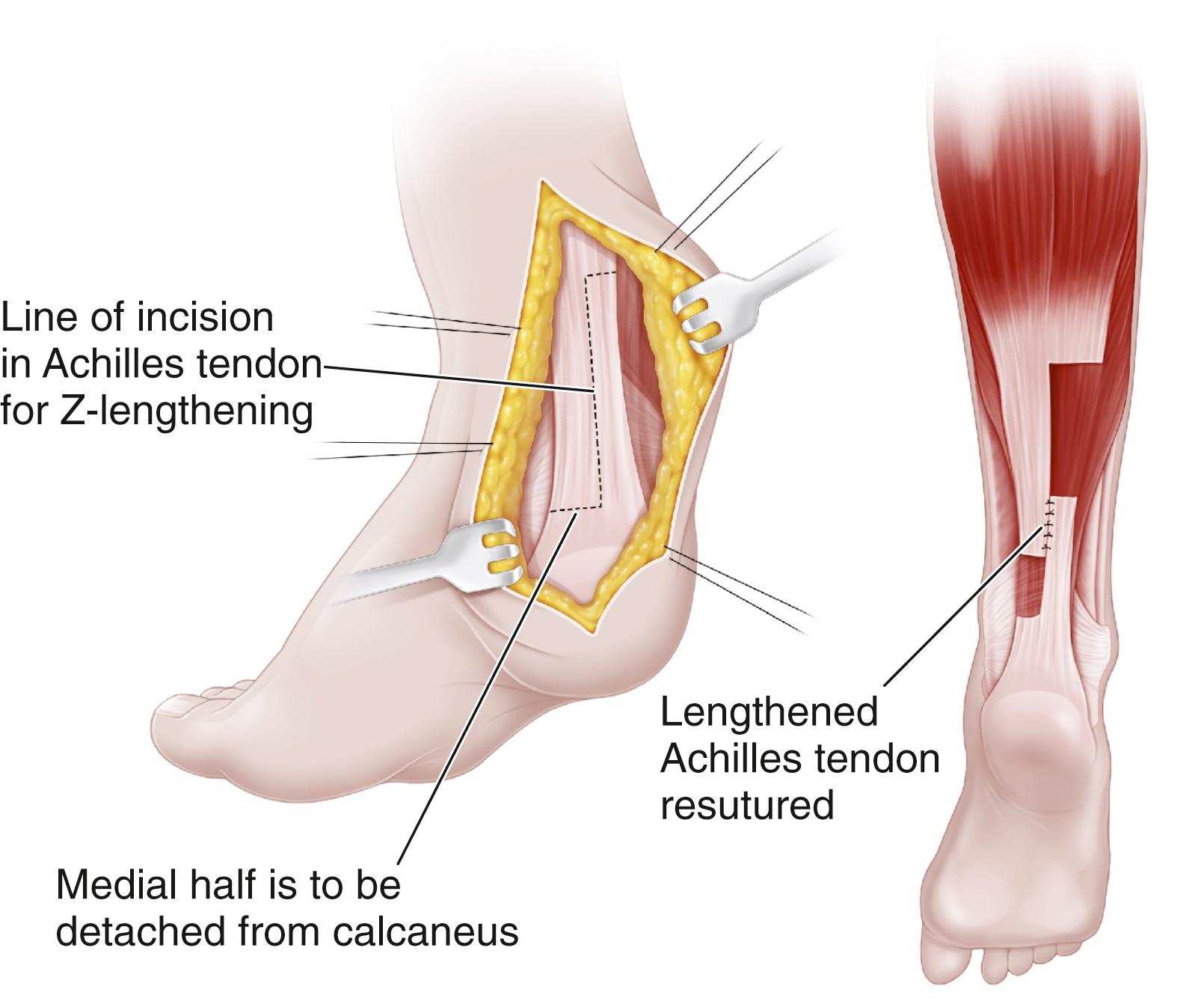
The percutaneous technique may require two or three cuts in the tendon. We prefer the two-cut technique described by White. Just proximal to the calcaneal insertion, a little more than half the tendon is divided medially by inserting a scalpel longitudinally into the center of the tendon, turning the blade out medially, and ballotting the tendon down onto the blade. The lateral half of the tendon is then divided by using a similar maneuver more proximally. The heel is inverted into varus, and the ankle is dorsiflexed to the neutral position (see Plate 31.1 ) . The tendon can be heard lengthening as the ankle is gently manipulated. It is not unusual to hear a pop as the tendon slides and lengthens, although we prefer more gradual and gentle lengthening to prevent overlengthening of the tendon. FLOAT NOT FOUND
It is important to check the integrity of the tendon after percutaneous lengthening. The calf is squeezed while the surgeon observes the ankle. If the ankle plantar-flexes when the calf is squeezed, the tendon can be presumed to be intact within the tendon sheath, and no sutures are needed. Steri-Strips are applied to the two stab wounds, followed by a cast. If no plantar flexion is noted, the tendon may have been completely separated by the lengthening procedure and open suture repair will be needed to reestablish continuity of the tendon. This is a very rare occurrence; usually, the percutaneous technique proceeds without complication. Again, a short-leg cast is applied and worn for 6 weeks, with weight bearing encouraged.
Hoke described a three-cut technique for percutaneous heel cord lengthening. After making one lateral and two medial cuts in the tendon, the ankle is dorsiflexed as the tendon slides on itself and lengthens. Postoperative care is identical to that after the two-cut technique.
After either heel cord lengthening or gastrocnemius recession, patients may tend to flex their knees after surgery. This is caused by muscle spasm in the hamstrings and gastrocnemius (which crosses the knee joint). We find that use of a knee immobilizer for the first few days to weeks can help reduce the amount of muscle spasm and prevent the development of a postoperative knee flexion contracture.
After the short-leg cast is removed, an AFO is prescribed postoperatively for most patients. Many children who had footdrop in the swing phase preoperatively will have improved anterior tibialis function in the swing phase postoperatively, while other continue to have footdrop after heel cord lengthening. Patients and families should be forewarned of the potential continued need for orthoses after surgery to support the foot and improve toe clearance in the swing phase. Many younger children benefit from the stability of an AFO postoperatively, whereas in most older children and teens, the goal of heel cord lengthening is to rid them of braces on their feet, so they are reluctant to use an AFO after surgery.
Complications are rare after heel cord lengthening or gastrocnemius recession. Recurrent equinus is the greatest risk; it occurs in approximately 15% to 35% and correlates strongly with the age of the patient at surgery. Children who undergo heel cord lengthening at 4 years or younger are particularly at risk for recurrence. , , It is impossible to know whether it is the greater amount of growth left in younger children after heel cord surgery that leads to the high rate of recurrence, or whether it is preselection of young children with the greatest tone to undergo surgery at an early age after failure of nonoperative treatment that leads to recurrent contracture. In a long-term study by Rattey and colleagues, 26% of patients who had undergone Z-lengthening of the Achilles tendon required repeated lengthening for recurrent equinus at an average follow-up of 10 years. The risk for recurrent contracture was greater in patients with hemiplegia (41%) than in those with diplegia (18%), and in those with more severe preoperative equinus contractures. Grant and associates found that an inability to actively dorsiflex the foot (i.e., lack of selective motor control of the tibialis anterior muscle) preoperatively increased the risk for recurrence. Repeated surgery is possible and common. With repeated lengthening, the Achilles tendon becomes scarred and adherent, so repeated lengthening must usually be done with the open technique. Patients who have undergone gastrocnemius recession have a higher rate of recurrence than do those treated by tendon lengthening.
Calcaneus deformity, defined as excessive dorsiflexion of the ankle during stance phase, may result for two reasons. First, the tendon may simply be overlengthened or may have lost continuity. When performing lengthening, the surgeon must be careful to bring the foot just to neutral. Overstretching of the tendon can lead to excessive length and a calcaneus gait, with poor push-off and a tendency for the development of progressive crouch of the knees.
The second reason that a calcaneus gait may occur after lengthening of the gastrocnemius or Achilles tendon is an unrecognized flexion of the knee during stance phase. If the knee remains crouched and the Achilles tendon becomes longer, the ankle sags into excessive dorsiflexion during stance phase and gait loses efficiency. A meticulous physical examination before surgery in which tone and range of motion of the knee are assessed is critical. If the popliteal angle is increased and the child exhibits excessive knee flexion during stance phase or an inability to straighten the leg as the heel makes contact with the ground, postoperative calcaneus will most likely develop if the knee is not surgically treated at the same time ( Fig. 31.24 ).
The incidence of calcaneus gait as a complication of heel cord lengthening averages approximately 5%. Segal and colleagues found calcaneus on kinematic graphs in 30% of patients. Although this figure seems high in comparison to previous studies, it does highlight the need to treat all levels of deformity during surgery and avoid overlengthening at all cost.
Although most patients in whom a calcaneus gait develops have previously undergone equinus surgery, in a group of patients calcaneus will develop de novo. An adolescent diplegic patient is at greatest risk for calcaneus. Patients who gain weight (as in the adolescent growth spurt) and who have plantar flexor weakness were found on serial gait analysis to be most prone to the development of a calcaneus gait.
In our clinical practice we prefer percutaneous Achilles tendon lengthening as the surgical treatment of fixed equinus contractures and gastrocnemius fascial lengthening when the ankle can be dorsiflexed to nearly neutral with the knee flexed. Postoperative cast immobilization is used for 6 weeks, and AFOs are prescribed on an individual basis. We agree with the philosophy that “a little equinus is better than any calcaneus” and avoid overlengthening. Most important, we thoroughly evaluate the child for other joint involvement, and we typically perform surgery on multiple levels of the lower extremity at the same time (SEMLS). We do see some recurrent contractures with growth, and we reoperate when these contractures interfere with gait. Gastrocnemius fascial lengthening is performed in our center for milder dynamic equinus.
Equinovarus deformity of the foot results from muscle imbalance in which the invertors of the foot, specifically the posterior and anterior tibialis muscles, overpower the evertors (the peroneals). The gastrocnemius contributes equinus to the deformity. Patients walk on the lateral border of the inverted foot, and painful calluses may develop laterally over the fifth metatarsal. The deformity is most prevalent in patients with spastic hemiplegia ( Fig. 31.25 ).

Gait analysis shows an internal foot progression angle as a result of inversion of the foot. Ankle equinus is present, and footdrop may be evident during the swing phase. Pedobarographic assessment demonstrates decreased hindfoot pressure (secondary to equinus) and increased pressure beneath the lateral border of the forefoot and midfoot, specifically the fifth metatarsal ( Fig. 31.26 ).
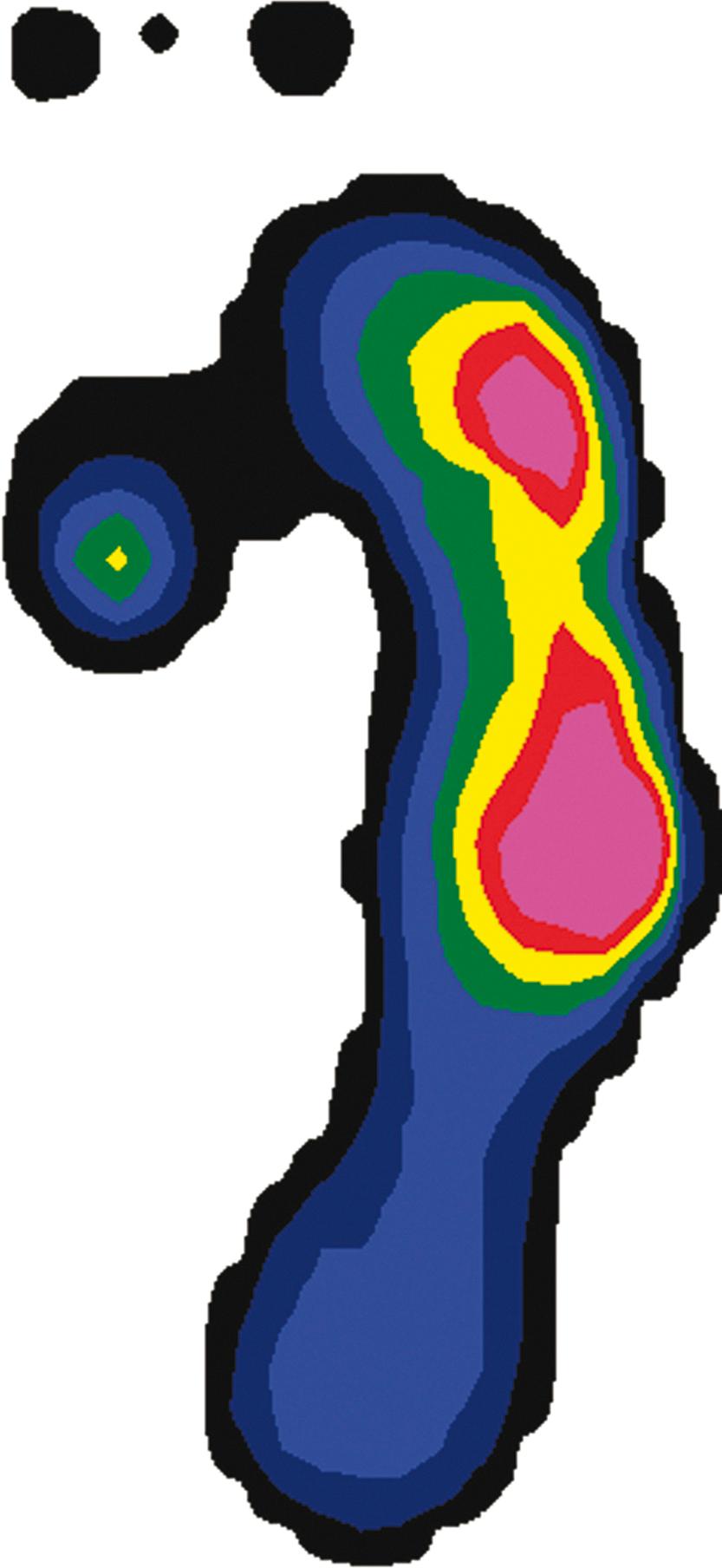
Nonoperative treatment is usually unsuccessful. A supple deformity can be braced, but if the muscles are very spastic, the orthoses can exacerbate the blisters or calluses over the lateral border of the foot. Botulinum toxin injections have been tried, but no published studies have shown long-term efficacy for equinovarus.
Surgery is indicated to improve foot contact and to relieve pain and skin changes. If the foot can be passively corrected with manipulation in the clinic to a neutral position, tendon surgery can be performed. Lengthening procedures and split transfers have been described, and more detail will be provided about these procedures. If the deformity is stiff and the foot cannot be manipulated into a plantigrade position, bone surgery will be necessary. In patients with spastic diplegia and quadriplegia, equinovalgus tends to develop later during childhood and adolescence. Young patients, specifically those younger than 8 years, who are diplegic or quadriplegic are most likely to have unsatisfactory long-term results after surgery for equinovarus. For this reason, nonoperative treatment options should be exhausted in these patients.
Surgical decision making for the soft tissue correction of spastic equinovarus focuses on distinguishing whether it is the anterior tibialis or the posterior tibialis that is the cause of the deformity. The evaluation should start in the clinic with a careful physical examination. The confusion test is helpful in this setting. The patient is asked to flex the hip against resistance while seated, and the ankle is inspected. In most children with CP the anterior tibialis fires while the hip is flexed; this is considered a positive confusion test ( Fig. 31.27 ). However, the reflex has been seen in some children without CP. The examiner should look for the position of the foot as the anterior tibialis fires. If supination of the forefoot is seen, the anterior tibialis is most likely contributing to the equinovarus deformity. If the foot dorsiflexes without supinating, the equinovarus is less likely to improve with surgery on the anterior tibialis.
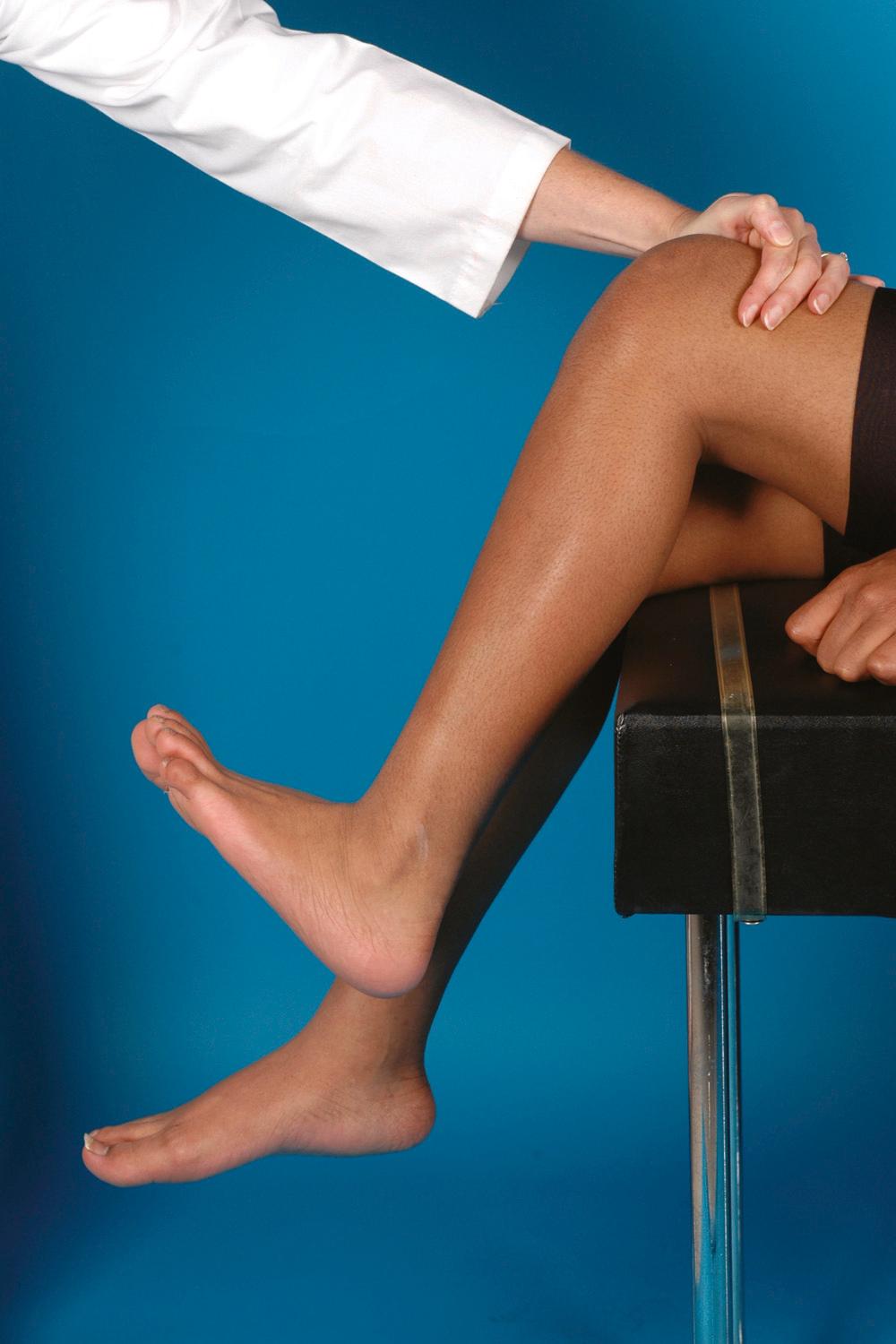
Next, the examiner feels for spasticity in the posterior tibialis muscle. Passive manipulation of the hindfoot into valgus while feeling the posterior tibialis tendon can help the physician appreciate tightness in the posterior tibialis. The examiner should look at where the varus appears to be located. Hindfoot varus is most probably caused by overpull of the posterior tibialis, whereas forefoot supination is more commonly caused by the anterior tibialis. The examiner should observe for tension in the anterior tibialis and posterior tibialis during gait; the spastic tendon may be visibly taut as the patient walks.
Gait analysis with dynamic EMG can help determine which muscle is acting inappropriately. , , Surface electrodes suffice for measuring anterior tibialis activity, but the posterior tibialis is deep, and a fine wire–needle electrode is necessary to measure its signal. The anterior tibialis should be quiet in midstance. Signal from the anterior tibialis throughout stance phase is indicative of overactivity ( Fig. 31.28 ). The posterior tibialis serves to stabilize the foot during stance phase. Early-onset signal and prolongation of the activity during the swing phase indicate an abnormality in control of the posterior tibialis. Nearly one-third of patients with equinovarus will show inappropriate activity of both the anterior and posterior tibialis muscles during gait.
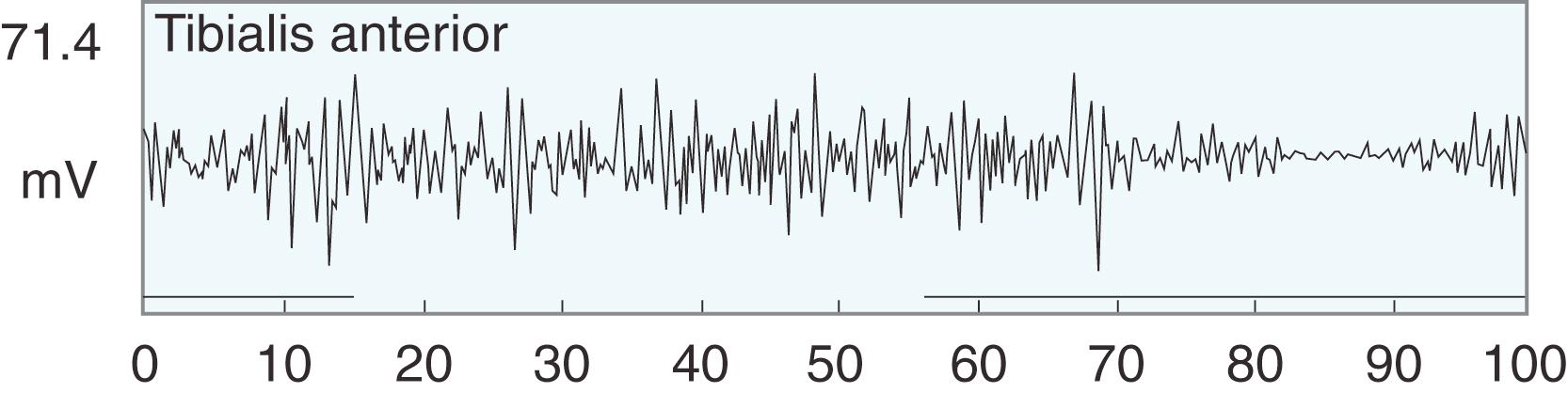
Some patients with equinovarus feet have preexisting footdrop during the swing phase. When this is seen, either in the clinic or during gait analysis, further weakening by anterior tibialis surgery may lead to significant footdrop postoperatively, and an AFO will be needed.
Surgical options for equinovarus deformity start with lengthening of the posterior tibialis tendon, usually done in conjunction with an Achilles tendon–lengthening procedure in young patients with mild varus in addition to equinus. The tendon may be Z-lengthened distally, or preferably intramuscular lengthening can be performed in the distal third of the calf ( Fig. 31.29 ). The patient is placed in a short-leg cast postoperatively for approximately 6 weeks. Complications consist of recurrence of the deformity and the development of postoperative valgus. The posterior tibialis is weakened by the lengthening, but rebalancing of forces around the foot does not occur to the same extent as with a tendon transfer. Yet in many milder cases this is sufficient to obtain a plantigrade foot.
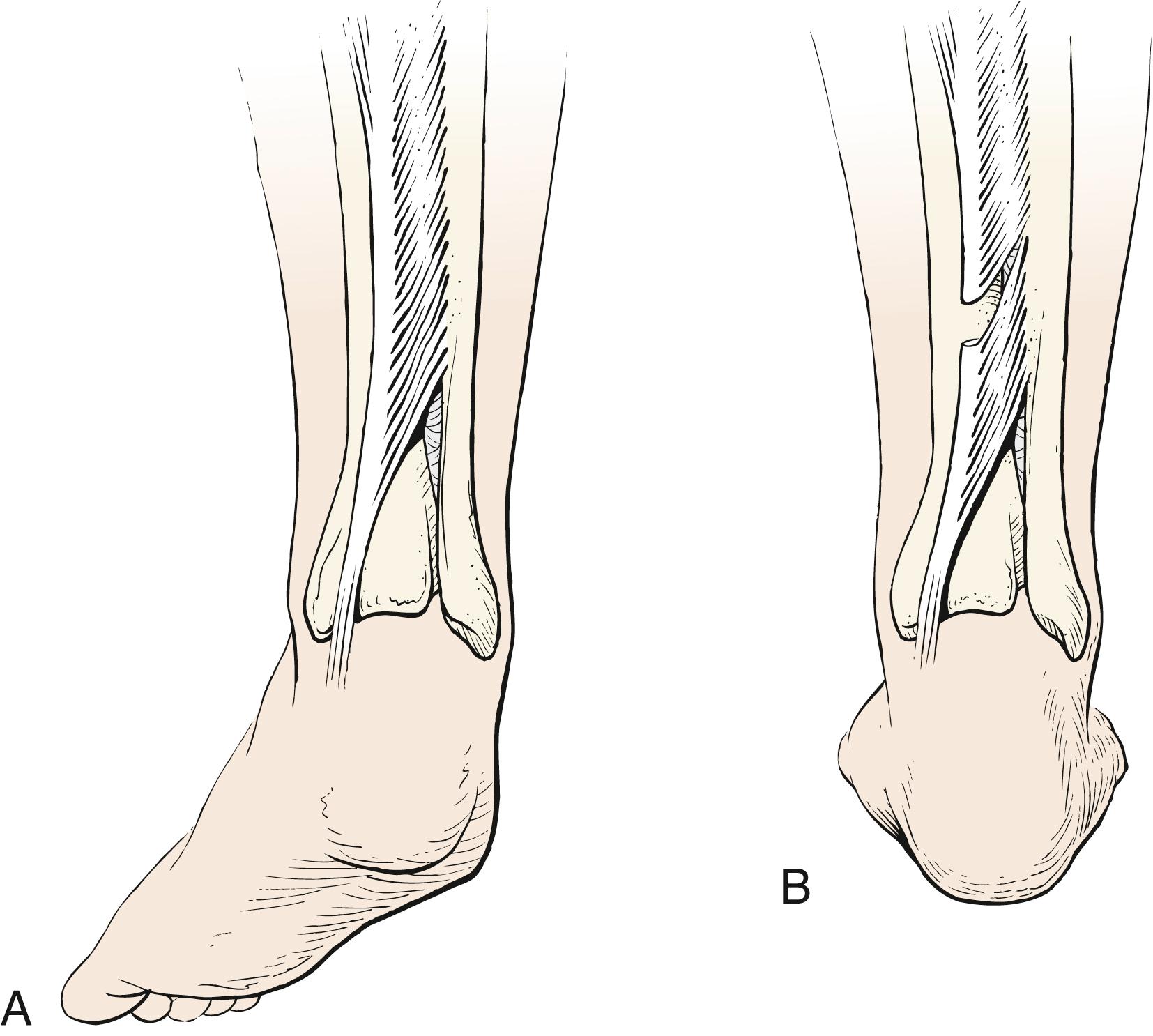
Anterior transfer of the entire posterior tibialis tendon has been performed as treatment of equinovarus. , Calcaneovalgus can be a disastrous result of this procedure and occurs in up to 68% of patients. , We do not perform this operation in children with CP at our hospital.
Overcorrection with complete tendon transfers led to the popularization of split tendon transfers in children with CP, one of the most common being that of Green. In this procedure the posterior half of the posterior tibialis tendon is detached from its insertion, split proximally to a level just proximal to the ankle, rerouted posterior to the tibia and fibula, and then woven into the tendon of the peroneus brevis ( Fig. 31.30 ). The remaining posterior tibialis tendon attached to the navicular is then balanced by the lateral half of the transferred tendon, which acts as an evertor. Four incisions were used in the original description of the operation. Usually, an Achilles tendon–lengthening procedure is also required to treat the equinus.
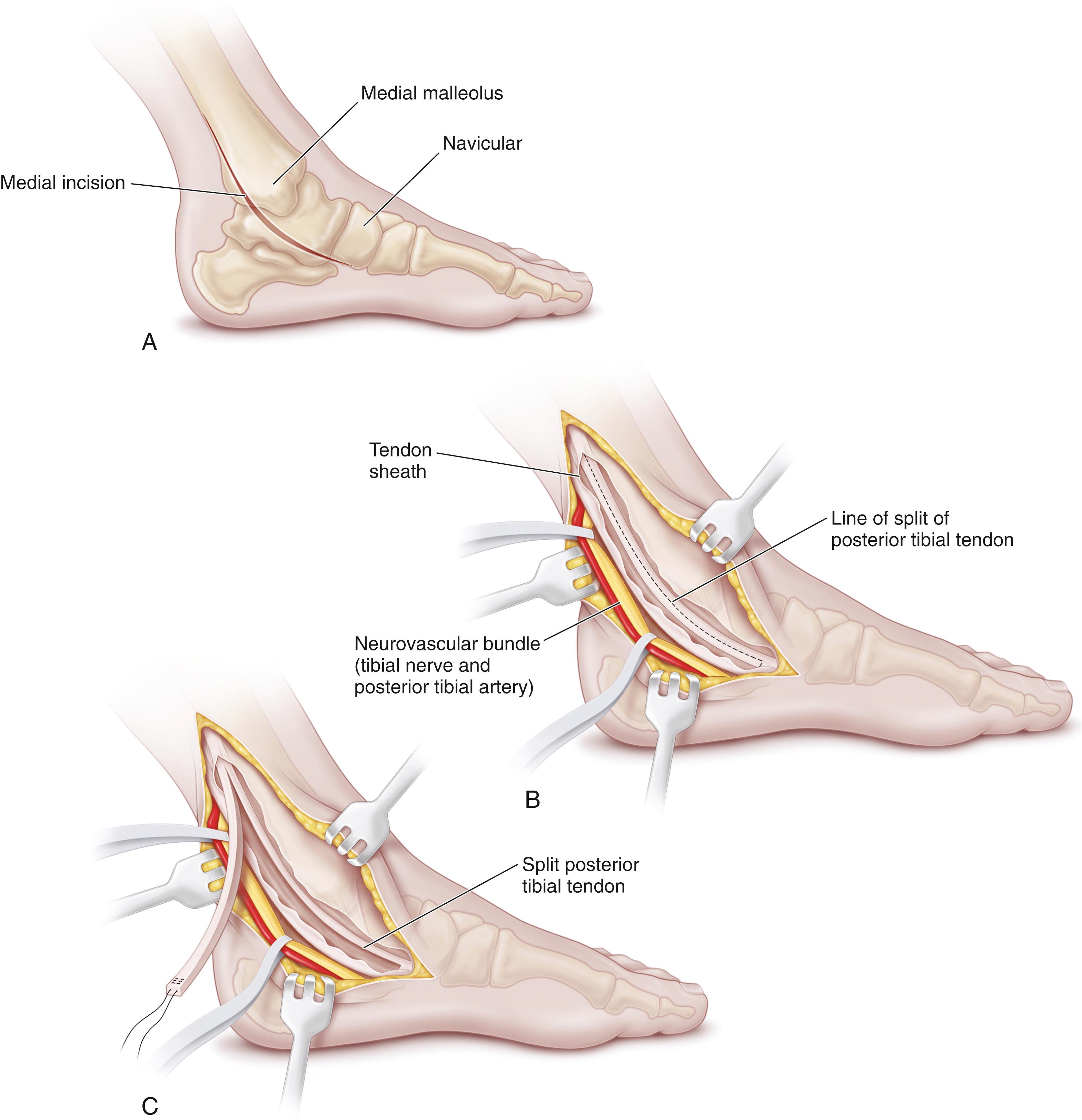
The prerequisite for a successful split posterior tibialis tendon transfer on gait analysis is swing-phase activity seen on dynamic EMG. Yet several authors have reported successful series of split posterior tibialis tendon transfers in which preoperative or postoperative EMG was not performed. ,
Often, patients can walk without orthoses after this tendon transfer. As with any tendon transfer, the deformity must be passively correctable preoperatively for postoperative correction to be expected. Recurrent varus may be a complication after split posterior tibialis tendon transfer and generally results from inappropriate selection of patients with a deformity that is too rigid. Green stated that overcorrection into valgus has not occurred in their patients, but we have occasionally seen overcorrection in older patients in our outpatient clinics several years after tendon transfer.
Transferring the anterior half of the posterior tibialis tendon anteriorly through the interosseous membrane to the dorsum of the foot is a variation of the classic posterior tendon transfer. It is believed that this split transfer might assist dorsiflexion with less calcaneus deformity than is the case with transfer of the entire tendon because the posterior half of the tendon remains intact. Although studies have shown promising early results, this procedure has yet to be adopted universally. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here