Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Coronavirus Disease 2019 (COVID-19) is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The virus infects its host via the spike subunit of SARS-CoV-2, which engages angiotensin-converting enzyme-2 (ACE2) on the surface of cells as an entry receptor ( Fig. 50.1 ). SARS-CoV-2 has tropism for the respiratory tract, given the high expression of ACE2 in multiple epithelial cell types of the airway. As a result, viral infection typically manifests as upper airway symptoms, pneumonia, and/or acute respiratory distress syndrome (ARDS). In addition, multiorgan injury with cardiac, renal, neurologic, and endocrine involvement has also been described. Pathophysiologic mechanisms include direct viral toxicity, endothelial cell damage and thromboinflammation, dysregulation of the immune response, and dysregulation of the renin-angiotensin-aldosterone system (RAAS).
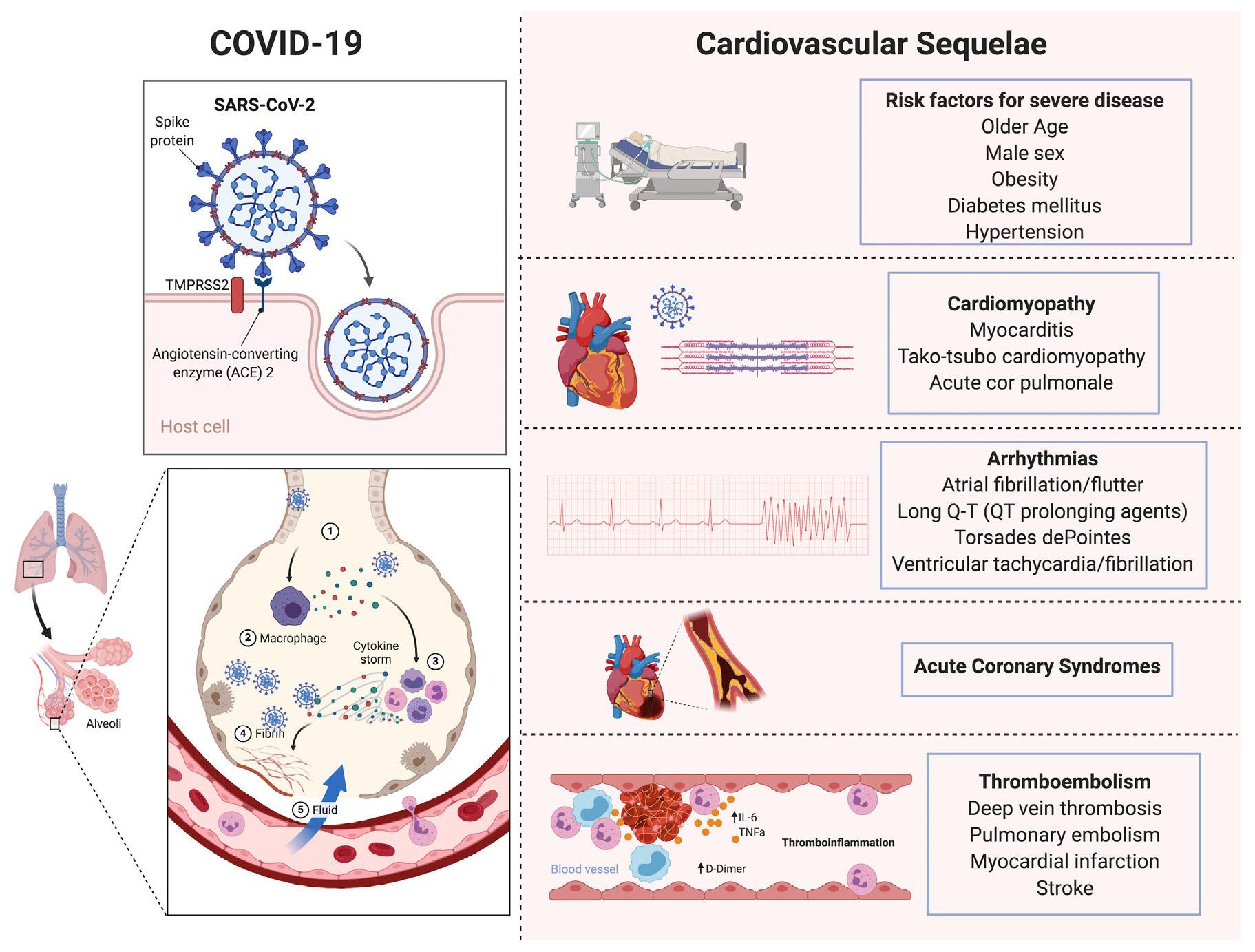
The heart and blood vessels have high concentrations of ACE2 and as such facilitate cardiovascular tropism of SARS-CoV-2. Cardiovascular sequelae of COVID-19 include myocardial injury, myocarditis, arrhythmias, thrombotic cardiac complications including myocardial infarction and stroke, biventricular cardiomyopathy, and/or acute cor pulmonale particularly in the setting of ARDS or pulmonary embolism. Arterial and venous thromboembolism have also been well documented. Furthermore, features of atypical Kawasaki disease have been noted in a small proportion of children and have been termed as multisystem inflammatory syndrome in children (MIS-C). It is thought to be immune-mediated and manifests with a spectrum of cardiovascular features, including vasculitis, coronary artery aneurysms, and cardiogenic shock.
Risk factors for severe illness with COVID-19 include older age, male sex, and comorbidities including hypertension, heart failure, arrhythmias, diabetes mellitus, obesity, chronic kidney disease, chronic lung disease, and immunocompromised states like solid organ transplant and cancer.
Preexisting cardiovascular disease predisposes patients to severe illness, and reasons behind this are not fully understood. It is hypothesized that dysregulation of the RAAS and alterations in multiple cytokines, chemokines, and adipokines, including increased proinflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukin (IL)-6, IL-8, leptin, and adiponectin, as observed in obesity and metabolic syndrome, all potentially exacerbate the inflammatory response seen in this disease. Furthermore, poor physiologic reserve at baseline can predispose patients with preexisting cardiac disease to more severe cardiovascular sequelae.
In published autopsy series of 39 patients, SARS-CoV-2 has been isolated from the myocardium in 24 patients, and 16 of them were found to have viral titers higher than 1000 copies per microgram RNA. Occasional case reports have documented the presence of a few interstitial inflammatory infiltrates, but these have typically not been reported in myocardial cells.
Lymphopenia is a cardinal finding of COVID-19. Other laboratory manifestations of COVID-19 may include elevated inflammatory markers including erythrocyte sedimentation rate, C-reactive protein, ferritin, interleukin-6, and lactate dehydrogenase. Coagulation indices including D-dimer and fibrinogen, and prolonged prothrombin time and partial thromboplastin time may also be observed.
Of note, cardiac biomarkers including troponins and B-natriuretic peptide may be elevated in patients with serious illness, particularly in the setting of a hyperinflammatory response. Elevation of troponins has been reported in at least one in five hospitalized patients with COVID-19, and in up to one in two among those with preexisting cardiovascular disease. Acute nonischemic myocardial injury is likely the predominant mechanism for troponin elevation in these patients, potentiated by acute heart failure, myocarditis, stress cardiomyopathy, or direct viral-induced injury. It may also be due to several primary noncardiac etiologies such as septic shock and pulmonary embolism. Myocardial infarction, potentially due to thromboembolism, has also been reported.
Both troponin and natriuretic peptide elevations in hospitalized patients have been associated with more severe disease and worse outcomes and as such can serve as important prognostic markers during initial triage and subsequent follow-up of these patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here