Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The increasing popularity of soft tissue augmentation requires knowledge of reversal techniques.
Adverse events due to filler injection can be totally or partially reversed depending on the filler type and timing.
Nonpermanent fillers are much more forgiving and more easily corrected.
Hyaluronidase can be considered a treatment “eraser” for many complications arising from the injection of hyaluronic acid, and small doses can be effective.
Vascular occlusion should be rapidly diagnosed and treated, especially if vision loss is involved, and large doses of hyaluronidase should be considered
Late-onset inflammatory reactions may require a multifaceted approach, particularly after complications due to permanent filling agents.
An emerging treatment for calcium hydroxylapatite filler is the use of sodium thiosulfate.
According to the most recent (2019) procedural survey by the American Society for Dermatologic Surgery (ASDS), the use of injectable fillers has increased by 78% since 2012 in the United States. The advent of new, nonpermanent soft tissue fillers, and the resurgence of older, permanent fillers, speaks to the increased demand of the public for soft tissue augmentation. As with all cosmetic procedures, the increase in number of procedures performed increases the chances for complications. Additionally, many of the soft tissue fillers on the market today are injected in locations that are off-label, many of which carry a higher probability of vascular occlusion or other complication. Thus, a profound knowledge of how to reverse filler complications is paramount. Soft tissue fillers can be divided into two categories according to biodegradability: biodegradable (absorbable) or nonbiodegradable (permanent). The innovation of cross-linking techniques with different hyaluronic acid (HA) concentrations determines where fillers may be placed and how long they may provide tissue correction ( Table 38.1 ). Any of the listed products can lead to early or delayed complications, but only HAs can truly be reversed with the use of hyaluronidase. Unlike biodegradable agents, particularly HAs, permanent fillers require a multifaceted approach and can be more difficult to correct in the event of misplacement or serious adverse reactions.
| Biodegradable Products | |||||
|---|---|---|---|---|---|
| Product | Manufacturer | Cross-Link Tech/Vehicle | Type Product and Concentration (mg/mL) | Approval by Country | Biodegradability/erasability |
| Juvéderm Ultra XC | Allergan | Hylacross | HA-24 | All | Absorbable/erasable |
| Juvéderm XC ultra plus | Allergan | Hylacross | HA-24 | All | Absorbable/erasable |
| Juvederm Volux | Allergan | Vycross | HA-25 | South, Central America/Canada/Europe | Absorbable/erasable |
| Radiesse | Merz | Carboximetilcelullose 70% | Calcium hydroxylapatite 30% | All | Absorbable/nonerasable |
| Sculptra | Galderma/Sinclair | Saline and Lidocaine | Poly-l-lactic acid (% depend on dilution) | All | Absorbable/nonerasable |
| RHA 2 | Teoxane SA | Preserved Network | HA-23 | United States/Europe | Absorbable/erasable |
| RHA 3 | Teoxane SA | Preserved Network | HA-23 | United States/Europe | Absorbable/erasable |
| RHA 4 | Teoxane SA | Preserved Network | HA-23 | United States/Europe | Absorbable/erasable |
| Juvéderm Voluma XC | Allergan | Vycross | HA-20 | All | Absorbable/erasable |
| Juvéderm Volbella XC | Allergan | Vycross | HA-15 | South America/Canada/Europe | Absorbable/erasable |
| Juvéderm Volift a | Allergan | Vycross | HA-17.5 | South, Central America/Canada/Europe | Absorbable/erasable |
| Juvederm Vollure XC | Allergan | Vycross | HA-17.5 | All (Vilify in other countries) | Absorbable/erasable |
| Belotero Hydro | Merz | No crosslink, contains glycerol | HA-18 | South, Central America/Canada/Europe | Absorbable/erasable |
| Belotero Balance | Merz | CPM | HA-22.5 | All | Absorbable/erasable |
| Belotero Volume a | Merz | CPM | HA-26 | South, Central America/Canada/Europe | Absorbable/erasable |
| Belotero Intense a | Merz | CPM | HA-25.5 | South, Central America/Canada/Europe | Absorbable/erasable |
| Belotero Soft a | Merz | CPM | HA-20 | South, Central America/Canada/Europe | Absorbable/erasable |
| Emervel Classic a | Galderma | OBT | HA-20 | South, Central America/Canada/Europe | Absorbable/erasable |
| Emervel Lips a | Galderma | OBT | HA-20 | South, Central America/Canada/Europe | Absorbable/erasable |
| Emervel Volume a | Galderma | OBT | HA-20 | South, Central America/Canada/Europe | Absorbable/erasable |
| Emervel Deep | Galderma | OBT | HA-20 | South, Central America/Canada/Europe | Absorbable/erasable |
| Restylane | Galderma | NASHA | HA-20 | All | Absorbable/erasable |
| Restylane Silk/Vital | Galderma | NASHA | HA-20 | All | Absorbable/erasable |
| Restylane Defyne | Galderma | NASHA | HA-20 | All | Absorbable/erasable |
| Restylane Refyne | Galderma | NASHA | HA-20 | All | Absorbable/erasable |
| Restylane Kysse | Galderma | NASHA | HA-20 | All | Absorbable/erasable |
| Restylane Lyft | Galderma | NASHA | HA-20 | All | Absorbable/erasable |
| Nonbiodegradable products (permanent) | |||||
| Silikon 1000 | Alcon | — | Polydimethylsiloxane | All (not aesthetic uses) | Nonabsorbable/nonerasable |
| Belafill | Suneva Medical | Bovine collagen | PMMA + bovine collagen | North America and Canada | Nonabsorbable/nonerasable |
Reversers are necessary secondary to improper placement on depth of filler, overcorrection, vascular occlusion, and late-onset immune-mediated reactions. Looking from the perspective of filler permanence, only HA and collagen can be completely erased by two different enzymes: hyaluronidase and collagenase. An emerging treatment for dissolving calcium hydroxylapatite (CaHA) fillers is the use of sodium thiosulfate (STS), although its use is not as well studied as hyaluronidase and collagenase. If we look at all the adverse events after filler application, especially with nonabsorbable fillers, there are complications that can be partially reversed or at least treated with a variety of substances, lasers, or oral medicines (those will be mentioned but not discussed in this chapter). This chapter will focus on the use of substances, procedures, or technology that can alter or change not only the permanence of filler itself, but also change the elements involved in a specific adverse event.
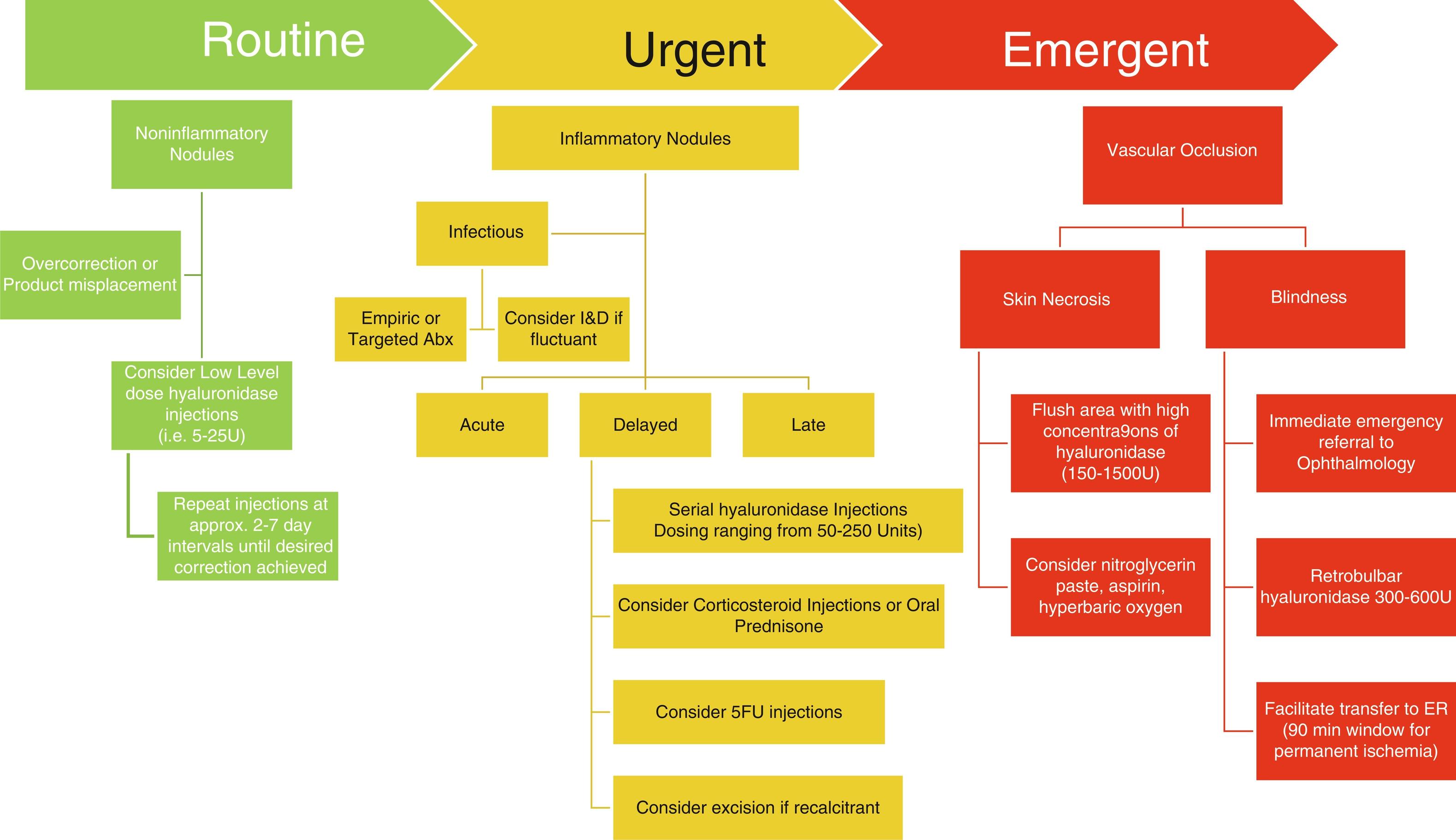
Adverse effects following soft tissue augmentation are generally classified as early onset (up to 2 weeks after injection), late onset (> 14 days to 1 year), or delayed onset (> 1 year) ( Box 38.1 ). Late- or delayed-onset reactions are more commonly associated with nonabsorbable filling agents but have been more recently described with HA, CaHA, and poly-l-lactic acid (PLLA). Early-onset adverse events are easier to treat and more common than late-onset ones. According to reversibility classification, HA and collagen injection may be a clear advantage over other substances ( Figs. 38.1–38.5 ).
Early onset (immediate to 14 days)
Injection site reactions including purpura and edema
Noninflammatory nodules (overcorrection or misplacement of injected material)
Infection (bacterial, viral)
Hypersensitivity reaction/anaphylaxis
Vascular occlusion
Late onset (from 15 days to 1 year) and delayed (> 1 year)
Inflammatory nodules (immune-mediated vs infection/biofilm)
Persistent or recurrent edema
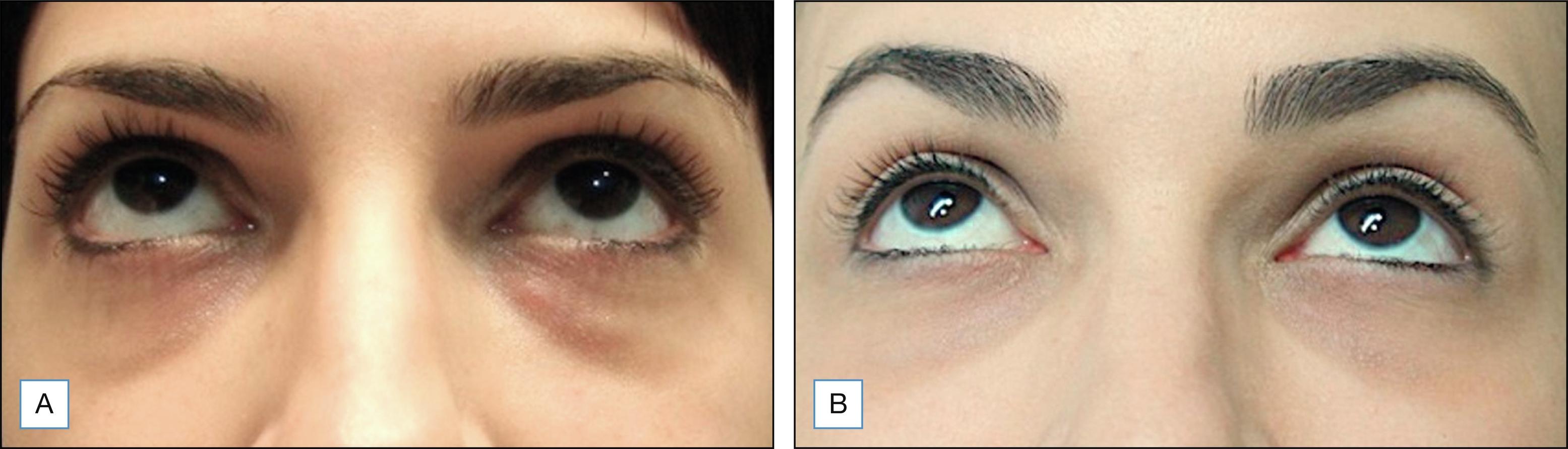
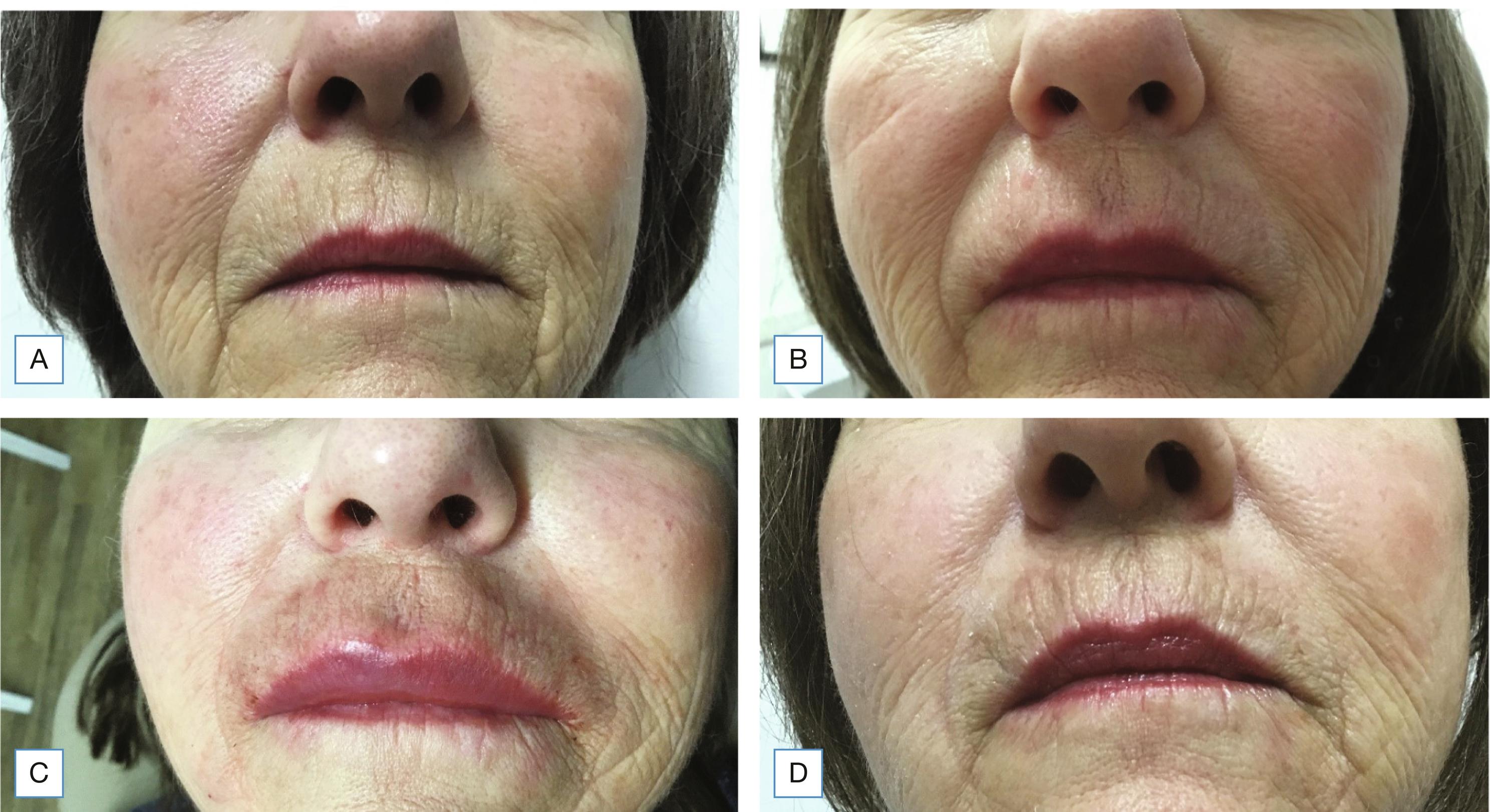
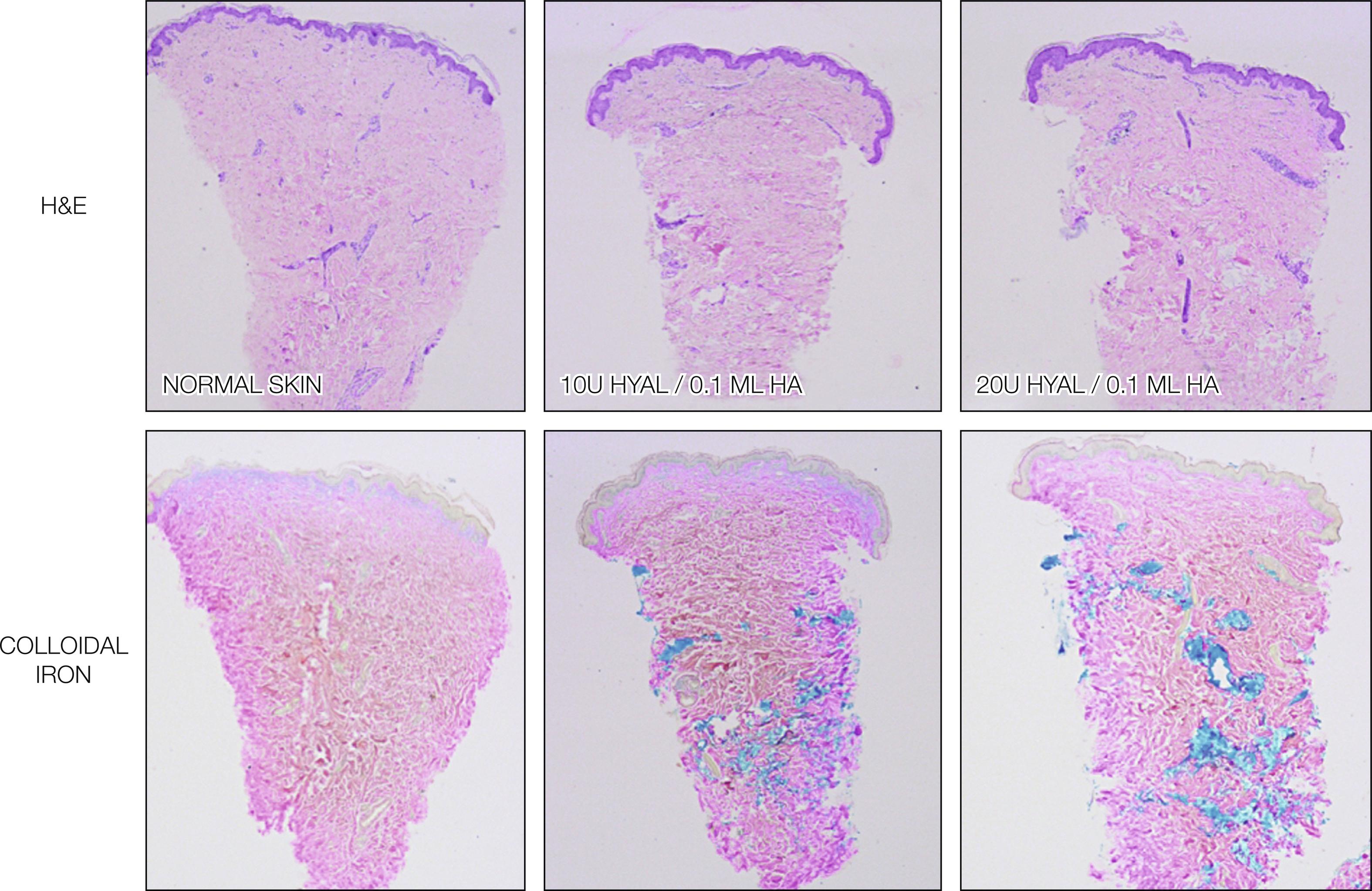
Expected early reactions are most commonly injection site reactions, including purpura and edema. Vascular lasers are effective in the treatment of postprocedure purpura. Infections are much less frequent, with herpes virus being the most common. Hypersensitivity and angioedema are very rare but can occur with all fillers composed of foreign body material. Anaphylaxis reaction symptoms are pruritus, erythema, urticarial lesions, angioedema, nausea, vomiting, breathlessness, dizziness, syncope, hypotension, or even cardiorespiratory arrest. The physician must be prepared to deal with anaphylaxic reactions. Epinephrine is the most important drug in this clinical emergency, maximally effective when injected promptly. H1-antihistamines are not drugs of choice in initial anaphylaxis treatment because they do not relieve life-threatening respiratory symptoms or shock, although they decrease urticaria and itching. Diphenhydramine can be administered, and glucocorticoids remain in use for anaphylaxis because they potentially prevent biphasic anaphylaxis, reducing the risks of late symptoms. If symptoms are mild, oral prednisone can be prescribed, but more severe cases need intravenous methylprednisolone. It is important to have an oxygen cylinder and β2 agonist medication, such as salbutamol, in the clinic in case of hypoxemia. After clinical stabilization, the physician must decide whether the patient should be taken to the hospital.
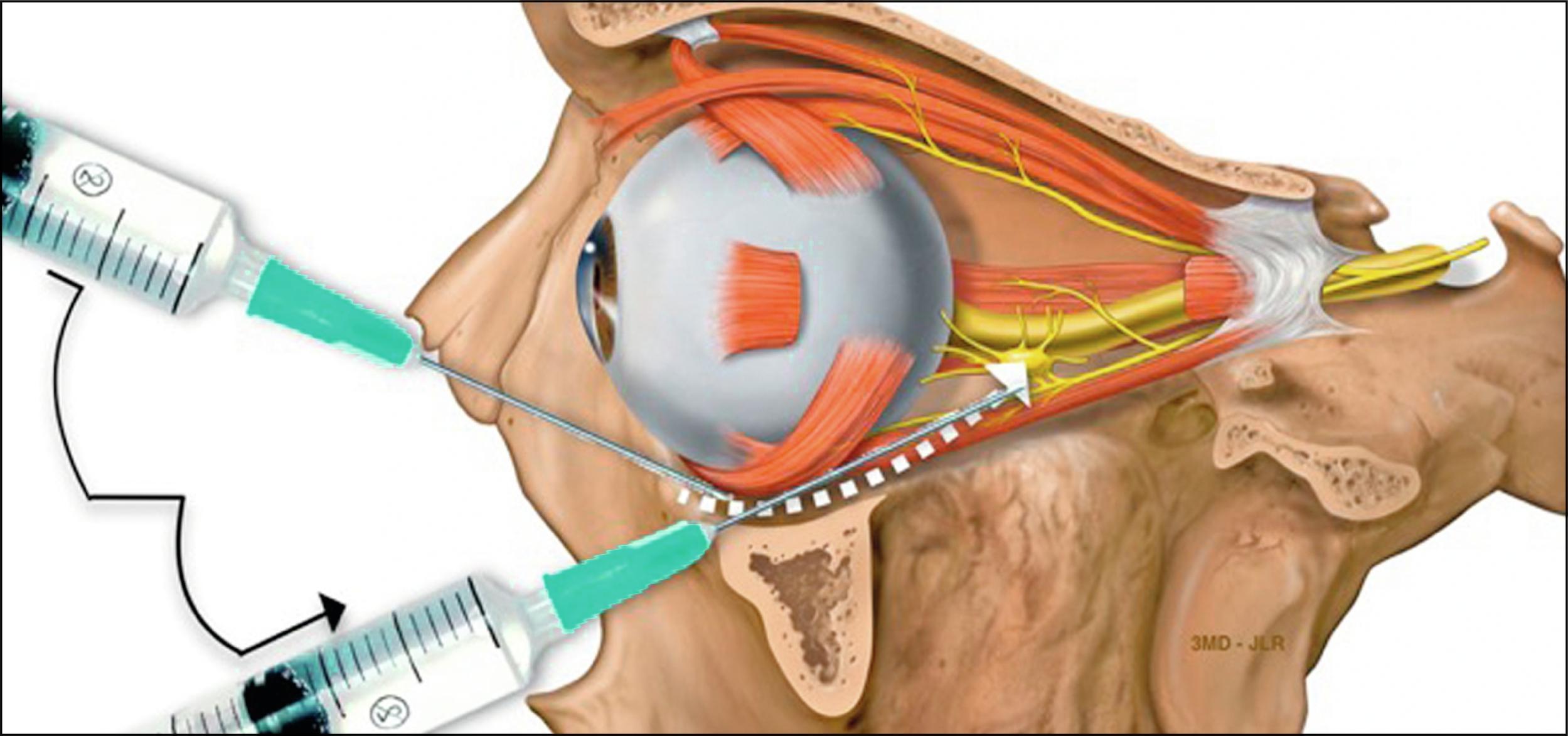
Among all the adverse events related to filler placement, intravascular injection and its consequences are most concerning and can be devastating. Chapters 35 and 37 discuss the various types of vascular occlusion events and present the relevant anatomical considerations. This section will review the treatment protocols for reversers in more detail. The mechanism of action of hyaluronidase in the setting of HA vascular occlusion is bathing the artery in hyaluronidase, which can then readily diffuse across the vessel wall and be carried by arterial flow to the embolism. Selective angiography has also been described, but this technique is highly specialized and requires interdisciplinary care.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here