Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
There are several pulmonary diseases discussed elsewhere in this book that, among the many effects they have upon the patient, convey a recognized predisposition to lung cancer development. These diseases include many forms of diffuse pulmonary fibrosis (idiopathic pulmonary fibrosis, connective tissue disease–associated pulmonary fibrosis, asbestosis), congenital adenomatoid malformations, juvenile tracheobronchial squamous papillomatosis, pleomorphic adenoma, and other acquired and congenital cystic lung lesions. Readers are referred to the suggested references at the end of this chapter for further information.
The 2015 World Health Organization (WHO) classification of lung tumors describes four entities that are regarded primarily as precursors of malignancy in the lung: squamous dysplasia/squamous cell carcinoma in situ (SD/SCCIS), atypical adenomatous hyperplasia (AAH), adenocarcinoma in situ (AIS), and diffuse idiopathic pulmonary neuroendocrine cell hyperplasia (DIPNECH). It is believed that SD/SCCIS and AAH/AIS precede a large proportion of primary lung carcinomas, and certainly define two separate pathways of lung carcinoma development: SD/SCCIS, the essential part of central bronchial squamous carcinogenesis, leading to squamous cell and perhaps some other carcinomas, and AAH/AIS, the basis of peripheral lung adenocarcinogenesis. DIPNECH is the least common and least important of the four lesions and is included in the WHO classification due to its association with peripheral carcinoid tumors.
Lesions characterized by cytologic and architectural atypia of squamous epithelium, occurring as single or multiple foci within tracheobronchial epithelium and which are the precursors of squamous cell carcinoma
Occurs in approximately 40% of heavy tobacco smokers
More common in lungs also bearing squamous cell carcinoma
None
More common in males than females
No apparent racial predilection
Disease of adults
No specific features
Not radiologically detectable
Risk factor for invasive carcinoma, but no data to enumerate risk
Progression not inevitable, but more likely with higher-grade disease
Smoking cessation is recommended
Treatments are under investigation
Usually none, but may appear as ill-defined areas of roughened mucosa
Occurs in central cartilaginous airways, often on the spur of bifurcations
AFB will demonstrate lesions more easily
By definition, this lesion occurs in a squamous-type epithelium
Graded as mild, moderate, or severe dysplasia, or carcinoma in situ
Grading based upon distribution of basaloid and/or atypical squamous cells and mitoses within the epithelium
Carcinoma in situ is characterized by the most marked cytologic atypia and lack of maturation in a chaotic epithelium
Some dysplastic lesions retain a layer of differentiated columnar epithelial cells on the surface
Atypical squamous cells may be found in samples taken at bronchoscopy or in sputum
Grading based on the degree of nuclear structural abnormality and cytoplasmic tinctorial characteristics
Distinction between in situ and invasive carcinoma is difficult
BCH
SM
Chemotherapy- or irradiation-induced epithelial cell injury
Epithelial reactive changes associated with inflammatory processes
Invasive carcinoma in small samples
Bronchial SD/SCCIS incorporates a range of recognized entities, all of which show cytologic atypia within a squamous-type epithelium lining conducting airways in the lung. Mild SD, moderate SD, severe SD, and SCCIS represent a biologic continuum and should be considered together with changes in respiratory epithelium that may precede their formation, namely, basal cell hyperplasia (BCH) ( Fig. 30.1 ) and squamous metaplasia (SM) ( Fig. 30.2 ). Goblet cell hyperplasia (GCH) is included for completeness, although its role as a precursor of malignancy is controversial. This spectrum of change is the likely precursor of most central squamous cell carcinomas and possibly other neoplasms, including small cell lung carcinomas.
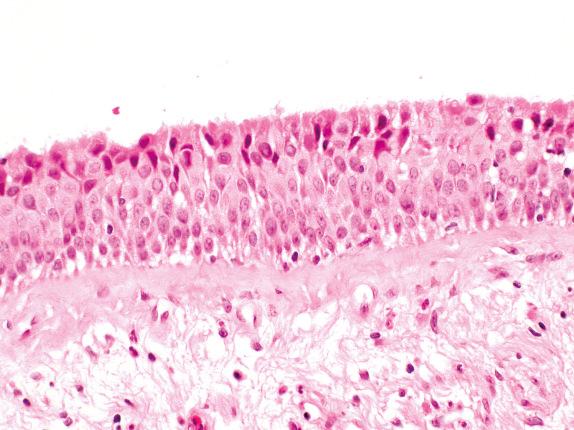
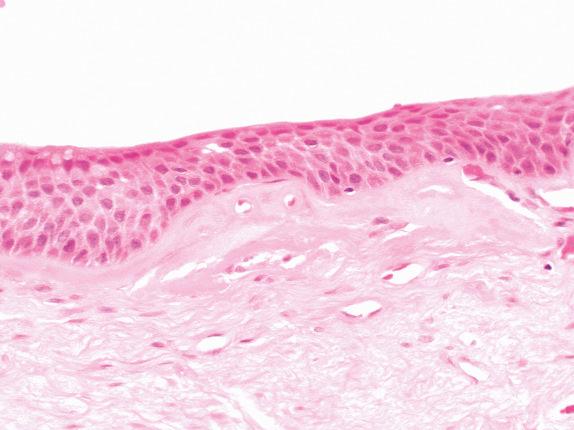
Whereas BCH, GCH, and SM are associated with a variety of chronic irritants (tobacco smoke, asthma, chronic infection, irradiation, vitamin A deficiency, air pollution), the presence of SD/SCCIS is strongly associated with heavy tobacco use or radiation exposure. With the exception of GCH, which is responsible for the disease-defining mucus expectoration of chronic bronchitis, the other changes are asymptomatic, which partly explains why early primary lung carcinoma is so difficult to detect. The strong association with carcinogen exposure mentioned earlier allows identification of populations at risk for developing SD/SCCIS, a key factor in creating strategies for lung cancer screening. Past smoking demographics probably account for SD/SCCIS being more commonly described in males. Both the length and intensity of the smoking history correlate with the frequency of SM and with the extent and degree of dysplasia in SD/SCCIS. Good data are rare, but approximately 40% of heavy smokers will have SD/SCCIS, most will have BCH or SM, and SD/SCCIS is more frequent in lungs where invasive squamous cell carcinoma has already developed. These data argue for a causal association between cigarette smoking and subsequent evolution from BCH/SM to SD/SCCIS to invasive squamous cell carcinoma.
SD/SCCIS cannot be detected radiologically, another reason why early lung carcinoma detection is so problematic. Rarely, a circumferential non-obstructive lesion may compromise airway drainage through loss of mucociliary function, leading to distal pneumonia.
In surgically resected lung or at autopsy examination, the vast majority of SD/SCCIS lesions are invisible on gross examination. SCCIS is frequently found on the spurs of bronchial carinae, occasionally giving the mucosa a pale, granular, opaque character with loss of the mucosal rugae and pits. It is believed that the genetic changes driving this disease occur in clonal patches that are widespread in the bronchial mucosa, a so-called field effect. Such patches of morphologically normal mucosa contain tens of thousands of basal epithelial cells and measure approximately 1 mm 2 . Studies have shown that patches of SCCIS range from approximately 4 to 12 mm in diameter and up to 4 mm in thickness. Studies using autofluorescence bronchoscopy (see later) have shown dysplastic lesions of 1 to 3 mm in diameter.
The lack of gross features means that most SD/SCCIS lesions are also invisible by standard white-light bronchoscopy. About 30% to 40% of SCCIS lesions may be visible to an experienced bronchoscopist as flattened or, less often, nodular patches of mucosa lacking normal luster, perhaps granular, red, and with an abnormal vascular pattern. Abnormal bronchial mucosa loses green autofluorescence when viewed under blue light using special sensors. Autofluorescence bronchoscopy (AFB) increases the detection rate by up to sixfold for all grades of SD/SCCIS lesions, but is nonspecific, because many lesions detected at AFB show hyperplastic changes, inflammation, or no significant pathology.
The 1999 WHO lung tumor classification was the first to include descriptions of, and criteria for, the diagnosis of mild, moderate, and severe bronchial SD and SCCIS. Previously, some rather complex systems of classification were proposed, based on either histologic material from autopsy examination or cytologic preparations. The current classification assumes a full-thickness squamous epithelium and uses criteria similar to those for grading dysplasia in other sites such as the cervix, considering a range of cytologic and architectural changes distributed in the lower, middle, and upper thirds of the metaplastic epithelium. It is recognized that this is a biologic system, such that this division into different grades is artificial. Within the bronchial tree of any person with SD/SCCIS, the degree of atypia will vary even within a single high-power microscopic field, and all the proposed defining criteria for a particular grade of SD will not necessarily be present.
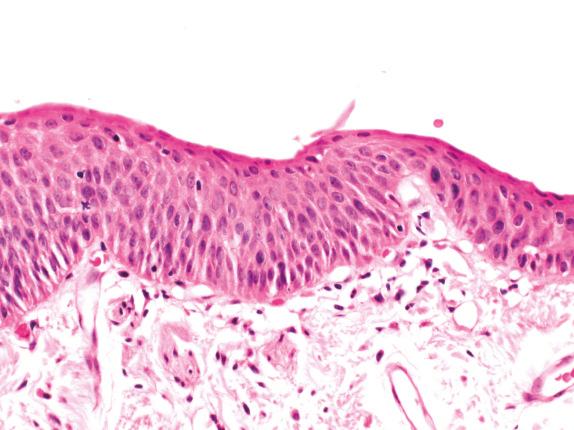
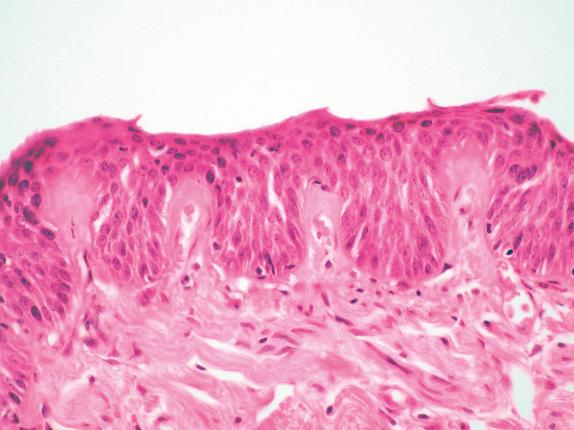
Mild SD is difficult to recognize ( Fig. 30.3 ). Expansion of the basal cell zone into, but not beyond, the lower third of the epithelium is probably the best criterion. Cytologic atypia is minimal, and rare mitotic figures should be basal.
Moderate SD may show basal cell expansion with vertically orientated nuclei extending no further than the middle third of the epithelium. More atypia is allowed. Mitoses can occur off the basal layer, but are confined to the lower third of the epithelium ( Fig. 30.4 ).
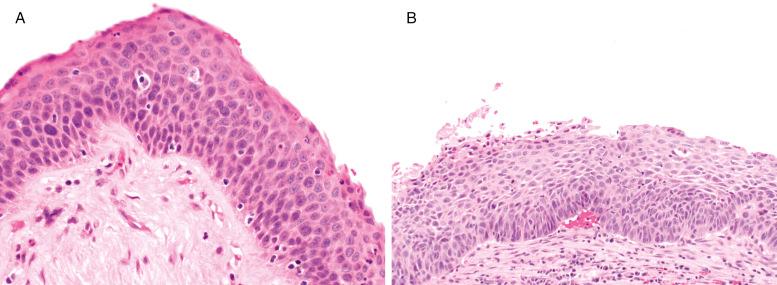
Severe SD shows a marked increase in atypia. Cell crowding extends into the upper third of the epithelium; yet there is evidence of surface flattening; the epithelium clearly has a “top.” Mitoses extend into, but not beyond, the middle third of the epithelium ( Fig. 30.5 ).
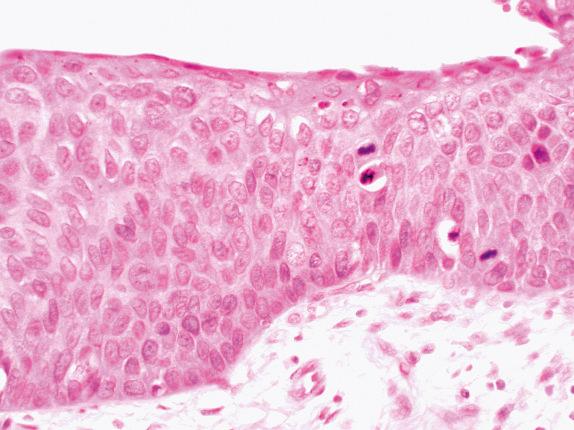
The best way to think of SCCIS in this classification is “chaos.” The malignant-looking squamous epithelium has no “top.” Inverted, it would look more or less the same, with atypia and mitoses throughout ( Fig. 30.6 ).
Cytopathologists may be able to distinguish severe SD/SCCIS vs. lower-grade dysplasia based on the presence of nuclear pleomorphism, coarse/uneven chromatin, thickened nuclear membranes, and thickened cytoplasm.
Studies of interobserver diagnostic reproducibility for the WHO five-category classification (SM, mild-moderate-severe SD, and SCCIS) found a mean weighted kappa of 0.55 (random agreement 0, perfect agreement 1.0), with no improvement after simplification into a three-category classification of metaplasia/mild dysplasia, moderate dysplasia, and severe dysplasia/SCCIS. Studies on alternative simplification schemes, for example, low-grade dysplasia vs. high-grade dysplasia/SCCIS, have not been reported but would be useful.
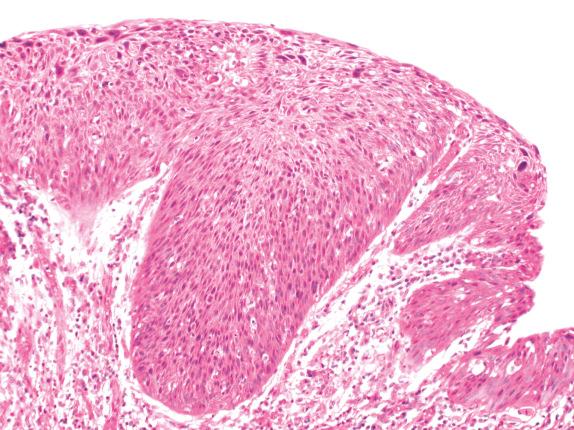
Occasionally a localized squamoproliferative lesion with a papillary structure, derived from sprouts of subepithelial capillaries invaginating into the overlying epithelium, may be found in airway mucosa. Although these lesions are sometimes covered by respiratory-type epithelium, most show a squamous epithelium with varying degrees of cytologic atypia, hence their classification as angiogenic squamous dysplasia ( Fig. 30.7 ). Papillary squamoproliferative lesions with koilocytic atypia due to human papillomavirus (HPV) infection are unexpected and should raise the possibility of upper airway squamous papillomatosis with lower airway involvement.
The Cancer Genome Atlas (TCGA) data on invasive squamous cell carcinoma indicate that there is both loss of function of tumor suppressor genes (due to allelic loss, promoter hypermethylation, coding sequence mutations, alternative splice transcripts) and/or gain of function of oncogenes (due to gain-of-function coding sequence mutations or wildtype allele amplification). The most frequent changes that occur during squamous cell carcinogenesis include loss of function (mutation) of p53, loss of function (deletion, promoter hypermethylation, mutation) of CDKN2A (p16, p14), up-regulation of PI3K and RTK signaling pathways, amplification of SOX2 and p63, and frequent copy number variation. No recurring fusion genes (analogous to ALK or ROS1 translocations in lung adenocarcinoma) were noted.
Studies on SD have shown early copy number variation (3p, 9p, 17p allelic losses; 3q ( SOX2, p63) amplification), CDKN2A (p16, p14) promoter hypermethylation, and p53 coding sequence mutation. Some of these genetic and epigenetic changes may occur in a stepwise process that correlates with, and perhaps determines, the morphologic changes that appear as SD/SCCIS evolves into invasive carcinoma. It may be that there are several different pathways at a genomic level through which SD/SCCIS/invasive squamous cell carcinoma may develop.
Ultrastructural studies have nothing to offer in terms of diagnosis. Immunohistochemistry is equally unhelpful. There is plentiful literature on a range of markers, but none is of any proven diagnostic benefit. The prior odds of high risk (HR) HPV as an etiologic agent are probably <5%, but HR HPV in situ hybridization (ISH) could be considered in nonsmokers with squamous carcinomas.
SM may occur in the lung in other diseases, and where inflammation is prominent, a degree of reactive atypia is common ( Fig. 30.8 ). In bronchiectasis or tuberculous cavities, there may be SM with atypia, and atypical SM can be seen in the regenerating hyperplastic epithelium of the proliferative phase of diffuse alveolar damage (DAD). The context in which such atypical squamous epithelium is found is important in reaching the correct diagnosis. Physicians must be cautious of making a diagnosis of SD/SCCIS outside the context of epithelium lining an intrinsically normal bronchus. There are pitfalls even in this situation. Mucosa around areas of chronic ulceration or in airways draining an area of chronic infection may show atypical SM associated with marked mucosal inflammation and should not be classified as SD/SCCIS. Chemotherapy and/or radiotherapy injury can cause bizarre epithelial atypia, so that clinical history should be known and considered. Likewise, viral infections can cause epithelial atypia, but these viral cytopathic effects are usually focal, involving single cells or small groups.
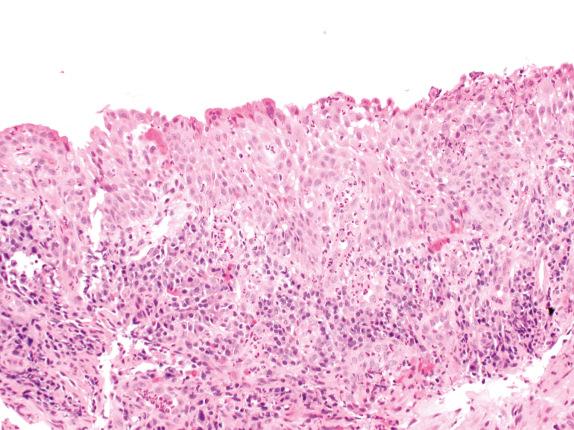
Although published criteria for SD/SCCIS are welcomed and will likely improve diagnostic reproducibility, there are still problems to reckon with. The criteria are based on a full-thickness squamous epithelium with a sufficient number of cell layers present to allow its separation into thirds. It is the authors’ view that true full-thickness keratinizing SM as a basis for supervening dysplasia is uncommon. More often, squamous change with prickle cells develops in a background of BCH, and even high-grade dysplasia may supervene within this basal cell population while respiratory columnar cells persist on the epithelial surface ( Fig. 30.9 ). BCH, which is all but full-thickness, may be confused with severe SD or SCCIS by the unwary. BCH shows a regular cell population without atypia. Not infrequently, atypical epithelium may be very thin or may have shed its superficial cells during tissue processing, making grading extremely difficult. When strict application of the WHO criteria is inappropriate or impossible, a more pragmatic approach that assesses the recognizable characteristics of the lesion is best. In these situations, classifying lesions as low- or high-grade dysplasia seems appropriate. Some studies have considered moderate and severe dysplasia together as “high-grade” dysplasia, and there is some molecular evidence to justify this practice.
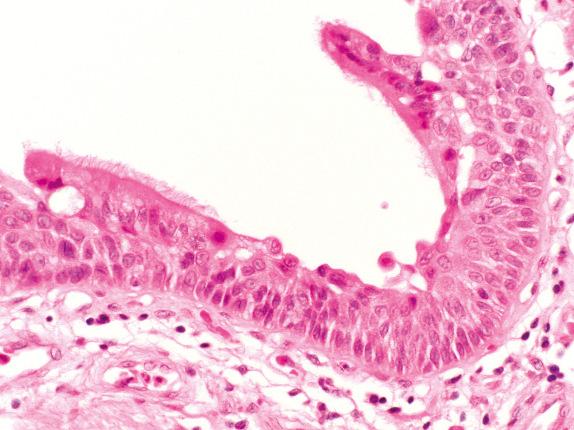
In diagnostic bronchial biopsy specimens with SCCIS, care must be taken not to miss evidence of bronchial mucosal invasion. Isolated nests or small groups of severely atypical squamous cells, background desmoplasia, necrosis, irregular outlines to sheets of malignant-looking squamous epithelium, and vascularization of such epithelium all favor a diagnosis of invasive carcinoma. The basement membrane may be very hard to identify and, even in SCCIS, may be variably thickened or thinned. Knowledge of the bronchoscopic findings may help; for example, was there a tumor mass seen and biopsied, or was the biopsy from a vague area of mucosal irregularity? Likewise, knowledge of the radiographic findings may help; for example, does computed tomography (CT) show a mass effect or postobstructive changes? Sometimes, when invasion cannot be ruled out, a diagnosis of “at least squamous cell carcinoma in situ” is appropriate. SD/SCCIS may extend from the bronchial surface into bronchial gland ducts and even replace the glands themselves. A misdiagnosis of invasive carcinoma may be avoided by appreciating the retained lobular architecture of the glands.
It can be challenging to distinguish a solitary pulmonary metastasis—for example, in a smoker with a prior head and neck primary squamous cell carcinoma—from a new primary lung squamous cell carcinoma. This distinction affects staging and would likely affect patient management. Because SCCIS represents the precursor lesion for invasive squamous cell carcinoma, the finding of SCCIS in continuity with invasive squamous cell carcinoma resolves this problem, supporting a diagnosis of lung primary.
Predicting the likelihood of progression from SD/SCCIS to invasive squamous cell carcinoma has become an area of considerable interest, particularly with renewed interest in lung cancer screening and improved detection of SD/ SCCIS using AFB. Proper longitudinal studies of SD/SCCIS in humans are difficult, even with the improved localization afforded by AFB surveillance. A review of such studies found that roughly 40% of severe SD persisted or progressed to SCCIS; that roughly 85% of SCCIS persisted, recurred after local treatment, or progressed to invasive carcinoma; and that roughly 50% of SCCIS cases progressed to invasive squamous carcinoma within 2.5 years. Follow-up studies of patients with squamous atypia in sputum have found that up to 50% of patients eventually develop invasive squamous cell carcinoma. Relatively speaking, fewer patients have been followed up by repeated AFB and biopsy, but such studies would be valuable. Results have so far been highly variable, and many confounding factors are at work. Some generalizations may be drawn from the data already published:
Progression of SD/SCCIS to invasive squamous cell carcinoma is not inevitable, and lesions may wax and wane or completely regress. Smoking cessation may hasten, but does not guarantee, regression.
If SD/SCCIS does progress to invasion, this probably takes years rather than months.
Each stage of SD/SCCIS may persist for many months or years, but more advanced disease progresses faster and is less likely to regress.
There is a body of work reporting on molecular biologic markers that may predict outcome in SD/SCCIS. Although there are data to suggest that abnormal expression of the proteins P53, P16, BAX, BCL2, FHIT, and cyclins D1 and E, as well as some genetic abnormalities, may have a predictive role, none of these, so far, reliably predict outcome for a given patient. Detection and management of SD/SCCIS pose problems for those caring for these high-risk patients, especially when prediction of outcome is so uncertain. “Watch and wait” may be appropriate in many situations, as chemoprevention strategies are still the subject of early trials. Field cancerization and problems localizing SD/SCCIS make surgical resection an infrequent option, but some data suggest that photodynamic therapy may have a role in some cases.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here