Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Upper gastrointestinal (GI) endoscopy is routinely performed as part of the evaluation of patients with GI symptoms. Newer techniques such as double-balloon enteroscopy and capsule endoscopy also allow for complete visualization of the small intestine. Gastroenterologists may identify and sample a variety of polyps, nodules, excrescences, and subtle abnormalities in the mucosa during the course of the procedure that, before the endoscopic era, would have gone largely unrecognized. Many of these lesions are clinically asymptomatic and occur in the duodenum, which is amenable to upper endoscopic examination. This chapter discusses the clinicopathological features and differential diagnoses of the most common or important lesions that may cause the clinical impression of an intestinal polyp ( Box 21.1 ).
Hyperplasias and heterotopias
Brunner gland hyperplasia/hamartoma
Gastric heterotopia
Pancreatic heterotopia
Inflammatory lesions
Crohn’s disease–associated inflammatory “pseudopolyp”
Inflammatory polyp, CMV associated
Inflammatory polyp, not otherwise specified
Xanthoma
Inflammatory fibroid polyp
Peutz-Jeghers polyp
Juvenile polyp (juvenile polyposis syndrome and PTEN hamartoma tumor syndrome)
Polyp of Cronkhite-Canada syndrome
Benign epithelial neoplasms
Adenoma
Pyloric gland adenoma
Hyperplastic polyp
Neuroendocrine neoplasms
Well-differentiated neuroendocrine neoplasm
Gangliocytic paraganglioma
Neuroendocrine carcinoma
Mesenchymal tumors
Gastrointestinal stromal tumor
Leiomyoma
Lipoma
Hemangioma
Neurofibroma
Granular cell tumor
Ganglioneuroma
Schwannoma
Kaposi sarcoma
Lymphangiectasia
Lymphoid lesions
Nodular lymphoid hyperplasia
Diffuse large B-cell lymphoma
Mantle cell lymphoma (lymphomatous polyposis)
Low-grade B-cell lymphoma (follicular, MALT-type, Mediterranean fever)
T-cell lymphoma (gluten sensitivity enteropathy–associated lymphoma)
Metastases
As in other areas of the GI tract, inflammatory lesions of the small intestine may produce polypoid masses. Small polyps are often incidental, whereas larger polyps may be symptomatic as a result of hemorrhage or luminal obstruction. Inflammatory pseudopolyps associated with Crohn’s disease are the most common inflammatory polyps of the small intestine; they are commonly encountered in the terminal ileum, where they demonstrate varying degrees of villous architectural distortion, pyloric gland metaplasia, and active inflammation ( Fig. 21.1 ). They can be quite numerous and can even carpet the small bowel with finger-like projections.
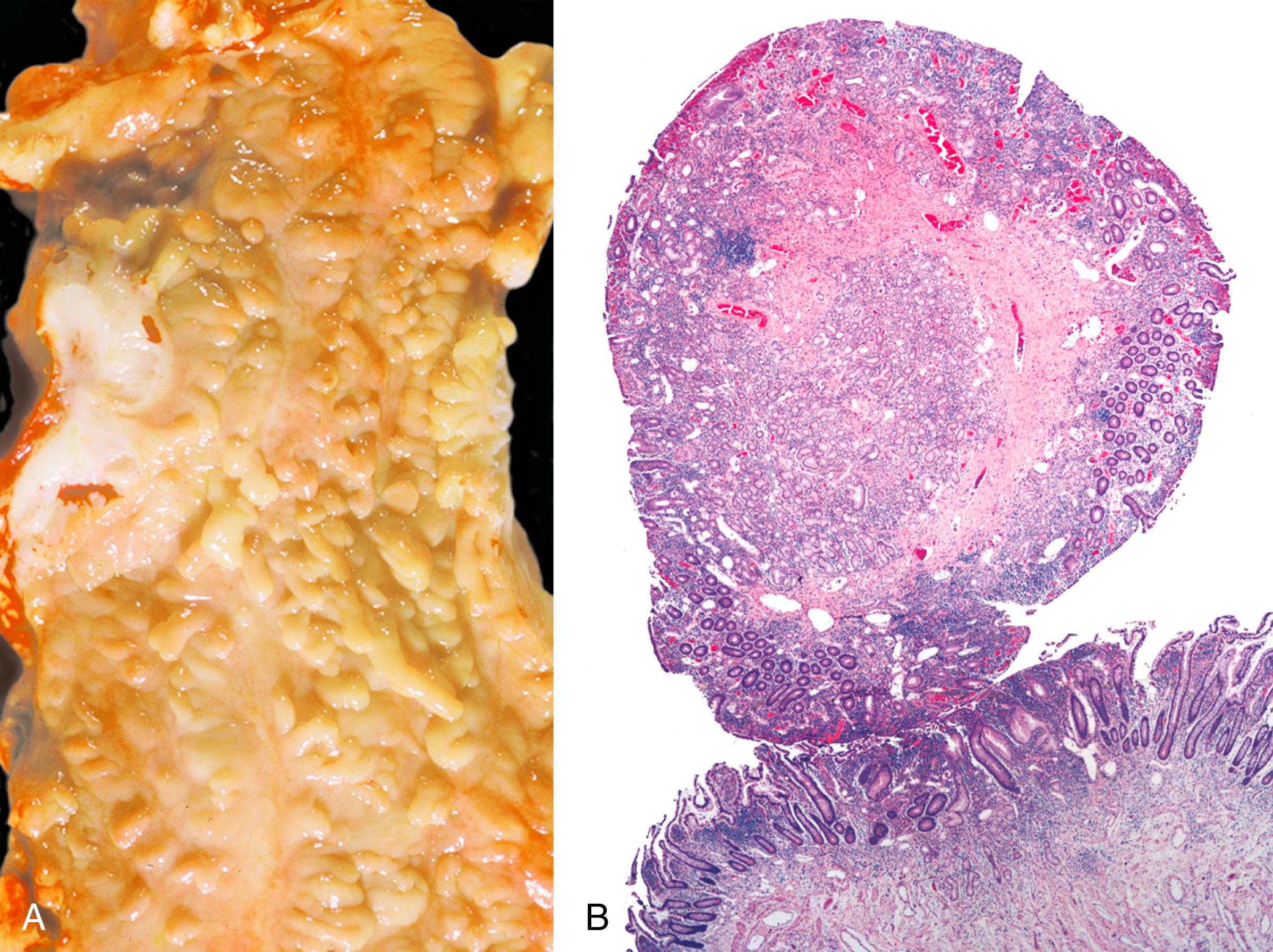
Inflammatory-type polyps of the small intestine may arise secondarily from other types of disease. Cytomegalovirus infection can rarely manifest as isolated GI inflammatory polyps, often in immunocompromised hosts ( Fig. 21.2 ). , Endometriosis of the bowel wall and submucosa may induce inflammatory changes including inflammatory-type polyps. Changes mimicking Crohn’s disease have also been reported. , Endometriosis can also involve the mucosa, resembling dysplasia and even carcinoma ( Fig. 21.3 ). Well-differentiated neuroendocrine tumors (NETs) may also induce an inflammatory reaction in the mucosa away from the main tumor, likely related to mucosal prolapse. These lesions contain numerous ectatic capillaries with admixed smooth muscle bundles and fibrosis.
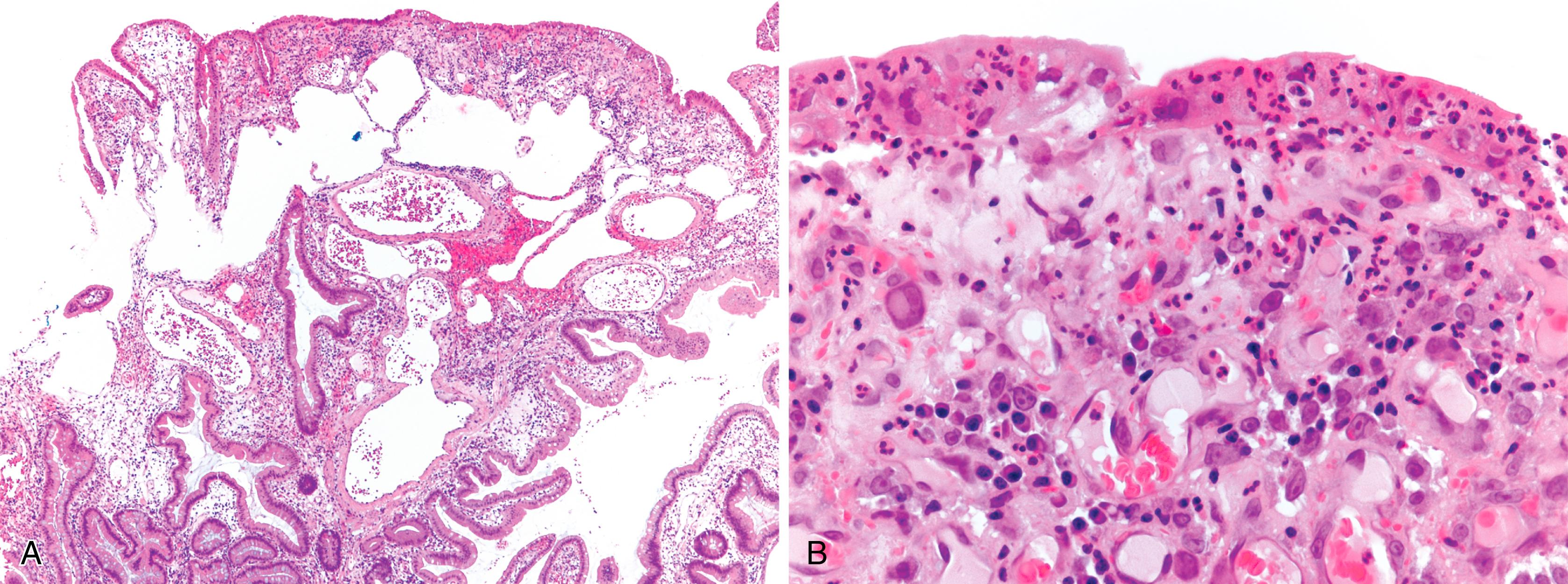
Xanthomas can also occur in the duodenum, although they are rarer than gastric xanthomas. They are characterized by collections of pale and foamy histiocytes in the lamina propria. Histiocytic differentiation can be documented by immunohistochemical staining with CD163, CD68, or both.
Inflammatory fibroid polyps (described in Chapter 30 ) are also less common in the small bowel compared with the stomach ( Fig. 21.4 ). Similar to their gastric counterparts, they often demonstrate mutations in the PDGFRA gene.
Brunner glands are lobular collections of tubular glands within the duodenum, predominantly located in the duodenal submucosa. In the first part of the duodenum, however, Brunner glands commonly transgress the muscularis mucosae and extend into the lamina propria. Brunner glands within the lamina propria are less frequently observed in the middle to distal duodenum.
Occasionally, Brunner glands proliferate, creating small polypoid excrescences or imparting a nodular appearance to the mucosal surface ( Fig. 21.5 ). These Brunner gland proliferative lesions are most commonly encountered in the duodenal bulb, usually as an incidental finding at endoscopy performed for other indications. However, they may be seen as one of a constellation of findings, including villous shortening and gastric foveolar mucous cell metaplasia of the villous epithelium, that are indicative of peptic injury of the duodenum (peptic duodenitis). Proliferative Brunner gland lesions have also been associated with end-stage renal disease and uremia. ,
The nomenclature of Brunner gland proliferative lesions is not well established, and a number of diagnostic terms, including Brunner gland hyperplasia, Brunner gland adenoma, and Brunner gland hamartoma, have been used. Histologically, it is difficult to distinguish hyperplasia from hamartomas and adenomas, and the distinction between these diagnoses is arbitrary. No well-documented cases of either true glandular dysplasia or carcinoma arising in proliferative Brunner glands have been reported. Careful review of the limited literature reports of Brunner gland adenocarcinoma and dysplasia reveals that the dysplastic glandular epithelium involves the surface epithelium with likely secondary involvement of the underlying hyperplastic Brunner glands. In other cases, the adenocarcinomas appear to arise from pyloric gland adenomas and not from hyperplastic Brunner glands. , Therefore the term Brunner gland adenoma is potentially misleading, and we prefer to use the term Brunner gland hyperplasia to limit confusion among our clinicians and to highlight the nonneoplastic nature of the entity.
Some authors distinguish between Brunner gland hyperplasia and Brunner gland hamartoma based on the size of the lesion. However, the size cutoff between these two entities is arbitrary and not well established in the literature. The distinction is of no clinical significance, and a diagnosis of Brunner gland hyperplasia/hamartoma is usually sufficient in large polypoid lesions.
Brunner glands are composed of neutral, mucin-secreting, cuboidal to columnar cells with basally located nuclei arranged in lobules containing thin, fibrous septa. Brunner glands are histologically indistinguishable from pyloric glands of the distal gastric mucosa. Indeed, biopsies of the gastroduodenal junction may demonstrate a gradual transition of these epithelial types. The predominantly submucosal location of Brunner glands helps identify the biopsy as duodenal in origin.
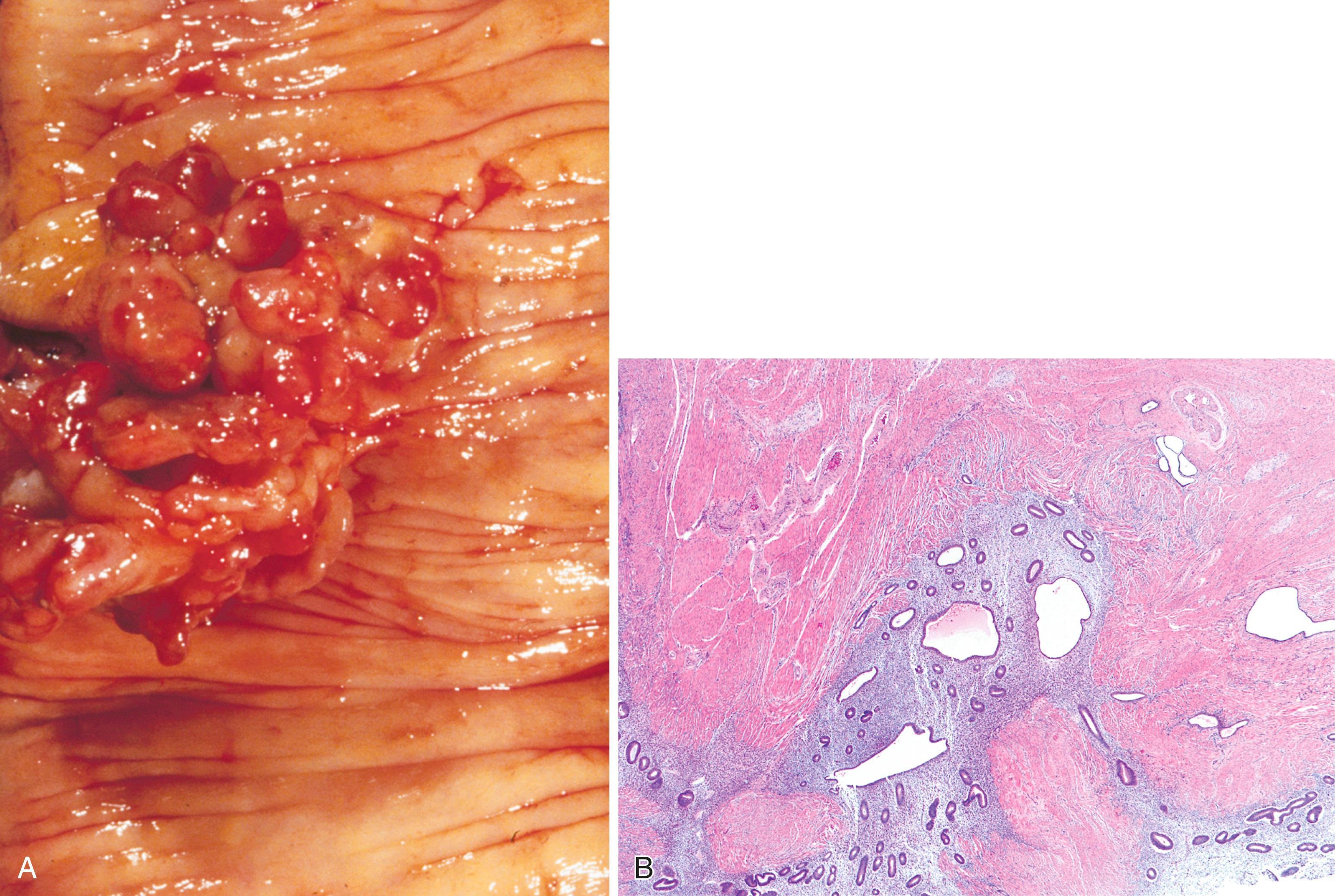
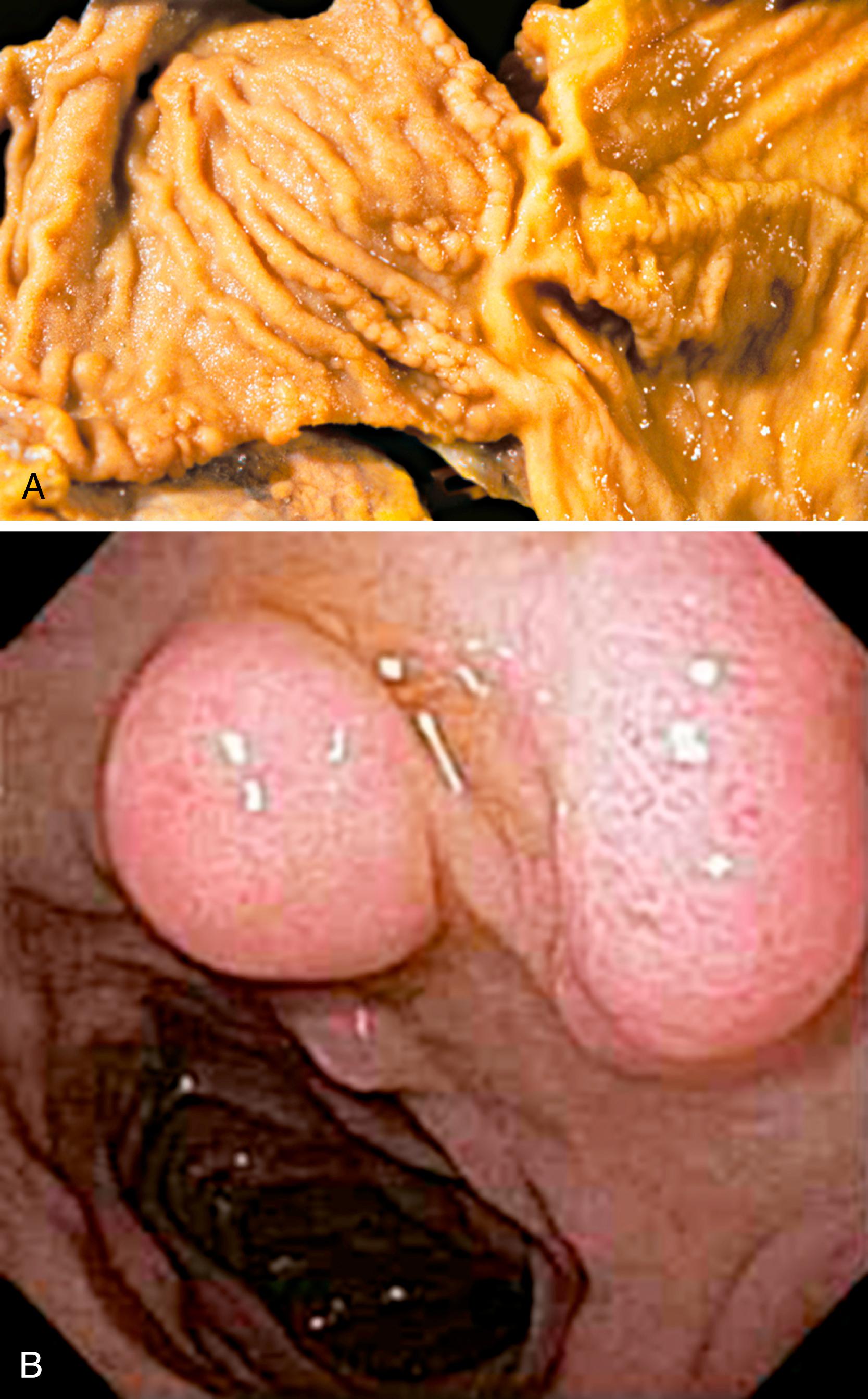
The diagnostic criteria for Brunner gland hyperplasia are subjective because Brunner glands may be normally seen in the lamina propria of the duodenum, particularly within the duodenal bulb. In addition, proliferation of Brunner glands is commonly observed in peptic-type duodenal injury. We reserve the term Brunner gland hyperplasia for those endoscopically visible duodenal nodules that are found to contain lobules of Brunner glands within the mucosa in at least 50% of the length of a biopsy specimen ( Fig. 21.6A ). Brunner gland hyperplasia may manifest as solitary or multiple nodules that are typically small (<1 cm) and characterized by lobules of glands that are increased in both size and number. The lobules extend into the mucosa and are separated by delicate fibrous septa. Cystic dilation of Brunner glands may occur, and rare cases of Brunner gland cysts have been reported. , The cells are cytologically bland with abundant neutral mucin and small, basally located nuclei with minimal to absent mitotic activity.
Large polyps (>2 cm) composed of Brunner glands may produce symptoms of obstruction, intussusception, melena, and anemia requiring endoscopic resection. Such polyps typically display more abundant fibromuscular stroma, likely the result of mucosal prolapse-type changes. Those who advocate for distinction between Brunner gland hyperplasia and hamartoma note that hamartomas consist of Brunner glands admixed with large ducts and bands of smooth muscle, while hyperplastic lesions are composed only of Brunner glands. For such large polyps, however, the diagnosis of Brunner gland hyperplasia/hamartoma may be appropriate, as it recognizes the relatively arbitrary distinction between these two diagnoses.
In mucosal biopsy specimens, Brunner glands are commonly crushed, imparting a spindle cell appearance to the epithelial cells ( Fig. 21.6B ). Crushed Brunner glands may be mistaken for histiocytic collections, raising concern for Whipple disease, particularly given their intense positive reaction on periodic acid–Schiff (PAS) staining. The spindled appearance of crushed Brunner glands may also raise concern for a mesenchymal lesion. Careful histological evaluation with comparison of adjacent partially crushed lobular collections of Brunner glands will allow for identification of this distortion artifact.
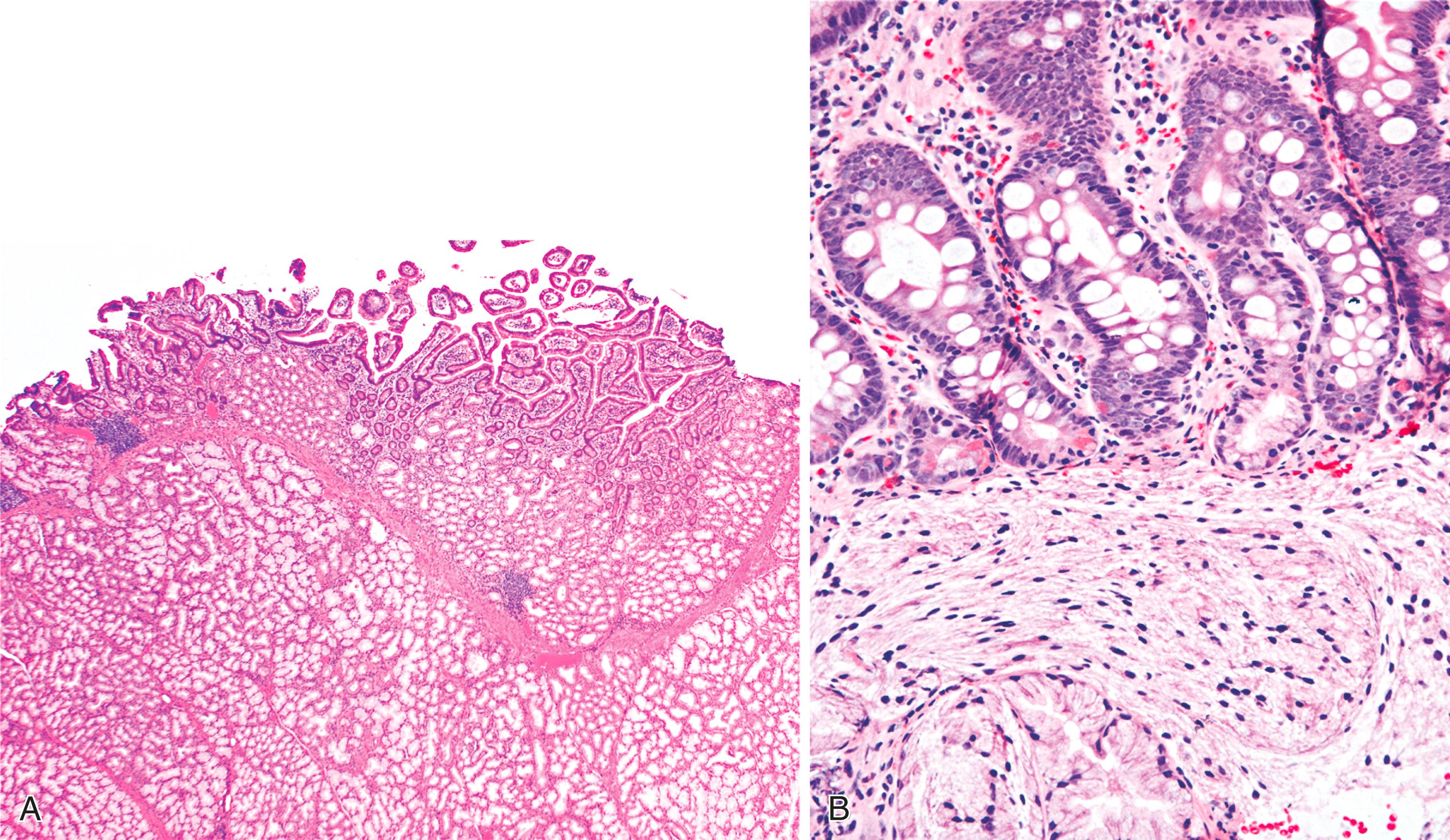
Histological features of peptic-type injury of the duodenum include any of the following: active inflammation within the lamina propria or epithelium, Brunner gland hyperplasia ( Fig. 21.7A ), gastric foveolar metaplasia of the surface epithelium ( Fig. 21.7B ), hemorrhage, and edema. Nodular duodenitis is frequently observed in patients with a history of peptic ulcer disease. In the setting of histological findings indicative of peptic-type duodenal injury, the presence of Brunner gland hyperplasia should be attributed to peptic duodenitis.
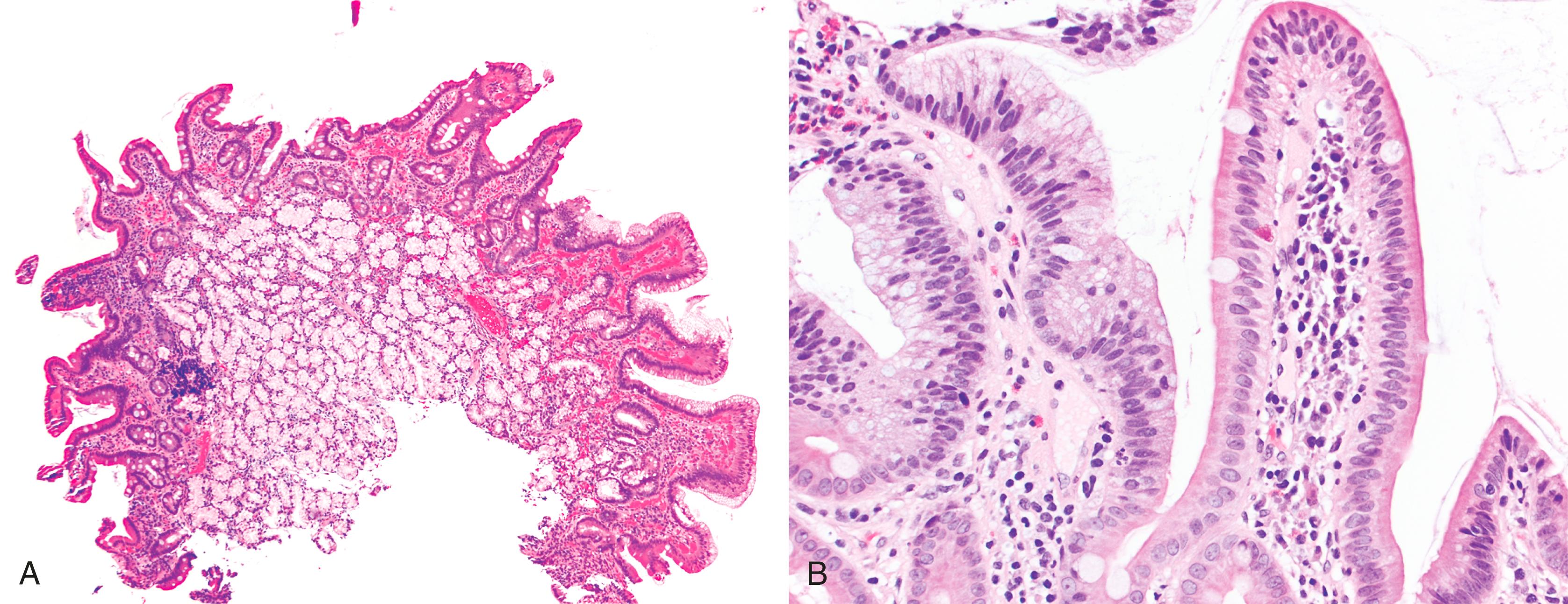
Pyloric gland adenoma is a relatively recently described adenoma subtype that can occur in the duodenum and not infrequently evolves into invasive adenocarcinoma. The distinction between pyloric gland adenoma and Brunner gland hyperplasia/hamartoma is difficult (see later discussion). Both express the mucin core peptide MUC6; however, pyloric gland adenomas usually also demonstrate labeling with MUC5AC, which is not typical of Brunner glands.
Gastric heterotopias in the duodenum are most commonly identified in the bulb and are usually incidental, small (<1.0 cm) nodules that may be multiple. Patients are typically asymptomatic, but larger polyps may manifest as masses and cause obstruction or intussusception. , Peptic ulceration resulting from heterotopic oxyntic glands in the proximal duodenum is rare because the acid secretions are quickly diluted by the alkaline duodenal contents derived from the pancreatobiliary system. However, rare cases of massive GI bleeding have been reported, particularly in patients with extensive heterotopias. , Gastric heterotopia is also associated with concurrent fundic gland polyps of the stomach, suggesting that the increased use of proton pump inhibitors (PPIs) in the general population may enhance its endoscopic detection.
Biopsies of gastric heterotopias display well-organized oxyntic glands composed of chief and parietal cells ( Fig. 21.8A ). The overlying surface often exhibits gastric foveolar-type mucinous epithelium with adjacent normal duodenal intestinal villi. Gastric heterotopia in the duodenum may harbor the same pathological processes that affect the stomach, including Helicobacter pylori infection ( Fig. 21.8B ). Large gastric heterotopias may exhibit secondary mucosal prolapse-type changes including prominence of the muscularis mucosae, submucosal fibrosis, cystic dilation of the oxyntic and mucinous glands, and surface epithelial hyperplasia.

Gastric heterotopia differs from gastric foveolar metaplasia, which is hypothesized to be a response to inflammation caused by peptic injury associated with gastric H. pylori infection ( Table 21.1 ). Histologically, peptic-type injury of the duodenum typically results in active inflammation within the lamina propria or epithelium and erosion or ulceration. The metaplastic gastric foveolar epithelium is not associated with the oxyntic glands characteristic of gastric heterotopia.
| Gastric Heterotopia | Gastric Metaplasia | |
|---|---|---|
| Clinical presentation | Usually incidental | Epigastric pain |
| Endoscopic features | Nodular mucosa | Duodenitis |
| Histological features | Oxyntic mucosa Normal duodenal mucosa |
Gastric foveolar (mucous) cells Villous blunting ± active inflammation Prominent Brunner glands |
| Clinical implications | None | Peptic injury |
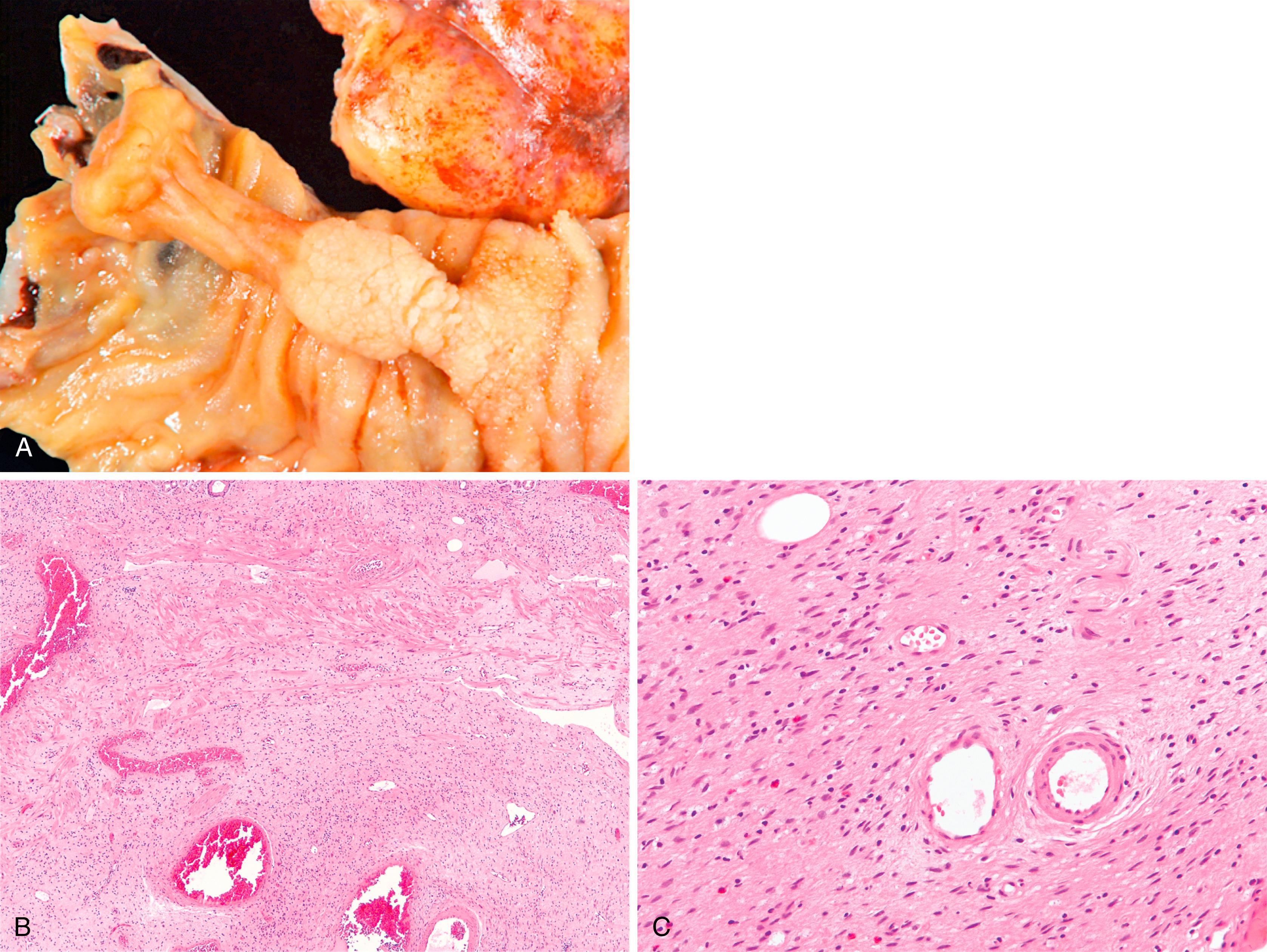
Pancreatic heterotopia represents the presence of pancreatic tissue outside the normal pancreas without any anatomic or vascular connection to the pancreas. It is synonymous with the terms pancreatic rest and ectopic pancreas . Pancreatic heterotopia of the small intestine is most commonly located in the proximal duodenum and can be seen either confined to the mucosa or within the muscularis propria. Pancreatic tissue may be normally identified in the region of the minor papilla and does not necessarily qualify as heterotopia. In most cases, pancreatic heterotopia is asymptomatic and only incidentally detected. Mural heterotopias develop most commonly in the periampullary region and are more likely to be symptomatic as a result duodenal obstruction, intussusception, and stricture or stenosis of the ampulla. Symptomatic mural pancreatic heterotopias often mimic a neoplastic process, leading to surgical resection, and an endoscopic biopsy diagnosis is not possible given the location of the lesion.
Pancreatic heterotopia may be composed of pancreatic acini, ducts, or islets, either alone or in combination ( Fig. 21.9 ). Most frequently, pancreatic ducts are identified, with a variable number of acinar cells and islets and no relationship between symptoms and the cell type present. Mural lesions are often histologically indistinguishable from normal pancreas. Most mucosal-based lesions are unassociated with inflammation, being present in otherwise normal mucosa of the duodenum. Rarely, ductal adenocarcinoma, mucinous neoplasms, and NETs have been reported to develop in pancreatic heterotopia.

A periampullary duodenal wall cyst (groove pancreatitis) is not often confused with mural pancreatic heterotopias. Although both may contain pancreatic ducts, acini, and islets, a periampullary duodenal wall cyst has characteristic clinicopathological features. Most patients are young to middle-aged men who report alcohol use and typically present with pain and vomiting caused by duodenal stenosis. Jaundice may also be seen late in the disease course as a result of compression of the common bile duct. The lesion typically involves the area of the minor papilla where pancreatic tissue may normally be identified. Fibrosis and inflammation occur in the “groove” between the superior aspect of the pancreatic head, the common bile duct, and the duodenum. In addition to groove pancreatitis, alternative names include cystic dystrophy of the duodenal wall and paraduodenal pancreatitis . Most cases are characterized by multiple or solitary cysts varying in size from 1 to 10 cm within the duodenal wall and pancreas ( Fig. 21.10 ). “Solid” types have also been described and are characterized by fibrotic thickening of the duodenal wall with small (<1 cm) cysts. A reactive myofibroblastic spindle cell proliferation is usually present in the duodenal wall, whereas the fibrosis in the groove area is more paucicellular and hyalinized. Cystically dilated ducts partially lined by ductal epithelium and containing inspissated eosinophilic material are characteristic ( Fig. 21.11 ). Squamous metaplasia, granulation tissue, calcium deposition, and giant cell reaction are also frequently encountered. , It has been speculated that this disorder is a localized form of alcohol-induced pancreatitis occurring in pancreatic tissue present in the area of the minor papilla.


By definition, hamartomatous polyps are a haphazard arrangement of normal stromal and epithelial elements. Hamartomatous polyps of the small bowel occur mainly in the setting of polyposis syndromes including Peutz-Jeghers syndrome (PJS), juvenile polyposis syndrome (JPS), and PTEN hamartoma tumor syndrome. The latter two syndromes have similar-appearing hamartomatous polyps. Correct classification of these polyps is critical to management because many of these syndromes carry unique risks of GI and extra-GI malignancy ( Table 21.2 ). Rarely, sporadic hamartomatous polyps resembling their syndromic counterparts occur. Other sporadic hamartomatous lesions have been described, including those made predominantly of mesenchymal elements such as adipose tissue, vessels, and nerves. There are numerous reports of neuromuscular and vascular hamartomas in the literature; however, these lesions are not hamartomas but are likely related to injury from nonsteroidal anti-inflammatory drugs (diaphragm disease) (see Chapter 12, Chapter 16 ). ,
| Syndrome | Polyp Type | Small Bowel Involvement | Histology | Dysplasia In Polyp ∗ | Gene Mutations | Risk of Small Bowel Adenocarcinoma |
|---|---|---|---|---|---|---|
| Peutz-Jeghers syndrome | Peutz-Jeghers polyp | Frequent (>90%) | Normal mucosal elements with arborizing smooth muscle | Rare; possible increased risk of dysplasia in nonpolypoid mucosa | STK11/LKB1 | 13% |
| Juvenile polyposis syndrome | Juvenile polyp | Infrequent (<20%) | Inflamed, edematous lamina propria, cystic glands, often ulcerated | Rare | SMAD4 and BMPR1A | ∼10% |
| PTEN hamartoma tumor syndrome | Multiple types: juvenile polyps, ganglioneuromas, lipomas, and adenomas | Variable (33%-66%) | Variable | Rare | PTEN | Unclear, likely low |
PJS is an autosomal dominant hamartomatous polyposis syndrome with a prevalence of 1 in 50,000 to 200,000 births. The majority of patients have a positive family history; however, 25% of cases are de novo. A diagnosis of PJS should be considered in patients with any of the following: (1) perioral or buccal mucocutaneous melanin pigmentation, (2) two or more histologically confirmed Peutz-Jeghers polyps, and (3) a family history of PJS. More than 90% of patients with PJS develop small intestinal polyps, most commonly in the jejunum, followed by the ileum and the duodenum. PJS patients are often diagnosed at an early age (approximately 20 years) because these polyps often cause abdominal pain, obstruction resulting from intussusception, and bleeding. , Surgical intervention often occurs in these settings. Indeed, patients are at risk for short gut syndrome resulting from the numerous GI surgeries undertaken to resect these polyps.
Patients with PJS have a significantly increased risk of intestinal adenocarcinoma, particularly of the colon (cumulative risk of 39%) but also of the small bowel (13% cumulative risk). , , For this reason, patients are enrolled in an endoscopic screening protocol at a very young age. Affected individuals are also at increased risk for other tumors, including breast (24% to 54% cumulative risk), pancreas (11% to 36% cumulative risk), stomach (29% cumulative risk), cervix (10% to 23% cumulative risk for adenoma malignum), ovary (21% cumulative risk for sex cord/stromal tumor with annular tubules and mucinous tumors), lung (7% to 17% cumulative risk), uterus (9% cumulative risk), and testis (9% cumulative risk for large-cell calcifying Sertoli cell tumor). , , , , The cumulative lifetime risk for any cancer approaches 90%.
Given the increased risk of cancer in these patients, an initial colonoscopy, upper GI endoscopy, and video capsule endoscopy is recommended at 8 years of age. If polyps are identified, surveillance colonoscopy, upper GI endoscopy, and video capsule endoscopy is recommended every 3 years. For those without polyps on this initial screen, repeat colonoscopy, upper GI endoscopy, and video capsule endoscopy is recommended at 18 years of age with continued surveillance every 3 years.
Peutz-Jeghers polyps have an irregular, multilobulated endoscopic appearance and lack the velvety texture typical of adenomas ( Fig. 21.12 ). Because they contain prominent smooth muscle, they are tightly anchored to the bowel. Unlike juvenile polyps, they rarely autoamputate.

In 1998, the gene responsible for the majority of sporadic and inherited cases of PJS was identified as STK11 (also known as LKB1 ), a serine/threonine kinase located on chromosome 19p13.3. , , STK11 is a tumor suppressor gene involved in a wide spectrum of cellular functions, including cellular proliferation, cell polarity, and apoptosis. , STK11 has been shown to interact with other tumor suppressors, including TP53 and PTEN . Approximately one-third of the mutations in PJS patients are caused by large deletions.
There have been case reports of sporadic Peutz-Jeghers polyps, but this is somewhat controversial. In a study at a tertiary care center, three patients with potential sporadic Peutz-Jeghers polyps were identified, but two of them had other features suggestive of PJS, although strict clinical criteria were not met. The authors concluded that if sporadic Peutz-Jeghers polyps do exist, they are extremely rare.
The hamartomatous polyps of PJS are histologically distinctive and consist of disorganized mucosa with prominent smooth muscle bundles. The smooth muscle fibers of the muscularis mucosae form thick cores and permeate the polyps in an arborizing fashion ( Fig. 21.13A ). The epithelium lining these polyps is similar to the normal mucosa in that there are goblet cells, absorptive enterocytes, endocrine cells, and Paneth cells ( Fig. 21.13B ). Rarely, metaplastic bone formation has been reported in these polyps. As much as 10% of Peutz-Jeghers polyps have foci of misplaced epithelium in which the epithelium is located within the submucosa, the muscularis propria, or both. This phenomenon is encountered more frequently in polyps larger than 3 cm in diameter ( Fig. 21.13C ).

The development of Peutz-Jeghers polyps is the subject of some controversy. Given the prominent smooth muscle in these polyps and the presence of misplaced epithelium, some authors suggest that patients with PJS are prone to mucosal prolapse. Indeed, the differential diagnosis of Peutz-Jeghers polyps, particularly in the colon, is mucosal prolapse polyps. However, hemorrhage and hemosiderin deposits, commonly seen in prolapse polyps, are not prominent in Peutz-Jeghers polyps.
The sequence of events that results in GI dysplasia and carcinoma is also unclear in relation to Peutz-Jeghers polyps. One occasionally encounters dysplasia and even carcinoma in a Peutz-Jeghers polyp; however, in a study of 2461 Peutz-Jeghers polyps from 63 patients, only 6 polyps contained foci of dysplasia. These results argue that the hamartoma-adenoma-carcinoma hypothesis proposed by some authors rarely occurs. Peutz-Jeghers polyps have also been shown to be polyclonal, arguing against the possibility of malignant potential. Given these results, it is possible that carcinoma develops in surrounding mucosa that may have an accelerated pathway to dysplasia and carcinoma. ,
In any case, when one encounters dysplasia in a Peutz-Jeghers polyp, the dysplasia should be classified according to a two-tier system (i.e., low-grade or high-grade dysplasia). Care must be taken not to interpret misplaced epithelium as invasive adenocarcinoma. The misplaced epithelium should not have desmoplastic stroma or an infiltrative growth pattern. The epithelial cells should have a similar cytology to other parts of the polyps, and the glands should be invested with lamina propria.
Juvenile polyps of the small intestine are rare and occur in two polyposis syndromes: JPS and PTEN hamartoma tumor syndrome. Juvenile-type polyps can also be seen in Cronkhite-Canada syndrome, an extremely rare, noninherited disorder associated with diffuse GI polyposis. , Juvenile polyps tend to be friable and undergo autoamputation. Endoscopically, they appear smooth and often have evidence of hemorrhage. As a result, patients typically present with complaints related to bleeding, such as occult blood loss, hematochezia, fatigue, or anemia.
First described in 1964, JPS is an extremely rare (1 per 100,000 to 160,000 births), autosomal dominant polyposis syndrome with variable penetrance and phenotypic heterogeneity. Diagnostic criteria include (1) at least five colorectal juvenile polyps, (2) any number of extracolonic juvenile polyps, or (3) any number of juvenile polyps in patients with a family history of JPS. Three clinical phenotypes of JPS have been described: infantile juvenile polyposis, juvenile polyposis coli, and generalized juvenile polyposis . Infantile juvenile polyposis syndrome, the most severe type, is characterized by early and diffuse involvement of the entire GI tract. Because these hamartomatous polyps have surface erosion, affected patients usually present with GI bleeding and anemia. JPS is also associated with a significantly increased risk of adenocarcinoma in involved GI sites. The colon is the site most commonly involved by disease, with the risk of colorectal cancer ranging from 17% to 22% by 35 years of age and nearing 68% by 60 years of age. The lifetime risk of developing carcinomas of the stomach, small intestine, or pancreas is approximately 10% to 15% ; however, in those with SMAD4 mutations, the risk of gastric cancer approaches 30%. Prevention of intestinal adenocarcinoma mandates intensive screening, with colonoscopy and upper GI endoscopy every 1 to 3 years, beginning 12 to 15 years of age. Severe GI bleeding may necessitate immediate surgery. Progression to dysplasia in a polyp is not necessarily an indication for surgery, provided that it can be completely removed. Given the increased risk of adenocarcinoma, genetic screening of family members of affected individuals is essential.
Mutations in two genes have been described in patients with JPS: SMAD4 (also known as MADH4 or DPC4 ) and BMPR1A (also known as ALK3 ). , Approximately 25% of patients with JPS have a negative family history, and their polyps presumably arise from de novo mutations. The proteins encoded by these genes are involved in the transforming growth factor-β (TGF-β) signal transduction pathway. Because the TGF-β signaling pathway mediates growth inhibitory signals, proteins in this pathway function as tumor suppressors. Mutations in SMAD4 have been shown to be more common in patients with upper GI polyps, whereas patients with BMPR1A mutations are more likely to present at a younger age than those with mutations in SMAD4 .
The term PTEN hamartoma tumor syndrome was coined to encompass all hamartoma syndromes that arise through mutations in the tumor suppressor, PTEN . This term encompasses Cowden syndrome, Bannayan-Riley-Ruvalcaba syndrome (BRRS), Proteus syndrome, and others. , Patients with these syndromes have a wide variety of GI, skin, and soft tissue tumors and an increased risk for carcinomas of the colorectum, thyroid, breast, and endometrium.
Cowden syndrome is 50% less common than JPS and is characterized by multiple hamartomatous tumors of endodermal, mesodermal, and ectodermal origin. The most distinctive lesions are mucocutaneous—in particular, trichilemmomas, acral keratosis, subcutaneous lipomas, palmoplantar keratoses, oral cobblestoning, and oral papillomas. Colonic polyps in Cowden syndrome are common and include ganglioneuromas, adenomas, inflammatory-type polyps, juvenile polyps, and less frequently lipomas and leiomyomas. Although rarer, similar polyps can occur in the small intestine and stomach, with frequencies ranging from 37% to 66%. , Current American College of Gastroenterology (ACG) guidelines recommend surveillance colonoscopy and upper GI endoscopy every 2 years beginning at 15 years of age.
BRRS is also associated with juvenile polyps of the GI tract (mostly limited to the ileum and colon); however, these patients also have neurological findings, including macrocephaly and slowed psychomotor development. Some BRRS patients also develop extraintestinal hamartomatous tumors similar to those seen in patients with Cowden syndrome.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here