Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
When coronary revascularization is considered, prognostic and symptomatic indications must be distinguished.
In general, percutaneous coronary intervention (PCI) for single-vessel disease is justified only if an improvement of symptoms can be anticipated or ischemia that comprises more than 10% of the left ventricle can be relieved.
With multivessel disease or left main coronary artery (LMCA) stenosis, the decision for PCI versus coronary artery bypass grafting (CABG) depends on the procedural risk, the ability to achieve complete revascularization, the presence or absence of diabetes mellitus, and the complexity of the coronary artery involvement, which can be gauged by the Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery (SYNTAX) score.
With multivessel disease or LMCA stenosis and low anatomic complexity (i.e., SYNTAX score below 23) in the absence of diabetes mellitus, the 5-year outcome of PCI is similar to that after CABG, provided that complete revascularization can be achieved. Thus PCI can replace bypass surgery in this setting.
In general, outcomes with CABG are superior to those with PCI in multivessel disease with intermediate-to-high anatomic complexity (i.e., SYNTAX score above 22) and CABG is the treatment of choice in this setting.
In the subset of patients with LMCA stenosis, there is no compelling evidence for superiority of CABG over PCI. Thus at least in patients with intermediate SYNTAX-Scores (23 to 32), PCI is a reasonable alternative to CABG.
In patients with multivessel disease, diabetes mellitus is the strongest predictor of superior outcomes with CABG. CABG is the treatment of choice in this setting. Nevertheless, PCI may be considered in selected patients with a low SYNTAX score.
In many instances, individualized decisions must be made jointly by the cardiac surgeon and the interventional cardiologist at institutionalized meetings of the heart team.
When the era of interventional cardiology began, with the pioneering work of Andreas Grüntzig on plain balloon angioplasty, percutaneous coronary intervention (PCI) was a treatment option only for isolated proximal coronary lesions that did not involve the ostium or the left main coronary artery (LMCA) stem. In the late 1980s, coronary stents were developed with the goal of reducing the risk of restenosis and achieving a more predictable acute result of angioplasty, thus avoiding the dreaded abrupt closure as a result of dissection. As shown subsequently, stents were successful in achieving this goal. Nevertheless, they created a new problem: subacute stent thrombosis. After intense research on periinterventional and postinterventional antithrombotic treatment, the concept of dual- or triple-antiplatelet therapy emerged, which significantly reduced the incidence of this complication. The use of coronary stents in conjunction with optimized antithrombotic treatment extended the spectrum of coronary lesions for which PCI was considered a reasonable treatment option and thereby led to a substantial expansion of interventional techniques. Because of the large number of patients who were being treated with coronary stents, restenosis as a result of neointima formation became a serious problem. Although various studies demonstrated that compared with plain balloon angioplasty, stents reduced the need for reintervention, restenosis rates continued to be relevant and ranged from just above 10% in the simplest lesions to more than 50% with diffuse disease in patients with diabetes.
Thus it is not surprising that the community of interventional cardiologists celebrated the advent of drug-eluting stents (DESs) as a major breakthrough, given that the initial studies suggested restenosis rates of zero. In the meantime, it has become clear that compared with bare-metal stents (BMSs), DESs reduce the need for target-vessel reintervention by around 80%, thus largely reducing—but not eliminating—the problem of restenosis. Subsequently, DESs led to another massive expansion of the proportion of patients treated with PCI. With the widespread use of these stents for PCI, reports appeared that pointed to a new problem that had not been seen with BMSs: late stent thrombosis. Yet a thorough reevaluation of the data from randomized studies—with uniform application of definitions for definite, probable, and possible stent thrombosis—failed to confirm these alarming initial reports. Nevertheless, a meta-analysis of studies with first-generation paclitaxel- and sirolimus-eluting stents suggested a slight increase in the risk of very late (>1 year) stent thrombosis after the placement of DESs compared with BMSs. In the meantime, DESs with improved design have replaced the first-generation paclitaxel- and sirolimus-eluting stents. For the most part, these next-generation DESs are more efficacious and safer than first-generation DESs. Specifically, a network meta-analysis based on 49 trials that included 50,844 randomly assigned patients showed a 2-year risk of stent thrombosis for the everolimus-eluting stents that was even lower than that for BMSs.
Despite the remaining problems of PCI, its use has increased exponentially over the past few decades. Initially, this increase has come at the expense of lone medical therapy. More recently, however, with the advent of DESs, a shift has been seen in patients with multivessel disease and other complex coronary anatomies from coronary artery bypass grafting (CABG) to PCI. This shift has been facilitated by both physician and patient preference for the supposedly easier approach to coronary revascularization, given the idea that the problem of restenosis has been largely solved. However, reasonable concern exists that this shift has led to the overuse of PCI and that in some patients, it may not yield the same outcome as CABG, which for a number of indications is an established treatment option with a well-documented survival benefit compared with that of medical therapy.
In comparing PCI with medical treatment alone or with bypass surgery, it is important to scrutinize the available evidence that PCI offers at least as great a benefit as CABG on the one hand or a greater benefit than medical treatment alone on the other. This review summarizes and discusses the currently available evidence so as to present a rationale for clinical decision making.
Pharmacologic therapy and coronary revascularization, by either CABG or PCI, are the mainstays of treatment for coronary artery disease (CAD). The prime objective of such treatment is improved survival (prognostic indication); other reasonable treatment goals are relief of symptoms and improved quality of life (symptomatic indication). In pursuing these goals, the prevention of myocardial infarction (MI) is a key issue that pertains to both survival and quality of life. In deciding on the optimal revascularization strategy in a patient with CAD, it is necessary to determine first whether a prognostic or symptomatic indication for coronary revascularization exists and then to choose the most appropriate revascularization modality. This chapter presents criteria for both of these elements in clinical decision making and focuses primarily on the prognostic indication for coronary revascularization. Based on a review of the general criteria for revascularization, the efficacy and safety of PCI compared with CABG are discussed, and the role of PCI in symptomatic indications for coronary revascularization is also addressed, predominantly in comparison with medical therapy alone. The focus is on stable coronary disease. Acute coronary syndromes (ACSs) that include MI are discussed in depth in other chapters.
Several factors must be considered in regard to clinical presentation. Among these are ST-segment changes, cardiac troponin release, and ischemic area, which can help the clinician discern the appropriate treatment.
The prognostic benefit of myocardial revascularization in patients with MI with or without ST-elevation or unstable angina with high-risk features is well established. The evidence is reviewed in detail in Chapters 18, Chapters 19
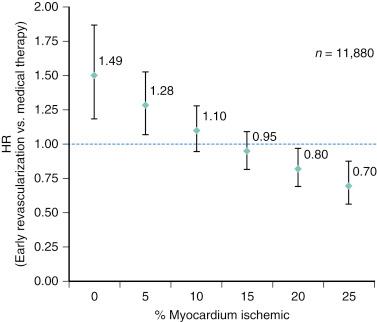
Among patients with chronic stable angina, those with severe angina, large or multiple perfusion defects on functional testing, or a low threshold for the induction of ischemia ( Box 2.1 ) have a poor prognosis, with an annual mortality risk greater than 3%. If these high-risk features are associated with double- or triple-vessel disease, patients benefit from revascularization irrespective of left ventricular (LV) function. In an analysis of 5303 patients in the Coronary Artery Surgery Study (CASS) registry, surgical benefit was greatest in patients who exhibited at least 1 mm of ST-segment depression and could exercise only into stage 1 or less. In the surgical group with triple-vessel disease and severe exercise-induced ischemia, 7-year survival was 81%, whereas it was 58% in the corresponding medical group. Likewise, in another registry that included 2023 patients with severe angina and two-vessel disease, 6-year survival was 76% in patients treated medically and 89% in patients treated surgically ( P < .001). Cox multivariate analyses showed that surgical treatment was a beneficial independent predictor of survival for patients with two-vessel coronary disease and Canadian Cardiovascular Society (CCS) class 3 or 4 angina. The Asymptomatic Cardiac Ischemia Pilot (ACIP) is a more recent trial that was designed to compare the efficacy of medical therapy versus revascularization. In ACIP, 558 patients with angiographically documented CAD, mostly multivessel disease, and stable CAD were randomly assigned to medical therapy, adjusted either to suppress angina or to suppress both angina and evidence of ischemia during ambulatory electrocardiogram (ECG) monitoring or revascularization with either PCI or CABG. Revascularization was significantly more effective in relieving ischemia than either of the medical strategies. During 1-year follow-up, the ACIP trial appeared to show better outcomes in patients treated with revascularization; mortality was 4.4% and 1.6% in the two conservative groups, whereas none of the patients in the revascularization group had died during the 1-year follow-up period. The apparent benefit of revascularization was largely confined to patients with double- or triple-vessel disease. Expanding the series done in 2003 and 2006, a large observational study of patients who underwent exercise or adenosine myocardial perfusion single photon emission computed tomography (SPECT) analyzed the impact of the extent of inducible myocardial ischemia and of myocardial scar on the potential survival benefit of revascularization. The study included 13,969 consecutive patients: 12,329 were treated medically, and 1226 underwent early revascularization (501 CABG, 725 PCI) in the first 90 days after scintigraphy. During a median follow-up of 8.9 years, 3893 patients (27.9%) died. Hazard ratios for all-cause death that compared early revascularization with medical treatment were derived from Cox models adjusted for logistic-based propensity scores for revascularization, extent of ischemia and scar, and baseline characteristics. In 11,880 patients without large myocardial scars (<10% fixed myocardial defect), a progressive decrease in the hazard ratios was found with increasing extent of inducible ischemia ( Fig. 2.1 ). Thus the risk associated with conservative treatment increased as a function of percent myocardium ischemic, whereas the risk associated with revascularization decreased. Equipoise between revascularization and conservative treatment was reached at about 10% ischemic myocardium (see Fig. 2.1 ). Only in patients with extensive scar (≥10% fixed myocardial defect) was the ischemia treatment interaction not significant ( P = .469), and an advantage of revascularization over conservative treatment could not be established. In summary, in the absence of extensive myocardial scars, myocardial revascularization compared with conservative treatment improved long-term survival in patients with significant (>10% myocardium) inducible ischemia. Early myocardial revascularization was, however, harmful in patients with minimal inducible ischemia.
High-risk treadmill score
Stress-induced large or moderate-sized nuclear perfusion defect (particularly if in the anterior wall)
Stress-induced multiple perfusion defects with left ventricle dilation or increased lung parenchymal uptake of thallium-201 isotope
Echocardiographic wall-motion abnormality involving more than two segments, developing at a low dose of dobutamine (≤10 μg/kg/min) or at a low heart rate (120 beats/min)
Stress-induced echocardiographic evidence of extensive ischemia
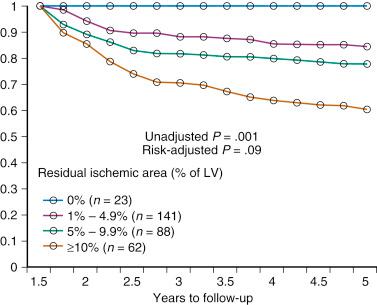
Consistent results were obtained in a nuclear substudy on 314 patients in the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial. In this substudy, the extent of residual posttreatment ischemia—assessed as a percentage of the left ventricle by myocardial perfusion SPECT—was a predictor of outcome: rates of death or MI ranged from 0% to 39% for patients with no residual ischemia to 10% or more residual ischemia despite treatment ( P = .001 [risk-adjusted P = .09]; Fig. 2.2 ). With respect to treatment, a 5% or more reduction in ischemic myocardium lowered the risk of death or MI ( P = .037 [risk-adjusted P = .26]), particularly if baseline ischemia was 10% or greater ( P = .001 [risk-adjusted P = .08]). PCI on top of optimal medical therapy increased the likelihood of achieving this goal. The findings of this substudy suggest that revascularization is indicated if, in addition to optimal medical therapy, it affords at least a 5% reduction in myocardial ischemia.
Thus, although adequately powered randomized trials to address the impact of severe angina or large perfusion defects on outcome in patients with chronic stable angina are lacking, the bulk of the currently available evidence suggests that these patients benefit from revascularization, particularly if more than one vessel is affected.
Until now our understanding of the anatomic conditions that constitute a survival benefit from coronary revascularization compared with medical therapy alone has been largely based on milestone studies performed during the 1970s. Soon after CABG was introduced in 1969, three randomized trials compared surgical revascularization with lone medical therapy: the Veterans Administration (VA) Study, the European Coronary Surgery Study (ECSS), and CASS. Although these studies are outdated in many respects, including a low use of arterial conduits and limited means of pharmacologic risk-factor modification and platelet inhibition, it is unlikely that they will ever be replicated. In concert with analyses of large registry databases, these early studies established the conditions under which CABG improves survival compared with medical therapy ( Box 2.2 ). A meta-analysis of the seven published randomized trials of CABG versus medical treatment alone for CAD identified left main CAD (diameter of stenosis ≥50%), multivessel disease, and involvement of the proximal left anterior descending coronary artery (LAD) as significant predictors of a survival benefit from CABG. In the cumulative experience of the seven studies, the VA study being the first, surgical revascularization for left main CAD was associated with a 65% relative reduction in mortality compared with medical therapy alone. Notably, in left main CAD, a survival benefit of surgery was reported irrespective of the presence or absence of spontaneous or inducible symptoms or signs of ischemia or reduced LV function. The same is also true for triple- or double-vessel disease involving the proximal LAD.
Left main coronary artery disease
Triple- or double-vessel disease involving the proximal left anterior descending coronary artery
Triple- or double-vessel disease in the presence of severe angina or large areas of ischemia on functional testing
Triple-vessel disease associated with impaired left ventricle function
In all other conditions, the indication for surgical coronary revascularization depends on a combination of anatomic and clinical criteria. If triple-vessel disease is associated with impaired LV function (LV ejection fraction <50%), surgical revascularization improves survival irrespective of LAD involvement. In the presence of severe angina or large areas of ischemia on functional testing, surgical revascularization of triple- or double-vessel disease is also indicated for both symptomatic and prognostic reasons even in the absence of LV dysfunction. Myocardial revascularization has never been shown to confer a survival benefit in patients with single-vessel disease. This is also true for isolated proximal LAD stenoses. Yusuf and colleagues’ meta-analysis that showed a survival benefit from surgery in patients with LAD involvement must be interpreted with the notion that this result was obtained in a cohort that had predominantly multivessel disease, and no dedicated subgroup analysis was undertaken for isolated LAD stenosis. More recently, the randomized Medicine, Angioplasty, or Surgery Study (MASS) trial compared medical treatment alone with plain balloon angioplasty or CABG in 214 patients with symptomatic, isolated, high-grade stenosis of the LAD. During a 5-year follow-up, no appreciable difference was found among the three treatment arms with regard to either death or MI. Although the power to detect small differences in event rates was low in MASS, its results are consistent with the current judgment that no prognostic indication exists for coronary revascularization in stable single-vessel disease. No study ever demonstrated that in patients with stable angina, the risk of subsequent MI could be reduced by either bypass surgery or PCI. The degree of stenosis is a notoriously poor predictor of subsequent events. Although the risk of subsequent MI is higher with high-grade stenoses than with low-grade stenoses, the latter are far more frequent. Thus the majority of infarctions are triggered by low-grade stenoses. Despite advances, our current means of identifying vulnerable plaques are limited.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here