Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A 64-year-old female presents to the Gynecologic Oncology clinic for a second opinion regarding a new diagnosis of uterine serous carcinoma. She originally presented to her gynecologist within the last 3 months complaining of postmenopausal vaginal spotting. Pelvic ultrasound demonstrated a 3 mm endometrial stripe and endometrial sampling was deferred. However, the spotting persisted, and hysteroscopy, dilation and curettage (D&C) was performed. Final pathology demonstrated uterine serous carcinoma in an endometrial polyp. Computed tomography (CT) of the chest/abdomen/pelvis demonstrated no obvious evidence of metastatic disease. Surgical staging was performed with minimally invasive total hysterectomy (TAH), bilateral salpingo-oophorectomy (BSO), and pelvic and aortic lymph node lymphadenectomies. Final pathology demonstrated serous endometrial cancer without lymphovascular space invasion. However, one of the right pelvic lymph nodes was positive for disease. The patient presents to clinic inquiring about additional recommended work-up and suggestions for adjuvant treatment.
Uterine corpus cancer is the third most common cancer in women in the United States, with approximately 65,620 new cases in the United States in 2020 and 12,590 estimated deaths. Endometrial cancers can broadly be classified into two groups. Cancers of endometrioid histology are common (about 80% of cases), are associated with obesity and estrogen exposure, and generally have good prognosis. Nonendometrioid cancers are more rare (~ 20%), are less hormone dependent, and have poorer prognosis.
Endometrial cancer incidence has been steadily rising in the United States and many other areas internationally, with an average annual percent change in the United States of 1.4%–3.1%. Analysis of the SEER database in the United States has suggested that this increase is largely driven by nonendometrioid cancers.
Uterine serous carcinoma is the most common form of nonendometrioid uterine corpus cancer. Data from the Surveillance, Epidemiology, and End Results (SEER) database from 1988 to 2001 suggest uterine serous carcinoma accounted for about 10% of uterine cancers but 39% of deaths. Uterine serous carcinoma is more likely to be stage III or IV at presentation (52% of cases), even compared with other high-risk endometrial cancer histologies (36% for clear cell and 29% for grade 3 endometrioid carcinoma). Median age at diagnosis is in the 60s ( Table 12.1 ).
| Endometrioid endometrial carcinomas | Non-endometrioid, including uterine serous, carcinomas | |
|---|---|---|
| Risk factors | Obesity Exposure to unopposed estrogen Insulin resistance |
Older age History of breast cancer |
| Pattern of recurrence | Locoregional: Vaginal/pelvic | Distant: Extrapelvic |
| Precursor lesion | Complex atypical hyperplasia / endometrial intraepithelial neoplasia | |
| Histologic grade | Largely grade 1/2 | Always high grade |
| Stage I/II at diagnosis | 70%–80% | 50%–60% |
| 5-year OS for stage I/II disease | 70%–95% | 50%–85% |
| Molecular features | PTEN inactivation Microsatellite instability |
TP53 mutation HER2 amplification Mutations in PIK3CA, PPP2R1 , FBXW7, CHD4 |
When first described, uterine serous carcinoma was noted for its histological similarity to ovarian papillary serous carcinoma. Prominent features included marked nuclear pleomorphism, complex papillary architecture, fibrous stalks, prominent tumor necrosis, and frequent psammoma bodies. Additionally, disease was often noted within the myometrium, ovarian lymphatics, or vascular channels without evidence of gross disease.
Later studies also observed that within uterine serous carcinoma, different histological subtypes are apparent, including disease admixed with other histologies such as endometrioid or clear cell and disease confined to endometrial polyps. Overall, the authors found that these tumors were clinically similar in their aggressive behavior and shared hobnail-shaped cells with high-grade nuclei with foci of papillary or glandular architecture. These findings led them to propose renaming the disease to uterine serous carcinoma. Subsequent studies have found that mixed adenocarcinomas with serous components, even if they contained some endometrioid or clear cell components, should be treated similarly to uterine serous carcinoma as the presence of a serous component was associated with worse prognosis.
The relationship between myometrial invasion and disease severity also differs in uterine serous carcinoma compared to endometrioid endometrial cancer. While extrauterine spread is rare in low-grade endometrioid endometrial cancer with limited myometrial invasion and small tumor size, early studies found that patients with non-invasive uterine serous carcinoma had a high risk of stages III–IV disease and high rates of death from disease. Subsequent studies have affirmed these findings. Noninvasive or early invasive forms of uterine serous carcinoma have been variably termed “intraepithelial serous carcinoma” and “endometrial intraepithelial carcinoma” or “superficial serous carcinoma,” respectively, and have previously been felt to represent earlier forms of disease. However, in one study, 3 of 9 (33%) patients with noninvasive disease and 15 of 31 (48%) patients with early invasive disease had extrauterine spread. Considering these groups together as “minimal uterine serous carcinoma” to highlight the invasive potential of these uterine lesions, 5-year survival was 94% for patients without extrauterine spread vs 56% with extrauterine spread. Thus, even for apparently polyp-confined disease, complete surgical staging with detailed pathological scrutiny to rule out extrauterine spread is advised. Though noninvasive disease does not rule out extrauterine spread, larger studies have found that depth of myometrial invasion does predict worse outcome, with 38% of patients with noninvasive disease having stages III–IV disease vs 80% of patients with depth of invasion ≥ 50%.
Efforts at identifying a true precursor lesion to uterine serous carcinoma led to description of a distinct entity termed endometrial glandular dysplasia in 2004. This is morphologically distinct from both benign endometrium and uterine serous carcinoma, has molecular features shared with subsequently diagnosed uterine serous carcinoma, and can be diagnosed by EMB or D&C. The clinical implications of this pathological entity are still unclear, but point to key steps in the development of uterine serous carcinoma.
New capabilities in molecular characterization have further helped elucidate the biological underpinnings of uterine serous carcinoma. Mutations in TP53 are the most common genetic alteration in uterine serous carcinoma, present in over 90% of cases. While TP53 mutations are never observed in resting endometrium, they occur in 43% of endometrial glandular dysplasia and 72% of serous endometrial intraepithelial carcinoma, suggesting an early role of TP53 in uterine serous carcinoma carcinogenesis. An even earlier lesion termed “p53 signature glands” may represent an even earlier form of disease identifiable only by immunoassay rather than histological change. As TP53 is the most commonly mutated gene in human cancer, improved understanding of the biology of uterine serous carcinoma may yield insights into other cancer types, and vice versa. Other recurrently mutated genes in uterine serous carcinoma include PIK3CA , FBXW7 , and PPP2R1A.
Risk factors for uterine serous carcinoma include African-American race, older age, and multiparity. Overall, uterine serous carcinoma is approximately twice as common in non-Hispanic black women than in other populations (non-Hispanic white, Hispanic, Asian) in a US study population from 2000 to 2011. In addition, while rates of low-grade endometrioid endometrial cancer decreased slightly in non-Hispanic whites (annual percent change or APC = − 0.8) and increased slightly in non-Hispanic blacks (APC = 1.0), rates of uterine serous carcinoma are increasing in all racial groups. However, the increase in rate is more pronounced in minority populations, with rates of increase ranging from APC = 2.8 in non-Hispanic Whites to 3.8 in non-Hispanic blacks to 4.5 in Hispanics and 9.0 in Asians. Non-Hispanic blacks face worse outcomes in endometrial cancer even controlling for histology and stage of disease, and this trend sadly is also true in uterine serous carcinoma. Non-Hispanic white, Asian, and Hispanic populations experience 5-year survival rates of about 80%, 40%, and 20% for localized, regional, and distant disease. Analogous figures for non-Hispanic black populations are approximately 70%, 30%, and 10%. The reasons behind the rising rates and racial disparities seen in uterine serous carcinoma are poorly understood and likely multifactorial.
An area of much controversy is the association between BRCA1/2 alterations and risk of development of uterine serous carcinoma. Past studies have linked a history of breast cancer with increased risk of subsequent development of uterine serous carcinoma. BRCA1 alterations are disproportionately common in patients with uterine serous carcinoma and may predispose to both breast and uterine cancers. Specifically, loss-of-heterozygosity events have been seen in uterine serous carcinoma in a BRCA1 germline carrier, suggesting a causal role for this mutation. That said, the risk of developing uterine serous carcinoma in women who are germline positive for BRCA remains undefined. The recommendation for a hysterectomy at the time of risk reducing bilateral salpingo-oophorectomy (RR-BSO) in women with germline BRCA1 mutation remains controversial but should be discussed with patients.
The oncogenic potential of the human epidermal growth factor receptor 2 (HER2) is well known. HER2, encoded by the gene ERBB2 , is receptor tyrosine kinase. Among all four HER family proteins, HER2 has the strongest catalytic kinase activity and functions as the most active signaling complex of the HER family after dimerization with other HER family members [1, 2]. Overexpression of HER2 leads to increased homodimerization (HER2:HER2) and heterodimerization (e.g., HER2:HER3), which initiates a strong pro-tumorigenic signaling cascade [3]. Overexpression of this oncogene plays an important role in the development and progression of certain aggressive types of breast, gastric and uterine cancers [4–7]. While the protein has become an important biomarker and target of therapy for approximately 30% of breast cancer patients, has also been identified that 25%–30% of women with uterine serous carcinoma also overexpress HER2.
Serous carcinoma of the endometrium most commonly occurs in atrophic uteri and can present with grossly identifiable tumor or as microscopic disease, often involving endometrial polyps. Myometrial invasion as well as adnexal involvement may be grossly evident or only identified microscopically.
Uterine serous carcinoma can demonstrate glandular ( Fig. 12.1 A), papillary ( Fig. 12.1 B) or solid architecture. Although there can be substantial variability in the cytologic features, marked cytologic atypia is a hallmark of serous carcinoma ( Fig. 12.1 A ). The tumor cells may have eosinophilic cytoplasm or cytoplasmic clearing, sometimes with hobnail features. Many, if not all, of the tumor cells show nuclear pleomorphism, hyperchromasia, and prominent eosinophilic nucleoli. Mitotic figures, including abnormal mitotic figures, are easily identified, as are smudged nuclei. When serous carcinoma arises in an endometrial polyp, the tumor cells line the surface of the polyp and may show extension into underlying glands with no associated stromal invasion. This phenomenon can also be seen in the native endometrium. This lesion was previously called serous endometrial intraepithelial carcinoma. However, given that the purely intraepithelial carcinoma can be associated with metastatic disease, it is now recommended to simply refer to it as serous carcinoma. Uterine serous carcinomas are all considered high grade, thus the use of “high grade” to further describe these tumors is considered redundant and not recommended. Lymphovascular invasion can be focal or extensive. Careful attention to exclude other mixed components including endometrioid carcinoma, clear cell carcinoma and sarcoma is necessary.
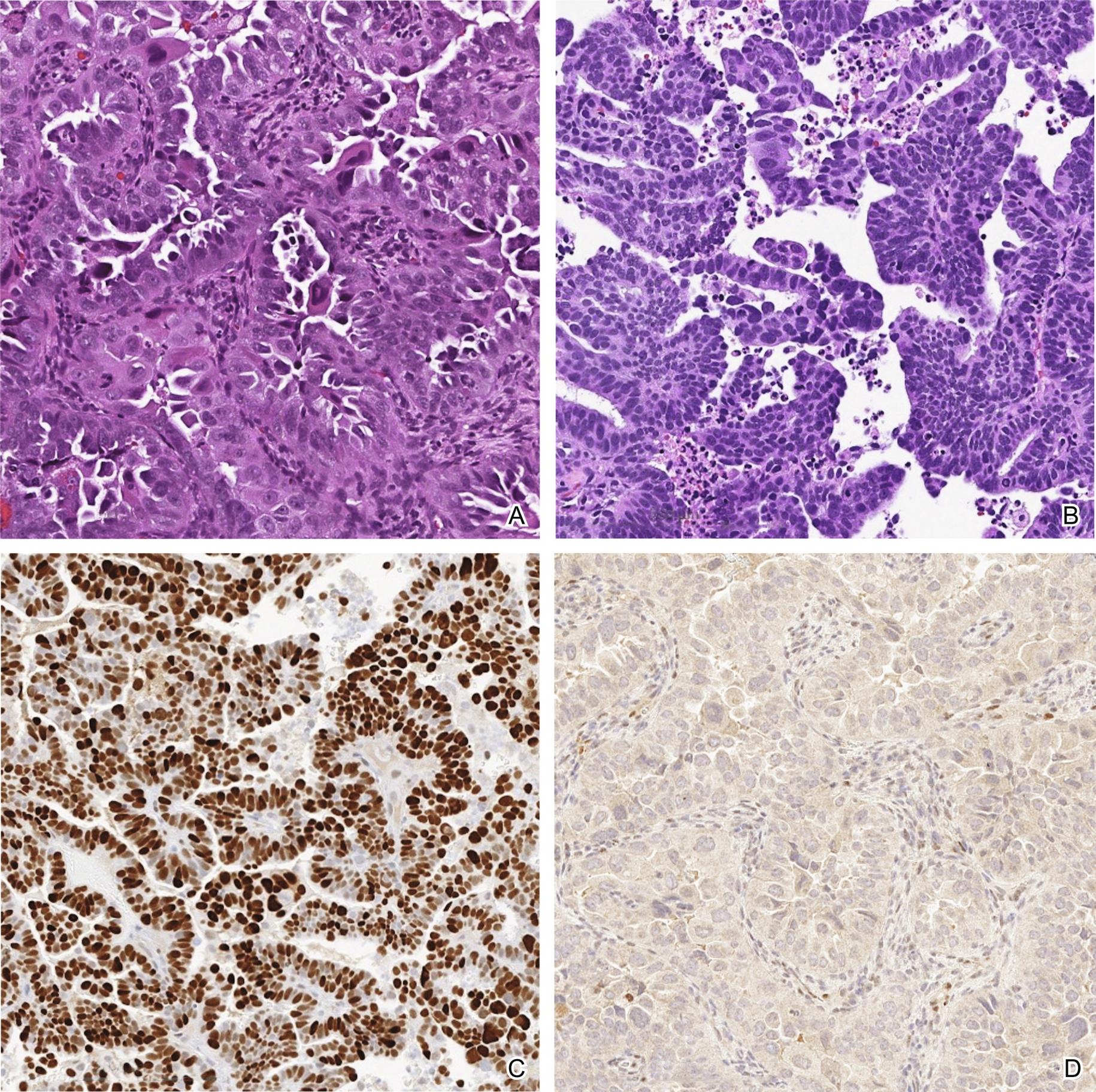
Serous carcinomas express epithelial markers such as keratins and EMA, PAX-8, and hormone receptors though the latter show decreased expression compared to endometrioid carcinomas. The most common pattern of aberrant expression of p53 is diffuse strong nuclear staining in > 75% of the tumor cells ( Fig. 12.1 C). However, in a subset of cases, there is complete loss of staining consistent with a null-phenotype ( Fig. 12.1 D). In the latter, positive staining in internal controls is necessary to ensure authenticity of the absent staining. More recently diffuse cytoplasmic staining has also been recognized as a pattern of aberrant p53 expression.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here