Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The functions of the fetal kidney in utero remain little understood. Prior to delivery, the placenta regulates fetal homeostasis of salt and water, and fetal acid-base balance in the context of the acid load generated by intrauterine metabolism and growth. The fetal kidney in utero generates the amniotic fluid that contributes to fetal pulmonary development and provides mechanical protection to the enlarging fetus. Parturition marks the abrupt beginning of the fetal transition to extrauterine life, accompanied by transfer of the responsibility for organismal homeostatic regulation from the maternal placenta to the fetus’ own regulatory mechanisms, including those of the kidney. This transition is accompanied by initiation of breathing and by rapid increases in blood pressure, cardiac output, and renal blood flow, as well as by the start of postnatal nephron maturation. Although human nephrogenesis is complete by 36 weeks gestation, the neonatal kidney continues to mature in many ways during the first 1 to 2 years of life before it achieves adult-level function as scaled to body mass. This chapter will review the neonatal kidney’s handling of systemic acid-base balance, after briefly reviewing urinary acidification mechanisms of the adult kidney. Acid-base regulation by the neonatal and postnatal kidney has been the subject of several recent reviews.
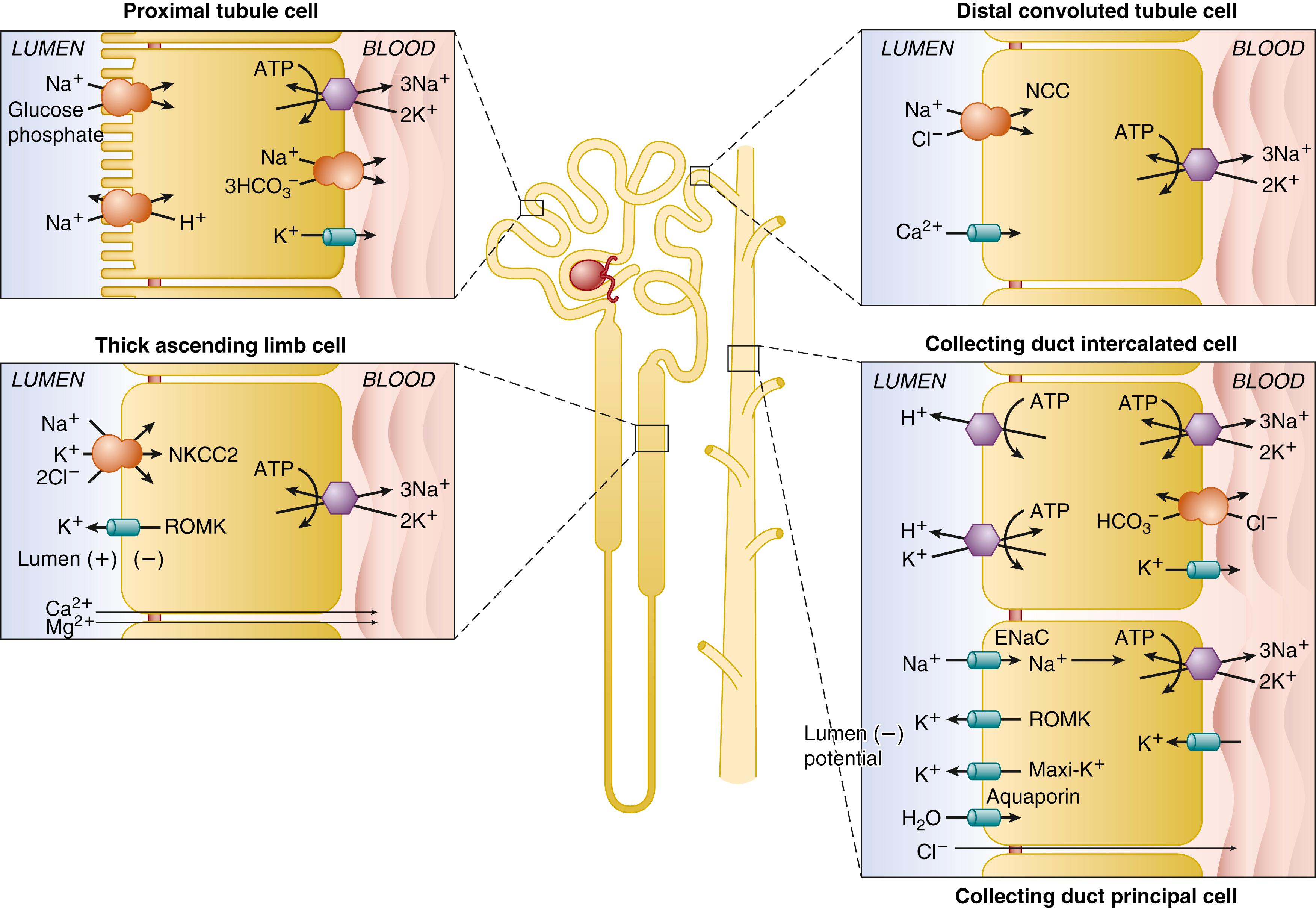
Acid-base buffering in the body is accomplished by the volatile CO 2 /HCO 3 − buffer system , with lesser contribution from fixed buffers, including phosphates, ammonium, citrate, and the proteins, proteoglycans, and numerous mobile buffers of cells and extracellular fluids. The 2 million glomeruli of the two human adult kidneys generate 150 to 180 L of glomerular filtrate each day. This filtrate contains 4000 mmoles of HCO 3 − that must be recovered to sustain systemic buffering and to provide one-carbon equivalents for biosynthetic pathways. Eighty percent of this HCO 3 − reclamation is accomplished by the proximal tubule, with the remainder reabsorbed by the thick ascending limb (TAL) and less quantity by downstream nephron segments ( Fig. 103.1 ). The adult body on a regular Western diet also generates approximately 50 to 70 mmoles of protons from metabolism that must be excreted by the kidney to avoid uncompensated neutralization of HCO 3 − , with its resultant loss to the environment through pulmonary exhalation of CO 2 . This process is mediated by secretion of H + into the urinary space by the type A intercalated cells of the collecting duct, which also reabsorb molar equivalent HCO 3 − newly generated in the process (see Fig. 103.1 ). The secreted proton equivalents are trapped by the major urinary buffer ammonium, with smaller buffering contributions from phosphate, citrate, and other organic ions.
HCO 3 − reabsorption by the proximal tubule is carried out largely in the convoluted segments S1 and S2 ( Fig. 103.2A ), whereas the proximal straight tubule (S3 segment) preferentially reabsorbs chloride. The proximal tubule reabsorbs 70% to 90% of filtered HCO 3 − , mostly via transcellular ion transport. Paracellular transport across tight junctional barriers via claudins 10a and 17 accounts for most of reabsorbed Cl − , whereas claudin 17–assisted paracellular transport contributes up to 20% of reabsorbed HCO 3 − and to a portion of paracellular Cl − transport. As shown in Fig. 103.2A , the microvillar apical membrane of the proximal convoluted tubule secretes protons to acidify the urinary space. Proton secretion is mediated 65% by the sodium/proton exchanger NHE3/SLC9A3 and 35% by the complex multisubunit vacuolar H + -ATPase. The glomerular filtration rate (GFR) likely helps to regulate this bicarbonate reabsorption through both mechanisms, because both are stimulated by axial fluid flow. The secreted proton and the tubular fluid HCO 3 − are converted, with the catalytic assistance of the glyosylphosphatidylinositol-linked carbonic anhydrase IV of the apical membrane, to CO 2 , which diffuses into the proximal tubular cell, and OH − , which is instantaneously buffered in the tubular fluid.
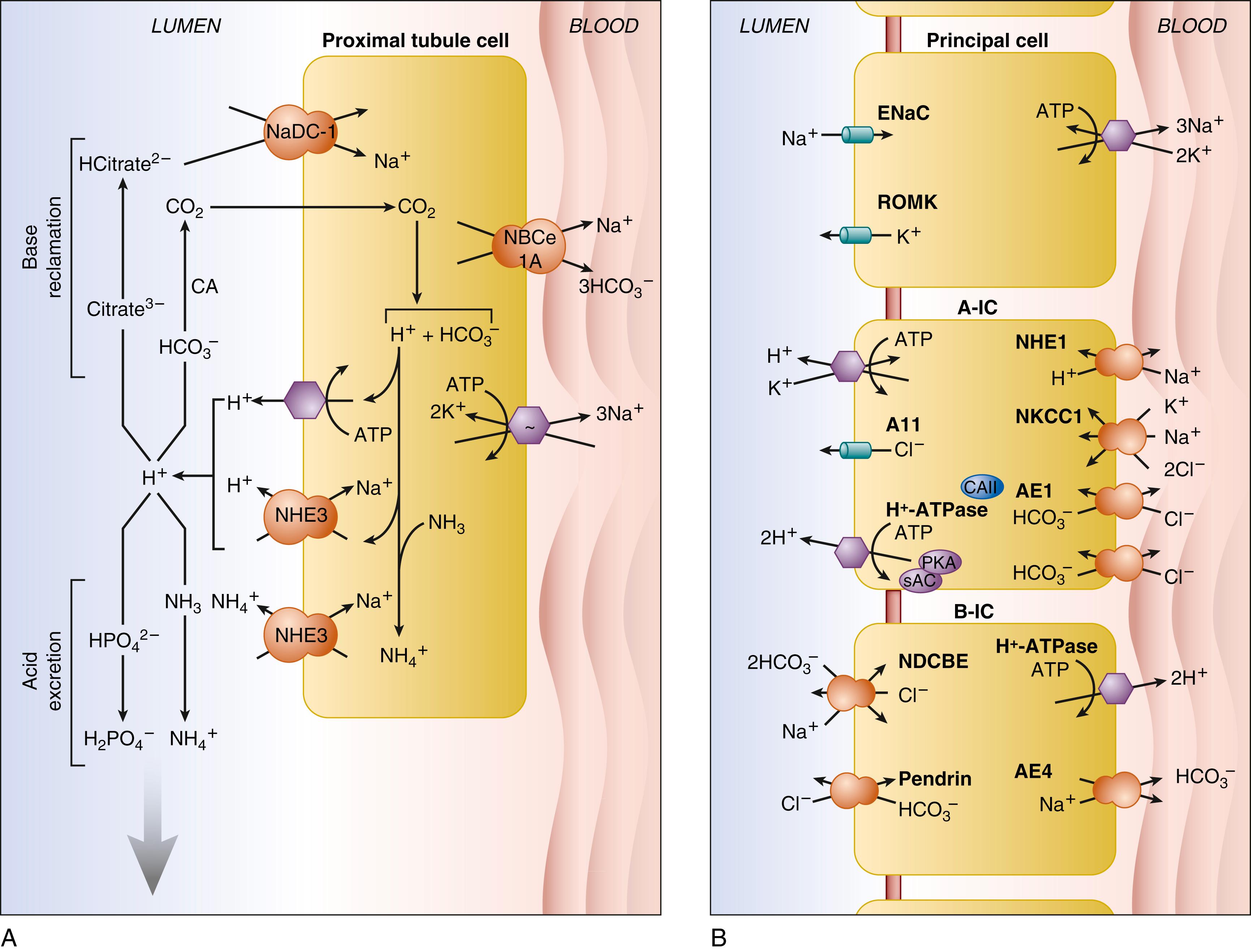
The CO 2 in the proximal tubular cell cytoplasm encounters abundant cytosolic carbonic anhydrase II, which regenerates intracellular HCO 3 − . This newly regenerated HCO 3 − serves as substrate, along with cytosolic Na + imported from the tubular fluid by the action of NHE3 and other proximal tubular sodium/solute co-transporters, for the bicarbonate efflux transporter of the proximal tubular basolateral membrane, NBCe1A/SLC4A4. NBCe1 operates with a stoichiometry of 2 to 3 HCO 3 − per 1 Na + . This stoichiometry, in the presence of proximal tubular cells’ highly negative intracellular voltage and elevated intracellular concentrations of Na + and HCO 3 − , favors efflux of Na + and HCO 3 − from cytosol into basolateral interstitial fluid, and on across the fenestrated endothelial cells of peritubular capillaries back into the systemic circulation. Additional contributors to the electrochemical gradient favoring HCO 3 − reabsorption from lumen to blood include the basolateral membrane electrogenic Na + ,K + -ATPase and basolateral membrane K + channels. By the end of the proximal tubule, luminal pH has decreased from the original glomerular filtrate value of 7.4 down to 6.8 to 6.9.
Proximal tubular HCO 3 − reabsorption can be inhibited by the carbonic anhydrase inhibitor diuretic, acetazolamide. Potent and specific diuretics are not available clinically for NBCe1A or for the voltage-dependent (v)H + -ATPase. Tenapanor, a potent and specific inhibitor of gut NHE3 engineered to be nonabsorbable, has entered clinical use for treatment of systemic fluid overload by reduction of intestinal Na + uptake. ,
The proximal tubule also generates all the ammonium found in the final urine. Proximal tubular cells take up glutamine and shuttle it to the mitochondria, where each glutamine undergoes two sequential deaminations by glutaminase and glutamate dehydrogenase. The two ammonium molecules generated exit the mitochondria as NH 3 via AQP8 into the cytosol. Two bicarbonates are also generated. The cytosolic pH (alkalinized by elevated HCO 3 − and NH 3 ) has been postulated to stimulate NHE3 to carry NH 4 + from cytosol into luminal fluid. (However, NH 4 + is excreted by alternate pathways in two models of NHE3 knockout mouse. , ) Proximal tubular ammonium production and secretion are enhanced during metabolic acidosis by increased levels of glutaminase and glutamate dehydrogenase secondary to stabilization of the messenger RNAs (mRNAs) encoding these enzymes in response to the fall in intracellular pH. The normal increase in proximal tubular NH 3 generation in response to hypokalemia requires expression of Na + /HCO 3 − co-transporter NBCe1.
The proximal tubule also reabsorbs nearly all the phosphate filtered by the glomerulus. Reabsorption at the proximal tubular luminal membrane occurs primarily via the electroneutral NaPi-2c/SLC34A3 (1 HPO 4 2− per 2Na + ), with smaller contributions by electrogenic NaPi-2a/SLC34A1 (1 HPO 4 2− per 3Na + ), electroneutral Pit-2/SLC20A2, and paracellular phosphate reabsorption. Inhibition of gut luminal NHE3 by the NHE3 inhibitor, tenapanor, can be used to control intestinal phosphate uptake during maintenance hemodialysis through reduction of paracellular phosphate absorption. Although the identities of the phosphate exporters of the basolateral membrane have not yet been confirmed, , XPR1/SLC53A1 is a strong candidate.
In response to systemic acidosis, the proximal tubule increases uptake of glutamine (largely via basolateral membrane SNAT3/SLC38A3), with increased generation of ammonium by increased activity of glutaminase and glutamate dehydrogenase. Na + /H + exchanger NHE3 is translocated from its vesicular storage sites under the apical membrane to fuse with the apical microvillar membrane, where it is subject to further activation. Citrate reabsorption by apical Na + /citrate co-transporter NaDC-1/SLC13A2 is increased in response to acidosis, whereas phosphate reabsorption is decreased through direct inhibition and internalization of NaPi-2c and -2a. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here