Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Surgical pathologists are often challenged when evaluating neoplastic lesions of the eye and ocular adnexa. Most pathologists are accustomed to focusing their attention on the diagnosis and prognostic features that guide therapy. The diagnosis of neoplasms of the eye and adnexa adds an additional dimension: The pathologist must provide information that also helps the surgeon to preserve vision, a therapeutic goal that may assume equal importance to the treatment of the neoplasm itself. For many patients, the fear of losing vision is at least as intense as the fear of losing their lives. Most ophthalmologists have encountered patients who do not hesitate to exclaim, “Doctor, I'd rather be dead than be blind!”
The ophthalmic surgeon's need to preserve vision when treating malignancies requires the pathologist to adjust standard procedures for the gross examination of tissues and the construction of pathology reports. For example, little normal tissue is typically resected around ophthalmic malignancies in the interest of preserving vision. Therefore, although histopathologic diagnosis of basal cell carcinoma of the eyelid or squamous dysplasia of the conjunctival epithelium may not be difficult, communicating precise information about margins of resection can be exceptionally challenging. To cite another example, a commonly used classification of conjunctival melanomas and their precursors is different from that used for cutaneous melanoma—not because ophthalmic pathologists are uneducated or contrary—but because the classification used in the conjunctiva was designed to assist ophthalmologists in the clinical differential diagnosis and management of these conditions.
Surgical pathologists will also encounter familiar neoplasms with unfamiliar behaviors within and around the eye. Although cutaneous and conjunctival melanomas originate in an epithelial compartment, uveal melanomas originate in mesenchymal compartments—the choroid, ciliary body, and iris—and there are thus no counterparts to in situ melanoma in the uvea.
Some neoplasms, while seldom encountered in a practice of general surgical pathology, are encountered commonly if the pathologist works with ophthalmologists with busy practices. The eyelid contains the highest concentration of sebaceous glands in the body and, in our laboratory, sebaceous carcinoma is encountered far more frequently than squamous cell carcinoma. Thus pathologic changes that suggest the diagnosis of bowenoid actinic keratosis in the eyelid may well represent the pagetoid intraepidermal distribution of sebaceous carcinoma.
Many orbital and eyelid neoplasms are discussed elsewhere in this book. Therefore this chapter focuses on those entities that either are unique to the eye and its adnexae or require the pathologist to consider the impact on vision as well as longevity in describing findings. This chapter is organized by anatomic location. An introductory section to each anatomic site provides information about functional anatomy so that the pathologist understands the rationale behind the resection technique employed. This information should also assist pathologists during the gross examination of tissues, which is the first step in the overall process of examining tissues.
Although most ophthalmic neoplasms do not develop preferentially in men or women, a distinctive age distribution is seen for these tumors, and as at other sites, clinical context is important. In this chapter, discussions of specific tumor types emphasize pitfalls in the histologic differential diagnosis of ophthalmic neoplasms and techniques by which pathologists can supply with reports that can be used to guide therapy.
Protection of the cornea is among the major functions of the eyelid. Neoplasms or surgical interventions that distort the eyelid may lead to imperfect eyelid closure, painful corneal exposure, and possibly corneal infection and ulceration.
The eyelid skin and adjacent skin at the medial canthus along the bridge of the nose are non–hair-bearing (glabrous) areas. For this reason, large resections of eyelid skin necessitate replacement by eyelid tissue from the opposite eyelid (e.g., a resection of large amounts of tissue from the lower eyelid may necessitate borrowing tissue from the upper eyelid of the same eye) or a skin graft from supraclavicular or retroauricular skin. Large resections of eyelid tissue and tissue from around the eyelid may require painstaking reconstruction; for this reason oculoplastic surgeons tend to resect tumors with only a minimum amount of normal tissue surrounding the lesion.
Fortunately surgeons are able to resect nearly one-third of the lower eyelid and accomplish primary closure without grafting. By severing the lateral canthal ligament (a trivial surgical maneuver), surgeons are able to resect nearly half of the lower eyelid and still accomplish primary closure.
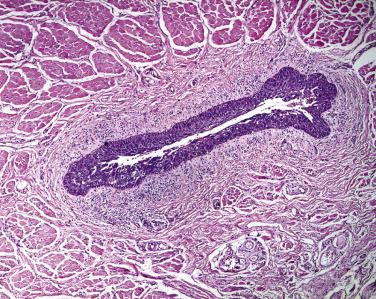
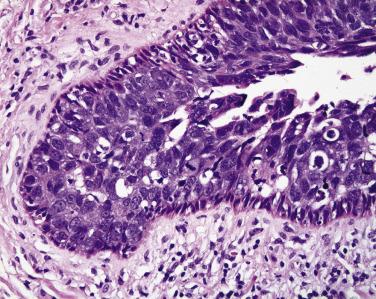
Most neoplasms of the eyelid begin at the mucocutaneous junction of the eyelid ( Fig. 29.1 ). The dermis of the eyelid contains only loosely arranged collagen; no analogue to the reticular dermis exists in the eyelid. Furthermore, no subcutaneous fat is present in the eyelid. If a pathologist encounters adipose tissue in an eyelid biopsy sample, either the adipose tissue has prolapsed through a defect in a fascial plane—the orbital septum—or the surgeon (and the tumor) has penetrated the orbital septum—an anatomic firewall separating the eyelid from the orbit. Apocrine glands are present within the eyelid and may give rise to simple cysts (apocrine hidrocystomas), which are relatively common or, rarely, to malignant neoplasms, including mucinous carcinoma ( Figs. 29.2 and 29.3 ), a locally invasive neoplasm that seldom metastasizes but may invade locally and requires total excision. Sebaceous glands may be identified near the eyelid margin in association with the eyelash (Zeis gland) or within the tarsus (meibomian glands). Sebaceous carcinoma may originate from either Zeis ( Fig. 29.4 ) or meibomian glands.
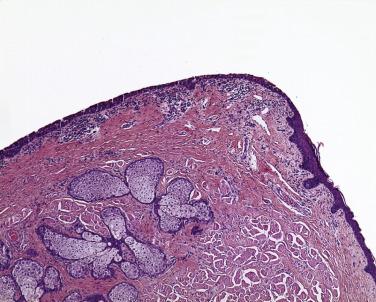
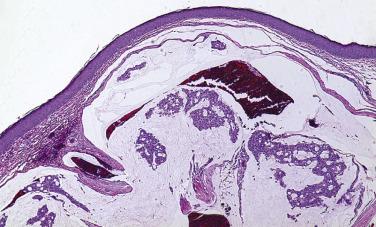
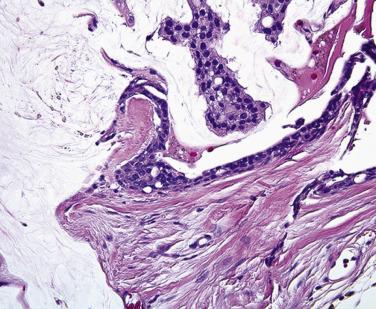
The lacrimal punctum is situated near the medial canthus of both the upper and lower eyelids. Tears, which are produced continually by accessory lacrimal glands embedded in the eyelid (akin to minor salivary glands in the buccal mucosa—the main lacrimal gland generates tears in response to pain and emotional triggers) collect at the punctum, which forms a miniature elevated basin drained by the lacrimal canaliculus. Surgical sacrifice of the punctum of the upper eyelid may be performed with little consequence to the patient, but sacrifice of the lower punctum or the puncta of both eyelids may result in an overflow of tears onto the periocular tissue (epiphora), which is terribly distressing to patients and may interfere with their ability to function well in both work and recreational activities. Ophthalmic surgeons strive to avoid sacrificing the lacrimal puncta and the canaliculus when resecting tumors. Surgical pathologists should therefore check for the presence of the lacrimal puncta and the lacrimal canaliculus ( Figs. 29.5 and 29.6 ) during the gross and microscopic examination of eyelid tissue.
Although basal cell and squamous cell carcinomas and melanomas may develop in the eyelid in childhood and young adulthood in extraordinary circumstances such as xeroderma pigmentosum, most eyelid tumors develop in adults. Thus a lesion that appears to represent a squamous cell carcinoma of the eyelid in a child should prompt serious consideration of pseudoepitheliomatous hyperplasia in the differential diagnosis.
It is also helpful to understand that although basal cell carcinoma is the most common malignancy of the eyelid (with a tendency to develop in the sun-exposed lower eyelid and medial canthus), squamous cell carcinoma seems to be far less common in the eyelid than sebaceous carcinoma.
Ophthalmologists have been encouraged to refer patients with periocular cutaneous malignancies to dermatologic surgeons who practice Mohs microsurgery. The rationale behind this practice is based on the perceived advantage of resecting periocular malignancies under frozen-section control, thus minimizing the amount of tissue resected. Also, some ophthalmic plastic surgeons argue that it is in the patient's best interest that the surgeon who performs the resection should be different from the surgeon who performs the reconstruction, to avoid the resecting surgeon compromising on the amount of tissue resected to facilitate an easier path for reconstruction.
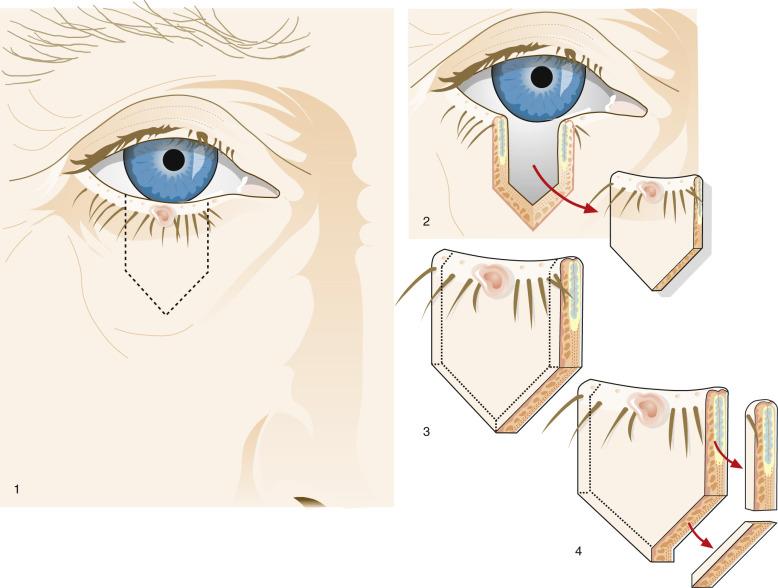
Little difference exists between the resection techniques used by Mohs surgeons in removing tumors of the eyelid margin and ophthalmic surgeons. Thus most ophthalmologists are well qualified to perform both the resection and the primary closure of the eyelid of most tumors in this location. Many ophthalmologists will insist on frozen-section control of small tumors of the eyelid margin. Because the resection technique involves the removal of a pentagon of eyelid tissue ( Fig. 29.7 ), the ophthalmologist may require the examination of four margins for each tumor resection. Few ophthalmologists are aware of the time required to perform four frozen sections, and some surgeons order the frozen sections to comply with perceived standards of care but may be tempted to proceed with the reconstruction before learning the results of the pathologist's examination, which invariably produces tension between surgeon and pathologist.
In general, most periocular neoplasms of the eyelid margin can be resected safely and the margins monitored on fixed tissue. The surgeon may choose to close the eyelid defect primarily, and if the margins are positive for tumor after primary closure, excision of the scar may suffice to remove residual microscopic neoplasm. Thus the principles of gross examination of an eyelid resection are similar, regardless of whether the examination is performed during a frozen section or on fixed tissue.
Most eyelid neoplasms originate at or near the mucocutaneous margin. Therefore the pathologist must take sections parallel to the surgical resection margin (see Fig. 29.7 ); the two side margins should be submitted separately (labeled medial or lateral; or, in corresponding ophthalmic terms, nasal or temporal, respectively). Some ophthalmic surgeons obtain a debulking layer and submit margins separately. Histologic sections of an eyelid neoplasm should never be obtained using a cruciate sampling strategy of the type often used for elliptical skin resections for cancer.
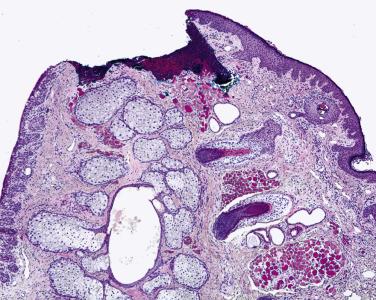
It is important that the histologic sections contain the mucocutaneous junction of the eyelid. Improper embedding of the block may lead to an incomplete sampling of this important landmark ( Fig. 29.8 ), necessitating reembedding and the cutting of additional sections.
Most eyelid neoplasms will be familiar to surgical pathologists and require no special consideration because they affect periocular skin (see Chap. 23 ). However, some eyelid neoplasms are worthy of special mention here.
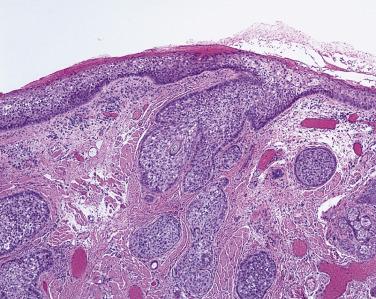
The histologic diagnosis of basal cell carcinoma is seldom problematic. However, the presence of slender cords of tumor entrapped between thick bundles of collagen ( Fig. 29.9 )—basal cell carcinoma of the sclerosing type (sometimes referred to as morpheic basal cell carcinoma)—should prompt the pathologist to mention this feature in the pathology report. It may be exceptionally difficult for surgeons to gauge the extent of tumor involvement in the eyelid and periocular skin by clinical examination. The identification of a sclerosing basal cell carcinoma by the pathologist may prompt the surgeon to perform a wider resection or excision under frozen-section control, a thoroughly justifiable use of frozen sections.
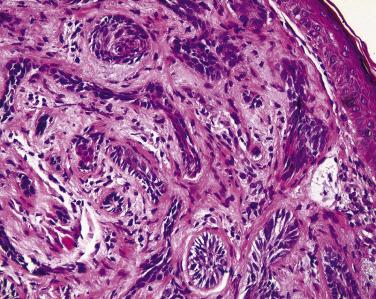
Sebaceous carcinoma may originate from either the glands of Zeis (see Fig. 29.4 ) or the meibomian glands. These tumors can present clinically as a discrete mass (often simulating a chalazion, a focus of lipogranulomatous inflammation) or with diffuse eyelid involvement (often simulating blepharitis). Many ophthalmologists will therefore submit recurrent chalazia to the pathologist with a clinical diagnosis of rule out sebaceous carcinoma and suspect the infiltrative form of sebaceous carcinoma in patients with unilateral thickening of the eyelid with loss of eyelashes; blepharitis is typically a bilateral condition. Sebaceous carcinoma may be more commonly encountered in women and tend to affect the upper eyelid. It is possible that early detection and treatment is helpful in lowering the mortality from this disorder. Sebaceous carcinoma may be associated with the Muir-Torre syndrome.
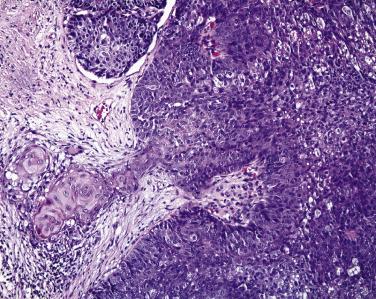
Histologically nodular sebaceous carcinoma may mimic a variety of other cancers and even some benign conditions. Typically lobules of tumor contain cells with foamy cytoplasm and the cytoplasmic vacuoles are usually small ( Fig. 29.10 ). The nuclei of tumor cells are typically very basophilic and may appear smudged. Many atypical mitotic figures may be encountered. The intracytoplasmic vacuolization may be quite subtle, and condensation of cells at the periphery of the lobule may impart the impression of basal cell carcinoma. Zones of central necrosis (comedo pattern) may be encountered. In rare cases, sebaceous carcinoma may mimic Merkel cell tumors histologically. Squamous differentiation may be seen in some tumors ( Fig. 29.11 ), and if this feature is prominent the unsuspecting pathologist may be lured into an erroneous diagnosis of squamous cell carcinoma.

There is considerable ongoing interest in the use of immunohistochemistry to facilitate the separation of sebaceous carcinoma from squamous cell carcinoma, basal cell carcinoma, and other entities in the differential diagnosis. Attention has focused on the expression of epithelial membrane antigen (EMA), adipophilin (ADP), Ber-EP4, p53, ERBB2 (formally known as Her2/neu), and perforin. The sensitivity and specificity of these markers is evolving in the literature. Fortunately the application of immunohistochemistry is replacing the procedurally cumbersome demonstration of lipid with Oil Red O staining.
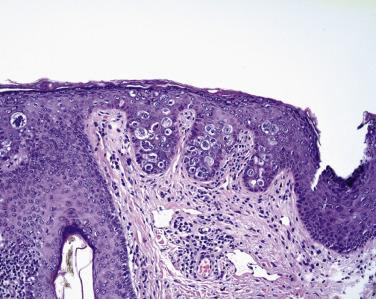
Perhaps the most challenging diagnostic aspect of sebaceous carcinoma is encountered in cases in which the tumor spreads diffusely throughout the eyelid epithelia—the epidermis and conjunctival epithelium. With pagetoid spread in the epidermis ( Fig. 29.12 ), pathologists may be tempted to consider melanoma in the differential diagnosis, but primary melanomas of the eyelid, especially those of the superficial spreading type, are extremely uncommon. Pagetoid spread through the epidermis may mimic actinic keratosis of the bowenoid type. In the conjunctiva, which normally lacks a superficial cornified layer of keratin, sebaceous carcinoma may replace the entire thickness of the conjunctival epithelium so that, instead of pagetoid spread as seen in the epidermis, a pattern more reminiscent of squamous dysplasia or carcinoma in situ may be encountered ( Fig. 29.13 ). It is possible to separate intraepithelial sebaceous carcinoma in the conjunctiva from intraepithelial squamous neoplasia; intraepithelial sebaceous carcinoma cells are typically noncohesive and appear to fall apart from one another. Although this histologic feature may be confused with acantholytic variants of squamous dysplasia and carcinoma in situ, identification of cells with subtle intracytoplasmic vacuolation and intensely basophilic and smudged-appearing nuclei tips the differential diagnosis toward sebaceous carcinoma. Replacement of the conjunctival epithelium by sebaceous carcinoma can also mimic papillary conjunctivitis ( Fig. 29.14 ).
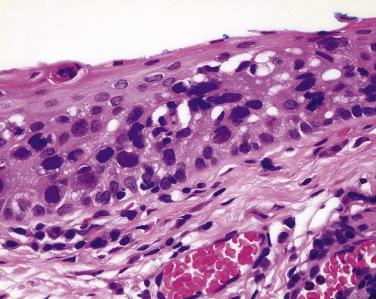
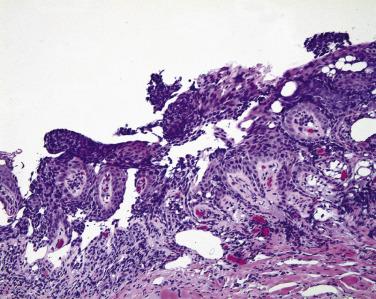
Often the conjunctival epithelium that is diffusely infiltrated by sebaceous carcinoma separates from its basement membrane (simulating the subepithelial bulla of ocular cicatricial pemphigoid) or sloughs entirely ( Fig. 29.15 ). Because of the nonspecific inflammation in the subepithelial connective tissue that often accompanies intraepithelial sebaceous carcinoma, immunoglobulin and complement deposits may be detected by immunofluorescence. Thus when ophthalmologists take a biopsy of the conjunctiva to rule out ocular cicatricial pemphigoid, they are encouraged to submit one piece of conjunctival tissue fixed in formalin to exclude the possibility of sebaceous carcinoma and a second piece for immunofluorescence studies.
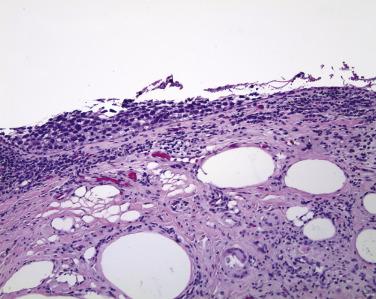
The conjunctival epithelium that is infiltrated by sebaceous carcinoma may slough, either leaving only a trace of basal epithelial cells adherent to the basement membrane in a tombstone-like configuration or exposing a denuded basement membrane. Thus when checking the eyelid for margins, the pathologist who notices no epithelium lining the conjunctiva should note this feature in the report and add a comment that the absence of epithelium in the context of the overall diagnosis of sebaceous carcinoma most likely indicates involvement at this site.
Sebaceous carcinoma may spread through the eyelid and down the lacrimal punctum into the canaliculus (see Figs. 29.5 and 29.6 ), into the lacrimal sac, and into the paranasal sinuses. Sebaceous carcinoma may also spread through the conjunctival epithelium into the ductules of the lacrimal gland and thereby retrograde into the orbit. Sebaceous carcinoma may evolve in multiple sites within the tarsus, resulting in skip lesions. Therefore in examining a resection specimen of eyelid with nodular sebaceous carcinoma without pagetoid spread, the pathologist may be comfortable in commenting on the adequacy of the resection margins. However, in the context of pagetoid spread, the pathologist should remain circumspect and may wish to comment in the report about areas of denuded epithelium that may represent positive margins and may wish to call to the ophthalmologist's attention the possibility of skip lesions so that a clear margin may not represent the limits of the extent of tumor spread.
Box 29.1 summarizes parameters to consider when constructing a surgical pathology report. Histologic features associated with an adverse outcome include tumor diameter greater than 10 mm; pagetoid spread; an infiltrative pattern with invasion into vessels, lymphatics, and soft tissue of the orbit; simultaneous involvement of both upper and lower eyelids; poor differentiation; and multicentric origin. Sebaceous carcinoma tends to spread first to the regional lymph nodes of the eyelid—the parotid and submandibular lymph nodes.
Location of the tumor (upper eyelid vs lower eyelid)
Some pathologists suggest that tumors of the upper eyelid are associated with a more aggressive clinical course
Size of the nodular component, if present (in mm)
Gland of origin
Zeis, Meibomian, or both
Tumors that originate from Zeis glands are frequently associated with pagetoid spread
Presence or absence of infiltrative growth pattern or comedo necrosis pattern
Both of these characteristics have been associated with an aggressive clinical course
Pagetoid spread
The detection of pagetoid spread may trigger the performance of map biopsies of the conjunctiva
Multicentricity, if detected
The presence of multicentricity may challenge the assumption that a lack of tumor at a margin truly indicates a complete resection
Notes: The absence of any epithelium in the conjunctival component of the resection may indicate the replacement of epithelium by tumor cells that have sloughed. Such margins may therefore be interpreted as positive despite the absence of any epithelium.
Treatment of sebaceous carcinoma requires total extirpation of both invasive and intraepithelial components of the disease. To gauge the full extent of the disease, ophthalmic surgeons may take small incisional biopsy samples in multiple locations on the surface of the conjunctiva (map biopsies), in addition to biopsies of the eyelid. If the conjunctiva is positive for intraepithelial tumor, the ophthalmologist may resect tumor in the eyelid and may choose to treat the conjunctival component of the disease by chemotherapy (mitomycin C) delivered by eye drops or cryotherapy.
Nevi commonly appear at the eyelid margin and are typically removed by shave biopsy. Nevi may recur at the site of shave biopsy, and the histologic appearance of recurrent nevus of the eyelid may be worrisome, although the lesion is biologically benign. Histologic evidence of a small congenital nevus may be detected quite deep within the substance of the eyelid tarsus, a feature that may again provoke anxiety despite its inconsequential prognostic significance ( Fig. 29.16 ). Melanomas of the eyelid margin without concomitant involvement of the conjunctiva are very uncommon. These lesions are seen more typically in conjunction with melanomas or melanoma precursors of the conjunctiva and therefore are discussed later. The pathology report should include measurement of the depth of invasion.
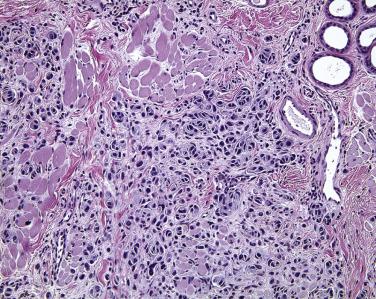
The histology of normal conjunctiva varies with topographic location on the eye. It is important that surgical pathologists appreciate the variability in appearance of the conjunctiva by topologic zone, to avoid overdiagnosis of conjunctival dysplasia.
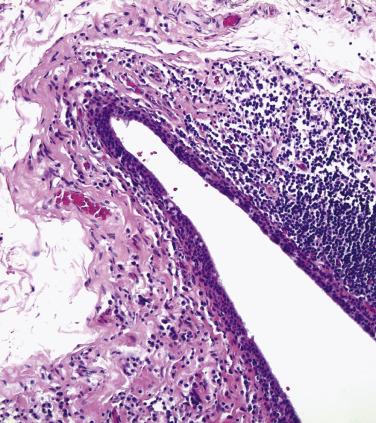
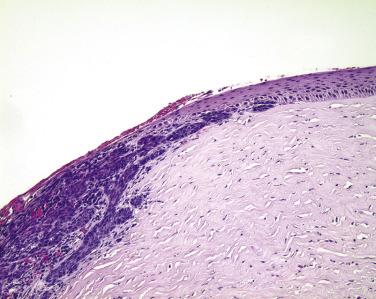
The conjunctiva lining the undersurface of the eyelid—the palpebral conjunctiva—is characterized by a pseudostratified columnar epithelium with goblet cells. The palpebral conjunctiva is very tightly tethered to the underlying tarsus ( Fig. 29.17 ). Therefore melanocytic lesions of the palpebral conjunctiva may appear clinically to be flat, despite deep invasion into the fibrous stroma of the eyelid. By contrast, the subepithelial collagen of the conjunctiva in the fornix is arranged loosely, allowing this tissue to distend under conditions of edema. The epithelium in the fornix is also a pseudostratified columnar epithelium, rich in goblet cells ( Fig. 29.18 ). Accessory lacrimal glands are present in the fornix and at the top of the tarsus in both the upper and lower eyelids, and the ductules of the main lacrimal gland pierce through the fornix superiorly and laterally. The fornix is normally populated by mucosa-associated lymphoid tissue. The distribution of goblet cells decreases over the bulbar conjunctiva—the component of the conjunctiva covering the eyeball—such that virtually no goblet cells are detected normally at the limbus, the junction between the cornea and sclera ( Fig. 29.19 ).
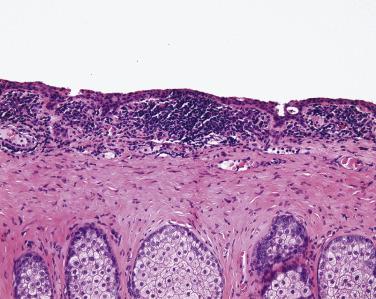
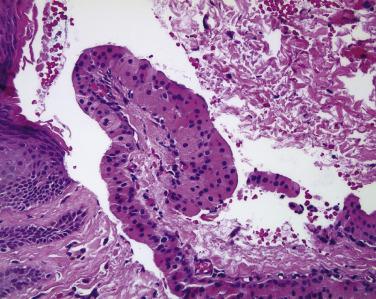
The conjunctival caruncle and the plica semilunaris (the human analogue of the nictitating membrane) are both situated in the medial corner of the conjunctiva and are unique histologically. The caruncle is covered by nonkeratinizing squamous epithelium, often containing goblet cells, but the subepithelial connective tissue contains adnexal structures such as pilosebaceous units and sweat glands. Thus neoplasms of the caruncle may include not only squamous papillomas and carcinomas, nevi, and melanomas but also sebaceous adenoma and carcinoma, arising in the sebaceous glands of the caruncle, and oncocytomas, arising from sweat glands ( Fig. 29.20 ). The plica semilunaris is invested with a high number of goblet cells, and tumors of this region are identical to those seen elsewhere in the conjunctiva.
Surgical pathologists who are unaccustomed to examining ophthalmic tissue will no doubt be perplexed by the small size of most conjunctival biopsy specimens, even for the most aggressive malignancies. Excisional biopsies for squamous cell carcinoma and even melanoma may include the narrowest of margins, perhaps 1 mm or less, a territory of normal tissue surrounding cancer that would be considered an inadequate and inappropriate margin of safety in most surgical oncologic disciplines. However, ophthalmic surgeons are required to balance the need to extirpate completely conjunctival malignancies with the pressing need to conserve conjunctival tissue to maintain the integrity of the ocular surface and vision. Large conjunctival resections, especially involving the fornix, reduce the number of goblet cells and thereby compromise surface mucin, which is essential for the adherence of the aqueous component of tears to the corneal epithelium. Patients who are subjected to large surgical resections of conjunctival tissue may likely suffer from dry eye, a condition that when severe may be painful and predispose to corneal opacification and ulceration because of the iatrogenic reduction in goblet cells, the loss of accessory lacrimal glands, and possible compromise of the ductules of the main lacrimal gland. It is therefore critical that pathologists make mention of the involvement of resection margins for all conjunctival lesions. Many ophthalmologists who resect conjunctival malignancies with narrow margins resort to adjunctive nonsurgical therapies (e.g., cryotherapy or topical chemotherapy) to ensure complete extirpation of these lesions.
Most neoplasms of the surface of the eye, both squamous and melanocytic in origin, originate at the limbus. The surgical resection of tumors at this site is difficult, and pathologists must be familiar with the histology of the limbus to interpret these biopsies correctly and guide the surgeon in therapy. Most conjunctival malignancies, squamous and melanocytic, do not invade the corneal stroma. Instead, neoplastic squamous epithelium and melanocytes typically migrate into the corneal epithelium but remain above the epithelial basement membrane and the underlying Bowman layer. The Bowman layer is a particularly effective barrier against the penetration of malignancies from the epithelium into the stroma (see Fig. 29.19 ). Therefore the surgeon who performs a shave biopsy of the cornea (known in ophthalmic parlance as lamellar keratectomy) compromises this barrier. Should the neoplasm recur at the limbus, which is a not-infrequent development, the iatrogenic absence of the Bowman layer provides unimpeded access of neoplastic cells to the underlying stroma. The surgical management of malignancy of the corneal stroma, which is usually preventable, may require corneal transplantation.
Ophthalmologists are instructed to resect limbal malignancies in two stages. They are first taught to inject anesthesia beneath the conjunctiva to balloon the tissue away from the surface of the eye. The adhesion of conjunctival tissue to the surface of the eye after this maneuver usually indicates invasion of the neoplasm into the episcleral connective tissue or the sclera. Ophthalmologists may submit very thin slices of superficial scleral tissue for histologic examination. Ophthalmologists should then resect tumor up to the limbus, without resecting any corneal tissue. After instillation of topical anesthesia, involved corneal epithelium is removed by chemical-mechanical debridement. A cotton-tipped applicator is soaked in absolute alcohol and wiped over the corneal surface, and the epithelium usually detaches as a sheet. Most ophthalmologists wrap this gelatinous tissue in filter paper and fix the tissue in neutral buffered formalin. Identification of tissue at the surgical pathology bench may be difficult, and application of eosin to the filter paper usually facilitates identification of the small transparent tissue specimen.
The main debulking layer of a conjunctival neoplasm at the limbus requires precise orientation at the gross examination bench. It is helpful for the ophthalmologist to tag a margin of the specimen with a suture to assist the pathologist in orientation. If the pathologist is uncertain about the orientation of the specimen, the surgeon should be invited to the gross examination bench to assist the pathologist. Sections should be taken perpendicular to the limbus so that all margins of resection can be adequately sampled ( Fig. 29.21 ).
Most conjunctival neoplasms develop in adults, and outside the context of xeroderma pigmentosa, these tumors are rarely encountered in children and adolescents. The patient's age should play a key role in assisting the pathologist with the differential diagnosis of conjunctival pigmented lesions. For example, conjunctival lymphoma is exceptionally uncommon in children, and the presence of a rich lymphoid infiltrate in the conjunctiva of a child or adolescent should immediately prompt a search for a component of nevus—the inflamed conjunctival nevus of puberty (also known as inflammatory juvenile nevus), discussed later. Junctional nevi of the conjunctiva are seldom encountered by pathologists, even in children. Well-documented examples of conjunctival junctional nevi no doubt exist, but the junctional phase of the evolution of conjunctival nevi must be exceptionally short, with most conjunctival nevi having a subepithelial component. (The term subepithelial is used instead of intradermal because the microarchitecture of the connective tissue of the conjunctiva is distinct from the dermis—a point that is underscored later in the discussion of conjunctival melanoma.) Therefore the presence of melanocytes within the conjunctival epithelium, either individually dispersed or in nests, in a patient who is not a young child should be diagnosed as primary acquired melanosis with atypia (or alternatively as conjunctival melanocytic intraepithelial neoplasia), a melanoma precursor, and never as a junctional nevus.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here