Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Prostatic carcinoma and benign prostatic hyperplasia (BPH) are the two principal conditions that involve the prostate; they account for more than 90% of all prostatic disease. Prostate cancer is the most common and second leading cause of cancer deaths in men in the United States. Early detection of cancer and better understanding of the premalignant conditions offer practical methods of reducing morbidity and mortality. Over 90% of malignant epithelial tumors of the prostate are acinar-type adenocarcinomas, whereas about 10% differ from the conventional histology and are regarded as variants.
In general, the morphologic diagnoses of prostatic lesions, particularly separating benign from malignant neoplasms, are relatively straightforward. However, several benign proliferative processes and normal histologic and anatomic structures of the prostate that exhibit a small glandular pattern, with or without cytologic atypia, can be mistaken for malignancy if one is not aware of their morphologic appearances. Most of the benign lesions mimicking cancer occur in the same age group as adenocarcinoma. They do not have specific clinical manifestations, and most are encountered during examination of transurethral resections of prostate (TURP) or in suprapubic or retropubic prostatectomies of patients with a clinical diagnosis of BPH. However, these lesions are occasionally seen in needle biopsy specimens of the prostate.
The classification of prostatic tumors, tumorlike lesions, and prostatic hyperplasia, as proposed by the recent World Health Organization (WHO) Classification on Tumors of the Urinary System and Male Genital Organs booklet, is outlined in Table 14A.1 . Benign prostatic lesions can be divided into epithelial and stromal lesions. Epithelial lesions include the usual type of BPH, basal cell hyperplasia, clear cell cribriform hyperplasia, atrophy and postatrophic hyperplasia, sclerosing adenosis, hyperplasia of mesonephric remnants, verumontanum mucosal gland hyperplasia, and nephrogenic adenoma.
| EPITHELIAL TUMORS | |
| Glandular Neoplasms | |
| Adenocarcinoma (acinar) | 8140/3 |
| Atrophic | |
| Pseudohyperplastic | |
| Microcystic | |
| Foamy gland | |
| Mucinous (colloid) | 8480/3 |
| Signet ring–like cell | 8490/3 |
| Pleomorphic giant cell | |
| Sarcomatoid | 8572/3 |
| Prostatic intraepithelial neoplasia, high grade | 8148/2 |
| Intraductal carcinoma | 8500/2 |
| Ductal adenocarcinoma | 8500/3 |
| Cribriform | 8201/3 |
| Papillary | 8260/3 |
| Solid | 8230/3 |
| Urothelial carcinoma | 8120/3 |
| Squamous neoplasms | |
| Adenosquamous carcinoma | 8560/3 |
| Squamous cell carcinoma | 8070/3 |
| Basal cell carcinoma | 8147/3 |
| NEUROENDOCRINE TUMORS | |
| Adenocarcinoma with neuroendocrine differentiation | 8574/3 |
| Well-differentiated neuroendocrine tumor (carcinoid tumor) | 8240/3 |
| Small cell neuroendocrine carcinoma | 8041/3 |
| Large cell neuroendocrine carcinoma | 8013/3 |
| MESENCHYMAL TUMORS | |
| Stromal tumor of uncertain malignant potential | 8935/1 |
| Stromal sarcoma | 8935/3 |
| Leiomyosarcoma | 8890/3 |
| Rhabdomyosarcoma | 8900/3 |
| Leiomyoma | 8890/0 |
| Angiosarcoma | 9120/3 |
| Synovial sarcoma | 9040/3 |
| Inflammatory myofibroblastic tumor | 8825/1 |
| Osteosarcoma | 9180/3 |
| Undifferentiated pleomorphic sarcoma | 8802/3 |
| Solitary fibrous tumor | 8815/1 |
| Solitary fibrous tumor, malignant | 8815/3 |
| Hemangioma | 9120/0 |
| Granular cell tumor | 9580/0 |
| HEMATOLYMPHOID TUMORS | |
| Diffuse large B-cell lymphoma | 9680/3 |
| Diffuse lymphocytic leukemia/small lymphocytic lymphoma | 9823/3 |
| Follicular lymphoma | 9690/3 |
| Mantle cell lymphoma | 9673/3 |
| Acute myeloid leukemia | 9861/3 |
| B lymphoblastic leukemia/lymphoma | 9811/3 |
| MISCELLANEOUS TUMORS | |
| Cystadenoma | 8440/0 |
| Nephroblastoma (Wilms tumor) | 8960/3 |
| Rhabdoid tumor | 8963/3 |
| Germ cell tumors | |
| Clear cell adenocarcinoma | 8310/3 |
| Melanoma | 8720/3 |
| Paraganglioma | 8693/1 |
| Neuroblastoma | 9500/3 |
| METASTATIC TUMORS | |
| TUMORS OF THE SEMINAL VESICLES | |
| EPITHELIAL TUMORS | |
| Adenocarcinoma | 8140/3 |
| Squamous cell carcinoma | 8070/3 |
| MIXED EPITHELIAL AND STROMAL TUMORS | |
| Cystadenoma | 8440/0 |
| MESENCHYMAL TUMORS | |
| Leiomyoma | 8890/0 |
| Schwannoma | 9560/0 |
| Mammary-type myofibroblastoma | 8825/0 |
| Gastrointestinal stromal tumor, NOS | 8936/1 |
| Leiomyosarcoma | 8890/3 |
| Angiosarcoma | 9120/3 |
| Liposarcoma | 8850/3 |
| Solitary fibrous tumor | 8815/0 |
| Hemangiopericytoma | 9150/1 |
| MISCELLANEOUS TUMORS | |
| Choriocarcinoma | 9100/3 |
| Seminoma | 9061/3 |
| Well-differentiated neuroendocrine tumor | 8240/3 |
| Lymphomas | |
| Ewing sarcoma | 9364/3 |
| METASTATIC TUMORS | |
a Morphology code of the International Classification of Diseases for Oncology (ICDO) and the Systematized Nomenclature of Medicine. http://snomed.org .
The most common stromal lesion is stromal hyperplasia; rare lesions include leiomyoma, atypical stromal hyperplasia and the closely related atypical leiomyoma (also known as bizarre or symplastic leiomyoma), stromal tumor of uncertain malignant potential, phyllodes-type atypical hyperplasia, postoperative spindle cell nodule, and inflammatory myofibroblastic tumor (pseudosarcomatous fibromyxoid tumor). Stromal sarcoma and other sarcomas of the prostate occur in adults and are relatively rare; rhabdomyosarcoma is the most common pediatric tumor.
Although two putative premalignant lesions of the prostate were initially described—prostatic intraepithelial neoplasia (PIN) (also termed intraductal dysplasia, primary atypical hyperplasia, large acinar dysplasia, acinar-ductal dysplasia) and atypical adenomatous hyperplasia (AAH) (also known as adenosis, small-gland hyperplasia)—the latter has not been proved to be a premalignant lesion. The first is characterized by cytologic atypia, although the normal architecture is maintained, whereas the second shows architectural atypia characterized by a neoacinar (small gland) proliferation without cytologic atypia. These lesions are discussed in the mimickers of carcinoma of the transition zone (see later discussion). Another potential premalignant condition is a subset of intraductal carcinomas, which will be discussed later.
McNeal first described this premalignant lesion as early as 1965 and in subsequent years, but his concept of preneoplasia was not fully embraced until the mid-1980s. In 1986 McNeal and Bostwick provided further evidence of the premalignant nature of PIN (originally termed intraductal dysplasia) and introduced a grading system. A National Institute of Health (NIH) workshop on premalignant lesions of the prostate was held in March 1989 in Bethesda, Maryland, and at this meeting PIN was adopted as the preferred term.
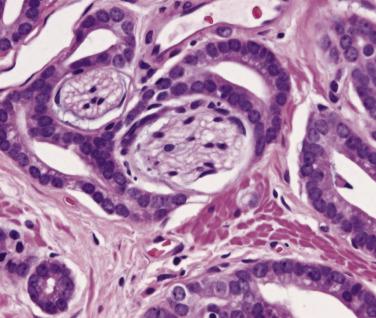
Microscopically, PIN can be distinguished from normal or hyperplastic glandular epithelium on low-power magnification because the affected glands or ducts most often depict striking hyperchromasia and nuclear stratification. On low-power examination the triad of too dark (hyperchromatic), too thick (nuclear stratification), and too complex (luminal complexity) should raise suspicion for PIN ( Fig. 14A.1 ). On high-power examination, the triad of nucleomegaly, prominent nucleoli, and hyperchromasia/chromatin alterations is diagnostic ( Fig. 14A.2 ). Depending on the severity of nuclear changes, PIN is subdivided into low and high grades.
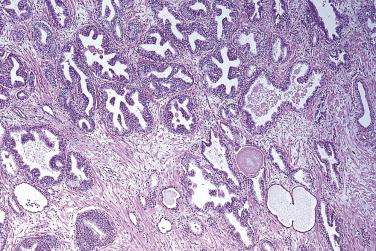
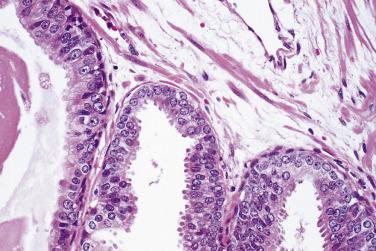
Although the consensus meeting in Bethesda, Maryland, agreed to divide PIN into two types, low grade and high grade, the histologic description here (for the purposes of better understanding PIN), is given according to the original grading system proposed by McNeal and Bostwick. Low-grade PIN (grade 1) is characterized by a slight increase in cellularity, some variation in nuclear size, hyperchromasia, and presence of small nucleoli. Grade 2 PIN consists of nuclear stratification with some nuclear enlargement and presence of nucleoli. These changes are generally focal, especially the nucleoli, which are not present in all of the cells. It is often very difficult to differentiate low-grade PIN (grade 1) from grade 2 PIN. In contrast, the epithelial cells of grade 3 PIN look cytologically malignant, as carcinomas in situ at many sites commonly appear. It is characterized by a marked increase in cellularity, with nuclear stratification and hyperchromasia. The nuclei are enlarged, a feature that can best be recognized by comparing the epithelial cells of PIN with those of the adjacent normal epithelial cells and stromal endothelial cells or fibroblasts ; however, some cases do not exhibit this feature, owing to fixation or preparation artifact or simply because there is no nuclear enlargement. The hallmark of high-grade PIN is the presence of large nucleoli (few in PIN grade 2, numerous in PIN grade 3), which are larger than 1 micron, and a halo is often present around them. Mitotic figures are uncommon in PIN but may be seen ( Table 14A.2 ).
| Diagnostic Criteria | Low-Grade PIN (PIN1) | High-Grade PIN (PIN2 and PIN3) |
|---|---|---|
| Architecture | Crowding, stratification, irregular spacing | More variation with four patterns: tufting, micropapillary, cribriform, and flat |
| Nuclei | Slightly increased with more size variation (anisonucleosis) | Markedly enlarged with less size variation |
| Chromatin | Normal | Increased |
| Nucleoli | Rarely prominent | Occasionally to frequently large and prominent |
PIN is found predominantly in the peripheral zone of the prostate (75%–80%), rarely in the transition zone (10%–15%), and extremely rarely in the central zone (<5%), and this distribution parallels the frequency of the zonal predilection for prostatic carcinoma. The frequency of high-grade PIN in needle biopsy series ranges from 5% to 16% and in TURP specimens between 2.3% and 4.2%.
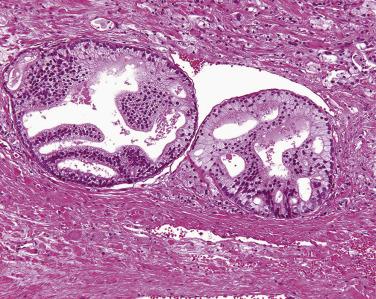
PIN may involve part of the lumen of a duct or the entire unit. At the onset, the epithelial proliferation is manifest as increased cellularity and pseudostratification, but as the process progresses, intraluminal papillae or cribriform architecture may develop. PIN exhibits four major architectural patterns: tufting, micropapillary, cribriform, or flat. The most common are the papillary and tufting patterns, less frequent is the cribriform pattern, and the least frequent is the flat pattern. Not uncommonly these patterns may coexist. Other rare patterns or features include those with mucinous cytoplasm, signet ring forms, small cell undifferentiated features, foamy gland type ( Fig. 14A.3 ), and inverted (hobnail) PIN features. Except for the foamy gland type, these architectural variations are more commonly seen in high-grade lesions than in low grade.
McNeal in 1969 described the multifocality of this process ; this observation has since been corroborated by many others. In a study of whole-organ sections of the prostate gland obtained from patients undergoing cystoprostatectomy for bladder cancer, Troncoso and colleagues found PIN in 89 of 100 prostates. Most often PIN was multifocal, and prostates with carcinoma had a greater number of foci of PIN than prostates without cancer. In another study, more than 10 foci of PIN were found in 71% of the prostates with carcinoma and a significant proportion displayed more than 20 foci of PIN.
In 1986, McNeal and Bostwick, studying 100 serially blocked prostate adenocarcinomas and 100 benign prostates obtained at autopsy, provided strong evidence supporting the contention that high-grade PIN (referred to as intraductal dysplasia) is a precursor of invasive carcinoma. They reported that high-grade lesions (grade 3) were more common in prostates with carcinoma (33%) than in those without carcinoma (4%) and concluded that, in the majority of prostate cancers, PIN may be the antecedent lesion. This finding was confirmed by the study of Troncoso and colleagues, in which high-grade PIN (grade 3) was found in 44 of 61 (72.1%) prostates with carcinoma and only 17.9% without carcinoma. Because high-grade PIN was closely associated with carcinoma, it had a predictive value for the latter. On occasion, carcinoma may arise directly from an area of high-grade PIN.
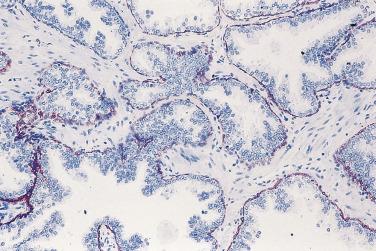
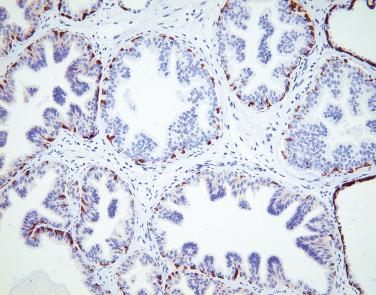
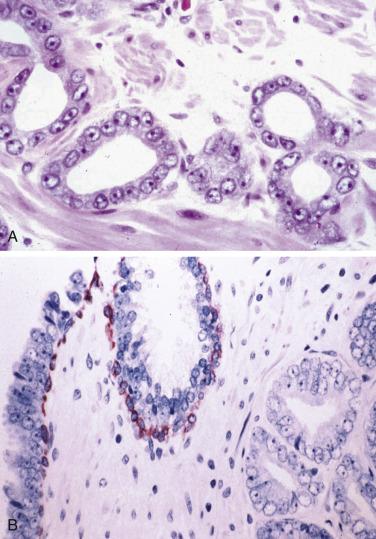
In contrast to prostatic adenocarcinoma, the basal cell layer is retained in low-grade and high-grade PIN ( Figs. 14A.4 and 14A.5 ), although it may be focally absent in high-grade PIN (see Fig. 14A.5 ). Bostwick and Brawer, using high molecular weight cytokeratin (HMWCK) specific for basal cells (clone 34βE12) in a series of PIN and adenocarcinoma, found that the basal cell layer was present in PIN but variably lost in higher grades, especially in acini adjacent to adenocarcinoma. These findings led these authors to postulate that early invasion of adenocarcinoma occurs commonly in association with high-grade PIN and that early invasion is characterized by disruption of the basal cell layer. McNeal and associates coined the term transitive glands for these glands. When high-grade PIN is adjacent to small atypical glands, it is difficult to decide whether the small glands represent budding or tangentially sectioned glands from adjacent high-grade PIN or invasive cancer next to high-grade PIN. Kronz and colleagues reported that PIN with adjacent small atypical glands appears to be a greater risk factor than high-grade PIN alone in predicting invasive cancer on repeat biopsy. Although age and predominant pattern of associated high-grade PIN may be helpful in predicting which men with this lesion will have cancer on repeat biopsy, they cannot be used reliably; therefore all men with PIN and adjacent small atypical glands are recommended to undergo repeat biopsy.
The importance of high-grade PIN as a predictive marker for prostate cancer has lost some of its past value. The routine extended core needle biopsies (usually 12) of the prostate generally map the entire prostate with significant accuracy, and even if a focus or two of high-grade PIN is found, the chances of finding a clinically significant cancer are minimal to such extent that many urologists prefer to follow patients on a clinical basis with repeat serum prostate-specific antigen (PSA), ultrasound, and digital rectal examination (DRE) rather than an immediate repeat biopsy.
In a multicenter European study of 626 patients with diagnosis of PIN, the incidence of prostate cancer in subsequent biopsies was 12.9%, which was not much different from that in normal controls. It is currently recommended to manage patients with a biopsy result of focal high-grade PIN as that of a benign biopsy.
Three basal cell markers are most commonly used in the diagnosis of PIN: HMWCK (34βE12), p63, and cytokeratin 5/6 (CK5/6). Several immunohistochemical markers have been used to correlate the relationship between high-grade PIN and invasive carcinoma, including KA4, Ulex europaeus lectin (UEAl), and vimentin, as well as the argyrophilic nucleolar organizer regions (AgNOR) and static DNA flow cytometry, and all of these are supportive of the precursor potential of high-grade PIN. Additional support for the precursor potential of PIN comes from many other studies, including immunostains for PSA, prostate acid phosphatase (PAP), CD57, collagen IV, collagenase, metalloproteinase and matrilysin expression, epidermal growth factor receptors, proliferating cell nuclear antigen (PCNA), Ki67, MIB1 proliferative activity, p53 tumor suppressor gene protein, c- myc gene expression, bcl- 2 oncoprotein, and so on.
More importantly, similar cytogenetic abnormalities (involving 7q, 8q, 10q, 16q) and numeric chromosomal changes are noted in high-grade PIN and cancer. Wu and associates demonstrated that a significantly higher α-methylacyl coenzyme A racemase (AMACR)/P504S-positive rate (56%) was found in isolated high-grade PIN glands adjacent to cancer (distance <5 mm) compared with those with cancer far from the PIN (distance >5 mm; 14%, P <0.0001). High-grade PIN glands adjacent to cancer also showed a higher AMACR/P504S intensity than did those away from cancer ( P <0.001). These data indicated that PIN strongly positive for AMACR/P504S might be more closely associated with cancer than PIN negative or weakly positive for AMACR/P504S; this study provides additional evidence to link high-grade PIN as a precursor lesion to prostatic adenocarcinoma. It has also been suggested that patients with AMACR-positive high-grade PIN are up to 5.2 times more likely to have a diagnosis of prostate cancer in subsequent biopsies.
No specific molecular marker for PIN exists, although the majority of the invasive prostate cancers have recurrent gene fusions involving the ETS family of transcription factors, ERG, ETV1, ETV4, and ETV5 fused to TMPRSS2 or other upstream partners. Among these aberrations, TMPRSS2-ERG fusion is the most prevalent, occurring in up to 50% of prostate cancers, and has been reported in up to 15% of high-grade PIN. Therefore TMPRSS2-ERG fusion is believed to be an early genetic event in human prostate cancer.
The most critical differential diagnosis for benign and low-grade lesions includes differentiation of grade 1 PIN (low-grade PIN) versus grades 2 to 3 PIN (high-grade PIN) and the histologic pattern of the central zone benign glands. For high-grade lesions, the differential diagnosis of high-grade PIN includes prostatic ductal adenocarcinoma, cribriform acinar adenocarcinoma, intraductal carcinoma, and urothelial carcinoma in situ involving prostatic ducts. Other processes also enter in the differential diagnoses, but these are easily ruled out. They are briefly discussed in the following section.
The differential diagnostic features between low-grade and high-grade PIN are listed in Table 14A.2 . Unfortunately, no clear-cut histologic criteria differentiate grade 1 PIN (low-grade PIN) and grades 2 to 3 PIN (high-grade PIN). Thus the differentiation is subjective and pathologists may wrongly call a lesion high-grade PIN simply because of the presence of hyperchromasia or some nucleoli. In a study by Sakr and colleagues, the authors submitted 30 prostate needle biopsy slides containing glands with variable degrees of architectural atypia, including 22 cases that had been diagnosed as high-grade PIN, to 11 urologic pathologists. After receiving variable answers for many of the cases, the authors concluded that although the morphologic criteria used by urologic pathologists to diagnose high-grade PIN were similar, the application of criteria in borderline cases varied considerably. This accounts in part for the differing prevalence rates of high-grade PIN in the literature and has significant clinical implications for individual patients.
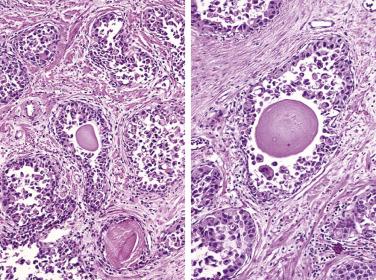
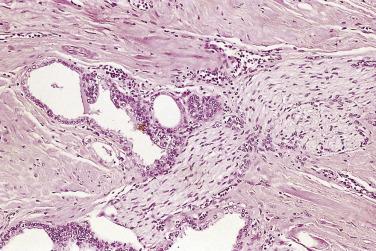
The central zone, often included in the biopsy directed to the base of the prostate, shows changes that may easily be confused with high-grade PIN. The central zone is characterized by having far more stroma between glands than in the peripheral or transition zones. Often, nuclear stratification makes the ducts appear abnormal, as if containing high-grade PIN. However, the nuclei of the epithelial cells are monotonously round, without cytologic atypia, although occasionally a nucleolus may be seen. Secondary formations of small glandular structures simulating so-called Roman arches or forming a cribriform pattern are not uncommon in the central zone. In addition, intraglandular papillations may occur, but these contain a central fibrovascular core ( Fig. 14A.6 ) and no atypical epithelial features. In contrast, the papillations of high-grade PIN do not have a central fibrovascular core.
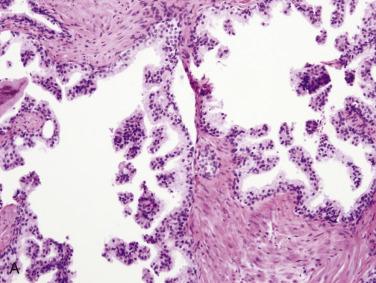
Transitional metaplasia generally occurs in the central large ducts close to the urethra, but may be seen in the peripheral zone, where it may be confused with low-grade PIN. The presence of oval to slightly elongated nuclei, often with frequent nuclear grooves, is characteristic of transitional cell metaplasia.
Of the high-grade tumors, urothelial carcinoma in situ (CIS) of prostatic ducts and lesions with an intraductal cribriform pattern are the main differential diagnoses.
Urothelial CIS involving ducts is a high-grade carcinoma that may show central, comedo-type necrosis, a feature not seen in high-grade PIN. The cells of urothelial CIS show significant nuclear anaplasia manifest by considerable variation in size and shape, hyperchromasia, and often brisk mitotic activity. These cells are positive for GATA3 and negative for PSA, PAP, and AMACR/P504S.
Lesions that share an intraductal cribriform pattern include high-grade PIN, microacinar cribriform adenocarcinoma, ductal adenocarcinoma, and intraductal carcinoma. The boundaries between the last three entities are not clear cut. Cribriform adenocarcinoma is similar to ductal adenocarcinoma, and when the patterns overlap, it is difficult to separate them. As a general rule, the cells of ductal adenocarcinoma are cuboidal to tall columnar, with obvious anaplasia, whereas those of cribriform adenocarcinoma are monotonously round and lower grade than ductal adenocarcinoma.
Prostatic ductal adenocarcinoma simulates high-grade PIN, and the distinction may not always be possible in needle biopsy specimens. Ductal adenocarcinoma frequently involves the transition zone around the verumontanum area and displays a back-to-back complex glandular architecture, true papillary formations, frequent necrosis, and marked nuclear anaplasia, including frequent mitoses. The presence of comedo necrosis generally rules out high-grade PIN.
High-grade PIN generally expands the duct without significant irregularities of its outline. When the cribriform proliferation is florid, with marked expansion of the ductal or acinar unit involved, the lack of a basal cell layer is in favor of a cribriform or ductal adenocarcinoma rather than high-grade PIN. The presence of basal cells would support a diagnosis of high-grade PIN, but caution should be exercised because retrograde extension of a ductal or cribriform adenocarcinoma into a duct or acinus may occur (intraductal carcinoma). Therefore careful interpretation of the presence of the basal cell layer is advised.
The term intraductal carcinoma has been used when ductal or acinar units are replaced by cribriform carcinoma. McNeal and Yemoto suggested that ductal involvement in adenocarcinoma is a common entity that is usually present within the tumor but is rare away from the invasive aspect of it. In this context they used the term intraductal carcinoma of prostate (IDC-P) and thought that the lesions were part of the evolution of prostate cancer rather than a precursor lesion. IDC-P is defined as malignant epithelial cells filling large acini and prostatic ducts, with preservation of basal cells forming either (1) solid or dense cribriform patterns or (2) loose cribriform or micropapillary patterns with either marked nuclear atypia (nuclear size 6 times normal or larger) or comedo necrosis.
Using these criteria for IDC-P (defined as high-grade PIN with cribriform, solid, or comedo patterns), Wilcox and associates studied whole-mount sections of prostate from 252 patients with pT3N0 prostate carcinoma for the presence of IDC-P and correlated the findings with Gleason score, total tumor volume, surgical margin status, seminal vesicle involvement, and disease progression. Patients with IDC-P had higher Gleason score and total tumor volume and were more likely to show seminal vesicle involvement and disease progression than those without IDC-P. Therefore the presence of IDC-P is generally associated with high-grade invasive adenocarcinoma and rarely poses a diagnostic problem. Furthermore, Guo and Epstein also demonstrated that IDC-P on prostate biopsies is frequently associated with high-grade cancer and poor prognostic parameters at radical prostatectomy, as well as potentially advanced disease after other therapies. Together these findings suggest that IDC-P represents an advanced stage of tumor progression with intraductal spread of tumor. Consideration should be given to offering definitive treatment in patients with IDC-P on biopsy even in the absence of documented invasive cancer. Shah and colleagues used the term atypical cribriform lesions of the prostate (ACL) to denote intraductal cribriform glands characterized by cytologically malignant cells with partial or complete retention of the basal cell layer. In this study, they separated ACLs into an isolated finding without associated invasive adenocarcinoma (isolated ACL), and ACLs associated with invasive cancer that is almost always a high-grade, high-volume prostate adenocarcinoma (cancer-associated ACL). The latter was further defined as an ACL intermixed with, or within 3 mm of the border of, an infiltrative adenocarcinoma and was categorized as ACL-adenocarcinoma, and consequently considered to be equivalent to IDC-P. On the other hand, isolated ACLs were considered equivalent to cribriform high-grade PIN or a questionable lesion between high-grade PIN and IDC-P on the limited samples, requiring further workup.
The study by Shah and colleagues encompassing 117 consecutive prostatectomy specimens found that ACL-adenocarcinomas and isolated ACLs were present in 21 (17.9%) and 15 (12.8%) of the specimens, respectively. The Gleason score of the ACL-adenocarcinoma was 7 or higher. A branching contour was present in 36 of 43 ACL-adenocarcinomas, but in only 1 of 23 isolated ACLs ( P <0.001). The dense cribriform and solid patterns were present in 6 (14.0%) and 4 (9.3%) ACL-adenocarcinomas, but none of the isolated ACLs. Comedo necrosis was present in 14 in 43 (32.6%) ACL-adenocarcinomas and in none of the isolated ACL ( P = 0.001). Pleomorphic nuclei or giant nuclei (at least 6 times larger than adjacent nuclei) were present in 12 (27.9%) ACL-adenocarcinomas but in none of the isolated ACL ( P = 0.005).
The authors conclude that if a biopsy contains a small number of ACL glands with low-grade morphology, it should be diagnosed as ACL, essentially a lesion in which distinction between cribriform high-grade PIN and IDC-P is not possible. It is strongly recommended that a repeat biopsy should be performed to rule out adenocarcinoma. In contrast, if a biopsy contains ACL with a large focus, architectural complexity with large branching glands, pleomorphic or giant nuclei, or comedo necrosis, the biopsy should be diagnosed as IDC-P and definitive therapy recommended.
There have been a number of studies evaluating the significance of IDC-P at radical prostatectomy (RP). Presence of IDC-P was associated with significantly higher rate of lymph node metastasis. Two studies have shown that in multivariate analysis, IDC-P was significantly associated with more frequent biochemical recurrence and lower cancer-specific survival. Furthermore, in evaluating 901 RP specimens, Miyai K et al have shown that two groups of IDC-P exist, namely IDC-P associated with adjacent invasive carcinoma (regular IDC) and IDC-P associated with invasive cancer at distant site or no associated cancer (precursor IDC). The regular IDC-P had significantly higher Gleason score, higher pathologic stage, and lower 5-year biochemical-free survival than precursor IDC-P. The authors concluded that IDC-P represents not only intraductal spread of preexisting high-grade invasive carcinoma, and that at least a subset of IDC-P could be a precursor lesion of invasive carcinoma.
Occasionally lesions do not fulfill all of the criteria for IDC-P, yet are worse than typical high-grade PIN, but the distinction can be difficult based on morphologic criteria alone. It has been shown that ERG rearrangement detected by ERG break-apart fluorescence in situ hybridization (FISH) is more common in IDC-P than in cribriform high-grade PIN.
The diagnostic scheme of large cribriform gland lesions is depicted in Fig. 14A.6A .
The differential diagnosis of high-grade PIN and infiltrating cribriform carcinoma involving ducts was discussed by Kronz and colleagues. They reported four clinicopathologic findings (two clinical and two histologic) to be helpful in differentiating cribriform high-grade PIN from infiltrating cribriform carcinoma: positive digital rectal examination ( P = 0.02), positive transrectal ultrasound ( P = 0.02), bilateral atypical cribriform glands ( P = 0.02), and detached cribriform glands ( P = 0.04). Other findings, including number of cribriform glands, largest size of cribriform glands, necrosis, and stromal fibrosis, failed to distinguish between the PIN and IDC-P.
Normal structures, such as seminal vesicles and ejaculatory ducts, may mimic PIN. Ejaculatory duct and seminal vesicle epithelium usually show scattered large pleomorphic nuclei, often with intranuclear inclusions and cytoplasmic lipofuscin pigment, although PIN epithelium may also show pigment that is usually finer and less conspicuous. Pleomorphism is unusual in high-grade PIN. In case of doubt, immunostains can be helpful as ejaculatory duct and seminal vesicle epithelium are negative for PSA and PAP, and positive for PAX8 and GATA3.
Infarction and inflammation common to the transition zone may exhibit marked degrees of epithelial atypia. Therefore caution should be exercised if infarction or inflammation (particularly acute) is present. The characteristic architectural features of PIN are usually not present in reactive prostatic epithelium.
Numerous studies have shown that the diagnosis of high-grade PIN in a biopsy confers a greater risk (30%–35%) of finding carcinoma in any subsequent biopsy, although this number has decreased in more recent contemporary series, particularly since the advent of extended biopsy (>10 biopsies). Unfortunately, two-thirds of patients do not show invasive carcinoma on repeat biopsy, and some of these patients undergo yearly repeat biopsies. Urologists and pathologists are becoming aware of this problem. Although there is good interobserver reproducibility for the recognition of high-grade PIN, this is not the case for low-grade PIN. Thus some low-grade PINs (grade 1) are probably wrongly diagnosed as high-grade PINs. Numerous articles on high-grade PIN are currently addressing other concomitant factors that may help select patients at higher risk of having invasive carcinoma.
In analyzing 245 men with an initial biopsy diagnosis of high-grade PIN ( Fig. 14A.7 ), Kronz and associates showed a 32.2% incidence of prostate carcinoma on follow-up biopsy. The only independent histologic predictor of an invasive cancer diagnosis was the number of cores with high-grade PIN. The more biopsy cores with high-grade PIN, the more likely the patient were to have an invasive adenocarcinoma in repeat biopsy. Netto and Epstein reported a 39% risk of finding invasive adenocarcinoma on repeat biopsies of patients in whom the initial biopsies demonstrated widespread high-grade PIN, defined as high-grade PIN present in four or more biopsy cores. Their findings support the need for a repeat biopsy in this subset of patients. If cancer is not found on the first two follow-up biopsies, it is unlikely to be found in the future. Using the histologic pattern of high-grade PIN as a predictor of carcinoma on subsequent biopsy has been inconsistent. Bishara and colleagues reported a higher risk of carcinoma with the tufting or flat category (31.9%) compared with the micropapillary or cribriform category (22.0%), whereas another study suggested that a higher risk of carcinoma is associated with micropapillary and cribriform high-grade PIN.
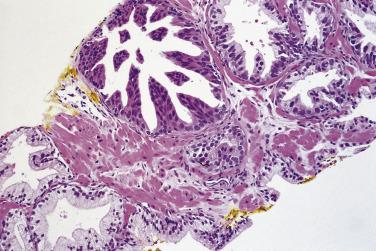
Is the diagnosis of high-grade PIN becoming obsolete? Probably not! However, the rules of the game might be changing with the introduction of extended biopsies, so-called saturation biopsies with sampling from 10 or more sites in contrast to traditional 6-site sextant biopsies. These biopsies allow for extensive mapping of the prostate in such a way that the chances of finding an invasive carcinoma are very high. In a study of high-grade PIN diagnosed with 12-site biopsy of 31 patients who were followed for 3 years before a repeat biopsy was done, the authors reported that 8 patients (28.8%) had invasive carcinoma on repeat biopsy, 11 (35.5%) again showed high-grade PIN, and 12 (38.7%) showed no disease. The authors suggested that repeat biopsies at delayed intervals for patients with diagnosis for high-grade PIN are recommended regardless of changes in the PSA level. Other authors believe that patients diagnosed with high-grade PIN on extended biopsies should be followed and a repeat biopsy should be done only if there are clinical indications for it. The final decision to perform a repeat biopsy on a patient with high-grade PIN diagnosed with 10 or more biopsy sites should be agreed jointly between the patient and his urologist.
Adenocarcinoma of the prostate is by far the most common malignancy, which accounts for more than 21% of all malignancies in men, and is the second leading cause of death after lung carcinomas in the United States. The American Cancer Society estimated that, in the United States, 180,890 males would be diagnosed with prostate cancer (21%) and 26,120 would die of their disease (8%) in 2016. As a result of aggressive screening and better treatment, both the incidence and mortality have significantly decreased in the past few years. Among 19,316 routine autopsies on men older than 40 years of age, 1589 cases (8.2%) of adenocarcinoma were found.
The incidence of adenocarcinoma is quite variable among races and in different countries, with the highest reported incidence in North America, Australia, and Scandinavian countries. Prostate cancers are relatively rare in Asian populations, but recent data indicate that the incidence is rapidly increasing. In the United States, the incidence and mortality is higher in the black population than in other races. A geographic difference in the incidence exists in Europe, with a higher incidence in the countries of northern and western Europe and lower in the countries of eastern and southern Europe. In addition to geographic, ethnic, and racial differences, genetic factors are responsible for the different incidences as well. Despite significant advances in the understanding of premalignant epithelial lesions and new clinical techniques with enhanced early detection of cancer using transrectal ultrasound and serum levels of PSA, important questions remain regarding the cause and prevention of adenocarcinoma, as well as the appropriate treatment of early-stage, particularly small-volume clinically significant prostate cancer and hormone-refractory advanced cancer. Adenocarcinoma continues to be a great clinical paradox; for example, in spite of the high incidence, more men will die with prostate adenocarcinoma rather than from the cancer itself. Autopsy studies have shown that the incidence of adenocarcinoma increases with age. Studies have found an incidence of 42% to as high as 50% of prostate adenocarcinoma in whole-mount sections of entire prostate from cystoprostatectomy specimens in patients with bladder urothelial carcinoma.
Conventional acinar adenocarcinoma represents over 90% of prostate carcinomas. The majority are multifocal (60%–90%) and exhibit an acinar or mixed acinar and ductal growth pattern. The remaining 10% are composed of variants of adenocarcinoma, which will be discussed separately.
Carcinomas may arise in any zone of the prostate, but the relative distribution is different in each zone; 68% of the carcinomas arise in the peripheral zone, 24% in the transition zone, and 8% in the central zone. Adenocarcinoma rarely can arise from ectopic prostate tissue.
The various patterns of growth are well described in the past by Mostofi and Price and Gleason ; they include acinar, fused acinar, cribriform, papillary, trabecular, and solid, representing different Gleason scores. The fact that prostate cancers have a high incidence but a low fatality has indicated that prostatic adenocarcinomas are relatively indolent, and most patients survive for a long time after the diagnosis. Local spread of adenocarcinoma occurs through extraprostatic extension or seminal vesicle invasion. Distant metastatic spread occurs when carcinoma invades into lymphovascular spaces. The most common sites of metastasis are regional pelvic lymph nodes, bone, and lung. Rare sites include the liver and testis. Metastasis to the testis is uncommon, and unilateral involvement is more common than bilateral involvement.
The majority of prostate adenocarcinomas are not difficult to diagnose; however, problem areas do exist. First is the separation of well-differentiated adenocarcinoma from the vast number of benign or atypical small-gland proliferations. Second is the threshold for recognizing extremely small foci of cancer in needle biopsies. Finally, at the extreme of the histologic spectrum, depending on the morphology, a very poorly differentiated adenocarcinoma of the prostate may be difficult to distinguish from inflammatory infiltrates, metastatic carcinoma, and urothelial carcinoma involving the prostate.
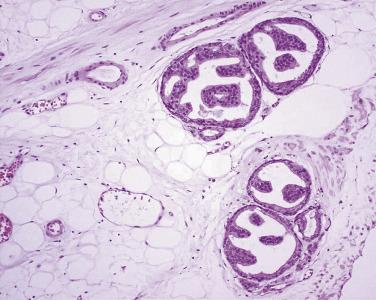
The principal criteria for diagnosis of well-differentiated adenocarcinoma include a small-gland proliferation recognized as being discrete or focally infiltrative on low-power examination, the presence of a single cell lining with complete absence of the basal cell layer, nucleomegaly, and presence of large nucleoli. The small-gland proliferation usually exhibits well-formed acinar structures, with acini arranged in a back-to-back fashion ( Fig. 14A.8 ) and a circumscribed or focally infiltrative margin. The size of the nucleoli is critical; in carcinoma, nucleoli are often prominent—being at least 1 micron in diameter ( Fig. 14A.9 ). Although nucleolar size usually is not measured, prominent nucleoli can be recognized by their distinct cherry red color on medium-power (10× or 20×) magnification. The number of nucleoli varies from one to several per nucleus. When nucleoli are multiple, they are generally small. A single cell lining (i.e., lack of basal cell layer) is also a requisite for the diagnosis of well-differentiated adenocarcinoma of the prostate. Several other features have been shown to be helpful for diagnosis of carcinoma—intraluminal crystalloids ( Fig. 14A.10 ), blue mucin ( Fig. 14A.11 ), glomerulations ( Fig. 14A.12 ), mucinous fibroplasia (collagenous micronodules) ( Fig. 14A.13 ), and circumferential perineural invasion ( Fig. 14A.14 ). Intraluminal crystalloids and blue mucin are not pathognomonic, but they are frequently associated with carcinoma. In contrast, mucinous fibroplasia, glomerulation, circumferential involvement of nerves, and glands in fat tissue ( Fig. 14A.15 ) are pathognomonic for adenocarcinoma. For low-power screening of prostate cancer in biopsy or TURP specimens, the rule of “three toos” (too small glands, too crowded glands with back-to-back arrangement, and too clear glands) is very useful in identifying carcinoma ( Fig. 14A.16 ). To confirm the diagnosis of carcinoma, three diagnostic criteria, including nuclear enlargement, prominent nucleoli, and lack of basal cells, should be present ( Fig. 14A.17 ). Aydin and colleagues suggested that mitotic figures and apoptotic bodies were more commonly seen in cancer than in benign mimickers of adenocarcinoma.
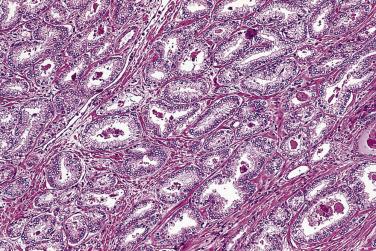


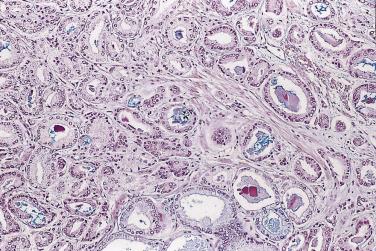
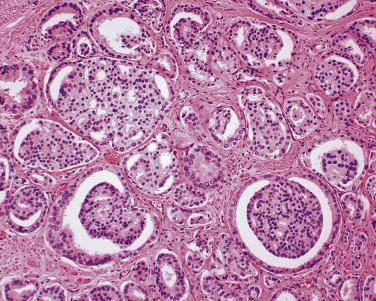
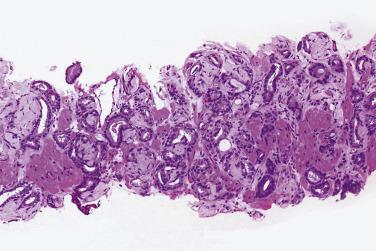
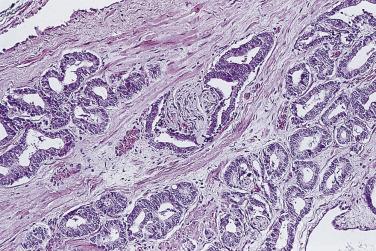
Previous studies have indicated that the overall false-negative prostate biopsies—that is, biopsies classified as benign but containing adenocarcinoma or atypical suspicious glands (atypical small acinar proliferations [ASAP])—were estimated to be 2.4% (1.1% for prostate cancer and 1.3% for ASAP). Clinicopathologic features at the time of initial biopsy and of subsequent prostate cancer diagnosis did not differ between patients with a false-negative or true benign biopsy result . Relatively low number of atypical glands (<10 glands), intense intermingling with benign glands, and lack of architectural disorganization were the most prominent risk factors for a false-negative diagnosis. Another potential pitfall was misinterpretation of prostate cancer variants, such as foamy gland, pseudohyperplastic, microcystic, or atrophic-type adenocarcinoma. It is suggested that routine examination of at least one level of a prostate biopsy at high magnification and awareness of histologic prostate cancer variants might reduce the risk of missing or misinterpreting a carcinoma during prostate biopsy evaluation.
Many sampling methods are used to examine radical prostatectomy specimens, with intent to provide the important and relevant pathologic information. In one study, Kim and associates compared five sampling methods for radical prostatectomy specimens, including (1) alternating slices, (2) alternating slices plus biopsy-positive posterior quarters, (3) every posterior half, (4) every posterior half plus one midanterior half, and (5) alternating slices plus peripheral 3-mm rim of the remaining prostate, and correlated the findings with prognostic parameters. They found that submission of alternating slices plus peripheral 3-mm rim of the remaining prostate was the most efficient and effective way to examine the prostate.
Crystalloids may be found within the lumina of malignant acini, in glands adjacent to the carcinoma (usually glandular structures exhibiting PIN or AAH and, rarely, in benign glands), and in sclerosing adenosis of the prostate. They are generally elongated, may be rhomboid or needlelike, and stain brightly eosinophilic with the usual hematoxylin and eosin (H&E) stain (see Fig. 14A.10 ). By electron microscopy they are made up of electron-dense material that lacks the parallel periodicity of crystals, and so they are referred to as crystalloids. They have been observed in up to 72.5% of carcinomas, predominantly in the well-differentiated type within neoplastic acini or in adjacent benign glands, but they have also been observed in cribriform carcinomas and, rarely, in poorly differentiated carcinomas. In needle biopsies in which crystalloids are present in benign glands, the likelihood of finding cancer on subsequent biopsies is low.
Intraluminal mucin occurs fairly frequently in well and moderately differentiated adenocarcinomas of the prostate. Neutral mucin may be found in normal glands, but acid mucins are present predominantly in prostatic adenocarcinoma. The mucin in carcinoma is present in the lumina of the glands, where it may be stained with mucicarmine or Alcian blue stains (see Fig. 14A.11 ); the intraluminal mucin appears to be wispy and basophilic on H&E staining. The cytoplasm of the neoplastic cells usually does not stain; this parameter may be helpful when trying to rule out a metastatic adenocarcinoma. However, intracytoplasmic acid mucin may be found in benign hyperplastic glands. Sclerosing adenosis, PIN, and AAH may also show intraluminal acid mucin; therefore intraluminal mucin alone is not diagnostic of carcinoma. Hence there is little utility in performing histochemical stains for acid mucin in routine diagnostic practice, but the observation of blue mucin in H&E-stained sections should alert the pathologist to rule out adenocarcinoma.
Glomerulation (cribriform formations) (see Fig. 14A.12 ), mucinous fibroplasia and collagenous micronodules (rounded, dense hyaline material within and adjacent to neoplastic acini) (see Fig. 14A.13 ), circumferential perineural invasion (see Fig. 14A.14 ), and glands in fat tissue (see Fig. 14A.15 ) are considered to be diagnostic features for malignancy because they have not been described in benign prostatic glands. Among a survey of genitourinary pathologists, features considered to be pathognomonic for cancer were glomeruloid bodies (58%), collagenous micronodules (64%), circumferential perineural invasion (84%), and glands in fat (36%). When none of these diagnostic features were present, 39% of pathologists required a minimum of 2 to 10 glands (median 3) to diagnose cancer, whereas the others had no lower limit.
It is important to realize that perineural involvement by benign prostatic glands may rarely be seen in prostate biopsy. Ali and Epstein evaluated 27 needle biopsy cases with perineural involvement and observed four patterns of perineural involvement: indentation, 14 (51.8%); tracking, 8 (29.6%); wrapping, 7 (25.9%); and intraneural, 4 (14.8%). Cases with partial or complete atrophy account for the majority of benign cases with perineural involvement. Immunohistochemical stains with HMWCK and p63 may not be helpful for diagnosis because some foci may lack basal cells. The patterns most closely mimicking cancer were intraneural and incomplete perineural encirclement. Therefore perineural involvement by benign atrophic glands may cause significant diagnostic difficulty, especially with negative basal cell immunostaining. Careful attention to the morphologic features of suspicious glands and comparison with the adjacent or distant benign glands and the extent of glands with perineural involvement are necessary.
Corpora amylacea in the prostate are a frequent finding in benign acini but can be seen rarely (0.4%) in adenocarcinoma. Thus the presence of corpora amylacea cannot be used to exclude malignancy.
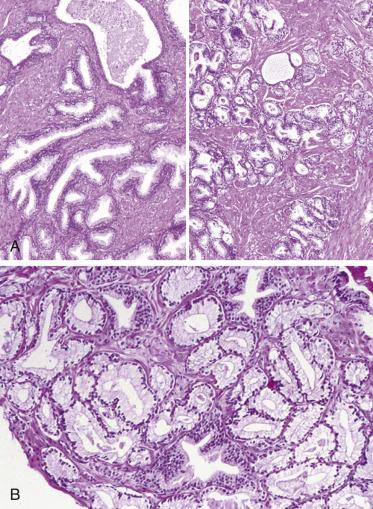
Prostate adenocarcinomas arising from the transition zone account for approximately up to 15% of prostate carcinomas, and they often have morphologic patterns different from that of typical peripheral zone carcinoma. One of the patterns, referred to as clear cell (tall columnar) carcinoma or pseudohyperplastic pattern of adenocarcinoma, occurs predominantly in the transition zone. It is a well-differentiated carcinoma that may mimic benign glandular hyperplasia in the transition zone. It is more likely to be seen in transurethral resectates, but may be seen in core needle biopsies, particularly from the base of the prostate. This tumor is made up of small to midsize glands that are lined by a single row of epithelial cells. The cells are generally tall columnar, but may be cuboidal, and characteristically have clear or amphophilic cytoplasm ( Fig. 14A.18 ). When the glands present greater variation in size or are cystically dilated, the resulting configuration may be confused with BPH.
Whether transition zone tumor morphology is unique or specific to this location is not well defined. Although this morphologic feature is much more commonly seen in the transition zone, Garcia and colleagues showed that this pattern, rarely, can be the dominant pattern or more commonly a focal or minor pattern or associated with other morphologic features in the peripheral zone. Because the nonfocal “transition-look” morphology can be seen in up to 35% of peripheral zone tumors, caution should be exercised in assigning zone of origin based on this histologic appearance, especially in limited samples such as needle biopsy.
Anterior-predominant prostatic cancers are much less common than posterior-predominant ones, accounting for 15% in one study. Among them, 49.2% and 35.5% were in the anterior peripheral zone and transition zone, respectively, and the remainder were either indeterminate or present in both zones. It is still unclear whether any differences in biology and behavior exist between tumors of transition zone and anterior peripheral zone origin.
Another well-differentiated carcinoma, foamy gland carcinoma, is characterized by small glands with foamy cytoplasm and pink luminal secretions ( Fig. 14A.19 ). It is believed to be a distinctive histologic variant of prostatic adenocarcinoma. Although it is assigned a Gleason score of 3+3=6 because of its deceptively benign histologic appearance, this tumor was reported in the past to be associated with aggressive behavior, although a larger subsequent study showed no prognostic difference. In addition, foamy gland carcinoma occasionally consists of cribriform, fused or poorly formed glands, cords or single cells, or solid sheets ( Fig. 14A.20 ), and the tumor cells can have more prominent nucleoli and increased mitotic figures; these tumors should be graded as Gleason pattern 4 or 5. A unique subset of high-grade foamy gland carcinoma can pose diagnostic difficulty when tumors exhibit scattered or rare relatively bland foamy glands embedded in an extensive densely sclerotic desmoplastic stroma.
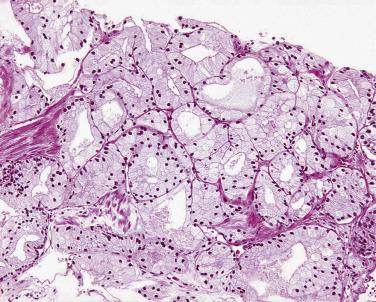
Pseudohyperplastic prostatic adenocarcinoma is another deceptive subset ( Fig. 14A.21 ), which shows features resembling benign hyperplastic glands architecturally, such as papillary infoldings, large branching glands, and corpora amylacea. Within the pseudohyperplastic foci, features helpful in establishing a malignant diagnosis are nuclear enlargement, pink amorphous secretions, occasional to frequent nucleoli, and crystalloids. Other features associated with malignancy (mitoses, blue-tinged mucin, adjacent high-grade PIN, and perineural invasion) can be seen but are infrequent. Similar to acinar carcinoma, immunohistochemical stains for HMWCK, CK5/6, and p63 show an absence of basal cells in the pseudohyperplastic areas, supporting the diagnosis of cancer. It is critical to be aware of this entity and apply immunohistochemical stains for basal cell markers when necessary, particularly on biopsy specimens.
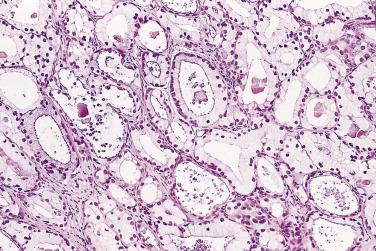
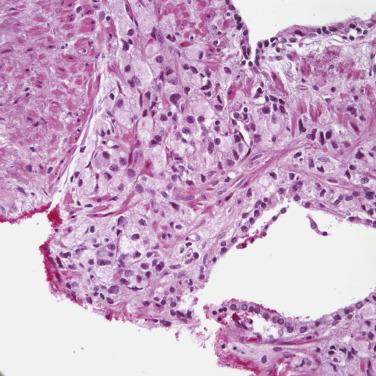
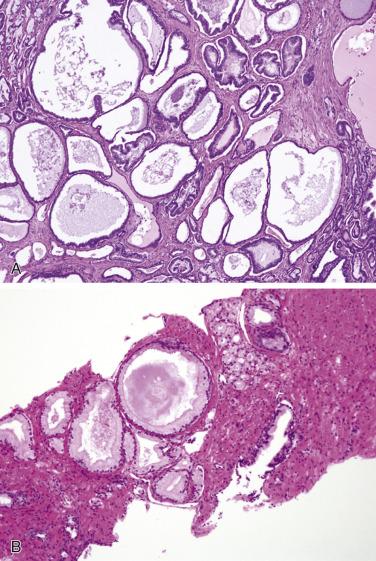
More recently, cystic change in adenocarcinoma of the prostate has been described that may mimic benign cystic atrophy. This tumor is referred to as microcystic adenocarcinoma of the prostate. It is defined by cystic dilation and rounded expansion of the malignant gland profile, with a flat luminal cell lining layer ( Fig. 14A.22A ). In one study, an incidence of 11.2% of microcystic carcinoma was identified in review of 472 consecutive radical prostatectomies for prostate cancer. The microcystic component was typically adjacent to usual small acinar adenocarcinoma and can be seen on biopsy (see Fig. 14A.22B ). Atrophic features were seen consistently in all cases. Gleason pattern 3 was the predominant grade of the tumor. Intraluminal crystalloids and wispy blue intraluminal mucin were seen in all cases; these are helpful features for diagnosis. The overexpression of AMACR and complete loss of basal cells as evaluated by HMWCK (34βE12), CK5/6, and p63 immunohistochemistry provide confirmatory evidence for the diagnosis.
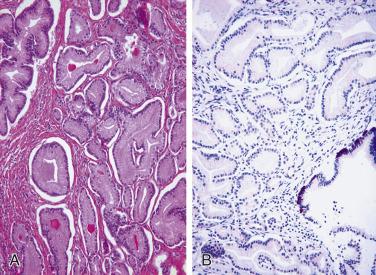
For well-differentiated adenocarcinoma, several differential diagnoses occur in the transition zone and exhibit small-gland proliferations, including atypical adenomatous hyperplasia (adenosis), sclerosing adenosis, basal cell hyperplasia, and clear cell cribriform hyperplasia of the prostate. Different from small acinar adenocarcinoma, a common and consistent finding among these benign small-gland proliferations is the presence of basal cells, which can be highlighted by immunohistochemical stains for HMWCK (34βE12), CK5/6, or p63. Other lesions such as nephrogenic adenoma, mesonephric and verumontanum mucosal gland hyperplasia, and normal anatomic structures (i.e., prostatic atrophy, ejaculatory duct and seminal vesicle epithelium, and Cowper glands) may simulate adenocarcinoma. These entities are considered in the discussion of carcinoma mimics (see later discussion).
Adenocarcinoma presenting with an atrophic pattern (atrophic variant of adenocarcinoma) may be misdiagnosed as a benign lesion ( Fig. 14A.23 ). Although this type of carcinoma must be kept in mind when atrophic-like changes are present, the atrophic variant is usually accompanied by conventional acinar adenocarcinoma. In difficult cases, a panel of immunostains demonstrating positive AMACR staining, along with negative staining with basal cell markers, is helpful in making the diagnosis of atrophic prostatic cancer rather than benign atrophy.

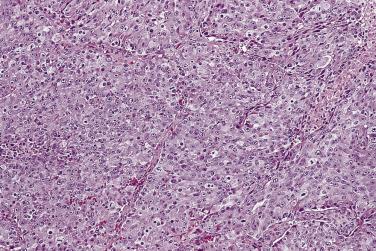
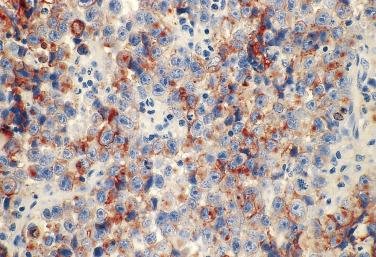
For adenocarcinoma with high-grade nuclear features, prostatic involvement by urothelial carcinoma associated with either invasive bladder carcinoma or primary urothelial carcinoma in the prostatic ducts and acini enters into the differential diagnosis. Even poorly differentiated adenocarcinoma of the prostate generally retains monotonous cytomorphology, and not infrequently a faint acinar pattern; the nuclei, although variable in size, do not show extreme variations, the chromatin is finely dispersed, and the nucleoli are prominent in most cells ( Fig. 14A.24 ), but mitoses are rare. In contrast, prostatic urothelial carcinoma often exhibits significant nuclear pleomorphism, coarse chromatin, nuclear hyperchromasia, and frequent mitoses ( Fig. 14A.25 ). PSA is usually positive in prostate adenocarcinoma ( Fig. 14A.26 ), although it may be negative or focally positive in patients who have received hormonal treatment. PAP is generally positive in prostate adenocarcinoma as well, but the staining may be focal. CK7 and CK20 are typically negative in adenocarcinoma but positive in urothelial carcinoma, as are GATA3, thrombomodulin, and uroplakin II or III, although they can be negative because of loss of antigenicity in high-grade urothelial carcinoma. Prostatic xanthoma may mimic high-grade prostatic adenocarcinoma or adenocarcinoma treated with hormone therapy because of its solid growth with cords and individual cells infiltrating the prostatic stroma. In addition, xanthoma cells may rarely be positive for PSA, PAP, and AMACR. However, these cells are uniformly positive for CD68 and negative for CK. Therefore careful attention to the morphology and immunohistochemical stains with both CD68 and CK (CAM5.2) will be helpful in the differential diagnoses of prostatic xanthoma versus high-grade prostate carcinoma.
One of the most challenging areas for surgical pathologists since the 1990s has been the accurate diagnosis of cancer on contemporary 18-gauge needle biopsy. Two aspects have been the separation of cancer from its many benign mimickers and establishment of a sensible threshold for diagnosing cancer in extremely small numbers of atypical glands. Although many criteria have been defined for the diagnosis of prostate cancer in numerous previous publications, it is still difficult to apply such criteria when dealing with a small number of atypical glands. A meticulous systematic approach is necessary. The process should involve evaluation of (1) architectural features; (2) cytologic features; (3) clues that may assist in the diagnosis, such as bluish acid mucin, crystalloids, collagenous micronodules, glomerulation, and circumferential perineural invasion, as mentioned earlier; and (4) presence of associated high-grade PIN. Caution is warranted if marked inflammation, budding from apparent benign glands, and artifacts of crushing or thick sections are present.
Immunohistochemistry plays an important role in the diagnosis of prostate cancer in some situations. PSA and PAP are two well-known prostate tissue–specific antigens used to establish whether a tumor is of prostatic origin. However, these two antibodies cannot be used to differentiate between benign and cancerous prostate tissue. Two markers, P501S (prostein) and prostate-specific membrane antigen (PSMA), have been introduced as prostate cancer markers, although PSMA may be more useful for prognosis than diagnosis. P501S immunostaining shows a perinuclear cytoplasmic (Golgi) pattern in prostate cancer, including poorly differentiated and metastatic cancers. It has been shown that a small subset of metastatic prostate carcinoma exhibits significant differences in staining intensity and extent for PSA and P501S; therefore use of both markers may result in increased sensitivity for detecting metastatic prostate cancer. However, because some in situ and invasive adenocarcinomas of urinary bladder origin may also show immunoreactivity for P501S and PSMA, caution is warranted when using them to differentiate an adenocarcinoma from the prostate or urinary bladder. The level of PSMA expression has been explored as a useful prognostic marker in predicting PSA recurrence. In addition to these markers, NKX3.1 (an androgen-related tumor suppressor gene) and proPSA (pPSA) (precursor form of PSA) have been evaluated and used to diagnose adenocarcinoma. It was shown that one of two types of proPSA, native [-5/-7] pPSA, is even better than PSA and PAP to detect metastatic adenocarcinomas. Therefore a panel of markers, including proPSA, may be used to confirm the adenocarcinoma in a metastatic setting. Markers considered to be useful for urothelial carcinomas include HMWCK, p63, thrombomodulin, uroplakin II and III, GATA3, and S100P (placental S100), which can be used to differentiate them from high-grade prostate adenocarcinoma. It has been recommended by the International Society of Urologic Pathology (ISUP) that PSA and GATA3 should be used as the first line panel, with P501S, NKX3.1, p63, and HMWCK being additional markers to distinguish high-grade prostate adenocarcinoma from urothelial carcinoma.
The loss of basal cells is the hallmark of prostate adenocarcinoma; therefore immunohistochemical staining of basal cells has been invaluable in establishing the diagnosis of prostate carcinoma. According to one study, the most commonly used markers in diagnosing prostate cancer are HMWCK (34βE12) (91%), p63 (58%), and, less commonly, CK5/6.
The greatest use for basal cell markers is demonstration of basal cells in small biopsy samples, in which suspicious foci are often very small; therefore the appropriate use of tissue levels is critical for diagnosis. It is recommended in many institutions to prospectively store intermediate levels on gelatinized unstained slides for possible immunostains. In addition, interpretation of basal cell immunostains has to be done judiciously and in conjunction with morphology. One must insist on a good internal control and absolute lack of staining in the entire focus in question before making a diagnosis of carcinoma. Both 34βE12 and p63 are highly specific for basal cells and are negative in areas of adenocarcinoma. The marker p63 is more sensitive than 34βE12 in staining benign basal cells, particularly for TURP specimens, offering a slight advantage over 34βE12 in diagnostically challenging cases. A cocktail of basal cell markers composed of 34βE12 and p63 has been developed, which not only increases the sensitivity of basal cell detection but also reduces the staining variability. Therefore, in difficult cases, use of both 34βE12 and p63 separately or as a cocktail will allow a more definitive diagnosis of cancer. Furthermore, it has been observed that prostate cancer may rarely show focal nonspecific staining for HMWCK, a phenomenon that may lead to false-negative diagnoses. Similarly, rare aberrant expression of p63 in adenocarcinoma has also been observed in a nonbasal distribution pattern, leading to the erroneous diagnosis of atrophy or atypical basal cell proliferation. It is critical that the diagnosis of adenocarcinoma is based on the morphology and confirmed by the absence of basal cells verified by immunohistochemical staining with both p63 and HMWCK.
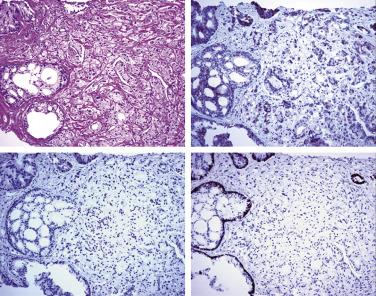
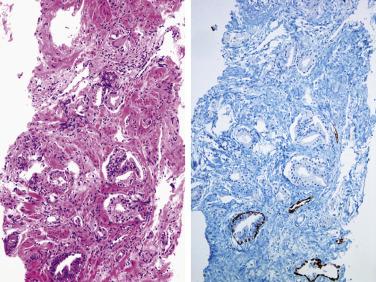
A positive tumor marker for prostate cancer, AMACR, monoclonal (P504S) or polyclonal (AMACR-p), is quite often used in conjunction with H&E histology and basal cell markers in the workup of difficult prostate needle biopsy specimens. However, it is not known how often a positive AMACR staining is used merely to support a malignant diagnosis that could otherwise be established based on routine H&E histology and negative basal cell staining. It has been shown that 64% of cases of high-grade PIN are positive for AMACR. In addition, benign prostatic lesions, such as nephrogenic adenoma and AAH, as well as nonprostatic tumors, can be positive with AMACR. Furthermore, approximately 20% of adenocarcinomas are AMACR negative, so use of AMACR alone is dangerous for the diagnosis of adenocarcinoma. To improve the sensitivity and specificity of the diagnosis of adenocarcinoma, a cocktail of two antibodies (p63/p504S) also has been used. Combined use of these two antibodies on the same tissue section, p504S as a positive marker and p63 as a negative marker for cancer, has the potential to improve diagnostic performance ( Fig. 14A.27 ). The cocktail is particularly useful in resolving atypical small acinar proliferations and reduces the percentage of ambiguous diagnoses and the need for additional biopsies. In addition, the cocktail was very useful in highlighting adenocarcinoma associated with high-grade PIN, flat and cribriform high-grade PIN, and distorted foci of minimal adenocarcinoma. Any of the three commonly used basal cell markers (34βE12, CK5/6, and p63) or a combination of the two basal markers with AMACR (p504S) either in a double or triple cocktail (PIN-4™ Cocktail: 34βE12+ p63 + P504S) is recommended by ISUP for the workup of small foci of atypical glands suspicious for carcinoma.

It has been recognized that the sensitivity of AMACR staining may vary in specimens from different pathology laboratories, possibly related to differences in fixation and processing. Therefore each laboratory must optimize the conditions for specimen processing and staining. For small foci of cancer on needle biopsy, AMACR may be negative. AMACR is also a useful diagnostic marker for the variant adenocarcinomas, such as foamy gland ( Fig. 14A.28 ) and pseudohyperplastic prostate cancer, although the rate of positivity is lower than that in acinar adenocarcinoma.
AMACR overexpression has been reported in many other tumors, such as colorectal, ovarian, breast, bladder, lung, and papillary renal cell carcinomas, as well as in lymphoma and melanoma; thus it is not a very useful marker in workup of a metastatic tumor. Locally advanced adenocarcinoma can involve adjacent colorectal tissue, and conversely advanced colorectal carcinoma can extend into the prostate. In such cases, in addition to the morphologic differences, immunohistochemical studies with a panel of markers, including PSA, PAP, CDX2, and CK20, should help establish the diagnosis. Caution should be taken when using AMACR and CDX2 staining because colorectal carcinoma can be positive for AMACR, and prostate cancer can be positive, rarely, for CDX2.
PCA3 (also known as DD3 and PCA3DD3 ) is one of the most prostate cancer–specific genes ever described. Overexpression has been detected in the majority of prostate cancers, including distant metastases, but only a negligible level of expression is noted in nonmalignant prostate tissue and none at all in other nonprostate tissue or malignancy. By in situ hybridization assay, Popa and colleagues detected expression of PCA3 in formalin-fixed, paraffin-embedded tissue. PCA3 RNA is detected in most prostate cancers and high-grade PIN, but rarely in normal glands, except those located in the immediate proximity of neoplastic glands.
Circumferential perineural invasion by glandular epithelium is one of the diagnostic features for adenocarcinoma ( Fig. 14A.29 ), although perineural indentation or cuffing by benign or atrophic prostatic glands can be seen. The glands in the latter cases appear totally benign and are present at only one edge of the nerve ( Fig. 14A.30 ). It has been postulated that neural cell adhesion molecule (N-CAM), one member of the immunoglobulin superfamily of adhesion molecules, may play an important role in perineural invasion and metastasis in prostate cancer.
The term ASAP differs from AAH, and should not be confused with it. ASAP is used to describe atypical glandular proliferations that do not reach the threshold to be diagnosed as cancer either qualitatively or quantitatively. This diagnosis does not imply a specific pathologic entity, but only implies that the glandular changes are suspicious for malignancy. Lesions that may be given such a diagnosis are a focus of small glandular proliferation with lack of prominent nucleoli, lack of nuclear enlargement, artifactual distortion, too few glands to be sure, depletion of tissue with inability to do basal cell stains, small glands associated with inflammation, and so forth ( Fig. 14A.31 ). The frequency of this diagnosis in contemporary large prostate biopsy series is quite variable, ranging from less than 1% to 23%, with an average around 5%. On repeat biopsy of patients with a diagnosis of ASAP, invasive carcinoma is identified in 17% to 60%. In one study by Chan and Epstein invasive carcinoma was identified in 48.9% of patients on repeat biopsy when the patient had a diagnosis of atypical glands suspicious for malignancy (AGSM or ASAP). Furthermore, the percentage of invasive carcinoma on repeat biopsy rose to 61% in patients with a diagnosis of AGSM (or ASAP), favoring malignancy. Given the high percentage of carcinoma identified in patients with a diagnosis of ASAP, it is recommended that patients with this diagnosis undergo immediate repeat biopsy regardless of the serum PSA level. It is also advisable to sample the suspicious areas more extensively, with two or more biopsies, because of the high chance of finding invasive carcinoma. The Progensa assay of prostate cancer gene (PCA)3 calculates a risk score based on the ratio of urine PCA3 mRNA to urine PSA mRNA, measured from urine specimens taken after “attentive” digital rectal examination, to generate a PCA3 score. PCA3 test is currently FDA approved to determine if men with a negative prostate biopsy should undergo repeat biopsy. Using a Progensa score of 25 as a cutoff, to optimize sensitivity, a negative result has been shown to have a negative predictive value of 90% for cancer on rebiopsy. Men with a negative test can thus avoid being rebiopsied. Most recently, PCA3 was validated in a prospective, blinded National Cancer Institute (NCI)–sponsored Early Detection Research Network cohort, where it was shown to significantly improve the positive predictive value for an initial biopsy (at a score >60) and the negative predictive value for a repeat biopsy (at a score <20).
Immunohistochemical staining with basal cell markers (34βE12 and p63) and AMACR is very useful and should be performed before a diagnosis of ASAP is rendered, particularly for ASAP that falls short of carcinoma qualitatively or quantatively due to the presence of artifact. In small lesions, immunostaining with cocktails (AMACR/p63; PIN4 stain) on a single slide would be superior to using either marker separately because of easier interpretation or a high chance of the loss of tissue in small lesions.
The previously used Whitmore and Jewett staging system (categories: A, B, C, and D) has largely been replaced by the new TNM system, which includes more parameters such as PSA levels and new revised grading groups ( Tables 14A.3 and 14A.4 ). In the following discussion, the Whitmore and Jewett categories (A, B, C, and D) are placed before the TNM system initials. Stage A/T1 disease is an unsuspected carcinoma found in a prostatectomy specimen or TURP examined for clinically benign disease. A1/T1a disease comprises cancers that involve up to 5% of the total TURP specimen; A2/T1b disease includes cancers involving more than 5% of the resected tissue. For TURP specimens, we recommend reporting both the number of microscopic foci of carcinoma and the percentage of carcinomatous involvement. With regard to the amount of prostatic chips to be examined in any TURP performed for benign disease, we follow Murphy et al's suggestion of submitting up to 12 g of any given specimen, which is enough to detect the majority of incidental carcinomas, and then submitting one additional cassette for each additional 10 g of tissue. If cancer is found in fewer than 5% of the prostatic chips, all of the remainder tissue of the prostate resectate should be processed for histologic examination.
| Stage | Definition of Criteria for Inclusion |
|---|---|
| A | No palpable tumor; incidental finding in operative specimen |
| A1 | Cancer involving = 3 chips |
| A2 | Cancer involving >3 chips |
| B | Palpable tumor confined to the prostate |
| B1 | Prostatic nodule = 2 cm confined to one lobe |
| B2 | Prostatic nodule >2 cm but confined to one lobe |
| B3 | Prostatic nodule involving both lobes |
| C | Extension beyond the prostatic capsule but without evident metastasis |
| C1 | Tumor <6 cm in diameter |
| C2 | Tumor = 6 cm in diameter |
| D | Metastatic disease |
| D1 | Pelvic lymph node metastases or urethral obstruction causing hydronephrosis |
| D2 | Bone or distant lymph node or organ or soft tissue metastases |
| DEFINITION OF PRIMARY TUMORS (T) | |
| Clinical (cT) | |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| T1 | Clinically inapparent tumor neither palpable nor visible by imaging |
| T1a | Tumor incidental histologic finding in 5% or less of tissue resected |
| T1b | Tumor incidental histologic finding in more than 5% of tissue resected |
| T1c | Tumor identified by needle biopsy in one or both sides, but not palpable |
| T2 | Tumor confined within prostate a |
| T2a | Tumor involves one-half of one lobe or less |
| T2b | Tumor involves more than one-half of one lobe but not both lobes |
| T2c | Tumor involves both lobes |
| T3 | Extraprostatic tumor that is not fixed or does not invade adjacent structures |
| T3a | Extracapsular extension (unilateral or bilateral) |
| T3b | Tumor invades seminal vesicle(s) |
| T4 | Tumor is fixed or invades adjacent structures other than seminal vesicles such as external sphincter, rectum, bladder, levator muscles, and/or pelvic wall |
| Pathologic (pT) | |
| pT2 | Organ confined |
| pT3 | Extraprostatic extension |
| pT3a | Extraprostatic extension (unilateral or bilateral) or microscopic invasion of bladder neck |
| pT3b | Tumor invades seminal vesicle (s) |
| pT4 | Tumor is fixed or invades adjacent structures other than seminal vesicles such as external sphincter, rectum, bladder, levator muscles, and/or pelvic wall |
| DEFINITION OF REGIONAL LYMPH NODES (N) | |
| NX | Regional nodes were not assessed |
| N0 | No positive regional nodes |
| N1 | Metastasis in regional node (s) |
| DEFINITION OF DISTANT METASTASIS (M) b | |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| M1a | Nonregional lymph node (s) |
| M1b | Bone(s) |
| M1c | Other site(s) with or without bone disease |
| AJCC PROGNOSTIC STAGE GROUPS a | |||||
| Group | T | N | M | PSA | Grade Group (see Table 14A.5 ) |
| I | cT1a-c, cT2a | N0 | M0 | <10 | 1 |
| I | pT2 | No | M0 | <10 | 1 |
| IIA | cT1a-c, cT2a | N0 | M0 | ≥10 <20 | 1 |
| IIA | cT2b-c | N0 | M0 | <20 | 1 |
| IIB | T1-2 | N0 | M0 | <20 | 2 |
| IIC | T1-2 | N0 | M0 | <20 | 3 |
| IIC | T1-2 | N0 | M0 | <20 | 4 |
| IIIA | T1-2 | N0 | M0 | ≥20 | 1-4 |
| IIIB | T3-4 | N0 | M0 | Any | 1-4 |
| IIIC | Any T | N0 | M0 | Any | 5 |
| IVA | Any T | N1 | M0 | Any | Any |
| IVB | Any T | Any N | M1 | Any | Any |
a When either PSA or Gleason is not available, grouping should be determined by T stage and/or either PSA or Gleason as available.
b When more than one site of metastasis is present, the most advanced category is used. M1c is most advanced.
In the clinical T1 stage, the TNM system includes a third category, T1c, which is assigned to patients with clinically inapparent carcinoma (nonpalpable, nonvisible) detected by prostatic biopsy because of an elevated PSA.
Palpable tumor, or tumor that is visible on ultrasound but confined within the prostate, is designated as stage B/T2. Because radical prostatectomy for stage T2 is performed with the intention of cure, the role of the pathologist is to confirm the diagnosis of carcinoma, grade the tumor, provide the pathologic stage, estimate the tumor volume, and evaluate seminal vesicle involvement and the surgical margins, including the apex and bladder cuff. Organ-confined tumors (pathologic stage T2) are subdivided into pT2a, T2b, and T2c depending on the extent of tumor involvement (half of one lobe or less, more than half of one lobe but not both lobes, and both lobes, respectively). If tumor is found beyond the confines of the prostate (e.g., the periprostatic connective tissue or the seminal vesicles), the tumor is pathologically upstaged to stage pT3 disease. Further management depends largely on the pathologic findings in the radical prostatectomy specimen.
Stage C/T3 or T4 (see Tables 14A.3 and 14A.4 ) disease refers to carcinoma extending outside the prostatic capsule to involve the seminal vesicles, bladder, rectum, or pelvic wall. Patients with clinical stage C/T3 or T4 disease in the past were generally treated with definitive radiation to the pelvis, which yielded a 5-year survival rate of 60% to 65%. Currently, the clinician decides whether radiation alone or in combination with hormonal manipulation is the best management. Surgery is usually limited to transurethral resection of the prostate if the patient experiences obstruction. Adjuvant chemotherapy is generally reserved for patients who are unresponsive to hormonal treatment. Microscopic bladder neck involvement by carcinoma was staged as T4 in the 2002 TNM staging system, but now is considered T3a in the 2009 seventh and most current 2016 AJCC TNM staging system, because some studies have shown that microscopic bladder neck involvement by tumor has a prognosis similar to that of tumor with extraprostatic extension, but differs significantly from the prognosis of tumor with rectal or pelvic wall involvement.
Although it is usually true that, when tumor invades adipose tissue on a biopsy or prostatectomy specimen, it is staged as pT3a, only small amounts of fat have been shown to be present in prostatic stroma, usually near the prostatic capsule. In one study, prostatic fat tissue was identified in 4% of prostate specimens when examining the prostate by whole-mount sections. Therefore caution must be exercised in diagnosing extraprostatic extension based only on identification of fat invasion, especially in a needle biopsy. The small size of foci of adipose tissue and its admixture with benign glands are useful morphologic clues in distinguishing it from true extraprostatic fat.
Stage D/N or M disease denotes metastatic adenocarcinoma. This stage is also subdivided in both staging systems and is subdivided into M1a (nonregional lymph node metastasis), M1b (bone metastasis), and M1c (other sites with or without bone) according to the metastatic sites (see Tables 14A.3 and 14A.4 ). Management essentially comprises hormonal management with blocking agents, orchiectomy, or both, thereby ablating any androgen activity. Tumor histology and expression of PSA and chromogranin A are heterogeneous in metastatic adenocarcinoma to the bone or other sites. Gleason grade of the primary tumor does not predict the histologic pattern of the metastases. Although more than 70% of tumor cells express PSA, the fraction of PSA-positive cells varies widely in separate metastases in some patients. Likewise, the fraction of neuroendocrine tumor cells in different metastases varies widely.
In an autopsy study of 1589 patients with prostatic adenocarcinoma, metastases were identified in 35%, with most frequent visceral locations being bone (90%), lung (46%), liver (25%), pleura (21%), and adrenal glands (13%). An inverse relationship was found between spine and lung metastases, suggesting that metastasis to the spine is independent of lung metastasis. The maximum frequency of spine involvement occurred in smaller tumors (4–6 cm) compared with the maximum spread to lung (6–8 cm) and liver (>8 cm), suggesting that spine metastases precede lung and liver metastases in many prostate cancers. A gradual decrease was seen in spine involvement from the lumbar to the cervical level (97% vs 38%), which is consistent with upward metastatic spread along spinal veins after initial lumbar metastasis. Loss of heterozygosity in at least one marker was more frequently identified in lymph node–positive than lymph node–negative adenocarcinomas, and 10q23.3 could be a marker for metastatic progression.
Several investigators designated tumor with certain characteristics, small size (≤0.5 mL), lack of Gleason pattern 4 or 5 component, and absence of extraprostatic or seminal vesicle invasion, as clinically insignificant cancer. Patients with these features may be candidates for active surveillance. In a study of 371 consecutive radical prostatectomy specimens evaluated by whole-mount sections, 16.7% had tumor volume less than 0.5 mL. Small-volume prostate cancers are often multifocal and bilateral, with predilection for the peripheral zone. Of these small-volume cases, 16% had Gleason pattern 4 and might therefore be clinically significant.
Several grading systems have been proposed for prostatic carcinoma in the past, such as those from the MD Anderson Cancer Center, Gaeta, and Mostofi. In 2005 and more recently 2014, the ISUP conducted two consensus conferences to update the prostate grading system. Additional changes have been made in 2014 and a modified Gleason system was adopted by the World Health Organization for the 2016 edition of Pathology and Genetics: Tumours of the Urinary System and Male Genital Organs .
The original Gleason system has been widely used by pathologists, urologists, and medical and radiation oncologists in the United States and throughout the world as the preferred grading system for prostate adenocarcinoma for many years. The Gleason scoring system is based primarily on the growth pattern of neoplastic glands on low-power magnification. The tumor is divided into five patterns based on the tumor differentiation, with 1 being best differentiated and 5 being worst differentiated. Because of the frequent heterogeneity of tumor differentiation, prostate cancers often have more than one pattern. In the original Gleason grading system, the two most common patterns (primary and secondary) are recorded. However, in the revised Gleason grading system, the score has been revised to the most common primary and worst patterns instead of the second most common pattern for biopsy grading. The sum of these patterns constitutes a score that ranges from 2 to 10.
Pattern 1 tumor is composed of closely packed, separate, single, round uniform glands with a well-defined margin. The tumors with pattern 1 display a well-defined nodule, relatively uniform gland size and shape, and back-to-back arrangement with minimal intervening stroma ( Fig. 14A.32 ). Unlike BPH or normal secretory cells, cells of pattern 1 cancer are uniformly large with even, straight borders laterally and luminally. The nuclei may sometimes appear benign but are fixed in a basal row dissimilar to the uneven distribution in BPH or normal glands. These subtle cytologic features are useful diagnostic clues, especially in small-needle biopsy samples, in which architecture may be difficult to interpret.
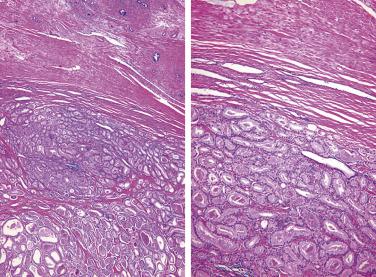
Pattern 2 is similar to pattern 1, but nodular margins are less well defined than in pattern 1, the glands are less uniform than in pattern 1, and the intervening stroma of pattern 2 is more obvious than in pattern 1 but less than in pattern 3 (>1 gland diameter) ( Fig. 14A.33 ). Recognizing patterns 1 and 2 is unreliable and almost never diagnosed in 18-gauge needle biopsy specimens; therefore diagnosis of pattern 1 or 2 tumor on needle biopsies should not be made. Patterns 1 and 2 as initially designated by Gleason may possibly represent AAH (adenosis) because of the lack of basal cell markers at that time. Because of the elimination of pattern 1 and 2 in the current revised Gleason system, the lowest pattern is pattern 3 in biopsy specimens. Pattern 3 prostatic adenocarcinoma consists of variably sized glands that are well formed as discrete units. Small well-formed glands and irregularly shaped glands are still considered pattern 3 ( Fig. 14A.34 ). A few poorly formed glands adjacent to well-formed pattern 3 glands may represent tangential section of well-formed glands ( Fig. 14A.35 ) and are still considered pattern 3. One major deviation from the original Gleason system is that any cribriform gland, regardless of size or shape, is considered pattern 4 ( Fig. 14A.36 ) because numerous studies have consistently shown that cribriform glandular pattern is an adverse prognostic feature.
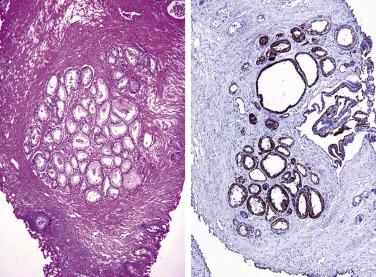


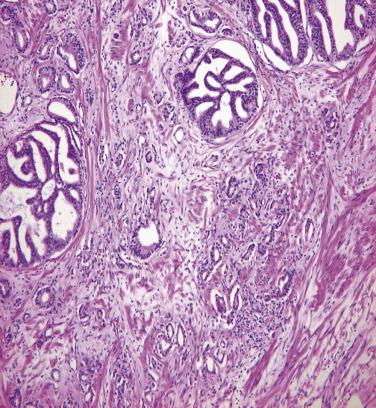
Collagenous micronodules (see Fig. 14A.13 ) were to be graded as Gleason 6 (3+3) in the consensus meeting. The delicate ingrowths of fibrous tissue seen with mucinous fibroplasia can result in glands appearing to be fused and resembling cribriform structures, although the underlying architecture is often individual discrete rounded glands invested by loose collagen. It was the consensus of the panel that an attempt should be made to subtract the mucinous fibroplasia and grade the tumor based on the underlying glandular structures. A more recent study by Kim and colleagues demonstrated that collagenous micronodules were more frequently associated with Gleason pattern 4 cancer warranting morphologic reappraisal, rather than the consensus assignment of Gleason pattern 3.
Pattern 4 based on the revised Gleason scoring system consists of three patterns: (1) cribriform glands regardless of size or shape ( Fig. 14A.37 ), (2) poorly formed glands, and (3) fused glands ( Fig. 14A.38 ). It is recommended that Gleason's original description of hypernephroid pattern not be used in pattern 4 cancers. The glandular proliferation may display a large papillary or cribriform architecture with ill-defined boundaries or confluent growth. It is recommended that glomeruloid glands regardless of morphology be assigned pattern 4 (see Fig. 14A.12 ), as this pattern is highly associated with cribriform pattern 4 or high-grade cancer. It is recognized that occasional poorly formed glands or fused glands between or adjacent to well-formed glands may not be sufficient for a diagnosis of pattern 4, as they may represent tangential sections of adjacent well-formed glands. The importance of pattern 4 either as a primary or a secondary pattern has been widely recognized. Tumors with Gleason score 7 or higher, that is, having at least Gleason pattern 4 as the primary or secondary pattern, behave worse than tumors with Gleason score 6 or less. Some studies have shown that among tumors with Gleason score 7, a significant difference exists in recurrence-free survival between patients with Gleason pattern 4+3 and those with Gleason pattern 3+4. Gleason pattern 5 tumor is the worst-differentiated prostate carcinoma and has essentially no glandular differentiation. There are two main histologic patterns: solid nests or cribriform carcinoma with unequivocal comedo necrosis (5A) ( Fig. 14A.39 ) and poorly differentiated prostate cancers that infiltrate the stroma as single cells, ill-defined cords, or solid sheets of tumor cells (5B) ( Fig. 14A.40 ). This pattern also had included the undifferentiated small cell carcinoma ( Fig. 14A.41 ). However, in the 2005 and 2014 ISUP consensus conferences, it was recommended that a grade is not provided for small cell carcinoma of the prostate because its clinicopathologic features are different from those of Gleason pattern 5 acinar-type carcinoma. A simple diagnostic algorithm of Gleason grading is depicted in Fig. 14A.42 .
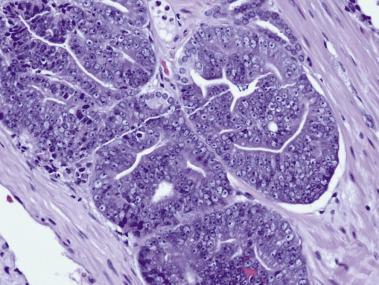

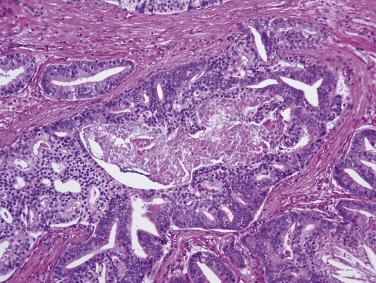
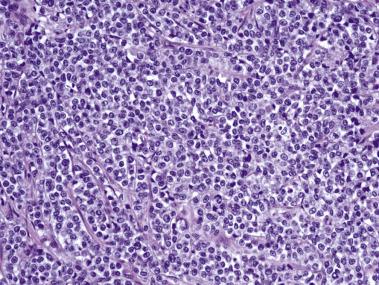
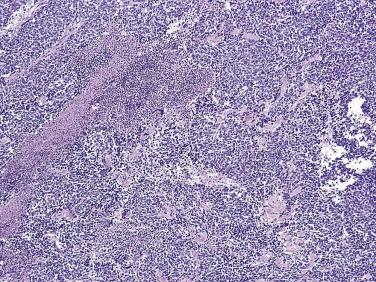

Gleason grading has proved to be a reliable and clinically significant prognostic factor in numerous studies. It correlates with extraprostatic extension, seminal vesicle invasion, and regional lymph node metastases. It is one of the key variables in several well-established prognostic models. Accurate Gleason score, along with pathologic stage and surgical margin, are critical information for the planning of post–radical prostatectomy management in patients with adenocarcinoma. Based on the multivariable model of 7869 radical prostatectomy at Johns Hopkins Hospital, a prognostic grade grouping system ( Table 14A.5 ) was proposed and then subsequently verified in a meta-analysis of more than 20,000 men treated with radical prostatectomy at five insititutions. The grade grouping system was adopted recently in the 2016 WHO classification. Although the fundamental pattern recognition of original Gleason scoring is still maintained, this novel system provides several benefits, including (1) more accurate stratification than the existing system because the criteria for patterns 3 and 4 are further defined and the prognostic significance of patterns 4 and 5 are recognized; (2) a simplified categorization of five groups rather than nine possible grades/scores; and (3) a more intuitive scale of grading group, starting at score 1 instead of 6.
| Grade Group | Gleason Score | Histologic Definition |
|---|---|---|
| 1 | ≤6 |
|
| 2 | 3 + 4 = 7 |
|
| 3 | 4 + 3 = 7 |
|
| 4 | 4 + 4 = 8 3 + 5 = 8 5 + 3 = 8 |
|
| 5 | 9 or 10 |
|
a For cases with >95% poorly formed/fused/cribriform glands or lack of glands on a core or at RP, the component of <5% well-formed glands is not factored into the grade.
b Poorly formed/fused/cribriform glands can be a more minor component.
Although some studies have shown correlation of expression of molecular markers with Gleason grading, currently no single marker has been shown to outperform or replace the Gleason grading system. Measurement of DNA content of prostate carcinomas has been compared with Gleason score. Gleason scores of up to 5 are usually associated with diploid tumors, whereas high-grade carcinomas (score 9–10) are generally aneuploid. Tumors with an intermediate grade (score 6–8) have both diploid and aneuploid patterns.
Recurrent gene fusions between the androgen-regulated gene TMPRSS2 and the ETS family of transcription factors ERG, ETV1, ETV4, and ETV5 have been identified as a common molecular event in prostate cancer development. TMPRSS2 gene encodes a serine protease enzyme in prostatic luminal secretory cells in response to androgen stimulation. Fusion of TMPRSS2 with an ETS transcription factor leads to androgen-dependent protein expression. Recurrent gene fusion events involving TMPRSS2 have been identified by FISH assays in up to 70% of prostate carcinomas. The most common gene fusion identified is TMPRSS2-ERG , which accounts for more than 90%.
The presence of a TMPRSS2-ERG fusion shows no consistent statistical association with tumor stage, Gleason grade, or recurrence-free survival. Several studies have shown some correlation with morphologic features, such as more frequent cribriform pattern and intraductal spread, signet ring cell morphology, and blue-tinged mucin with TMPRSS2-ERG gene fusion. In one study, Han and colleagues showed that 55% of the less frequent histologic variants of prostate carcinoma had ETS aberrations involving predominantly ERG . TMPRSS2-ERG fusion was identified in 83%, 71%, 50%, 33%, and 29% of mucinous, small cell, ductal, glomeruloid, and foamy gland prostate carcinomas, respectively. Notably, 88% of variant morphologies showed concordance of TMPRSS2-ERG fusion with the presence of associated acinar type, suggesting that variant morphology is clonally related to the latter. Another study by Guo and associates reported that TMPRSS2-ERG fusion is more prevalent in prostate cancer arising from the peripheral zone than the transition zone. However, the value of TMPRSS2-ETS gene fusion in diagnosis, prognosis, and therapy has not been clearly established for adenocarcinoma.
DNA microarray has been used to evaluate global gene expression in prostatic adenocarcinoma. In one early study, by different patterns of gene expression, Lapointe and colleagues identified three distinct subtypes (1, 2, and 3) of adenocarcinoma that have not been recognized histologically. Tumor recurrences were observed only among patients in subtypes 2 and 3, suggesting that subtype 1 may represent a clinically favorable subclass of prostate cancer. By using surrogate markers for the subtypes, assayed by immunohistochemistry in tissue microarray, the authors confirmed that subtype 1 (defined as zinc-α2-glycoprotein [AZGP1] positive and mucin 1 [MUC1] negative) was associated with significantly lower recurrence rates compared with combined subtype 2 and 3 cases (AZGP1−, MUC1+), independent of pathologic stage, tumor Gleason grade, and preoperative serum PSA levels. The same group subsequently identified hCAP-D3 as a second tissue biomarker for favorable prognosis in subtype 1 prostate tumors.
Numerous molecular markers have been evaluated as potential prognostic markers for tumor recurrence, metastasis, and survival. Some of them are nuclear MYC, NDRG1/Cap43, MMP14, inhibitor of DNA binding or inhibitor of cell differentiation (Id-1), nuclear forkhead transcription factor family (FKHR), urokinase plasminogen activator (uPA), Smad4, insulin-like growth factor (IGF), p16 (INK4a) , p53, PTEN, karyopherin α2, cysteine-rich secretory protein 3, OCT4, Aquaporin 5, among others. However, none of them have been proved to have consistent clinical value at the present time. As a result of the recent introduction of next-generation sequencing technologies, there will be undoubted expansion of our knowledge in the understanding of prostate cancer pathogenesis and progression. Currently there are several commercially available multigene tests for risk stratification in prostate cancer. In some cases, these tests may add value; however, none of these tests have been fully validated prospectively, and their clinical roles remain to be determined. The updated National Comprehensive Cancer Network (NCCN) Guidelines recommend “consideration” of molecular tests under certain circumstances, such as to help ascertain the likelihood of death from conservative management, of biochemical progression after radical prostatectomy or external-beam therapy, and of developing metastasis after radical prostatectomy or salvage radiotherapy. Several multigene expression assays such as Prolaris, Oncotype Dx for prostate, and Decipher are available to predict prostate cancer behavior. Pathologists continue to play an important role in the era of targeted therapy for prostate cancer.
Pathology reports should provide all clinical relevant pathologic variables for the appropriate management of patients with prostate cancer. Standardization of reports enhances communication between pathologists and clinicians who take care of patients with prostate cancer. In response to this need, various organizations have put forth tremendous efforts in developing guidelines and protocols for reporting prostate cancer in biopsy, TURP, and radical prostatectomy. Most notably are the checklists developed by the Association of Directors of Anatomic and Surgical Pathology (ADASP) and the College of American Pathologists ( https://www.cap.org/protocols-and-guidelines/cancer-reporting-tools/cancer-protocol-templates ).
For biopsy specimens, the consensus and checklist recommend the following information to be included in the report: Gleason score with primary and worst pattern, size and percentage of tumor, perineural invasion, extraprostatic extension, and lymphovascular invasion if present. With Gleason score, some variations of reporting exist among pathologists. No minimal size criteria exist for a Gleason score. When three patterns are present in the prostate biopsy, the score should include the predominant pattern and highest pattern of any amount (no longer is a minimal amount of >5% required). The ISUP recommended assigning individual Gleason scores to prostate cancer cores submitted in separate containers or multiple cores in the same container with site identifiers. However, scenarios in which multiple cores are submitted in the same container without site identifiers or labeled “left/right” are common. Even if multiple intact cores are submitted in the same container without specific identifiers, some authors recommend that individual cores with cancer should be graded and/or the worst Gleason score should be recorded. Data from Poulos and colleagues indicated that the highest Gleason score from all sites and the Gleason score from the site with the highest tumor volume on the needle biopsy are equally and significantly predictive of final Gleason score on radical prostatectomy. We also recommend that the highest Gleason score from all positive biopsy sites should be used when assigning an initial score in needle biopsies.
Although no universal consensus criteria for designated foci of tumor are available, the tumor is considered two separate foci if the distance between the tumors is more than 2 mm. It is recommended that separate Gleason scores be given to each biopsy site. Whereas Karram et al's study demonstrated that, for prostate cancer in which the needle biopsy grade is representative of the entire tumor, quantifying cancer extent on biopsy by measuring discontinuous cancer on biopsy from one end to the other, as opposed to “collapsing” the cancer by subtracting out the intervening benign prostate tissue correlates better with organ-confined disease and risk of positive margins. If multiple biopsy cores contain prostate cancer with different Gleason scores, each core should be assigned a separate Gleason score rather than an overall Gleason score, especially in cases with high Gleason score cancer on at least one core.
A study surveying members of the ISUP, in addition to reporting Gleason score, indicated that parameters most often reported include the following in descending order: percent involvement of the core by cancer (67%), the presence or absence of perineural invasion (38%), the number of cores with cancer (33%), and the length of core involvement (29%). However, the value of perineural invasion by carcinoma to independently predict pathologic stage in radical prostatectomy tissues remains uncertain. A few studies have shown that perineural invasion may predict extraprostatic extension or prognosis. In other studies, perineural invasion in radical prostatectomy specimens does not predict PSA recurrence. Despite the controversy on this issue, it is recommended that the presence or absence of perineural invasion be reported routinely in needle biopsies of the prostate. Although rare, lymphovascular invasion, particularly peritumoral lymphovascular invasion, is a significant prognostic factor in predicting nodal metastasis, advanced stage, and recurrence. Studies have demonstrated the significance of a minute focus of high-grade cancer in prognosis and therapeutic decision making. Therefore it is critical to recognize and report Gleason patterns 4 and 5 cancer.
Pathologists frequently sign out benign prostate needle biopsies as showing BPH when there are no histologic findings of carcinoma or high-grade PIN on biopsy. Although occasionally a stromal nodule, indicative of the transition zone, may be found in a core biopsy, glandular hyperplasia is not commonly seen on a core biopsy. Thus benign prostate biopsies should not be signed out as displaying BPH unless there is stromal hyperplasia associated with proliferating glands or a stromal nodule is present.
Extraprostatic extension is an important parameter for predicting biochemical recurrence and ultimate prognosis of prostate cancers (see Fig. 14A.15 ). Consequently, it has been assigned to pT3a in 2002 and the current 2016 TNM staging systems. A few studies have shown that the extent of extraprostatic extension defined by different criteria is an important prognostic factor in predicting biochemical recurrence and survival. The ways to describe extent of extraprostatic extension include measurement of radial distance of millimeters of cancer outside the capsule by ocular micrometer. PSA recurrence-free survival of 2 years and 4 years was 62% and 35%, respectively, for patients with a radial distance of less than 0.75 mm, compared with 35% and 18%, respectively, for those with a radial distance of 0.75 mm or greater. Epstein and colleagues proposed subcategorizing extraprostatic extension into focal and established categories. Focal extraprostatic extension was defined as only a few neoplastic glands and established extraprostatic extension as more extensive extraprostatic spread. Wheeler and associates classify extraprostatic invasion into two levels: If tumor was present outside the prostate to a depth of less than 1 high-power field (hpf) on no more than two separate sections, they were subclassified as level 3 focal (L3F); otherwise, in the remaining patients with more extensive extraprostatic tumor spreading, tumor was labeled as level 3 established (L3E).
Seminal vesicle invasion is a poor prognostic factor and is associated with more advanced tumor stages, nodal metastasis, local and distant recurrence, and survival. Seminal vesicle invasion is defined by invasion of the muscular wall of seminal vesicle by invasive prostate cancer ( Fig. 14A.43 ). Three types of invasion have been described by Ohori and colleagues. Type I involvement is direct spread along the ejaculatory duct complex into the seminal vesicles. Type II involvement is spread outside of the prostate, through the capsule, and then into the seminal vesicle. Type III involvement is characterized by the finding of isolated deposits of cancer in the seminal vesicle, with no contiguous primary cancer in the prostate. Overall, they found a more favorable prognosis for patients with type III seminal vesicle invasion than with other types, and patients with type III seminal vesicle invasion had a progression-free survival rate similar to that of extracapsular extension without seminal vesicle invasion. A study by Roethke showed that type I invasion was the most common with 48.6%, followed by type IIa (31.4%) and IIb (20%) invasion. Type III was not present. There was no statistically significant difference between the three groups regarding Gleason score, age, and prostate-specific antigen level.
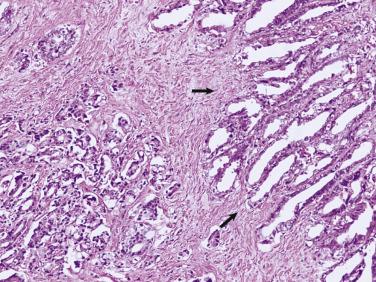
A positive surgical margin has been shown to be an independent prognostic factor in almost all studies. The incidence of a positive margin in contemporary series has been around 10%. With the advent of laparoscopic techniques, and particularly the rapid adoption of robotic-assisted radical prostatectomies in the United States, the incidence of positive margins is comparable to that in open perineal radical prostatectomy.
The positive margin is defined by tumorous glands that are transected at the inked margin and is often associated with cautery artifact. It was shown also that the length of a positive surgical margin (≤10 mm vs >10 mm) was an independent prognostic factor for biochemical recurrence in patients with prostate cancers after radical prostatectomy. Therefore it is recommended that the extent of positive margin (i.e., focal vs multifocal) and linear length should be reported in the pathology report.
Emerson and colleagues demonstrated that the closest distance between tumor and resection margin was not a significant predictor of PSA recurrence; therefore this information is not advocated as a standard part of the surgical pathology report on prostatectomy specimens.
Recently it was shown that the Gleason score of the tumor at the margin was strongly correlated with preoperative PSA, pathologic stage, and the Gleason score of the main tumor and lymph node status and was a strong predictive factor for biochemical recurrence. Therefore, in addition to reporting the positive margin, the Gleason score of tumor at the margin should also be reported.
The independent prognostic significance of tumor volume has not been demonstrated consistently. Tumor volume is correlated with pathologic stage and positive surgical margin status. At least in some studies, it has been shown that tumor volume as measured by maximum tumor diameter was a significant predictor of biochemical recurrence in patients with prostate adenocarcinoma.
Evaluation of extraprostatic extension and surgical margin status sometimes may not be straightforward. Evans and associates evaluated interobserver variability among a group of expert pathologists, concerning extraprostatic extension and surgical margin interpretation in radical prostatectomy specimens. Although urologic pathologists had good to excellent agreement when evaluating extraprostatic extension and surgical margin, significant interobserver variability was seen when there is no clearly definable prostatic capsule or significant crush or thermal artifact along the edge of the gland.
Vanishing cancer of the prostate is defined as no cancer in radical prostatectomy specimens, despite thorough sampling and careful examination in patients with adenocarcinoma diagnosed in needle biopsies or TURP specimens. The widespread use of PSA screening has led to considerable stage migration of prostate cancer. As a result, a greater frequency of low-volume disease has been found at radical prostatectomy. Previous data showed that about 4.2% of prostatectomy specimens showed no residual tumor after extensive examination. This phenomenon has been reported to be encountered even more frequently after neoadjuvant therapy or TURP. The Mayo Clinic radical prostatectomy database for 1966 to 1995 was searched for all cases in which there was no residual adenocarcinoma (pathologic stage pT0). Of the 6843 radical prostatectomies, 38 (0.6%) were identified in which no residual cancer was found despite careful sampling. The prognosis in such patients is excellent, indicating that there is no apparent clinical significance to microscopic foci of cancer that remain undetected after reasonably complete sampling. The inability to identify cancer in a prostate removed for biopsy-confirmed carcinoma does not indicate technical failure. Vanishing prostate cancer (pT0) is relatively uncommon in more contemporary radical prostatectomy series.
Several possible explanations exist for this finding, including misdiagnosis of the biopsy, very-low-volume disease, and downstaging as a result of neoadjuvant therapy, incomplete surgical removal, and specimen mix-up. When encountering a case with no residual cancer in the radical prostatectomy, it is recommended by Duffield and Epstein and Kosarac et al to (1) review the prostate biopsy, (2) perform immunostains on suspicious foci, (3) perform levels on blocks with high-grade PIN, (4) perform three levels on the posterior sextant and adjacent sextant region where cancer was identified on biopsy, and (5) flip the blocks in these regions and perform three additional levels. After this attempt, a critical question is raised as to whether the biopsy and subsequent radical prostatectomy in these patients are from the same patient. In a study by Cao and colleagues, polymerase chain reaction (PCR)–based microsatellite marker analysis was used to compare whether biopsy and prostatectomy specimens were from the same patient. The results confirmed that, in most cases of vanishing cancer, this reflects a chance sampling of a minute cancer and not a switch in specimens. However, specimen switch can occur, rarely, and if high-grade or extensive cancer is found in the biopsy sample with no or very minimal cancer in the radical prostatectomy specimen, the patient should be evaluated for identity with PCR-based microsatellite marker analysis.
Radiation treatment for prostate cancers is the most common cause of reactive atypia in the prostate. Cytologic atypia of nonneoplastic glands, including enlarged and hyperchromatic nuclei with prominent nucleoli ( Fig. 14A.44 ), have been reported in approximately three-fourths of radiated prostates. However, the architecture of the glands remains relatively preserved, a helpful feature in excluding carcinoma. Other radiation-induced changes include a decrease in the ratio of glands to stroma, atrophy, squamous metaplasia, basal cell hyperplasia, stromal fibrosis with atypical fibroblasts, foreign-body giant cells, intimal proliferation in arteries, and foam cells in vessel walls. The type of radiation affects the histologic changes as well. More cytologic atypia and stromal fibrosis are found in cases treated with interstitial radiotherapy (brachytherapy) or combined interstitial and external beam radiotherapy than external beam radiation. However, no correlation between the type of treatment and the effect on vessels has been found. There is no change over time in epithelial atypia in men treated with interstitial radiotherapy (brachytherapy) or combined interstitial and external beam radiotherapy. With external beam radiation, less epithelial atypia is present in biopsy samples taken more than 48 months after treatment compared with those with a shorter interval between biopsy and treatment. Radiation atypia in benign prostate glands may persist for a long time after the initial treatment, resulting in a significant pitfall in evaluating prostate biopsies, because prominent radiation effect can be detected in patients 6 years later after treatment with interstitial radiotherapy (brachytherapy). Radiation can induce tumor necrosis and fibrosis, but in many instances, residual viable tumor can still be identified after completion of treatment. When cribriform or fused glands, solid sheets, and cords are identified, the diagnosis can usually be made with confidence. In needle biopsies, distinguishing small acini with atypia induced by radiation from microacinar carcinoma can be a diagnostic challenge. A haphazard distribution of acini in the stroma should increase the suspicion for cancer. The absence of basal cells is helpful, but at times it is difficult to judge because of acinar shrinkage and distortion. The application of HMWCK, CK5/6, p63, or basal cell cocktail (34βE12 and p63) is very useful to identify residual basal cells, thus excluding carcinoma ( Fig. 14A.45 ). The nuclear atypia associated with radiation can mimic that of carcinoma, although the spotty, rather than diffuse, distribution of the atypia may be a clue to radiation atypia. The pleomorphism induced by radiation often exceeds that of cancer. On routine light microscopy, residual single or vacuolated tumor cells in the stroma may mimic stromal cells with vacuolar changes; in this situation, immunostains for PSA and pancytokeratin will be helpful in identifying residual tumor cells. It has been suggested that needle biopsy evaluation is of little value in the 12 months after radiation therapy because of the delayed manifestation of tumor cell death. Similar radiation changes have been reported in benign and malignant glands after three-dimensional conformal external beam radiation therapy. In one study, Martens and Keller showed that addition of AMACR in conjunction with HMWCK does not increase the recognition of prostatic adenocarcinoma in post–radiation prostate biopsies.
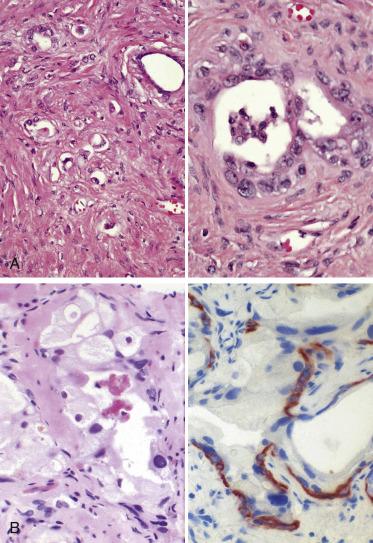
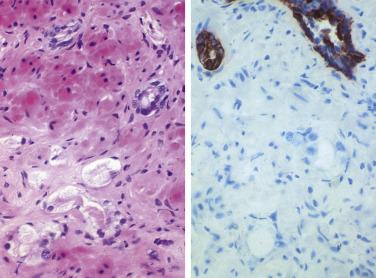
The morphologic effects induced by estrogen compounds such as diethylstilbestrol have been known for more than 50 years. Squamous metaplasia may be present, and individual tumor cells may display reduction of cytoplasm, nuclear pyknosis, and loss of nucleoli. Irregular “empty” spaces representing remnants of shrunken neoplastic glands may be seen, similar to the changes seen with androgen deprivation therapy. Recent studies have analyzed the effects of antiandrogen therapy, including total androgen blockade using drugs such as luteinizing hormone–releasing hormone agonists, flutamide, and cyproterone acetate. The effects of finasteride, a 5α-reductase inhibitor commonly used to treat hyperplastic and carcinomatous tissue, have been known for some time. Finasteride produces less pronounced morphologic changes than other androgen deprivation strategies. In radical prostatectomy specimens, the hormonal therapy leads to a reduction in tumor volume, decreased incidence of positive surgical margin, and an apparent downstaging effect, although the latter may be spurious.
Androgen deprivation has significant effects on both nonneoplastic and carcinomatous tissue. In nonneoplastic areas, pronounced glandular atrophy occurs, with shrinkage of the secretory cell compartment and basal cell prominence ( Fig. 14A.46 ). Foci of basal cell hyperplasia are common, and squamous and transitional cell metaplasia may also be seen. The frequency of high-grade PIN is significantly reduced following neoadjuvant hormonal therapy as compared with prostates not treated with hormonal therapy. Carcinomatous glands exhibit reduction in cytoplasmic quantity, cytoplasmic vacuolation, nuclear pyknosis, reduced gland diameter, and mucinous breakdown ( Fig. 14A.47 ). In many cases, prominence of collagenous stroma is seen, obscuring malignant glands. At times, complete disappearance of the neoplastic acini occurs, leaving acellular spaces, some of which may contain a mucosubstance, which can be highlighted with Alcian blue stain. This appearance has been likened to that of pseudomyxoma ovarii ( Fig. 14A.48 ). Collagenous micronodules (see Fig. 14A.13 ) are often present and likely represent organization of extravasated mucin. Stromal proliferation resulting in a hemangiopericytoma-like appearance may be seen. Nuclear changes include nucleolysis, nuclear pyknosis, and fragmentation. Paneth cell–like change is sometimes prominent in treated carcinoma, and this correlates with increased numbers of neuroendocrine cells. The architectural changes associated with androgen deprivation therapy lead to an apparent worsening of the Gleason score compared with the preoperative biopsy results. This change is likely spurious and related in great part to the cytoplasmic shrinkage and loss of lumina noted with the therapy . Studies utilizing DNA content and proliferating cell nuclear antigen have shown less proliferative activity in areas of apparently poor differentiation than in nontreated carcinomas with similar patterns, suggesting that the “upgrading” is artifactual. From a practical viewpoint, cases showing marked treatment effect should not be graded. In some situations, the neoadjuvant hormonal therapy has relatively little effect on tumor pattern, and in these situations a grade may be rendered, but caution should be exercised not to overinterpret areas that may superficially resemble Gleason grade 5 tumors. Some authors have advocated adjusting the Gleason score to compensate for the treatment effect, but this practice is not recommended because the grading of treated prostate cancer is not reproducible. In some cases, the residual carcinoma can be very difficult to appreciate, and immunohistochemical stains for pancytokeratin and PSA are necessary to look for carcinoma cells ( Fig. 14A.49 ). Rarely, stromal reactions, including a histiocytic infiltrate, may also mimic residual carcinoma cells, sometimes necessitating the use of a macrophage stain (CD68), pancytokeratin, and prostatic epithelial markers (PSA or PSAP). AMACR is frequently negative in hormone-treated adenocarcinomas, limiting its use in this situation. It has been shown that a triple cocktail stain using AMACR, 34βE12, and p63 can be helpful in making the diagnosis of residual carcinoma after hormonal therapy.
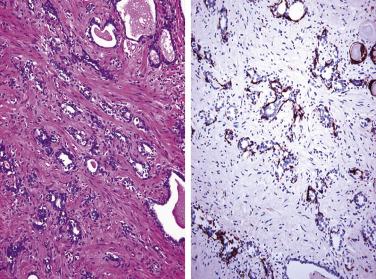
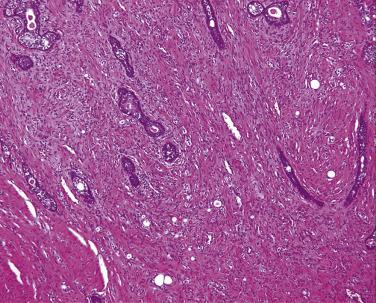
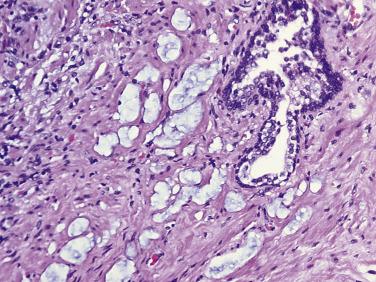
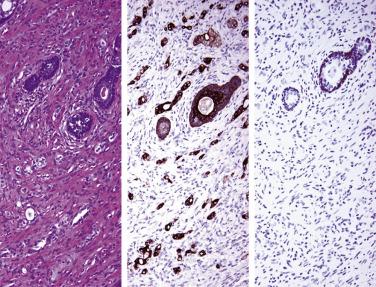
Cryotherapy is used as an option to treat locally advanced tumors in some patients. Post–cryotherapy biopsy samples show a variety of histologic changes depending on the timing of the biopsy with respect to treatment. Acute injury is manifest by necrosis, hemorrhage, and acute inflammation. Chronic changes include fibrosis, hyalinization, calcification, hemosiderin deposition, granulomatous inflammation, basal cell hyperplasia, transitional and squamous metaplasia, and (rarely) abscess formation. If present, residual carcinoma generally retains its original morphology and grade. Pathologic analysis of salvage prostatectomies performed after failed cryotherapy will reveal variable chronic changes, as well as residual carcinoma. A fatal complication from argon gas emboli during prostate cryosurgery has been described by Sandomirsky and colleagues.
Some morphologic changes, so-called in situ gene therapy effects, have been described after novel therapy of adenocarcinoma using the herpes simplex virus thymidine kinase gene (HSV-tk) followed by ganciclovir. In prostatectomy specimens, the prostate showed (1) various degrees of necrosis of cancerous foci, (2) loss of nuclear detail, (3) cytopathic changes in cancerous glands across the whole spectrum of Gleason grades, and (4) lack of necrosis of normal prostate glands, but often associated with an intense mononuclear infiltrate.
Variants of prostatic adenocarcinoma account for 5% to 10% of all adenocarcinomas. Recognition of these variants is important because many have a poorer prognosis than conventional acinar prostate adenocarcinoma. Furthermore, some variants have a deceptively benign appearance and will often have differential diagnoses that are different from acinar prostate carcinoma. Based on current understanding of their clinical behavior, Gleason grading is applied to these variants as well. Variants of adenocarcinoma with their proposed Gleason grading recommendations are listed in Table 14A.6 .
| Histologic Type or Pattern | Gleason Grade |
|---|---|
| Mucinous carcinoma | 4 or 3 (graded by underlying architectures) |
| Ductal adenocarcinoma | 4 or 5 (if with necrosis) |
| Signet ring–like cell carcinoma | 5 |
| Foamy gland carcinoma | 3-5 (graded by underlying architectures) |
| Pseudohyperplastic carcinoma | 3 |
| Microcystic carcinoma | 3 |
| Atrophic carcinoma | 3 |
| Pleomorphic giant cell carcinoma | 5 |
| Adenosquamous/squamous carcinoma | Not graded |
| Sarcomatoid carcinoma | Not graded |
| Urothelial carcinoma | Not graded |
| Small cell carcinoma | Not graded |
| Adenoid cystic/basaloid carcinoma | Not graded |
| Glomeruloid patterns | 4 |
| Collagenous micronodules | 3 (usually) or 4 (graded by underlying architectures) |
| Vacuoles | Subtract vacuoles from analysis |
| Mucin | Subtract mucin from analysis |
Since Boyd first reported a case of prostatic mucinous adenocarcinoma in 1882, approximately 60 cases of primary mucinous adenocarcinoma have been reported. The criteria for primary mucinous adenocarcinoma of the prostate are (1) at least 25% of the resected tumor must be composed of lakes of extracellular mucin and malignant glands, (2) nondilated glands containing mucinous material do not qualify as mucinous carcinoma, and (3) extraprostatic primary tumor sites should be excluded or tumor cells should be positive for PSA and PAP immunostaining. Using these criteria, Ro et al and Epstein et al reported that the incidence of mucinous adenocarcinoma was approximately 0.4% of all prostate adenocarcinomas. The morphologic features are similar to those of breast colloid carcinoma, with tumor cells floating within mucin lakes. More than two-thirds of conventional prostatic adenocarcinomas contain intraluminal mucin ; a tumor that has mucin confined within glandular lumina should not be classified as a mucinous carcinoma.
In the past, mucinous adenocarcinoma of the prostate was believed to be less aggressive than conventional acinar adenocarcinoma. They were less likely to metastasize to bone, were less likely to be associated with an elevated serum PSA level, and tended to arise from the “female” portion of the gland. However, subsequent studies found that their clinical symptoms, metastatic pattern, level of serum PSA, and response to hormonal treatment were similar to those of patients with conventional prostatic adenocarcinoma. Common metastatic sites include lymph nodes, bone, and lung. Interestingly, one case showed an endobronchial metastasis, which was confused initially with a primary bronchogenic carcinoma.
Histologically, primary mucinous adenocarcinoma of the prostate is always associated with conventional acinar adenocarcinoma ( Fig. 14A.50 ); no pure mucinous adenocarcinoma of the prostate has been reported thus far. Osunkoya and colleagues showed that the percentage of mucinous component ranges from 25% to 90% (mean 52%). The mucinous component displays extracellular mucinous lakes that often contain gyriform masses of tumor cells with occasional acini or well-formed glandular structures ( Fig. 14A.51 ), corresponding to Gleason pattern 3 or 4. In the 2005 ISUP consensus meeting, no consensus was reached relating to the grading of mucinous adenocarcinoma of prostate. Approximately half of the genitourinary pathologists assigned a Gleason score 8(4+4) for colloid carcinoma, whereas the other half thought the extracellular mucin should be ignored and the tumor graded based on architectural pattern, which may be Gleason pattern 3. In the 2014 ISUP consensus meeting, it was recommended that mucinous adenocarcinoma be graded based on its underlying growth pattern rather than pattern 4 for all cases. The patterns of the nonmucinous components varied between specimens and from area to area within the same tumor. The pattern is predominantly acinar, but other patterns such as cribriform, comedo, solid, and hypernephroid types can also be seen.

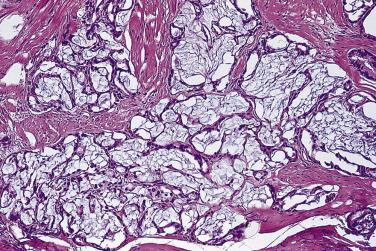
Before a diagnosis of primary mucinous adenocarcinoma of the prostate is made, metastasis from another site or direct extension of a mucinous carcinoma of the colon, bladder, prostatic urethra, or Cowper gland should be excluded. Several features are helpful to differentiate a mucinous carcinoma of prostate from nonprostatic origin, with the most characteristic feature being the presence of a conventional acinar component. Unlike those of the gastrointestinal tract or urinary bladder, the primary mucinous adenocarcinomas of the prostate usually do not contain columnar cells, goblet cells, or signet ring cells, a
a References .
although rare exceptions are reported. In addition, in prostatic mucinous carcinoma, tumor cells are floating in mucin lake–like mammary-type mucinous carcinoma rather than cells lining mucin lake (enteric-type mucinous carcinoma). Furthermore, intracytoplasmic mucin is not a feature of mucinous carcinoma of the prostate. Another condition that should be in the differential diagnosis is adenocarcinoma with focal mucin extravasation. The distinction between mucinous carcinoma and focal mucinous extravasation is the presence of epithelial elements floating within the mucinous matrix in mucinous carcinoma, whereas in mucinous extravasation only focal acellular mucin adjacent to the tumor glands is seen.
Immunohistochemically, mucinous adenocarcinoma is positive for PSA and PAP ( Fig. 14A.52 ) but usually is negative for carcinoembryonic antigen (CEA), CK7, CK20, and 34βE12. This profile allows easy distinction from urothelial-type adenocarcinoma or colorectal adenocarcinoma.

Primary signet ring–like cell carcinoma of the prostate is extremely rare, with only about 60 cases reported in the literature. The age of the patients reported has ranged from 50 to 80 years (mean 67.5 years), and the signs and symptoms are similar to those in conventional adenocarcinoma. However, patients with prostate signet ring–like cell carcinoma often are diagnosed with advanced-stage disease (pT3/T4) and metastasis and have a poor prognosis. Most patients died of disease between 32 and 60 months after the diagnosis, comparable with patients with high-grade prostatic adenocarcinoma.
Signet ring–like cell carcinoma grows as focal or diffuse sheets of undifferentiated cells with signet ring cell morphology, and thus this tumor is considered Gleason pattern 5. The signet ring cells are characterized by nuclear displacement to the edge by clear cytoplasm with nuclear indentation ( Fig. 14A.53 ). Although intracytoplasmic mucin has been described in a few cases, it is not usually present in the majority of cases. The tumor cells diffusely infiltrate the prostatic stroma ( Fig. 14A.54 ) in a pattern similar to that at other sites, such as the stomach, colon, and breast. All signet ring–like cell carcinoma of the prostate are associated with other forms of high-grade prostatic carcinoma, including solid, comedo, and cribriform types. Pure signet ring–like cell carcinoma has not been reported in the literature. Unlike disease in the other common sites, such as stomach, colon, breast, or urinary bladder, signet ring–like cell carcinoma of the prostate is usually not associated with mucinous adenocarcinoma. b
b References .
The signet ring cells are positive for prostatic markers, including PAP and PSA, but CEA is negative.
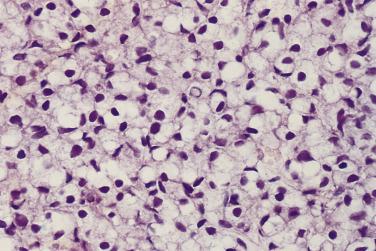
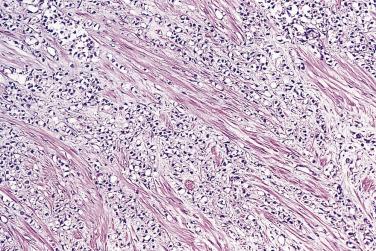
The differential diagnosis of signet ring–like cell carcinoma of the prostate includes tumors with a signet ring cell appearance and nonneoplastic processes. Adenocarcinoma may contain vacuoles and can mimic true signet ring–like cell carcinoma. Vacuoles can be seen within even Gleason pattern 3 tumors, and therefore to avoid the diagnosis of signet ring–like cell carcinoma, the tumor should be evaluated as if vacuoles were not present, by evaluating only the underlying architectural pattern. Distorted lymphocytes within areas of chronic inflammation and fibromuscular stroma can have strikingly vacuolated signet ring cell–like morphology, mimicking signet ring–like cell carcinoma. The diagnosis can be confirmed by its positive immunoreactivity with leukocyte common antigen (LCA) and negative immunostaining with CK, PSA, and PAP. Before a diagnosis of primary signet ring–like cell carcinoma of the prostate is made, metastasis from other distant sites or direct extension of a urinary bladder tumor with similar morphology should be excluded. Signet ring cell carcinomas of the urinary bladder or other organs typically produce mucin and can be highlighted with routine mucin stains. Another finding that is helpful in establishing a diagnosis of primary prostatic signet ring–like cell carcinoma is the presence of other histologic patterns of prostatic adenocarcinoma, particularly conventional acinar carcinoma. Prostate cancer after hormonal treatment can have signet ring–like tumor cells as well. Clinical history and histologic findings associated with hormonal treatment, such as squamous metaplasia of uninvolved glands and pyknosis of tumor nuclei, should provide clues to the correct diagnosis ( Fig. 14A.55 ).
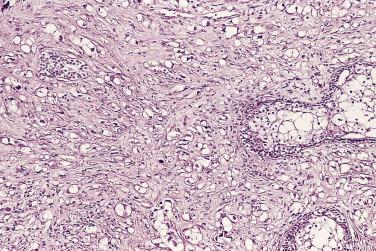
Ductal adenocarcinoma of the prostate arising in or near the prostatic verumontanum was first described in 1967 by Melicow and Pachter. They believed that the histologic pattern was similar to that of endometrioid adenocarcinoma of the uterus and suggested that this tumor originated in the prostatic utricle, a structure homologous to the female uterus and vagina. On the basis of histochemical and ultrastructural findings this tumor is believed to be a ductal adenocarcinoma of prostate, thus the terminology of ductal endometrioid carcinoma should be avoided.
Ductal adenocarcinoma shows two distinct patterns of growth. Type A pattern shows exuberant papillary growth characterized by papillary fronds; the tumor cells lining a central fibrovascular stalk range from a single layer to stratified tall columnar epithelium ( Fig. 14A.56 ). They have basally located nuclei and contain large prominent nucleoli and abundant eosinophilic cytoplasm ( Fig. 14A.57 ). Mitoses are common (>10 mitotic figures per 10 hpf) in some cases, but rare cases may have fairly bland cytologic features. Subnuclear vacuoles and secretory activity manifest by prominent intraluminal apocrine snouts are frequently observed. Type A tumors are frequently seen when the tumor grows into the prostatic urethra or within centrally located large ducts. Type B is characterized by intraductal papillary growth associated with complex glandular or cribriform with slitlike spaces, or solid and comedo-carcinomatous patterns ( Fig. 14A.58 ). This type of tumor is usually located deep in the prostate, beneath an intact or partially denuded urothelium. The tumor cells are similar to those seen in type A tumors. The two growth patterns (types A and B) coexist in approximately half of cases, and the two components tend to merge into each other. Conventional acinar adenocarcinoma is frequently associated with ductal adenocarcinoma, but it is usually focal and of lower grade. In our opinion, when this histology is seen as the pure or predominant pattern in a tumor (>80%), such a diagnosis is appropriate. In needle biopsy specimens, ductal adenocarcinoma histology may not be representative of the tumor within the prostate gland, and hence if ductal adenocarcinoma pattern is present, the diagnosis may be worded as adenocarcinoma of prostate with ductal adenocarcinoma histology ( Fig. 14A.59 ). All ductal adenocarcinomas show immunoreactivity for PSA and PAP. CEA can be positive, but it is usually weak and focal. Because CDX2 and CK20 can be positive in ductal carcinoma, differential diagnosis from metastatic colon cancer requires special caution. It has been shown that AMACR positivity is lower in ductal or cribriform Gleason 4 acinar adenocarcinoma than in noncribriform Gleason pattern 4 acinar adenocarcinoma, although the difference is not statistically significant. Basal cell staining in noncribriform acinar prostatic carcinoma is rare, but remnants of basal cells are identified by p63 or HMWCK immunostaining in a significant minority of both ductal and cribriform acinar prostatic adenocarcinoma, albeit in a patchy fashion. This result may suggest intraductal spread of tumor. The consensus has been that ductal adenocarcinoma should be graded as Gleason score 8(4+4) and that the term be retained to denote the unique clinical and pathologic findings. When necrosis is associated with this tumor, pattern 5 should be assigned.
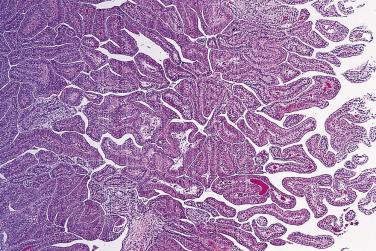

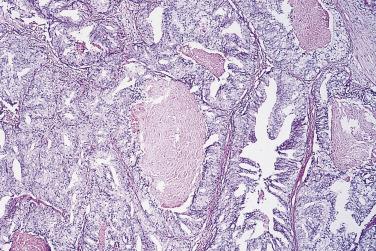
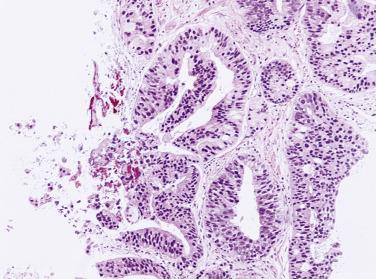
The age of patients with this tumor has ranged from 50 to 86 years (mean 65 years). Patients with ductal adenocarcinoma present more frequently with urinary obstruction and hematuria than patients with conventional adenocarcinoma. Serum levels of PAP and PSA in patients with ductal adenocarcinoma are frequently normal but can be elevated. The most frequent metastatic sites include pelvic lymph nodes, lung, and bone. However, the tumor is more likely to metastasize to viscera before it goes to bone. Bone metastases are usually osteoblastic but may be mixed lytic and osteoblastic. Unusual metastatic sites include penis and testis. Some objective responses to hormonal therapy have been observed, further supporting the contention that this tumor is a variant of prostatic adenocarcinoma.
Melicow and colleagues originally believed that this tumor usually presents as a low-stage tumor and behaves less aggressively than the conventional prostatic adenocarcinomas, which was supported by a few subsequent studies. It was postulated that this is due to its central or periurethral location and propensity to produce early symptoms with most patients diagnosed with disease limited to the prostate. However, several large studies demonstrated that these tumors may be more aggressive than previously thought and can present at a more advanced stage (T3 or any T with N1, M1).
The 5-year survival rate of 30% and median survival time of 46.3 months in our series were similar to those reported in the literature; Bostwick and colleagues reported that the mean survival of their 10 patients was 37 months and the crude 5-year survival rate was 15%; Dube and associates reported a 5-year survival rate of 42.8%. Christensen and associates demonstrated that prostatic ductal adenocarcinoma presented at a more advanced pathologic stage than clinically suspected and with a higher rate of recurrence after radical prostatectomy compared with stage-matched conventional acinar adenocarcinomas. One study suggested that finding prostatic ductal adenocarcinoma on needle biopsy implies more advanced cancer with a shortened time to progression. Samaratunga and colleagues demonstrated that ductal adenocarcinoma of the prostate had a much higher incidence of extraprostatic extension (pT3) compared with acinar adenocarcinoma.
The differential diagnoses for ductal adenocarcinoma include high-grade PIN, papillary adenoma of the urethra, prostatic-type urethral polyp, and urothelial carcinoma of the prostatic duct. c
c References .
High-grade PIN lacks the confluent growth, comedo necrosis, mitotic activity, and high degree of nuclear atypia commonly associated with ductal carcinoma. The similarities between high-grade PIN and ductal adenocarcinoma can be striking. Tavora and Epstein reported a series of ductal adenocarcinoma closely resembling high-grade PIN. PIN-like ductal adenocarcinoma differs from high-grade PIN by the presence of cystically dilated glands, a greater predominance of flat architecture, and less frequently prominent nucleoli. Their results suggested that PIN-like ductal adenocarcinomas are often associated with Gleason score 6 acinar adenocarcinoma and behave similarly to Gleason score 6 acinar adenocarcinoma.
Urothelial carcinoma involving the prostatic ducts and acini should be differentiated from the solid form (type B pattern) of prostatic ductal adenocarcinoma. Prostatic ductal adenocarcinoma is positive for PSA and PAP, whereas urothelial carcinoma is negative. Some ductal adenocarcinomas exhibit a bland cytologic appearance and may resemble a benign papillary adenoma of the utricle. In these cases, clinicopathologic correlation may give the clue as to the true nature of the lesion. The cells of prostatic-type urethral polyp are usually small, with extremely bland cytology, and the glands show two cell layers.
In the recent WHO classification, neuroendocrine tumors of prostate consist of three categories (i.e., well differentiated neuroendocrine tumor, small cell neuroendocrine carcinoma, and large cell neuroendocrine carcinoma). Overall neuroendocrine tumors of prostate are rare; among them, primary small cell carcinoma of the prostate is the most common. It has been reported in many studies that small cell carcinoma of prostate is a highly aggressive neoplasm, and the majority have been demonstrated to show neuroendocrine differentiation. The age of most affected patients is over 50 years (median 67 years), but a few patients are younger. The symptoms and signs are similar to those of conventional acinar adenocarcinoma, but rare cases have been reported to be associated with a paraneoplastic syndrome, including Cushing syndrome, Eaton-Lambert syndrome, hyperglucagonemia syndrome, inappropriate secretion of antidiuretic hormone, malignant hypercalcemia, and thyrotoxicosis. In some patients, conventional acinar adenocarcinoma precedes the diagnosis of small cell carcinoma, whereas in other patients the small cell carcinoma presents as a de novo tumor. Those presenting initially with a conventional acinar adenocarcinoma had survival of 7 months to 9 years (median 24 months). After the diagnosis of small cell carcinoma, regardless of a history of adenocarcinoma, death followed within 1.5 years (median 5 months). However, treatment with chemotherapeutic regimens similar to those used for pulmonary small cell carcinoma has resulted in prolonged survival, particularly in patients with only regional lymph node spread.
The histologic features of small cell carcinoma of the prostate are similar to those seen in the lung and other extrapulmonary sites. The small cells are of either oat or intermediate types ( Fig. 14A.60 ). More than half of prostatic small cell carcinomas are associated with conventional acinar adenocarcinoma ( Fig. 14A.61 ) and can rarely be associated with sarcomatoid and squamous carcinoma components. Immunohistochemical and electron microscopic studies demonstrate that the cells of prostatic small cell carcinoma are heterogeneous, like those of pulmonary or extrapulmonary small cell carcinomas. The cells of some tumors contain dense-core neurosecretory granules and show positive immunohistochemical staining for neuroendocrine markers ( Fig. 14A.62 ), whereas others are poorly differentiated adenocarcinomas or possibly carcinomas arising from basal (reserve) cells. Its frequent association with conventional adenocarcinoma and its immunohistochemical and ultrastructural heterogeneity led to the thought that prostatic small cell carcinoma may arise from multipotential prostatic epithelium rather than specific neuroendocrine cells.

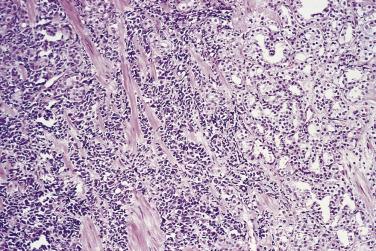
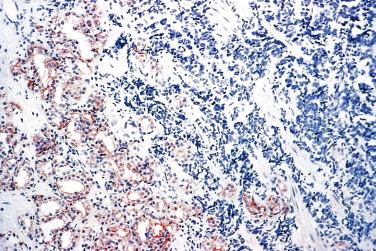
In a study by Wang and Epstein, prostatic small cell carcinomas were frequently negative for prostate tissue markers (P501S, PSA, and PSAP), and the majority were positive for neuroendocrine markers. Interestingly, almost half of the prostatic small cell carcinomas are positive for thyroid transcription factor-1 (TTF1). Expression of neuroendocrine makers (CD56, synaptophysin, and chromogranin), TTF1, CD44, PSA, and PSAP are the most helpful markers for differentiating small cell carcinoma of the prostate from high-grade prostate carcinoma. CD44 has been observed in 100% of prostatic small cell neuroendocrine carcinomas, but was negative in most of the small cell neuroendocrine carcinomas of nonprostatic origin. Small cell carcinoma of the lung, as well as extrapulmonary sites, including prostate, but not carcinoid tumor, have been reported to overexpress K homology domain containing protein, a member of the insulin-like growth factor messenger RNA-binding protein family also known as L523S and IMP3.
The levels of both PSA and PAP are useful clinical markers of disseminated adenocarcinoma of the prostate, but small cell carcinoma of the prostate is often not accompanied by significant elevation of these markers. Small cell carcinomas of the prostate have a tendency to metastasize early to the pelvic lymph nodes, lung, and liver. Unusual metastatic locations such as the omentum, vocal cord, temporal bone, axillary lymph node, and peripheral soft tissue have also been reported.
Since small cell neuroendocrine carcinoma of the prostate has unique clinicopathologic findings, different from those of high-grade acinar adenocarcinoma, including Gleason pattern 5 acinar adenocarcinoma, the consensus opinion is that small cell carcinoma of prostate should not be assigned a Gleason score.
Metastatic small cell carcinoma of the lung to the prostate must be considered in the differential diagnosis of primary small cell carcinoma of the prostate. Only a few cases of metastatic small cell carcinoma of the lung to the prostate have been reported. Since no morphologic difference exists between small cell carcinoma of the prostate and extraprostatic sites, careful clinicopathologic correlation is mandatory. One main helpful feature in making a diagnosis of primary small cell carcinoma of the prostate is the usually intimate association with conventional acinar adenocarcinoma. Lymphoma involving the prostate is rare but may mimic small cell carcinoma of prostate. Immunostaining for LCA and CK are helpful in the differential diagnosis. Embryonal rhabdomyosarcoma of the prostate has small, round blue cell histology but occurs in the pediatric population in the overwhelming majority of cases.
Well-differentiated neuroendocrine carcinoma (carcinoid tumor) of prostate is extremely rare. The diagnosis can only be made when the tumor is not closely associated with adjacent conventional acinar adenocarcinoma, and the tumor is negative for PSA but positive for neuroendocrine makers. Prostatic well-differentiated neuroendocrine carcinomas in the reported literature often present with locally advanced disease, some with regional lymph node metastasis, but they still have a relatively favorable prognosis. The other aggressive form of neuroendocrine tumor of prostate is large cell neuroendocrine carcinoma of prostate, which is exceedingly rare. In a study of seven cases by Evans and colleagues, six of the seven patients had a history of adenocarcinoma and were treated with hormone therapy. In rare situations, de novo large cell neuroendocrine carcinoma can occur. The mean patient age at diagnosis was 67 years. Large cell neuroendocrine carcinoma is characterized by solid sheets and ribbons of cells, with abundant pale to amphophilic cytoplasm, large nuclei with coarse chromatin and prominent nucleoli, brisk mitotic activity, and foci of necrosis. Foci of acinar adenocarcinoma were seen in six of the seven cases. Large cell neuroendocrine carcinoma is strongly positive for CD56, CD57, chromogranin A, synaptophysin, and AMACR (P504S). Six patients who had follow-up information died with metastatic disease at a mean of 7 months despite platinum-based chemotherapy.
Another form of neuroendocrine tumor of prostate is adenocarcinoma with Paneth cell–like change. This tumor is generally a microacinar carcinoma displaying scattered cells containing cytoplasmic eosinophilic granules. Paneth cell–like neuroendocrine cells were observed as either patchy isolated cells or diffusely involving glands or nests and can be associated with Gleason patterns 3, 4, or 5 ( Fig. 14A.63 ). The significance of Paneth cell–like neuroendocrine differentiation is unclear and remains to be elucidated.
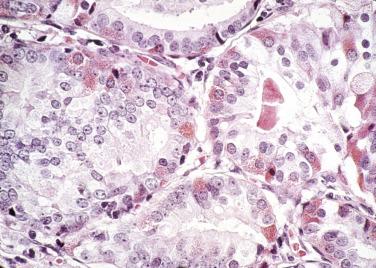
To study the influence of neuroendocrine differentiation in adenocarcinoma, expression of neuroendocrine markers, Ki67, and pololike kinase 1 were studied immunohistochemically. Analysis for PSA relapse-free survival showed an earlier progression in high-grade neuroendocrine carcinoma than in low-grade neuroendocrine tumors or adenocarcinoma with clusters of neuroendocrine tumor cells. Prominent or clustered neuroendocrine differentiation in adenocarcinoma may lead to increased proliferation and earlier tumor progression, whereas few or solitary neuroendocrine tumor cells have no prognostic impact.
Squamous cell carcinoma of the prostate is rare; it constitutes 0.2% of all malignant tumors of the prostate. Fewer than 100 cases have been reported in the literature. They are either pure squamous carcinoma or squamous carcinoma admixed with adenocarcinoma (adenosquamous carcinoma). In addition, urothelial carcinoma and sarcomatoid carcinoma have been reported to be associated with squamous and adenosquamous carcinoma. Clinically, primary squamous carcinoma and adenosquamous carcinoma cannot be distinguished from adenocarcinoma of the prostate. The presenting symptoms are similar to those of conventional adenocarcinoma. The diagnostic criteria for a pure squamous carcinoma of the prostate are (1) a clearly malignant neoplasm with disordered growth, cellular anaplasia, and invasion; (2) definite squamous features with evidence of keratinization, squamous pearl formation, and intercellular bridges; (3) lack of any glandular or acinar component; and (4) absence of primary squamous carcinoma elsewhere, particularly in the bladder. Most patients do not have a history of prior radiation or hormonal treatment, but some may have a history of prior treatment. The histogenesis of pure squamous carcinoma remains unclear; prostatic urethral epithelium, transitional epithelium of the periurethral duct, basal (reserve) cells of the prostatic acini, and common acinar cells have all been speculated to be the cells of origin. Prostatic squamous cell carcinoma associated with Schistosoma haematobium infection has been reported.
In contrast, adenosquamous carcinoma ( Fig. 14A.64 ) usually develops several years after radiation or hormone therapy for prostate acinar adenocarcinoma. Bennett and Edgerton offered two hypotheses for its occurrence: (1) The influence of hormone and/or radiation therapy alters the nature of adenocarcinoma to a mixed-cell type, or (2) a second adenocarcinoma involving periurethral ducts, concomitant with the acinar adenocarcinoma, develops a squamous component under the influence of radiation or hormonal therapy.

Even with metastatic disease, the serum PAP and PSA levels are usually not elevated in patients with squamous carcinoma. Bone metastases for squamous cell carcinoma are usually osteolytic, whereas they are usually osteoblastic in most adenocarcinomas. Overall, squamous carcinoma of the prostate is a highly aggressive tumor. Various therapeutic modalities have been used, and none have proven to be very effective in controlling this cancer. Estrogen therapy is usually unhelpful. Before a diagnosis of squamous or adenosquamous carcinoma is made, other differential diagnoses should be ruled out. Benign squamous metaplasia of prostatic ducts and acini is a frequent finding in response to infarction and estrogen treatment. It can be differentiated from squamous carcinoma by the lack of invasion or cellular anaplasia. Bladder and urethral urothelial carcinomas may show squamous and glandular differentiation and may mimic a squamous or adenosquamous carcinoma. Identification of urothelial carcinoma in situ or conventional urothelial carcinoma is helpful for the correct diagnosis. Direct extension from a squamous carcinoma of the anal canal or metastasis from distant sites (e.g., the lung) also should be ruled out. The current consensus is that no Gleason grade is assigned to squamous or adenosquamous carcinoma of prostate.
The incidence of urothelial carcinoma (UC) of the prostate varies from 1% to 5% of all prostatic carcinomas, depending on the series. Despite the low incidence, it is a very important lesion because its biologic behavior is significantly different from that of prostate adenocarcinoma. The prognosis of prostatic UC is generally very poor, and it does not respond to hormonal therapy. Although UC may arise de novo in the prostate, most cases are associated with UC of the urinary bladder. Urothelial carcinoma of the urinary bladder can involve the prostate in at least two patterns: (1) mucosal pagetoid spread through the prostatic urethra, prostatic ducts, and acini, with or without stromal invasion ( Fig. 14A.65 ), and (2) direct invasion through the bladder wall at the bladder neck or extravesical tissue ( Fig. 14A.66 ). In primary UC of the prostate, the periurethral glands and ducts are affected initially. The UC may spread along the ducts and acini and subsequently invade the fibromuscular stroma of prostate. In secondary UC, bladder carcinoma can invade the prostate directly, either at the bladder neck area or posteriorly through extravesical tissue. Just as in primary UC of the prostate, the tumor grows initially as malignant pagetoid cells, spreads through the prostatic ducts or acini, and eventually may invade the prostatic stroma. UC may also spread intraepithelially to the ejaculatory duct, seminal vesicle, and urethral meatus ( Fig. 14A.67 ). Previously, several studies demonstrated that prostatic stromal invasion was a poor prognostic factor in patients with bladder cancer. In a whole-mount section study of 214 radical cystoprostatectomy specimens, Shen and associates examined detailed patterns of involvement of prostate by UC in patients with bladder cancer and showed that the presence of prostatic urothelial carcinoma in situ and degrees of prostatic invasion were associated with prognosis.
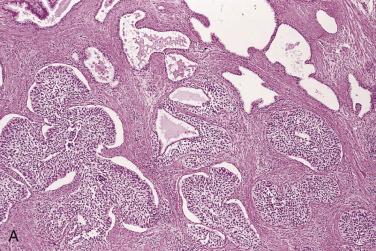
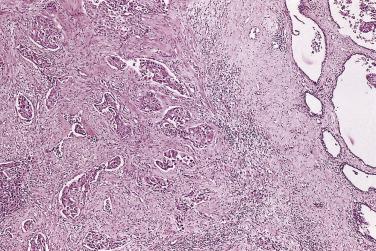
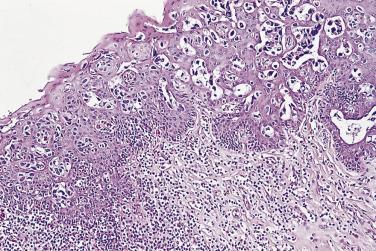
UC of the prostate typically shows a solid growth pattern, peritumoral inflammation, and frequent tumor necrosis. Furthermore, the tumor cells often exhibit much greater nuclear pleomorphism and more frequent mitotic activity than prostate adenocarcinoma ( Fig. 14A.68 ).
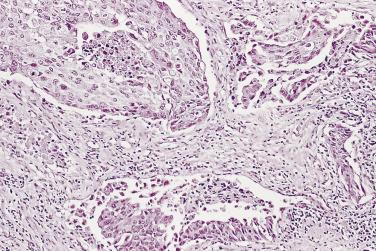
The treatments of UC and prostatic adenocarcinoma are entirely different, so distinction of high-grade solid adenocarcinoma of the prostate from prostatic UC is critical. Clinical history, careful microscopic evaluation, and immunohistochemical stains are all helpful for this differential diagnosis. By immunohistochemistry, urothelial carcinoma is negative for PAP and PSA, but usually positive for CEA, uroplakin II or III, GATA3, and thrombomodulin. Caution should be exercised when using CK7 and CK20 as markers for differential diagnosis, because some prostate cancers can be CK7+/CK20+ and some UCs can be CK7-/20-. It is recommended not to assign a Gleason grade for prostatic UC.
Transitional cell metaplasia can be differentiated from UC of the prostate by its bland cytology and prominent nuclear grooves, whereas UC is composed of much more anaplastic tumor cells with frequent mitoses.
Sarcomatoid carcinoma of the prostate is another uncommon variant of prostate adenocarcinoma with a biphasic appearance, containing carcinoma and a spindle or pleomorphic sarcomatoid component ( Fig. 14A.69 ). It can be difficult to differentiate from true sarcoma in situations in which either the conventional acinar adenocarcinoma component is minimal or the tissue sample is limited. Other terms such as carcinosarcoma, malignant mixed mesodermal tumor, spindle cell carcinoma, and carcinoma with pseudosarcomatous stroma have been used in the literature to describe this lesion. Strictly speaking, carcinosarcoma has been traditionally defined as a biphasic tumor with heterologous sarcomatous components. We and others prefer to use the term sarcomatoid carcinoma to describe all tumors showing biphasic growth with carcinoma and sarcomatous components, regardless of the presence or absence of heterologous elements.

Patients with sarcomatoid carcinoma of the prostate often present with obstructive symptoms and hence are frequently diagnosed in TURP specimens. The age of the patients with these tumors range from 58 to 85 years (mean 71 years). In our experience, these tumors are always associated with a high-grade prostatic adenocarcinoma that occurred either previously to, or simultaneously with, the sarcomatoid component. The majority of patients have metastatic disease at the time of diagnosis, and 50% have a history of prostate cancer treated with radiation or hormonal therapy. Sarcomatoid carcinoma in general has a poor prognosis, with aggressive local recurrence and short survival.
The sarcomatoid components are typically composed of spindle cells with large, pleomorphic, hyperchromatic nuclei. The pattern is that of high-grade sarcoma and in most cases similar to that of undifferentiated pleomorphic sarcoma of soft tissues ( Fig. 14A.70 ). The sarcomatoid component may also resemble fibrosarcoma or leiomyosarcoma. A heterologous sarcomatoid component (osteosarcomatous, chondrosarcomatous, rhabdomyosarcomatous) can be observed in up to one-third of cases. Immunohistochemical studies using antibodies against CK, PAP, and PSA can be helpful in proving the epithelial nature and prostatic glandular origin of this neoplasm (see Fig. 14A.69B ). The spindle cell components express CK in 20% to 25% of cases, and approximately one-third of the cases show focal reactivity for PSA, PAP, or both.

The differential diagnoses of sarcomatoid carcinoma include a variety of primary prostatic sarcomas, as discussed later. The distinction of primary sarcoma from sarcomatoid carcinoma lies in the absence of epithelial components (after extensive sampling) and usually the absence of a history of prostate adenocarcinoma. Other spindle cell lesions such as postoperative spindle cell nodule, phyllodes tumor, and inflammatory myofibroblastic tumor (pseudosarcomatous fibromyxoid tumor) should also be considered in the differential diagnosis. In these lesions, the spindle cells lack significant pleomorphism or atypical mitoses.
Several other uncommon morphologic variants of prostate adenocarcinoma have been described, including pleomorphic giant cell carcinoma, lymphoepithelioma-like carcinoma, urothelial-type adenocarcinoma, oncocytic carcinoma, renal-type clear cell carcinoma, tubulocystic clear cell adenocarcinoma, and PIN-like ductal carcinoma. Yanagisawa and colleagues described so-called stromogenic prostate carcinoma and suggested that stromal changes may be associated with aggressive behavior. The authors evaluated reactive stromal changes in prostate carcinomas in needle biopsy samples and graded them into four groups as grade 0 to 3 based on the percentage of reactive stroma: grade 0, with 0% to 5%; grade 1, 6% to 15%; grade 2, 16% to 50%; grade 3, 51% to 100%. Of 224 cases of prostate cancer, reactive stromal grading distribution was as follows: grade 0, 1 case (0.5%); grade 1, 149 cases (66.5%); grade 2, 59 cases (26.3%); and grade 3, 15 cases (6.7%). Reactive stromal grading in biopsies was correlated with adverse clinicopathologic parameters in the prostatectomy. Patients with reactive stromal grading 1 and 2 had better survival than those with 0 and 3 ( P = 0.003). Reactive stromal grading was an independent predictor of recurrence (hazard ratio = 1.953; P = 0.017). Reactive stromal grading is independent of Gleason 4+3 and 3+4 in patients with a Gleason score of 7. Therefore the authors concluded that quantitation of reactive stroma in prostate cancer and recognition of the stromogenic carcinoma in H&E-stained biopsies is useful to predict biochemical recurrence in patients with prostate carcinoma.
Although most small acinar proliferations simulating carcinoma are from the transition zone and are more frequently seen in TURP or suprapubic or retropubic prostatectomies, these changes are also seen on needle biopsies. It is conceivable that core biopsies targeted at or near the transition zone will have a chance to sample benign small acinar proliferations of the transition zone. Therefore it is important to be familiar with these lesions.
AAH, or adenosis of the prostate, is difficult to distinguish from well-differentiated adenocarcinoma of the prostate. Although McNeal and collegues referred to this lesion as a possibly premalignant lesion, to date convincing evidence for this association is lacking. In 1982, Brawn provided the only large study with clinical follow-up on patients with adenosis. One hundred and eight patients with AAH were followed for 5 to 15 years (average 7.7 years), and 7 developed carcinoma (6.5%). Carcinoma developed in 84 of 2263 patients with BPH (3.7%) who had been followed for a similar period. However, it is argued by many experts that some of the cases of AAH as illustrated by Brawn may represent well-differentiated carcinoma. Lopez-Beltran and colleagues investigated three-dimensional nuclear volume of AAH and compared it with BPH and well-differentiated prostate adenocarcinoma. Whereas significant differences were found between adenocarcinoma and AAH or BPH, the difference between AAH and BPH was not significant, suggesting that AAH is probably a histologic variant of BPH. However, genetic alterations in AAH lesions were found to be variable, and the association with adenocarcinoma is uncertain. Fortunately, AAH is uncommonly seen in needle biopsy samples because it is a lesion of the transition zone, unless the biopsy is performed in patients with marked BPH or if the biopsy is specifically targeted to the transition zone. AAH may be multifocal; its incidence in TURP specimens is 1.5% to 19.6% and up to 33% in radical prostatectomies.
AAH is a proliferation of small to midsize glands that are seemingly lined by a single row of epithelial cells with monotonous round nuclei and fine but dark chromatin pattern, showing neither nuclear atypia nor prominent nucleoli ( Fig. 14A.71 ). The basal cell layer is usually inconspicuous and attenuated, or obscured, and one may have to use immunohistochemistry for basal cell markers to prove the benignity of the lesion ( Fig. 14A.72 ). AAH may have intraluminal crystalloids, blue mucin, and a focally infiltrative pattern, making distinction from cancer even more treacherous.
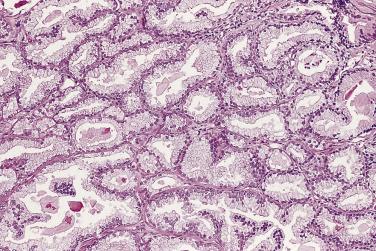
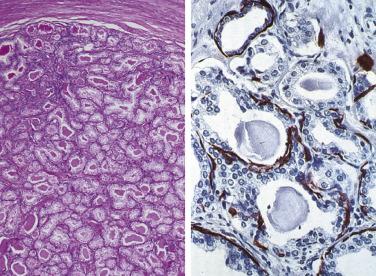
The differential diagnoses of AAH include well-differentiated adenocarcinoma and all other lesions with small glandular proliferations. By immunohistochemistry, in addition to maintaining basal cells, most AAH cases are negative for AMACR, but focal staining can be seen. In contrast, the majority of adenocarcinomas show diffuse AMACR staining. Lotan and Epstein reported a lesion termed diffuse adenosis of the peripheral zone, which occurs typically in younger patients (average 49 years). It is characterized by multiple foci of small, nonlobular, crowded but relatively bland acini on needle biopsy or in prostatectomy specimens at the peripheral zone. They suggested that diffuse adenosis of the peripheral zone should be considered a risk factor for adenocarcinoma, and those patients with this finding should be followed closely with repeat biopsy.
Basal cell hyperplasia (BCH) of the prostate gland is a common lesion in hyperplastic prostates being examined in TURP specimens. It is usually seen in the setting of BPH and thus occurs in the same age group as BPH (range approximately 63–80 years, mean 74 years); patients may present with urinary obstruction, although in most instances it is seen concomitantly with BPH. The other clinically and pathologically significant association is with antiandrogen therapy.
Microscopically, the degree of proliferation varies in extent; most foci of BCH show only individual or small groups of acini with proliferating basal cells, but lobular or diffuse areas of BCH are rather uncommon. BCH has a distinctive microscopic appearance, its recognition is not difficult in most cases, but it may mimic adenocarcinoma of small acinar pattern, particularly if one is not familiar with this histologic entity d
d References .
and when the case shows prominent nucleoli. This lesion has been described also as fetalization of the prostate simply because, on light microscopy, it resembles a fetal prostate. The presence of inflammation in the majority of BCH foci suggests that peripheral zone BCH in untreated patients may represent a stereotyped response to injury. BCH is characterized by a nodular growth of nests, tubules, and cords filled with proliferating, small, darkly staining basal cells. Most of the nests of BCH show vertical palisading of basal cells toward the periphery. The basal cells have scanty cytoplasm and oval to short-spindled hyperchromatic nuclei. Nucleoli are usually absent or, if present, small and inconspicuous ( Fig. 14A.73 ). Nuclear grooves are not present. Mitoses are absent or extremely rare, and no necrosis is present. The margin of the nodule is not encapsulated but is relatively well demarcated in most cases. e
e References .
BCH frequently merges into hyperplastic or normal prostatic glands. It has been reported to be associated with sclerosing adenosis and clear cell cribriform hyperplasia. For the sake of understanding the morphology, BCH can be classified as complete or incomplete on the basis of whether central lumina are absent or present. Complete BCH is characterized by solid nests of basal cells without luminal differentiation ( Fig. 14A.74 ), whereas incomplete BCH often shows significant central glandular differentiation with cuboidal or columnar secretory cells ( Fig. 14A.75 ); the lumina may contain neutral and acid mucin positive for PAS and Alcian blue stains. Scattered microcalcifications and corpora amylacea in the lumina are frequently seen. The stroma of BCH is usually more cellular than that of either nearby normal prostate or BPH and shows proliferation of fibroblasts or myofibroblasts. BCH is often found at the margin of infarcts, where it presumably represents a reaction to ischemia.

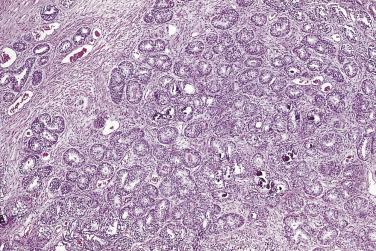
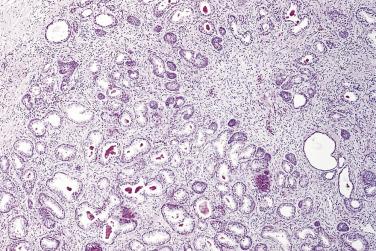
The basaloid cells of BCH show positive immunoreactivity for HMWCK, p63, and CK5/6, but show negative immunoreactivity for PSA, PAP, and AMACR. Neither basaloid nor secretory cells show immunoreactivity for S100 protein, muscle-specific actin, or vimentin, indicating lack of myoepithelial cell differentiation. In the areas of luminal differentiation (incomplete BCH), the luminal secretory cells are positive for PSA and PAP ( Fig. 14A.76 ). The incomplete BCH is more likely to be confused with carcinoma because some cases show a small acinar pattern with minimal basal cell proliferation. However, acini of incomplete BCH are composed of two cell types: the inner luminal layer of cuboidal to columnar cells and the outer layer of basaloid cells, whereas prostatic adenocarcinoma by definition does not contain a basal cell layer. The well-circumscribed growth pattern of BCH on low-power examination is the best clue for the diagnosis; in contrast, most adenocarcinomas have an infiltrative growth pattern. In addition, carcinoma shows nucleomegaly and prominent nucleoli. The basal cells of BCH may have nucleoli, but these are generally small and inconspicuous. However, an atypical variant of BCH characterized by nucleomegaly, pleomorphism, mitoses, or large prominent nucleoli (mean diameter 1.96 µm) has been described. The designation of atypical BCH has no clinical relevance or premalignant connotations, and the changes may in part be due to associated inflammation.
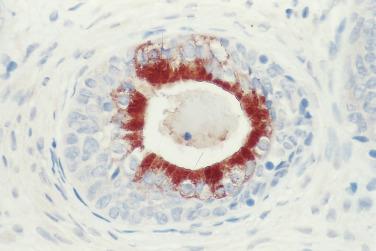
One important differential diagnosis of BCH is UC involving the prostate, which can be differentiated from BCH by its marked cellular pleomorphism, frequent mitoses, and necrosis. In addition, most patients with prostatic UC have a previous history of bladder carcinoma.
Other benign lesions, such as squamous or transitional cell metaplasia, also can mimic BCH, and although their distinction from BCH is not clinically meaningful, they can readily be separated in most instances because of fairly characteristic histology. Squamous metaplasia ( Fig. 14A.77 ) is seen usually in areas adjacent to an infarct or in patients who have received estrogen therapy. The cells of squamous metaplasia tend to have more cytoplasm and more distinct cell borders than basal cells in BCH. Transitional cell metaplasia ( Fig. 14A.78 ) is quite common in the transition zone around the prostatic urethra, although it may be fairly prominent and involve the peripheral zone. In a review of 103 consecutive biopsies it was seen in 34% of the specimens. The cells of transitional cell metaplasia are much larger and have more cytoplasm than the basal cells of BCH. The nuclear chromatin is diffusely and evenly dispersed in transitional cell metaplasia and more clumped in BCH, and the nuclei of transitional cell metaplasia have prominent nuclear grooves, not seen in the basal cells of BCH.
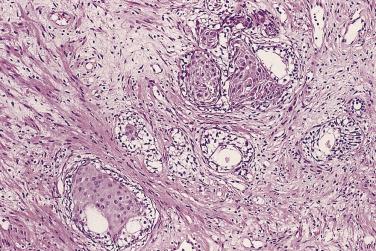
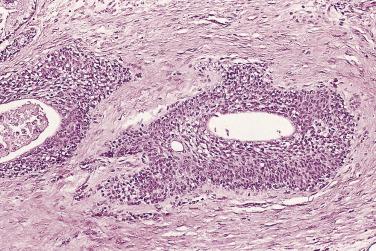
Rioux-Leclercq and Epstein described 25 cases of BCH with unusual features and identified four distinct groups: BCH with intracytoplasmic globules (5 cases), BCH with calcifications (8 cases), BCH with squamous features (3 cases), and cribriform BCH (9 cases), including 2 cases with globules. A total of 5 cases exhibited prominent nucleoli or cytologic atypia. Hyaline cytoplasmic globules have not been described in any other prostatic entity and appear diagnostic of BCH. Calcifications observed in BCH were psammomatous, differing from the fine stippled calcifications occasionally seen in areas of comedo necrosis within high-grade adenocarcinoma. BCH with squamous features differs from squamous differentiation in carcinomas (adenosquamous carcinoma) and from benign foci of squamous differentiation seen associated with either prostatic infarcts or with hormonal therapy. Whereas cribriform PIN and cribriform adenocarcinoma glands represent a single glandular unit with punched-out lumina, many of the glands within a focus of cribriform BCH appear as fused individual BCH glands. The use of basal cell markers can help in difficult cases. In cribriform BCH, it shows continuous multilayered basal cell staining in some of the glands. In contrast, cribriform PIN demonstrates an interrupted single layer of basal cell staining. Recognition of the architectural and cytologic features of unusual morphologies of BCH as well as basal cell staining pattern will facilitate its diagnosis and distinguish it from adenocarcinoma and high-grade PIN.
BCH in the peripheral zone can occur and is present in a significant minority of prostate needle biopsy (10.2%) and whole prostate samples (23%). The presence of prominent nucleoli in BCH can cause diagnostic concern for a neoplastic proliferation. In a prostate biopsy, the most helpful features for its diagnosis include solid nests, pseudocribriform glands, multilayering of cells, calcifications, and cellular stroma. Immunohistochemistry can be useful for highlighting the basal cell layers (HMWCK, CK5/6, and p63) and demonstrating negative AMACR staining.
Adenoid basal cell tumor can be considered an accentuated form of BCH characterized by larger and confluent nodules of proliferating basal cells up to 1 cm. The cells of adenoid basal cell tumor often contain nuclei two to three times larger than those of BCH and show some variation in size and shape. The chromatin is finely dispersed, and the nucleus contains occasional small nucleoli. The nests of adenoid basal cell tumor reveal areas of luminal differentiation with an occasional cribriform arrangement ( Fig. 14A.79 ). Individual nests are often invested by a thick basement membrane. Adenoid basal cell tumor is considered a locally aggressive lesion, but some lesions merge, in histology, with adenoid cystic and basaloid carcinoma.
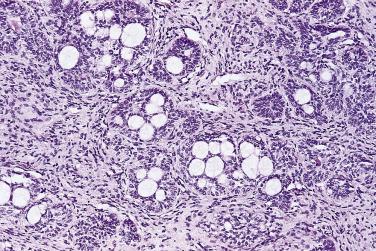
Adenoid cystic-like carcinoma and basaloid (basal cell) carcinoma of the prostate are very rare; in fact their occurrence has been challenged by some investigators because they lack the infiltrative growth or typical perineural invasion compared with typical adenocarcinoma. Adenoid cystic-like carcinoma is no longer regarded as a true adenoid cystic carcinoma but rather as prostatic basal cell carcinoma and within the spectrum of basaloid prostatic proliferations. It often contains typical BCH areas, and patients rarely if ever die of this disease. Furthermore, it has been argued that the reported cases are probably adenoid basal cell tumor or adenoid cystic carcinoma–like tumor. The tumor typically has been considered to have indolent biologic potential. However, one study of 19 patients found extraprostatic extension (26%), perineural invasion (26%) ( Fig. 14A.80 ), and distant metastasis (21%), suggesting that adenoid cystic carcinoma and basal cell carcinoma of the prostate are potentially aggressive and require ablative therapy in some cases.
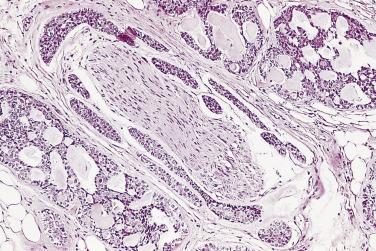
Bcl-2 expression and Ki67 index are elevated in basaloid carcinoma compared with typical BCH or atypical BCH, a fact that may aid in the diagnosis of basal cell proliferative lesions of the prostate. The marker p53 has not been discriminatory in separating benign from malignant basal cell lesions of the prostate. Ali and Epstein reported 29 cases of basal cell carcinoma of the prostate, including what others call adenoid cystic carcinoma of the prostate . Extraprostatic extension and metastasis occurred in several cases. Based on these findings, they concluded that basal cell carcinomas are rare tumors that predominantly have an indolent course; however, a small subset behaves aggressively, with local recurrences and distant metastases. The most common morphology among those with an aggressive behavior are tumors with large solid nests, central necrosis, high Ki67 index, and weak and less staining with basal cell markers.
Because basaloid tumors are frequently positive for p63, CK5/6, and 34βE12, it has been proposed that a relationship exists between the basal cells of the normal prostate gland, BCH, adenoid basal cell tumor, and adenoid cystic-like carcinoma, representing a continuum in the progression of hyperplasia to benign and then malignant neoplasia.
McKenney and colleagues reported cases with basaloid proliferations of the prostate showing morphologic patterns other than usual BCH. Most florid basaloid proliferations of the prostate fall into one of two categories. In the first, there is a clear association with nodular hyperplasia (adenoid cystic-like hyperplasia) and, although cytologic atypia and mitoses may be seen, they are present within a lesion that retains an orderly, vaguely nodular (noninfiltrative) pattern. The second group of cases (adenoid cystic-like carcinoma and basaloid carcinoma) shows a widespread, haphazard infiltrative growth pattern. Adenoid cystic-like carcinomas are biologically indolent after prostatectomy but have a low risk of distant metastasis. A lesion that may share similar features of basal cell proliferative lesion, called cylindroma of the prostate, has been reported and is characterized by a jigsaw-puzzle arrangement and heavy accumulation of basement membrane material around and within individual tumor islands, which is very uncommon in basaloid proliferations of the prostate. In the 2016 WHO classification, these tumors are described under basal cell carcinoma of the prostate with adenoid cystic-like carcinoma as a synonym.
Bishop et al recently studied MYB translocation of 12 prostate basal cell carcinomas, which is a characteristic molecular change in classic salivary adenoid cystic carcinomas. In these 12 cases, 7 cases exhibited a cribriform pattern reminiscent of salivary adenoid cystic carcinoma; 4 cases were purely solid and 1 a mixed pattern. MYB-NFIB fusion was detected in 2 of 7 (29%) adenoid cystic-like carcinomas but in none (0%) of the 5 prostate basal cell carcinomas with a prominent solid pattern. It appears that prostatic basal cell carcinoma may include separate entities: true adenoid cystic carcinoma with MYB translocation and basal cell carcinoma.
Clear cell cribriform hyperplasia (CCCH) of the prostate is a rare form of BPH. It was recognized by the WHO in 1980, and Gleason described it as florid benign papillary-cribriform hyperplasia of prostate in 1985. Similar to BPH, CCCH often has a nodular growth pattern and is characterized by a complex papillary–cribriform hyperplasia of clear cells involving acini. In most of the cases, a gradual transition from the usual type of benign hyperplasia to CCCH can be demonstrated. Its appropriate recognition as a benign condition is important to avoid confusion with malignant or premalignant lesions of the prostate, especially those with a cribriform–papillary pattern. The clinical features of CCCH are essentially those of BPH. Patients’ ages range from 58 to 88 years (mean 68 years). The clinical diagnosis is almost always BPH with a presenting symptom of urinary obstruction; the diagnosis is made during examination of TURP or prostatectomy specimens.
On histologic examination, CCCH may be focal or florid, involving part of one or several prostatic chips. On low-power examination, it has a nodular growth pattern. The acinar unit in CCCH is distended by a proliferation of bland-looking clear cells that have a papillary and cribriform arrangement ( Fig. 14A.81 ). A distinct basal cell layer occurs at the periphery of the dilated acini and consists of flat cells in close contact with the basement membrane. The proliferating cells are round, cuboidal to tall columnar, with a varying but usually abundant amount of cytoplasm that is most often clear ( Fig. 14A.82 ). They have uniform, small, round to oval nuclei, which are usually the same size as the nuclei of epithelial cells in adjacent hyperplastic acini. The nuclei at the periphery are slightly larger than those at the center of the proliferation, suggesting cellular maturation toward the center. Nucleoli are rarely seen, but, if present, they are small and inconspicuous. Mitotic figures are absent. The cribriform spaces are devoid of acid mucins or crystalloids. Scant glycogen may be present, but mucin typically is not found in the cytoplasm. The surrounding stroma of CCCH lacks desmoplasia. CCCH can be present as an isolated lesion or associated with BCH. Immunohistochemical stains demonstrate that the clear cells of CCCH show strong immunoreactivity for PSA and PAP. HMWCK (34βE12), CK5/6, and p63 stain the basal cell layer in a continuous fashion at the periphery in a majority of the cases ( Fig. 14A.83 ), although the staining can occasionally be focal.
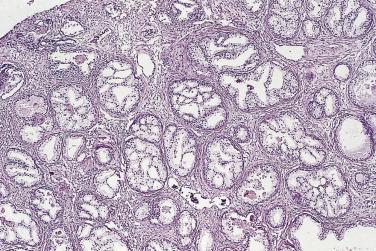
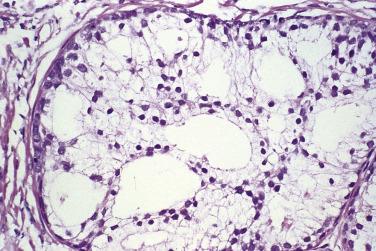
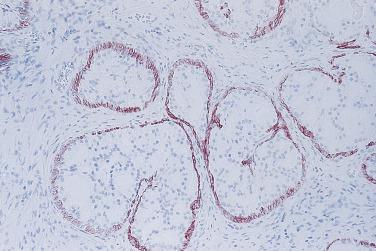
Cribriform adenocarcinoma, intraductal carcinoma, and high-grade PIN are the main lesions in the differential diagnosis of CCCH, but these lesions display malignant cytologic features. The key to the diagnosis of CCCH is the combination of bland cytologic features and architectural uniformity of the nodular growth pattern. Unlike that of carcinoma, the presence of the basal cell layer in CCCH is easily recognized by routine H&E staining, but may be facilitated by HMWCK, CK5/6, and p63 immunostains. Cribriform adenocarcinoma, on the other hand, is characterized by a diffuse growth pattern, nucleomegaly, prominent nucleoli, and occasional necrosis. Intraductal carcinoma and PIN show intraluminal papillary projections of secretory cells and have a cribriform pattern. High-grade PIN usually shows a spectrum of architectural patterns, including micropapillary, tufting, cribriform, and flat, and on high-power examination has nucleomegaly and prominent nucleoli. Intraductal carcinoma is almost always associated with a high-grade acinar carcinoma nearby. DNA flow cytometry also may be helpful to separate CCCH from cribriform carcinoma, intraductal carcinoma, and high-grade PIN, although realistically this is not necessary. CCCH invariably shows diploid DNA content, whereas cribriform carcinoma, intraductal carcinoma, and high-grade PIN may show aneuploid DNA content.
Sclerosing adenosis of the prostate has been reported under several different names, including adenomatoid tumor, fibroepithelial nodule, or pseudoadenomatoid tumor . Chen and Schiff first reported this lesion, highlighting the morphologic resemblance to adenomatoid tumor, and named it adenomatoid prostatic tumor . Hulman reported a similar lesion of the prostate and compared it with typical adenomatoid tumors, using histochemical and immunohistochemical methods; he concluded that the lesion was of prostatic origin and proposed the term pseudoadenomatoid tumor . The term sclerosing adenosis was coined by Young and Clement on the basis of the histologic resemblance to that of the breast.
It is importance to recognize sclerosing adenosis because of its remarkable resemblance to adenocarcinoma on histologic examination. Normal prostatic basal cells show no myoepithelial differentiation, as has been demonstrated by Srigley. Sakamoto and colleagues reviewed sections of the prostate from 263 patients and found 5 cases of sclerosing adenosis, with an incidence of 1.9%.
Sclerosing adenosis is not a premalignant condition. The lesion is usually small, ranging from 1.5 to 11 mm (mean 4.2 mm). It is seen in TURP and prostatectomy specimens (rarely in prostatic biopsies) and is localized to the transition zone, as are other proliferative benign lesions. Sclerosing adenosis is for the most part well circumscribed, but it is nonencapsulated. In certain cases, focal infiltration can be observed at the periphery.
On microscopic examination, sclerosing adenosis is characterized by an ill-defined nodule containing irregular, small acini and nests or clusters of cells embedded in a cellular stroma ( Fig. 14A.84 ). Some of these glands are slightly dilated, whereas others have a microacinar pattern. Assessment of the nature of the lining cells is frequently difficult. The basal cells are for the most part obscured or compressed, but are present in all cases, at least in some glands ( Fig. 14A.85 ). The luminal cells are larger and have clear to eosinophilic cytoplasm and basally located nuclei. The basally located cells tend to be flat to cuboidal, and their nuclei are oriented parallel to the basement membrane. One characteristic feature is the presence of intensely eosinophilic, thick basement membrane–like material surrounding some of the glands ( Fig. 14A.86 ). The lumina of the glands may have Alcian blue–positive acid mucin and crystalloids. The stroma is an integral component of this lesion and is typically very cellular, often myxoid, and made up of plump fibroblasts/myofibroblasts and smooth muscle cells.


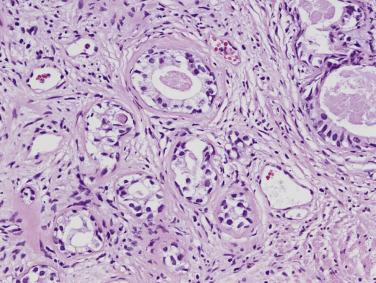
Cells within the glandular component of sclerosing adenosis demonstrate positive reactivity for pancytokeratin, HMWCK (34βE12), CK5/6, and p63, indicating prostatic basal cell origin. In addition, a distinct population of cells reactive for muscle-specific actin and S100 protein is identified within the glandular element and the stroma, suggesting myoepithelial differentiation ( Fig. 14A.87 ). Ultrastructural studies of sclerosing adenosis showed evidence of myoepithelial differentiation and concluded that myoepithelial differentiation is most likely a result of the metaplastic process of the prostatic basal cells. These findings have been confirmed by others. Apparently this is the only prostatic lesion that shows myoepithelial differentiation of basal cells. Cheng and Bostwick reported five cases of atypical sclerosing adenosis with significant cytologic atypia that may be easily mistaken for adenocarcinoma. They compared them with seven cases of classic sclerosing adenosis and found that, except for DNA aneuploidy in atypical form, there were no differences in their immunoprofile and clinical behavior.

Small acinar adenocarcinoma is the main differential diagnosis, particularly from atypical sclerosing adenosis. Unlike acinar adenocarcinoma, sclerosing adenosis is characterized by a proliferation of both glandular and stromal elements. The small, closely packed acini with an infiltrative pattern in sclerosing adenosis may be confused with neoplastic glands, and the proliferating stroma may be misinterpreted as a desmoplastic reaction. Sclerosing adenosis is a lobular proliferation at low power in which the acini typically show variation in size and shape, and at least some of the glands have a clearly visible double cell layer. The presence of basal cells can be highlighted by staining for HMWCK (34βE12), CK5/6, or p63; this is a critical feature for excluding a diagnosis of adenocarcinoma. However, the basal cell staining for these markers may be focal. In contrast to sclerosing adenosis, small acinar adenocarcinoma more often tends to infiltrate into the surrounding prostatic stroma. The nucleoli in carcinoma are usually prominent. The glands and compressed tubules of sclerosing adenosis are often surrounded by a thick hyaline basement membrane–like material (see Fig. 14A.86 ), which is not seen in carcinoma. The stroma in sclerosing adenosis is hypercellular and composed of plump spindle cells, whereas small acinar adenocarcinoma often does not elicit a hyalinized, hypercellular desmoplastic response.
Hyperplasia of mesonephric remnants is a very rare yet important small glandular proliferation within the prostate gland, the severity of the diagnostic pitfall being exemplified by a case misdiagnosed as cancer that resulted in a radical prostatectomy that did not show evidence of cancer. Bostwick and colleagues reported that mesonephric remnants are present in fewer than 1% of transurethral resections and are rarely identified in needle biopsy samples. The acini are lobular but can be infiltrative and may be architecturally mistaken for adenocarcinoma. This cytologically innocuous finding is probably underreported and interpreted as benign prostatic acini, but this is of no apparent clinical consequence. The lesion is almost always discovered incidentally and has no distinctive gross attributes. Microscopically, it is characterized by a lobular proliferation of small tubular structures lined by a single layer of epithelium or by infiltrating glands between muscle bundles and prostatic acini without a stromal desmoplastic response. Variation in the size of the tubules is seen, with occasional cyst formation, intratubular papillary proliferation, and eosinophilic secretions within dilated glands ( Fig. 14A.88 ). The cells are cuboidal with minimal cytoplasm and hence appear atrophic. The nonprostatic lineage of the lesional cells can be confirmed by their negative immunoreactivity for PSA and PAP; the lesional cells themselves may be positive for HMWCK (34βE12). Different from prostatic lineage, GATA3 and PAX8 are typically positive in these lesions. The nuclei are bland and lack prominent nucleoli. Confusion with cancer is compounded by the potential of mesonephric remnants to be associated with ganglia and nerves, simulating perineural invasion.
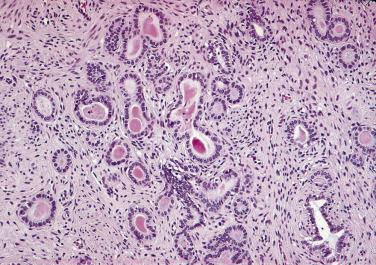
A previously unrecognized type of mesonephric hyperplastic lesion was described by Ayala and colleagues. The lesion consists of tubular structures that are slightly larger than normal ducts lined by cellular proliferation of elongated-fusiform epithelial cells with hyperchromatic nuclei, mimicking high grade PIN ( Fig. 14A.89 ). Morphologically, it may resemble epididymis on cross section. In core biopsies, this lesion can be confused with high-grade PIN, but the proliferating cells are negative for PSA, PAP, p63, or HMWCK.
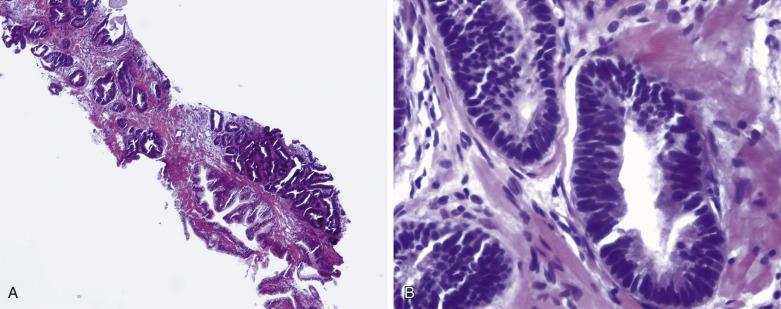
Verumonatum mucosal gland hyperplasia (VMGH) is a small-gland proliferation in the verumontanum and is found in 14% to 29% of radical prostatectomies. Subsequently it has been documented to occur in needle biopsy samples and has the potential to be misdiagnosed as carcinoma. VMGH has an expansile circumscribed growth pattern, with glands of small caliber arranged in a back-to-back fashion. Distinction from cancer is usually not a problem when attention is paid to the cytologic features at higher power. The glands show a basal cell layer and contain small, thickened, and dark-colored corpora amylacea or orange-brown secretions, and the nuclei lack nucleomegaly or prominent nucleoli ( Fig. 14A.90 ). Besides adenocarcinoma, the differential diagnoses include nephrogenic adenoma, hyperplasia of mesonephric remnants, and AAH. The glands of VMGH are positive for PSA and PAP, and the basal cells stain for HMWCK (34βE12), CK5/6, and p63. The distinction of VMGH from normal verumontanum glands is somewhat arbitrary, and the chief reason for awareness of this process is its potential to be misdiagnosed as adenocarcinoma. It has been reported that there is a significant association between the presence of VMGH and AAH.
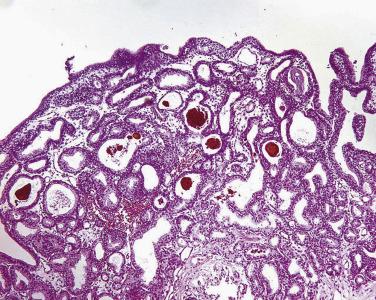
Nephrogenic adenoma is a benign lesion of the entire urinary tract from the renal pelvis to the urethra (see Chapter 12 ); it was first described in 1949 by Davis, who designated it a hamartoma of the urinary bladder, but Friedman and Kuhlenbeck shortly thereafter assigned its present name on the basis of its striking resemblance to the developing renal tubules and its possible neoplastic potential. Although nephrogenic adenoma occurs primarily in the bladder, the urethra is involved in 10% to 15% of cases and, less frequently, the ureter and renal pelvis. In the urethra, nephrogenic adenoma occurs in the bulbous or prostatic portion and may also occur in a urethral diverticulum. Although there is a strong male predominance among adults, there is a 3:1 female predominance of nephrogenic adenoma among children. The patients range in age from 4 to 83 years (mean 41 years). The most common associated conditions include previous surgery or trauma, infection, calculi, or a history of renal transplant. The lesion is usually solitary, but can be multifocal ; it is usually small (0.5–2 cm), but much larger lesions involving even the entire bladder surface have been reported. The lesion is discovered at the time of cystoscopy or is an incidental microscopic finding in about 20% of patients. The signs and symptoms in the remainder of the patients are nonspecific and include hematuria, dysuria, frequency, urgency, and suprapubic and flank pain.
Nephrogenic adenoma may be observed in TURP specimens as urethral mucosa, and suburethral tissue may be sampled during the TURP procedure. It may rarely be seen in prostatic needle biopsy specimens. When nephrogenic adenoma is present in the prostatic urethra and involves suburethral tissue and seemingly infiltrates the prostatic parenchyma proper, it may potentially be confused with prostatic adenocarcinoma because the common histologic manifestation is a proliferation of closely packed small glands.
Grossly, two-thirds of nephrogenic adenomas are papillary; the rest are polypoid or sessile. Microscopically, it is characterized by closely packed small tubular structures (endophytic component) associated with an edematous lamina propria. When present in deep periurethral tissue, it mimics infiltrative adenocarcinoma. The tubules of nephrogenic adenoma show considerable variation in size and shape, sometimes being cystically dilated. They are usually small and round to oval but may be convoluted, resembling collecting ducts or convoluted tubules of the kidney ( Fig. 14A.91 ). Papillary projections may be seen, either on the surface (exophytic component) or within the dilated tubules. The tubules, cysts, and papillae are lined predominantly by cuboidal or columnar cells. The cytoplasm is usually scant, but occasionally a moderate to large amount of eosinophilic or clear cytoplasm may be seen. The nuclei are round and small or pyknotic and usually lack nucleoli, although they may sometimes be conspicuous. In some areas they may have a hobnail appearance ( Fig. 14A.92 ). In rare cases, a diffuse proliferation of solid tubules or small tubules lined by single cells, the nuclei of which are compressed by intracytoplasmic lumina, resembling signet ring cell adenocarcinoma, has been described. Glycogen and mucin can be found in the cytoplasm in some cases. Despite the fact that nephrogenic adenoma may be seen between prostatic ducts, it must be remembered that it is not an invasive lesion. In the prostatic urethra there are submucosal glands, but also prostatic ducts emptying their products into the urethral lumen. Thus nephrogenic adenoma may be in contact with prostatic ducts, giving a false impression of prostatic invasion. Piña-Oviedo et al. have reported that rarely nephrogenic adenoma may have a predominant flat pattern, a major pitfall for mistaking a diagnosis of urothelial carcinoma in situ or invasive carcinoma. In addition, a rare fibromyxoid variant of nephrogenic adenoma has been reported.
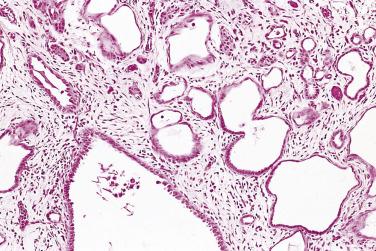

Although metaplastic origin based on the wide age range, site distribution, close association with trauma or inflammation, and evidence of transition from urothelium to nephrogenic adenoma has been entertained, ultrastructural and lectin-binding studies reveal some similarities between embryonic renal tubules and nephrogenic adenoma ; currently the belief is that nephrogenic adenoma represents implantation of renal tubular epithelium in the urothelial mucosa. Mazal and colleagues reported nephrogenic adenoma cases in patients who had renal transplants. All nephrogenic adenomas in 14 female recipients of transplants from male donors and 10 male recipients of transplants from female donors showed the same sex-chromosome status as the donor kidney, but not the same sex-chromosome status as the recipient's surrounding bladder tissue. The presence of aquaporin 1, PAX2, and lectin-binding capacity for peanut agglutinin, Lotus tetragonolobus agglutinin, and Sophora japonica agglutinin in nephrogenic adenomas indicates origin from renal tubular cells and not metaplastic proliferation of the recipient's bladder urothelium.
Some investigators have suggested that nephrogenic adenoma is a precursor or a benign counterpart of clear cell carcinoma of the genitourinary tract. Although a few cases of nephrogenic adenoma coexisting with a clear cell adenocarcinoma have been reported, a direct relationship between the two has not been proved. Nephrogenic adenoma can recur after conservative therapy, but in no case is it associated with unequivocal malignant change. The current consensus is that it is not a premalignant lesion.
The main differential diagnoses of nephrogenic adenoma when it involves the prostatic urethra include prostatic adenocarcinoma and clear cell adenocarcinoma of the urethra. The differentiation of nephrogenic adenoma from clear cell adenocarcinoma is generally not difficult, because the latter shows more diffuse growth and more appreciable cytologic atypia; occurrence of clear cell carcinoma in the male urethra is also very rare, with only three reported cases ( Fig. 14A.93 ). The small, closely packed tubules of nephrogenic adenoma may closely mimic prostate adenocarcinoma, given the presence of tubules, cords, and signet ring–like tubules, prominent nucleoli, underlying fibromuscular involvement, blue-tinged mucinous secretions, focal PSA and PAP positivity, and negative staining in some cases for HMWCK (34βE12). However, features useful in the diagnosis of nephrogenic adenoma include (1) the usual lack of nuclear atypia or nucleoli; (2) variation in tubular size; (3) the typical edematous or inflammatory stroma; (4) distinctive nephrogenic patterns, such as papillary and vascular; (5) adjacent urothelium; (6) thyroidization; (7) peritubular sheaths; (8) negativity for PAP and PSA; and (9) positivity for CK7, PAX8, and HMWCK (34βE12) in some difficult cases. Intracytoplasmic mucin positivity suggests nephrogenic adenoma, because the mucin in prostatic adenocarcinoma tends to be intraluminal.
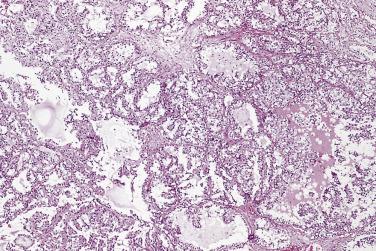
AMACR demonstrates strong cytoplasmic positivity in nephrogenic adenoma, indicating that it is not only a morphologic mimic of prostate adenocarcinoma but an immunohistochemical mimic as well. However, a recent study demonstrated that AMACR was not detected in nephrogenic adenoma using a biotin-free detection procedure, unlike adenocarcinoma. Therefore the authors concluded that AMACR can be used as a reliable marker for distinguishing nephrogenic adenoma from prostate cancer. McDaniel et al studied 63 cases of nephrogenic adenomas using PIN4 cocktail (AMACR, p63, and 34βE12) and GATA3. Although heterogenous staining patterns were seen within individual cases, use of the PIN4 cocktail effectively discriminates nephrogenic adenoma from prostate cancer because of the high frequency of coexpression of AMACR and 34βE12 in nephrogenic adenoma. GATA3 was not a useful marker in differentiating between nephrogenic adenoma and urothelial carcinoma. It has been shown that other markers (S100A1, PAX2, PAX8, CK7, and MUC1) are specific (in context) for nephrogenic adenoma but negative in normal prostate gland or adenocarcinoma.
Polypoid lesions containing prostatic tissue are occasionally found in the prostatic urethra, especially in the region of the verumontanum, and rarely in the membranous urethra, ureteric orifices, and trigonal area of the bladder. Patients with these lesions are usually young, in their third or fourth decade of life, and may present with hematospermia or hematuria closely associated with ejaculation. These lesions may be the most common cause of hematuria in young men. They have been termed adenomatous polyp of prostatic urethra, prostatic urethral polyp, papillary adenoma of prostatic urethra, and prostatic caruncle . The pathogenesis is still unclear, although ectopia secondary to developmental anomalies, prolapse of prostatic ducts, hyperplastic processes, mucosal redundancy, metaplasia, and neoplasia have been suggested.
These lesions exhibit papillary and glandular patterns ( Fig. 14A.94 ). They are covered by columnar prostatic epithelium or by a mixture of prostatic and transitional epithelium. Varying numbers of glandular structures are seen within the stroma of these polyps. They are morphologically identical to prostatic glands and typically are lined by two cell layers, composed of tall columnar epithelial cells with basally located nuclei and a surrounding layer of flattened basal cells. Occasionally, corpora amylacea are seen within the lumina. No cellular pleomorphism, necrosis, increased mitoses, or prominent nucleoli are seen ( Fig. 14A.95 ). Immunohistochemical staining for PSA and PAP is strongly positive, confirming the prostatic origin of this lesion. All of these lesions behave in a benign fashion, and they are not considered preneoplastic lesions. The main differential diagnosis includes ductal adenocarcinoma of the prostate. In contrast to the prostatic polyps, ductal adenocarcinoma usually exhibits nuclear anaplasia, prominent nucleoli, mitoses, and tumor necrosis.

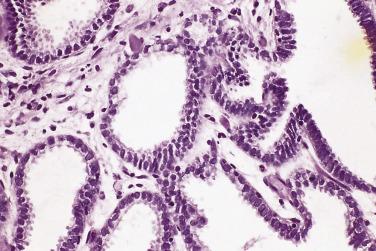
Tissue fragments derived from seminal vesicles and ejaculatory ducts are occasionally observed during examination of TURP or prostatic needle biopsy specimens, with a reported frequency of 3% to 23% in two studies. The histologic features of the seminal vesicle and ejaculatory duct are quite similar, and sometimes it is difficult to tell them apart in needle biopsy or TURP specimens. Both have tubuloalveolar glands as well as closely packed small acini, which are lined by pseudostratified, tall columnar secretory cells and a layer of small basal cells. When these structures are found in biopsy samples, most likely from the central zone, they are usually ejaculatory ducts rather than seminal vesicles. Unless the seminal vesicle is specifically targeted for a staging biopsy under ultrasound, it is too deeply seated to be readily sampled by either random needle biopsy or TURP procedures. Epithelium of the ejaculatory duct and seminal vesicle characteristically contains coarsely granular, yellow-brown lipofuscin pigment. It must be remembered, however, that prostatic adenocarcinoma also rarely contains intracytoplasmic lipofuscin pigment. The pigment, however, tends to be finer and less refractile than seminal vesicle pigment. The epithelial cells in the seminal vesicle and ejaculatory duct often have scattered large atypical, hyperchromatic nuclei; but nuclear detail is often not discernible because of a smudged chromatin pattern ( Fig. 14A.96 ). Neither nucleoli nor mitoses is readily apparent. Kuo and Gomez demonstrated monstrous (large and highly atypical) epithelial cells in 24 of 32 (75%) seminal vesicles they examined. A study on these monstrous cells, using multiple immunostains, indicated that the monstrous seminal vesicle epithelial cells reflected degenerative changes. Care should be taken not to mistake the monstrous epithelial cells as carcinoma. These structures often show small glandular structures arranged in a back-to-back pattern, and therefore they may be confused with a small acinar carcinoma of the prostate. The presence of monotonous nuclei with prominent nucleoli and the absence of a basal cell layer support a diagnosis of adenocarcinoma. Seminal vesicle secretions are fairly common and are composed of acid mucopolysaccharides. Inspissation of secretions appears to be associated with loss of acidity, presumably resulting in dense platelike secretions and crystallization. Awareness of both the crystalloid morphology in seminal vesicle tissue and the distinguishing features from prostatic crystalloids may be important when interpreting prostate needle biopsy samples, in which seminal vesicle epithelium may be confused for prostate carcinoma because of small acinar morphology with accompanying cytologic atypia and crystalloid morphology.
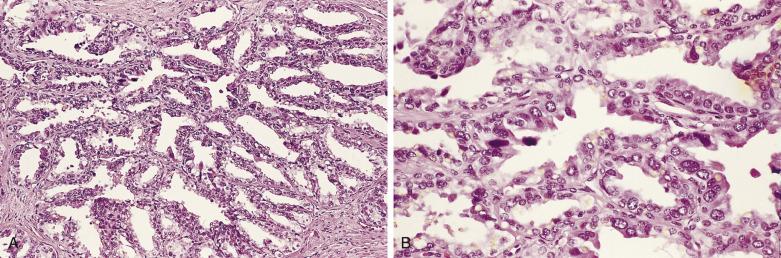
Seminal vesicle and ejaculatory duct epithelium are negative for PSA and PAP but are positive for MUC6, GATA3, and PAX8, which is helpful in the distinction from prostatic adenocarcinoma (MUC6, GATA3, and PAX8 negative). Zhai and colleagues reported that unlike prostatic gland or carcinoma, seminal vesicle epithelium was positive for PAX2 ( Fig. 14A.97 ) or PAX8, which is another useful marker to differentiate between prostate and seminal vesicle and ejaculatory system ( Fig. 14A.98 ).
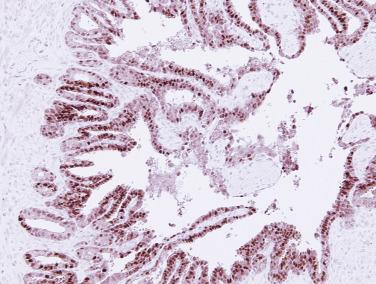
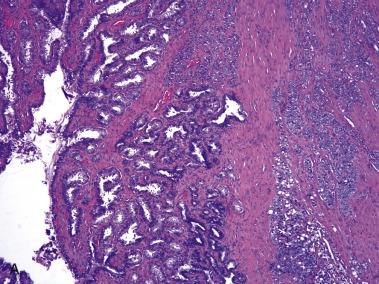
Tissue from bulbourethral Cowper glands may occasionally be present in TURP or needle biopsy specimens from the apex. Cowper glands are paired structures located in the urogenital diaphragm near the apex of the prostate and composed of lobules of mucinous acini in a back-to-back arrangement ( Fig. 14A.99 ). They are associated with excretory ducts and are frequently surrounded by skeletal muscle. Cowper gland may be confused with a well-differentiated adenocarcinoma of the prostate; however, the superficial resemblance of Cowper gland to minor salivary glands, the bland nuclear features, lack of prominent nucleoli, associated skeletal muscle, abundant intracytoplasmic mucin, and negative immunoreactivity for PSA and PAP point to the appropriate diagnosis.
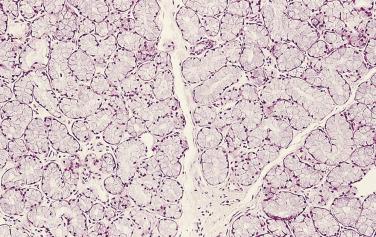
Mucous gland metaplasia of the prostate ( Fig. 14A.100 ) has been described and may resemble Cowper gland. The lesion, however, is located randomly within the prostate; it may be focally present within an acinus or involve groups of acini, but it is usually small.
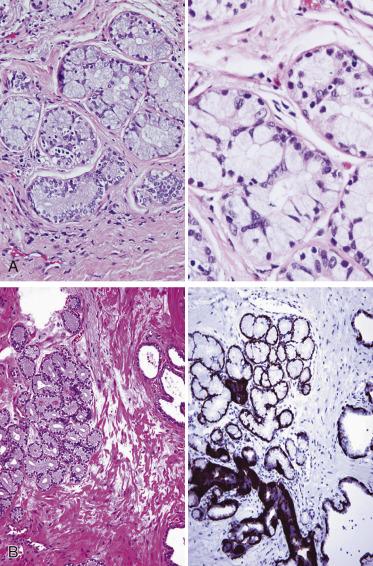
Another normal structure that may be present in prostate specimens and can be confused with carcinoma is paraganglionic tissue. Typically, paraganglionic tissue is present in periprostatic fat adjacent to lateral neurovascular bundles and, rarely, in lateral prostatic stroma. It consists of clusters of clear cells with a sinusoidal vascular pattern and Zellballen appearance, bland oval nuclear features, and clear cytoplasm, which is often abundant, and a close association with nerve fibers ( Fig. 14A.101 ). Occasionally, very large cells with large nuclei are present. If there is any doubt regarding its true nature, negative immunostaining for prostatic (PSA and PAP) and epithelial (keratin and epithelial membrane antigen) markers and positive staining for neuroendocrine markers chromogranin, and synaptophysin should resolve the diagnostic problem. Several cases of paraganglioma involving the prostate have been reported.
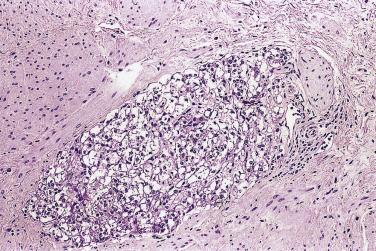
Prostatic atrophy is a common lesion in older men but also has been noted in adults as young as 20 years. Atrophy may coexist with hyperplasia, even within the same microscopic fields. It is usually located in the peripheral zone but may be present in the transition and central zones as well. In the vast majority of cases, the pathogenesis of atrophy is unknown, but it has been postulated to be a physiologic age-related phenomenon. Hormone treatment, inflammation, nutritional deficiency, local ischemia, and compression are thought to play important roles as well. Most patients with atrophy are asymptomatic, and atrophy is often found incidentally during the examination of needle biopsies or TURP specimens performed for BPH or prostate cancer.
On microscopic examination, atrophy may be diffuse or focal. Diffuse atrophy is typically seen in patients with androgen deprivation therapy. In contrast, focal atrophy is often sporadic, is sometimes associated with inflammation, and occurs as heterogeneous patches.
Histologically, atrophy can be classified as simple lobular, cystic, or sclerotic atrophy. Simple lobular atrophy is characterized by a collection of closely packed small acinar glands, usually situated around a central duct, with preservation of normal lobular architecture ( Fig. 14A.102 ). In cystic atrophy, the peripheral acini become cystically dilated ( Fig. 14A.103 ). In sclerotic atrophy, the lobular architecture may or may not be preserved. Atrophic acini are variably separated and compressed by sclerotic stromal elements, resulting in elongated and distorted glands separated by proliferating periacinar fibrocollagenous tissue ( Fig. 14A.104 ). This type of atrophy is usually seen at a later age than simple lobular atrophy. Elastosis of the prostatic stroma is frequently associated with atrophy.

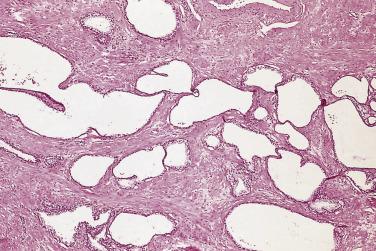
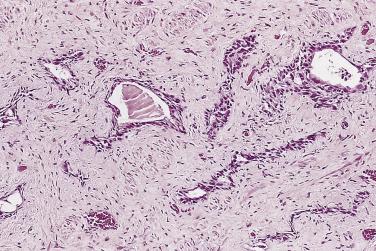
Another term, partial atrophy, is a more recently described subtype of atrophy. It has the architectural pattern of simple atrophy but with distinct cytologic features. In the involved acini, most of the luminal cells contain less cytoplasm than normal acini but more than that of typical atrophy. The cytoplasm is clear in many of the cells in partial atrophy ( Fig. 14A.105 ). In particular, more cytoplasm is present lateral to the nucleus, such that, unlike most simple atrophy and postatrophic hypoplastic lesions, partial atrophy does not seem basophilic at low magnification. Partial atrophy may show moderate nucleolar enlargement, which can lead to diagnostic confusion with adenocarcinoma. It is important to be aware that partial atrophy shares some of the immunohistochemical profile of adenocarcinoma when using triple cocktail stain for basal cell markers and AMACR; therefore recognition of the classic morphology of partial atrophy on routine H&E-stained sections is critical to avoid misdiagnosis of partial atrophy as adenocarcinoma. Herawi and colleagues reported that partial atrophy is among the most common benign changes that cause diagnostic difficulty and prompt consultation. The proliferative activity of partial atrophy is not significantly different from that of benign glands.
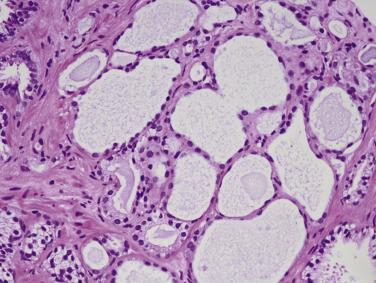
Gardner and Culberson described another pattern of atrophy giving the overall appearance of hypoplasia. They believed that it was not a true atrophy but was actually the postpubertal persistence of glands that retained the unstimulated appearance of neonatal or prepubertal prostate. Regardless of the pattern of atrophy, atrophic glands are lined by hyperchromatic, low cuboidal to flattened cells with a lesser amount of cytoplasm and bland nuclei ( Fig. 14A.106 ). Of particular note is the absence of prominent nucleoli in most cases; when nucleoli are present, they are invariably small and inconspicuous. Double layering of basal and secretory cells is generally present, but because the secretory cells lack abundant cytoplasm they appear similar to basal cells, making it difficult to recognize the two cell types. If any doubt exists, HMWCK (34βE12), CK5/6, or p63 staining specific for basal cells should be performed to confirm the presence of basal cells and thus rule out carcinoma ( Fig. 14A.107 ). Atrophy is sometimes accompanied by inflammation and periglandular fibrosis, and often scattered inflammatory cells are present in the adjacent stroma. When atrophy is seen in TURP or prostatectomy specimens, the diagnosis is usually straightforward; however, in needle biopsy specimens it may be difficult to differentiate it from adenocarcinoma. Sclerotic atrophy may cause the most diagnostic difficulty, especially if the stromal reaction is interpreted as tumor-induced desmoplasia. The most helpful features that point to a benign process are the low-power architecture, double cell layer, and bland cytology. A particularly challenging situation is the separation of atrophy from carcinoma with atrophic features (atrophic carcinoma). Appreciation of an infiltrative pattern and judicious use of immunohistochemistry (HMWCK, CK5/6, or p63) are helpful. Atrophic acini may, on occasion, be associated with secondary proliferative changes of the epithelium; this has been referred to by some researchers as postatrophic hyperplasia .
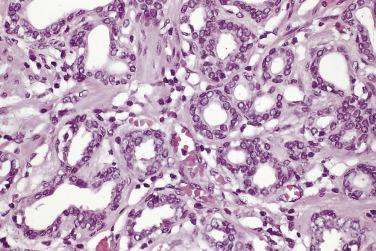
Postatrophic hyperplasia (PAH) may occur in a background of either simple lobular or sclerotic atrophy. Lobular hyperplasia develops from simple lobular atrophy and postsclerotic hyperplasia from sclerotic atrophy. When lobular atrophy undergoes secondary proliferative change, the entire lobule is usually involved, and tightly packed acini bud off from the small atrophic glands. When secondary hyperplasia occurs in a background of sclerotic atrophy, small acini bud off from the atrophic ducts, proliferating and penetrating into the adjacent dense sclerotic stroma.
PAH is a relatively common lesion, most often seen in the peripheral zone of the apical third of the gland. PAH may closely mimic carcinoma in needle biopsy specimens, in which it is seen in 2% to 3% of cases. Because PAH is a combination of two processes, atrophy and hyperplasia, the glands may be small and closely packed with scant cytoplasm, as with conventional atrophy, or may show a neoacinar pattern with proliferating acini with appreciable clear cytoplasm. A clue to the diagnosis is the variation in size and shape of the glands and the cytoplasmic content. Appreciation of bland cytology with no prominent nucleoli, the absence of overtly infiltrative growth or gland fusion, and the presence, at least focally, of a basal cell layer, will help avoid a misdiagnosis of carcinoma. Identification of basal cells with immunostains will help to confirm the benign diagnosis.
The relationship between PAH and prostatic adenocarcinoma has been controversial. Anton and colleagues showed a lack of association between the two lesions in a systematic topographic study. However, Franks and Liavag postulated that PAH associated with sclerotic atrophy might be a forerunner of small acinar adenocarcinoma, based on the observations that these two conditions often occurred together in the peripheral zone and that small acinar proliferation (regeneration) was frequently observed near the atrophy ( Fig. 14A.108 ). In contrast, McNeal reported that adenocarcinoma arises in glands with active epithelium that have never undergone any atrophy. Studies have demonstrated that the frequency of p53 mutation in PAH was similar to that of high-grade PIN, and the proliferative activity defined by MIB1 labeling and gain of chromosome 8 were significantly higher than in BPH or simple atrophy, suggesting a possible association of PAH with prostate adenocarcinoma.

Traditionally it has been stated that there is no skeletal muscle present in the prostate ; thus the finding of prostatic glands interspersed with skeletal muscle bundles in a biopsy or TURP specimen may raise the possibility of an adenocarcinoma extending beyond the prostate. In a normal human prostate gland, the skeletal muscle from the pelvic floor anchors to the prostate. The muscle fibers penetrate the apex anteriorly and laterally, extending upward toward the superior aspect or the base of the prostate, but are not present in the posterior aspect. Skeletal muscle fibers are thus commonly seen from the apex, anteriorly and laterally, up toward the base of the prostate. In these locations the skeletal muscle fibers are not uncommonly intermixed with the normal glandular architecture of the prostate. Therefore prostate samples taken from the anterior or anterolateral areas may have scattered skeletal muscle bundles among prostatic glands. Manley reported the presence of skeletal muscle fibers in 30.2% of prostatic specimens obtained by TURP. This can be a diagnostic problem to those who are not familiar with this normal anatomic finding. Absence of cytologic criteria for carcinoma and the presence of the basal cell layer in the glands are good evidence of benignity ( Fig. 14A.109 ). Furthermore, awareness of the presence of skeletal muscle fibers is also important to correctly stage the tumor. Presence of carcinoma involving striated muscle does not necessarily indicate extraprostatic extension of the tumor.
As indicated earlier, presence of benign prostatic glands around nerves (see Fig. 14A.30 ) has been well documented, emphasizing the potential pitfall of using perineural invasion as a single criterion for the diagnosis of adenocarcinoma of the prostate. McIntire and Franzini reported that benign glands were demonstrated in perineural spaces in 6 of 26 nonneoplastic prostates in autopsy specimens. However, when benign prostatic glands are found in the perineural spaces, the glands usually do not completely surround the nerve or invade it. However, Ali and Epstein reported that benign glands rarely show intraneural and almost complete perineural encirclement. Therefore perineural involvement by benign atrophic glands may cause significant diagnostic difficulty, especially with negative basal cell immunostaining. Careful attention to the H&E-stained morphologic features, comparison with the adjacent and distant benign glands, and evaluation of extent of perineural involvement are necessary.
Carcinomas of the prostate in general often encircle the entire nerve and penetrate it (see Fig. 14A.29 ). Nonneoplastic benign glands do not really invade the perineural space, but they simply appose the nerve, giving the impression of invasion. In addition, the cytologic features of nonneoplastic glands in perineural spaces do not meet the diagnostic criteria for carcinoma. The double cell layer of the benign glands is usually evident, and the cells do not contain prominent nucleoli.
Rectal tissue is often seen in transrectal needle biopsy specimens of the prostate gland. On rare occasions, distorted rectal glands can mimic prostatic adenocarcinoma. According to Schowinsky and Epstein, features that favor distorted rectal tissue rather than prostate tissue or carcinoma were the presence of lamina propria of rectal tissue, detached fragment from prostate tissue, associated inflammation, presence of goblet cells, and muscularis mucosae.
Multilocular cysts or cystadenoma of the prostate is the only bona fide benign epithelial neoplasm of the prostate, with less than 2 dozen cases being reported. It occurs in patients with a wide age range from 20 to 80 years (mean 64.5 years). Patients often present with obstructive voiding symptoms with or without a palpable abdominal mass. The tumor size ranges from 7.5 cm to 45 cm. Imaging studies are useful to determine the extent of the lesion. In all reported cases the diagnosis was made after surgical resection but the anatomic relationship of the mass to the native prostate varied. The masses are well circumscribed and partially adherent to adjacent organs. They are large, bosselated, multilocular, and cystic ( Fig. 14A.110 ). The cysts range from a few millimeters to about 4 cm and contain yellowish-brown or gray, semisolid to inspissated material. Microscopically they are composed of glands and cysts lined by cuboidal to low columnar epithelial cells with basally located nuclei ( Fig. 14A.111 ). Metaplastic squamous and transitional epithelium may be seen. The epithelial cells are positive for PSA and PAP by immunohistochemical staining. Complete surgical excision with preservation of normal pelvic structures is the treatment of choice. A case of cystadenoma of prostate diagnosed on needle biopsy has been reported, and the tumor was positive for AMACR. Because the tumor can have areas of small glands lined by a single cell layer and can be positive for AMACR, cystadenoma can be easily misdiagnosed as prostatic adenocarcinoma. Recently Paner et al described seven cases of cystadenocarcinoma of the prostate and most of them they reported represented a rare underrecognized pattern of prostatic adenocarcinoma, mostly within the histologic spectrum of the ductal variant.

Stromal lesions of the prostate are rare, but the diagnosis is often challenging because they encompass a broad array of benign and malignant processes. A subset of these lesions arises only within the prostate and generally represents entities that originate from the prostate epithelium or stroma, such as sclerosing adenosis (see earlier discussion), sarcomatoid carcinoma (see earlier discussion), stromal tumors of uncertain malignant potential (STUMP), and stromal sarcoma. Another subset of spindle cell tumors that involve the prostate is also found at other sites and includes solitary fibrous tumor, leiomyosarcoma, and neural lesions, among others. Finally, tumors may secondarily involve the prostate yet present as primary prostatic processes, such as gastrointestinal stromal tumors (GISTs). The utility of ancillary studies, including immunohistochemistry, is often limited, and the diagnosis is made based mainly on the morphologic findings on H&E section.
The most common stromal lesion of the prostate is benign stromal hyperplasia, which is easily recognized as it is usually associated with areas of glandular hyperplasia and seen in TURP specimens removed for clinical BPH. Other stromal lesions occur much more rarely in the prostate and include blue nevus, leiomyoma, and phyllodes-type tumor.
Two stromal lesions of the prostate, postoperative spindle cell nodule and inflammatory myofibroblastic tumor (pseudosarcomatous fibromyxoid tumor), have been increasingly recognized. These lesions may mimic sarcoma because of their high cellularity, cellular pleomorphism, and relatively high mitotic activity and may be erroneously diagnosed as such. Proper recognition is critical as their clinical behavior and management differ greatly.
Stromal hyperplasia (stromal nodule) occurs most often in the setting of BPH. It is usually microscopic in size and is seldom, if ever, confused with a malignant spindle cell lesion but may be confused with a leiomyoma. Stromal hyperplasia is characterized by a bland spindle cell proliferation devoid of glandular elements. The lesion is well demarcated from the surrounding prostatic tissue but is not encapsulated. The spindle cells are arranged in a fascicular or whorled pattern simulating leiomyoma. They frequently contain small, thick-walled blood vessels surrounded by the spindle cell component ( Fig. 14A.112 ). Myxoid change in the stroma can be seen and may be prominent, but nuclear atypia is minimal, and mitoses are infrequent. The distinction between a stromal nodule and a leiomyoma is somewhat arbitrary; leiomyomata are usually larger than 1 cm and often encapsulated. Park et al investigated histologic feature and immunoprofile of 149 stromal nodules of prostate, including solitary fibrous tumors (SFTs) and GISTs. They classified stromal nodules into four subtypes: (1) immature mesenchymal (4.7%), (2) fibroblastic (49.7%), (3) fibromuscular (35.6%), and (4) smooth muscular (10.1%). There were linear trends of the expression of growth factors such as VEGF, IGF1, FGF, and TGFß but only CD44 stem cell marker and androgen receptor as maturation progressed from immature mesenchymal to smooth muscular type. CD34 was positive in 55% of the stromal nodules. S100, c-KIT, and estrogen receptor were not expressed in any type of stromal nodule. These results suggested a possible link of SFT with prostatic stromal nodule, but the cells comprising the prostatic stromal nodules do not seem to be associated with GISTs.
Atypical stromal hyperplasia is defined by smooth muscle proliferations composed of large atypical cells with hyperchromatic smudged nuclei. Two types of atypical stromal hyperplasia have been described. One type is characterized by proliferation of atypical stromal cells infiltrating between normal prostate glandular structures; the other type is proliferative change forming a single nodule. The former type is known as atypical stromal proliferation, and the latter is known as atypical (bizarre or symplastic) leiomyoma. These lesions, whether they form a nodule or infiltrate between the normal glands, are characterized by large bizarre cells haphazardly arranged in a background of a stromal leiomyomatous proliferation ( Fig. 14A.113 ). The bizarre cells vary in size and shape, have hyperchromatic nuclei, and often exhibit multinucleate forms; mitoses are rare or absent, and necrosis is absent.
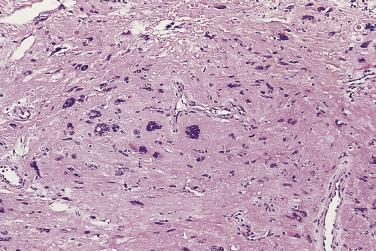
The reported patients’ ages ranged from 49 to 69 years (median 53 years). All patients presented with obstructive symptoms caused by prostatic enlargement, and treatment consisted of either TURP or suprapubic prostatectomy. Although the lesions may rarely recur after surgery, patients usually do not die of this disease.
If a symplastic lesion is found in a needle biopsy specimen, the diagnosis of STUMP should be made. The main differential diagnoses include stromal sarcomas and sarcomatoid carcinoma. Stromal sarcomas usually show high mitotic activity, tumor necrosis, and cellular pleomorphism. Similarly, the sarcomatoid component in sarcomatoid carcinoma is typically pleomorphic and mitotically active. The most important clue for the diagnosis is the presence of the associated carcinomatous component.
Leiomyoma of prostate is defined as a solitary, encapsulated mass of smooth muscle proliferation measuring at least 1 cm in diameter. Prostatic leiomyoma has not been universally accepted as a distinctive entity. Some researchers regard the majority, if not all, of the reported cases of leiomyoma to be stromal hyperplastic nodules. Fewer than 70 cases of solitary leiomyoma of the prostate have been reported. Most cases occurred in men older than 48 years, but a few patients were younger than 40. The tumors in relatively younger patients can be considered true neoplastic lesions, rather than hyperplastic, but there is no histologic criterion that defines the lesion as being neoplastic. The tumors can be quite large, up to 12 cm, and have a firm, whorled, white cut surface. Prostatic leiomyoma is histologically identical to leiomyoma occurring at other sites and is composed of eosinophilic spindle cells separated by variable amounts of collagen. The tumor cells are arranged in an orderly pattern of intersecting fascicles. The individual cells have blunt-ended nuclei with evenly distributed nuclear chromatin.
There are no reliable histologic criteria to distinguish a leiomyoma from a leiomyosarcoma in the prostate. However, infiltrative growth, hypercellularity, nuclear atypia, tumor necrosis, and increased mitotic activity are considered to be important, and a diagnosis of leiomyosarcoma can be made if two or more of these features are present in a tumor.
Pigmented lesions in the prostate are rare and include blue nevus and melanosis. In the prostate, the term blue nevus has been used when melanin is confined to the melanocytes in the stroma ( Fig. 14A.114 ), and the term melanosis has been variably applied for a grossly apparent lesion or microscopically conspicuous melanin pigment found both in the glandular epithelium and within melanocytes and histiocytes in the stroma ( Fig. 14A.115 ). Approximately 50 cases of prostatic blue nevus or melanosis have been reported since the first report by Nigogosyan and colleagues in 1963. About two-thirds of the pigmented lesions are blue nevi, and the rest are melanosis. A few cases of melanosis associated with prostatic adenocarcinomas have been reported.
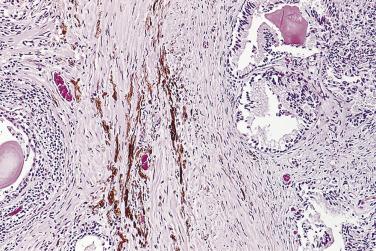

The histogenesis of melanin-containing lesions in the prostate is unknown. Most investigators believe that melanin is produced by migrating melanocytes in the stroma, which is then transferred to the glandular epithelium.
The age of recorded patients with blue nevus or melanosis ranged from 20 to 80 years (mean 68 years) and the finding was usually incidental, the most common clinical presentation being urinary tract obstruction; the usual clinical diagnosis was BPH. Occasionally, malignant melanoma was suspected intraoperatively because of diffuse dark staining of the prostate. Black to brown discoloration was visible in some lesions on gross examination. Microscopically, pigmented spindle cells with long dendritic processes are found scattered in the stroma, adjacent to the prostatic acini. The cytoplasm of the spindle cells contains finely granular brown or black melanin pigment. The pigment is positive for Fontana-Masson and Lillie ferrous iron stains for melanin. On ultrastructural analysis, the spindle cells contain different stages of melanosomes in white men and only mature-type IV melanosomes in black men. So far, only very few primary malignant melanomas of the prostate have been documented.
Lipofuscin pigment in prostatic glandular epithelium has been found in 10% to 100% of prostates by virtue of its positivity for Fontana-Masson, Ziehl-Nielsen, Luxol fast blue, and Oil Red O stains. The chances of finding pigment is greater if the entire gland is examined or if special stains are used for detection. This lipofuscin pigment is present in all zones of the prostate, in normal prostate, and in several pathologic conditions, including high-grade PIN and cancer. The lipofuscin pigment is usually fine, golden-yellow, either intranuclear or occurring throughout the cytoplasm; it may appear golden-brown or blue. It is finer and less refractile than seminal vesicle lipofuscin pigment. The pigment in the prostate is thought to represent wear and tear and aging pigment because of the accumulation of endogenous by-products within the cell. Awareness of lipofuscin pigment in the prostate is critical, particularly in scant biopsy samples. Glands with nuclear atypia and pigment should not be automatically interpreted as seminal vesicle or ejaculatory duct epithelium, and the possibility of PIN or cancer should be considered as well.
Prostatic lesions with morphology similar to fibroadenomas of the breast are encountered infrequently. Kafandaris and Polyzonis reported a dozen cases of prostatic lesions characterized by stromal and glandular hyperplasia without cytologic atypia, reminiscent of pericanalicular and intracanalicular fibroadenoma of breast. Cox and Dawson reported a prostatic lesion characterized by hypercellular fibromuscular stroma without atypia, surrounding irregularly shaped hyperplastic glands they designated cystadenoma–leiomyofibroma (giant fibroadenoma). Lesions with increased cellularity and nuclear atypia of the stroma have been referred to as phyllodes-type atypical prostatic hyperplasia or cystosarcoma phyllodes of the prostate, because they are considered analogous to phyllodes tumors of the breast. Less than 100 well-documented cases have been reported in the literature. These tumors occurred in adults between the ages of 22 and 78 years (mean 52 years) and generally caused urinary retention as a result of prostatic enlargement, hematuria, and dysuria; most of the patients were clinically diagnosed as having BPH. These lesions may grow to be up to 58 cm in greatest dimension.
The phyllodes-type tumor is histologically reminiscent of cellular fibroadenoma and phyllodes tumor of the breast. Because the phyllodes architecture may not always be present, some authors have preferred to designate these tumors mixed epithelial–stromal tumor of the prostate . A biphasic growth pattern occurs with epithelial and stromal cell elements displaying a varying degree of stromal cellularity and cytologic atypia ( Fig. 14A.116 ). The epithelial cells are cuboidal to columnar and have a well-formed basal cell layer; they are arranged in glands, slitlike spaces, or cysts, which are frequently compressed and distorted by hyperplastic stroma. Exuberant epithelial proliferation with leaflike papillary projections may also be seen. The stromal cells are spindle shaped, often in a myxoid matrix, and are more striking than the glandular component. Focal pure stromal hyperplasia devoid of glands is occasionally observed. The atypical stromal cells are present predominantly around the glands. Heterologous rhabdomyoblastic differentiation has been noted occasionally.
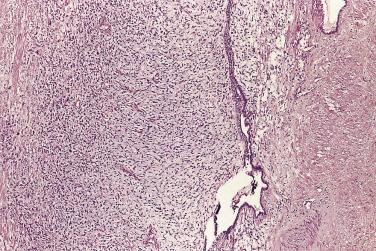
Bostwick and colleagues studied a large series of phyllodes tumors to define the relationship between histologic features and clinical outcome. Based on the cellularity, cytologic atypia, mitotic activity, the stroma to epithelium ratio, and necrosis, the lesions were classified into low-grade, intermediate-grade, and high-grade phyllodes tumors. Local recurrence was more frequent in intermediate-grade and high-grade tumors than low-grade tumor. Metastasis was rare in both low-grade and intermediate-grade tumors. Subsequently, a number of other studies have attempted to identify features associated with malignant behavior. Infiltrative growth, hypercellularity, mitotic figures in stromal cells, nuclear pleomorphism, stromal overgrowth, and tumor necrosis are found to be important factors in determining the malignant behavior. Rarely, rhabdomyoblastic or leiomyosarcomatous differentiation is seen in some malignant tumors. The qualifier “tumor of uncertain malignant potential” may be used for lesions in which histologic malignancy is not clearly evident. The malignant tumors tend to grow rapidly, recur after initial therapy, invade into the adjacent organs, or metastasize to distant organs, such as pelvic lymph nodes, lung, and liver ; at least two patients died of disease.
The distinction of benign phyllodes-type tumors from malignant phyllodes tumor and prostatic sarcomas can be achieved by their lack of mitotic figures in stromal cells, lack of stromal overgrowth, and absence of tumor necrosis. Sarcomatoid carcinoma of the prostate should also be considered in the differential diagnosis because of its biphasic nature. Although the spindle stromal component of sarcomatoid carcinoma may mimic phyllodes tumor, the glandular component in sarcomatoid carcinoma is clearly malignant.
All reported cases of phyllodes-type tumors have been treated surgically ; most investigators advocate surgical treatment similar to that for nodular hyperplasia. Chemotherapy and radiation therapy have been tried, and partial remission could be obtained.
A rare case of phyllodes tumor arising from the verumontanum area has been described.
Postoperative spindle cell nodule is a reactive fibroproliferative process that may resemble sarcoma histologically. Proppe and colleagues first described eight patients with postoperative spindle cell lesions in the genitourinary tract in 1984. All of these lesions had initially been interpreted as either sarcomas or suspicious for sarcoma, but follow-up indicated a benign course. The lesions occurred in the urinary bladder, prostate, or prostatic urethra; and all happened after a cystoscopic procedure for other diseases, such as BPH, prostatic adenocarcinoma, or TCC.
Patient ages ranged from 29 to 79 years (median 60 years). Most were asymptomatic, but hematuria and urinary obstruction could be the presenting symptoms. A postoperative spindle cell nodule may recur locally, necessitating reexcision, but no case of distant metastasis has been reported.
The postoperative spindle cell nodule is usually a small lesion ranging from 0.5 to 0.9 cm in greatest dimension, but it can be large (up to 4 cm in diameter). It is composed of intersecting fascicles of plump spindle cells, interspersed blood vessels, and a slight to moderate number of inflammatory cells. The nuclei of the spindle cells are uniform without atypia or pleomorphism. Mitotic figures range from 1 to 25 mitoses per 10 hpf in the reported cases ( Fig. 14A.117 ). Abnormal mitoses are not found. Nucleoli may be seen, but they are small or inconspicuous. The background may be edematous and focally hyalinized, depending on the interval between surgical manipulation and discovery of the lesion. In general, the margins of the lesion are ill defined and frequently show infiltrative growth, replacing collagenous stroma and destroying surrounding smooth muscle.
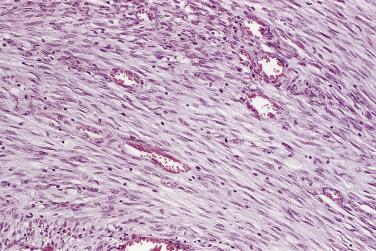
Immunostaining has demonstrated positivity for vimentin, smooth muscle actin (SMA), and desmin for postoperative spindle cell nodules. Interestingly, CK may also be positive in these lesions. On ultrastructural analysis, these spindle cells show fibroblastic or myofibroblastic differentiation.
Several benign and malignant neoplasms may simulate a postoperative spindle cell nodule histologically. The most important differential diagnosis is stromal leiomyosarcoma of the prostate. Unfortunately, mitotic count is not useful in differentiating a sarcoma from postoperative spindle cell nodule, because the latter may have frequent mitoses. Furthermore, leiomyosarcoma may not show significant cytologic atypia. The key feature in making a diagnosis of postoperative spindle cell nodule is the clinical history of surgery or prior instrumentation.
Other benign spindle cell lesions may be confused with a postoperative spindle cell nodule as well, such as inflammatory myofibroblastic tumor (pseudosarcomatous fibromyxoid tumor) and stromal nodule. Unlike postoperative spindle cell nodules, these lesions arise de novo without a history of previous surgery or instrumentation.
The differential diagnosis also includes malignant spindle cell tumors other than leiomyosarcoma: prostatic stromal sarcomas, rhabdomyosarcoma, malignant peripheral nerve sheath tumor, and fibrosarcoma. Undifferentiated pleomorphic sarcoma also has been described in the prostate. These sarcomas are markedly pleomorphic, and their differentiation from postoperative spindle cell nodule is usually not a problem. Another important differential diagnosis is sarcomatoid carcinoma. The spindle cell component of sarcomatoid carcinoma, however, is typically more pleomorphic, contains atypical mitoses, and may stain positively with CK in some cases and rarely with PSA and PAP.
Close communication with the clinician is essential to diagnose postoperative spindle cell nodule and avoid an erroneous diagnosis of sarcoma and unnecessary radical treatment.
An inflammatory myofibroblastic tumor similar to postoperative spindle cell nodule may occur without a history of surgical manipulation in the genitourinary tract. This lesion has been designated a variety of names, such as inflammatory pseudotumor, pseudosarcomatous fibromyxoid tumor, pseudosarcoma, and visceral form of nodular fasccitis. It is rare and poses considerable difficulty in diagnosis. It may occur anywhere along the urinary tract, especially in the urinary bladder (see Chapter 12 ) and prostate gland. Since it was first described by Roth in 1980 in the urinary bladder, approximately 50 cases in the bladder and prostate have been reported in the literature ; the prostate has been the site of involvement in less than 10 of the reported cases.
The histogenesis of inflammatory myofibroblastic tumor is not clear, but the histologic features, including the presence of granulation tissue–type vasculature and a prominent inflammatory cell component, along with a benign clinical course observed in most reported cases, strongly suggest a reactive or pseudoneoplastic process. It is histologically similar to nodular fasciitis, and Nochomovitz and Orenstein suggested that this lesion may represent a visceral form of nodular fasciitis. Subsequent studies have identified that its true nature is a neoplasm with frequent clonal alterations in chromosome 2p23 ( ALK ), with the potential for local aggressiveness, recurrence, malignant transformation, and metastasis.
Histologic examination reveals a myxoid lesion with atypical fibroblastic or myofibroblastic proliferation associated with a prominent inflammatory component and granulation tissue–type vasculature ( Fig. 14A.118 ). It has an ill-defined infiltrative pattern and may extend into the surrounding structures. The spindle cells may appear bizarre, including strap-shaped or tadpole-shaped cells with eosinophilic cytoplasm and prominent nucleoli ( Fig. 14A.119 ). Mitotic activity may be present but is not prominent, and abnormal mitoses are usually absent. f
f References .
By immunohistochemistry, anaplastic lymphoma kinase (ALK), CK, SMA, and desmin may be positive, but S100 and CD34 are usually negative. ALK gene alterations can be detected by FISH in 50% to 60%. Recently immunoglobulin G4 (IgG4)–related sclerosing inflammatory tumor, which is morphologically similar to inflammatory myofibroblastic tumor, has been reported in practically every organ. Presence of a lesion similar to inflammatory myofibroblastic tumor with abundant plasma cells in the lesion should raise the possibility of IgG4-associated sclerosing inflammatory tumor. It is characterized by the following morphologic triad: (1) dense lymphoplasmacytic infiltrate (>50 plasma cells/hpf and IgG4:IgG ratio >40%); (2) fibrosis, at least focally in a storiform pattern; and (3) obliterative phlebitis. IgG4-associated sclerosing inflammatory tumor of prostate has recently been reported.
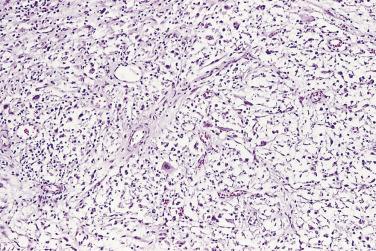
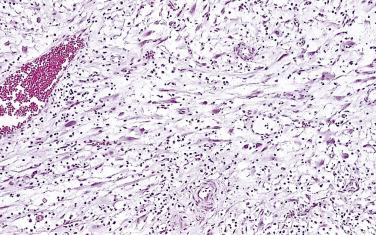
The most important differential diagnoses of myxoid lesions and their clinicopathologic features are summarized in Table 14A.7 . Other malignant neoplasms such as sarcomatoid carcinoma, malignant peripheral nerve sheath tumor, fibrosarcoma, inflammatory fibrosarcoma, and undifferentiated pleomorphic sarcoma also should be considered in the differential diagnoses. However, they usually have more pronounced cytologic atypia and greater mitotic activity, including atypical forms. The importance of recognizing inflammatory myofibroblastic tumors lies in that they are rare, have a more favorable prognosis than sarcoma, and usually do not need radical treatment. So far, metastasis is extremely rare, although it can be locally aggressive with recurrence. g
g References .
No universal agreement exists regarding treatment because only a handful of well-documented cases have been reported, and follow-up is limited. Given this experience, this lesion is classified as a tumor of uncertain malignant potential, and our general recommendation is conservative management with close follow-up.
| Features | PSFMT | PSCN | ML | MR |
|---|---|---|---|---|
| Cellularity | +/++ | +++ | +/++ | +/++ |
| Growth pattern | ND | Fascicles | ND | Cambium |
| Pleomorphism | + | − | −/+ | + |
| Mitoses per 10 hpf | 1-2 | 1-25 | 1->10 | 1->10 |
| Vessels | slit | ND | ND | ND |
| Keratin | −/+ | −/+ | −/+ | −/+ |
| SMA | −/+ | −/+ | + | − |
| Desmin | −/+ | −/+ | + | + |
| Myogenin | − | − | − | + |
| EM | F/MF | F/MF | SM | STM |
| Clinical course | Indolent | Indolent | R/M | R/M |
A few cases of pheochromocytoma, several cases of so-called hemangiopericytoma, solitary fibrous tumor, paraganglioma, granular cell tumor and approximately 30 cases of malakoplakia have been reported in the prostate. Some cases of hemangiopericytomas, in our opinion, may represent stromal sarcomas, and others may be solitary fibrous tumors of the prostate. Their morphologic features are similar to those arising in their more usual sites. Rare cases of prostatic xanthoma, which may mimic clear cell adenocarcinoma, have been described.
Nonspecific granulomatous prostatitis, usually associated with BPH, can clinically and, rarely, pathologically mimic high-grade prostatic carcinoma. Specific granulomatous inflammation may result from infectious agents (histoplasmosis, cryptococcosis, tuberculosis) after TURP ( Fig. 14A.120 ) or bacillus Calmette-Guérin (BCG) treatment for superficial bladder cancer ( Fig. 14A.121 ). Granuloma formation with destruction of ducts and acini and a chronic inflammatory reaction is the typical finding in chronic idiopathic granulomatous prostatitis; however, a lesion with a predominance of histiocytes and a paucity of other inflammatory elements can be confused histologically with a solid, high-grade prostatic carcinoma. Careful histologic evaluation with the aid of immunohistochemical staining (negative staining with CK, PSA, and PAP and positive reaction with CD163 or CD68) should help establish the correct diagnosis.
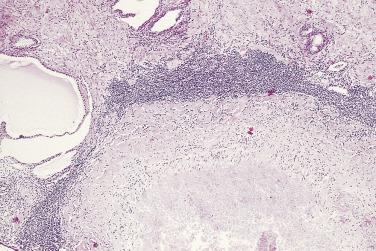

Sarcomas of the prostate are very rare, comprising less than 0.1% of all primary prostatic neoplasms. Most of the sarcomas of the prostate are sarcomas of specialized prostatic stroma, leiomyosarcomas, or rhabdomyosarcomas. Prostate sarcomas have a bimodal age distribution. Rhabdomyosarcoma, the most common sarcoma of the prostate, occurs frequently in infants, children, and young adults, whereas leiomyosarcoma, the second most common sarcoma of the prostate, occurs more commonly in older age groups (>60 years).
Rhabdomyosarcoma (see Chapter 24 ) is the most common sarcoma occurring in the prostate in children. Although at least 18 cases of rhabdomyosarcoma have been reported in adults older than 50 years of age, it typically occurs in patients younger than 20 years old. The genitourinary system, including the prostate, accounts for 21% of rhabdomyosarcomas in children and is the second most common site for rhabdomyosarcoma after the head and neck.
More than 80% of rhabdomyosarcomas of prostate are of the embryonal type (botryoid and spindle cell subtypes), and the remainder are of the alveolar subtype. The alveolar type tends to occur in a somewhat older age group (median 22 years). Pleomorphic rhabdomyosarcoma has also been reported in the prostate but is extremely rare. Recently a case of postradiation embryonal rhabdomyosarcoma has been reported in the prostate of a 20-year-old man with acute lymphatic leukemia who had been exposed to a cumulative radiation dose of 10 Gy across his entire body.
When the tumor is composed predominantly of round cells, immunohistochemistry may be necessary to confirm the diagnosis. Antibodies to muscle antigens, including myogenin, MyoD1, desmin, and muscle actin, are most commonly used in the diagnosis of rhabdomyosarcoma. Cytogenetic analysis may also be helpful (see Chapter 24 ).
Small cell carcinoma of the prostate and lymphoma may be difficult to differentiate from rhabdomyosarcoma on morphologic grounds alone in the adult population. Clinical presentation, as well as immunohistochemical and molecular genetic analysis of the tumor, will usually establish a definitive diagnosis. An inflammatory myofibroblastic tumor (pseudosarcomatous fibromyxoid tumor) is another consideration in differential diagnosis with embryonal rhabdomyosarcoma. It may have strap cells or tadpole-like cells set in a myxoid background, as in embryonal rhabdomyosarcoma; however, inflammatory myofibroblastic tumor does not have the cambium layer or the cells with cross-striations and does not stain with myoglobin, MyoD1, or myogenin.
Bladder-prostate rhabdomyosarcomas are often discovered relatively early because they produce symptoms and signs referable to the urinary tract, which lead to prompt medical attention. Rhabdomyosarcoma may be small and apparently limited to the prostate at the time of diagnosis, but may grow rapidly, invading adjacent soft tissue and bladder. In contrast, extraprostatic pelvic tumors often are large and infiltrative when they are first detected. Therefore rhabdomyosarcoma of the bladder-prostate region is said to have a better prognosis than those of the soft tissue of the retroperitoneum-pelvis outside the prostate. According to the results from Intergroup Rhabdomyosarcoma Study IV, which included 54 rhabdomyosarcomas in the bladder-prostate gland, the factors associated with a favorable outcome are younger age (1–9 years) at diagnosis, negative lymph nodes, and low stage at presentation based on multivariate analysis.
The prognosis of rhabdomyosarcoma in all locations, including the prostate, has improved considerably with contemporary combination therapy, including multidrug chemotherapy, surgery, and radiation, which led to a 5-year disease-free survival rate of 70% and overall survival rate of 75%. Metastases are less frequent since the advent of combination therapy.
Leiomyosarcoma is the most common sarcoma of the prostate in adults It accounts for 25% of prostatic sarcomas. Although most patients are in their fifth to eighth decades (median age 58 years), leiomyosarcoma may rarely affect patients younger than 10 years.
Leiomyosarcoma tends to be a large bulky tumor with a mean size of 9 cm. Its macroscopic findings are similar to those of its soft tissue counterpart. The tumor diffusely infiltrates the prostate and periprostatic soft tissue. Prostatic enlargement is readily detectable, and the clinical diagnosis is usually BPH or prostatitis.
Microscopically, it is similar to leiomyosarcoma of soft tissue or viscera (see Chapter 24 ) and consists of interlacing fascicles of spindle cells with blunt-ended nuclei and fibrillar eosinophilic cytoplasm. Nuclear atypia and mitoses are variable in extent but are always present ( Fig. 14A.122 ). Tumor necrosis and hemorrhage are frequently observed. Immunohistochemistry may be helpful to confirm smooth muscle differentiation. Most tumors are intermediate to high grade. At the low end of atypia there are no established criteria to distinguish leiomyosarcoma from a cellular or symplastic leiomyoma. We suggest making a diagnosis of leiomyosarcoma when the tumor has at least two of the following adverse histologic parameters: hypercellularity, pleomorphism, nuclear atypia, necrosis, infiltrative pattern, and high mitotic activity (>10 mitoses/10 hpf). However, reliable pathologic features in assessing the biologic behavior of smooth muscle tumors of the prostate have not been established because of the rarity of this tumor and insufficient follow-up of the reported cases. Differentiating GIST of the rectum extending into the prostate from leiomyosarcoma of the prostate may be difficult. Immunohistochemical stains for c-kit and DOG1 are helpful in this situation.
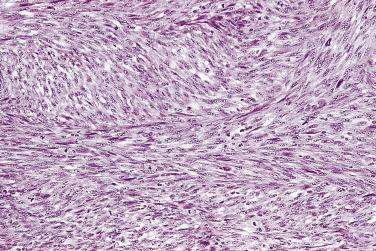
The reported 5-year survival rate for patients with leiomyosarcoma has been 50% to 60% ; however, the prognosis of leiomyosarcoma is encouraging with aggressive combination treatment involving radical surgery, preoperative and postoperative combination chemotherapy, and radiation therapy in selected patients. Ahlering and colleagues reported a 90% survival rate with these regimens. Local recurrence and visceral metastasis complicate half the cases.
Within the relatively rare category of adult soft tissue sarcomas that occur in the prostate, there is a group of sarcomas that are different from leiomyosarcoma and are thought to arise from specialized prostatic stroma, the latter assertion being supported by their immunoreactivity for progesterone and estrogen receptors. Like phyllodes tumors of the prostate, they represent a spectrum. Gaudin and associates proposed the term prostatic stromal proliferation of uncertain malignant potential (STUMP) for lesions defying unequivocal characterization as sarcoma and prostatic stromal sarcoma for tumors with stromal overgrowth, frequent mitoses, and necrosis. Histologically, the tumor is composed of primitive round, oval, or spindle cells, occasionally with a herringbone pattern, diffusely infiltrating the prostatic stroma ( Fig. 14A.123 ). The tumors are positive for CD34 and hormone receptors (progesterone and estrogen receptors) and stain variably for muscle markers (SMA, HHF35, and desmin). Herawi and Epstein reported a large series of specialized stromal tumors of the prostate, including 36 cases of STUMP, 7 cases of STUMP associated with sarcoma (5 low-grade and 2 high-grade sarcomas) either concurrently or subsequently, and 7 cases of stromal sarcoma (3 low-grade and 4 high-grade sarcomas). The histologic subtypes of STUMP did not correlate with the clinical behavior or likelihood of being associated with sarcoma. STUMPs can recur frequently, occur at a young age, often involve the peripheral zone where they can be adherent to the rectum, and can be associated with stromal sarcoma. It is important to recognize that these are neoplasms with unique local morbidity and malignant potential. Low-grade stromal sarcomas can locally invade and usually do not metastasize; high-grade sarcomas can metastasize and lead to death. Kim and colleagues reported a case of high-grade stromal sarcoma with rhabdoid features ( Fig. 14A.124 ); this tumor showed aggressive clinical behavior with short-term survival (7 months after diagnosis).
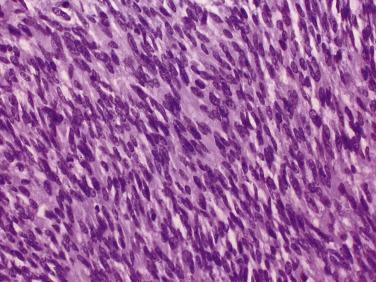

Fibrosarcoma in the prostate has been reported by Tannenbaum, but there is no detailed description of this lesion in the literature. Two cases of osteosarcoma of the prostate have been reported. Both were associated with adenocarcinoma of the prostate; one patient developed osteosarcoma de novo, and the other developed osteosarcoma after radiation therapy for carcinoma. These cases may be sarcomatoid carcinomas rather than primary osteosarcoma of the prostate. We have reported a series of 12 sarcomatoid carcinomas in which one patient had osteosarcoma as a sarcomatoid component.
A few cases of undifferentiated pleomorphic sarcoma of the prostate have been reported. Unlike undifferentiated pleomorphic sarcoma of soft tissue, these tumors displayed only moderate pleomorphism with less frequent mitoses. Although the degree of malignancy is difficult to evaluate from these case reports and the follow-up is relatively short, the prognosis appears to be poor.
One of the least common spindle cell tumors of prostate is solitary fibrous tumor. Herawi and Epstein reported their experience of 13 solitary fibrous tumors identified on either prostate needle biopsy or TURP. Mean patient age was 63 years. Urinary tract symptoms are common. Most were positive for CD34, Bcl-2, and CD99, but negative for c-kit. Approximately half of the cases were considered to be malignant. The most important differential diagnoses are rectal GIST and stromal nodule.
Several cases of angiosarcoma have been described in the literature. The patients’ ages ranged from 2 to 60 years (mean 34.5 years). One case occurred 10 years after radiation therapy for prostatic adenocarcinoma. All tumors were poorly differentiated, and the prognosis was dismal. Immunostaining with vascular markers is useful in differentiating angiosarcoma from other poorly differentiated sarcomas.
Other sarcomas reported in the literature include synovial sarcoma, malignant rhabdoid tumor, chondrosarcoma, malignant peripheral nerve sheath tumor, neuroblastoma, primitive peripheral neuroectodermal tumor, malignant perivascular epithelioid cell tumor, and GIST.
Malignant lymphoma of the prostate, either as a primary tumor or secondary to systemic involvement, is very rare. The incidence and histologic features of malignant lymphomas are not well documented. In one study, malignant lymphoma involving the prostate and pelvic lymph nodes was identified in 0.6% of prostate surgical specimens. Over 150 well-documented cases of prostatic lymphoma have been reported. Patients’ age range from 14 to 86 years (mean 61 years). There are no specific symptoms or signs for lymphoma. Urinary obstructive symptoms, hematuria, or both are the usual presenting symptoms. The clinical diagnosis is almost always BPH, prostatitis, or carcinoma, even in patients with a previous diagnosis of lymphoma.
Lymphomas involving prostate in reported series include the entire spectrum of malignant lymphomas seen in other sites. The predominant types are large B cell, follicular, mixed cell, small lymphocytic, and mantle cell lymphomas. There are only a few reported cases of nodular, T-cell, undifferentiated Burkitt, and non-Burkitt lymphomas. Hodgkin disease, lymphomatoid granulomatosis, and angiotropic lymphoma have also been reported in the prostate.
The criteria for diagnosing primary lymphoma of the prostate proposed by King and Cox include tumor limited to the prostate and adjacent tissues and absence of lymph node involvement. In addition to the previously mentioned criteria, Bostwick and Mann required that the major presenting symptoms be limited to the prostate and that there be a disease-free interval of at least 1 month to allow completion of staging procedures.
The prognosis of lymphoma involving the prostate is generally poor regardless of the patient's age, the tumor stage, type of lymphoma, or treatment regimens used. In a large series of 62 prostatic lymphoma reported by Bostwick and colleagues, 27 patients died of lymphoma. There was no statistical difference in mean survival in patients with primary prostatic lymphoma versus secondary involvement of prostate by lymphoma (9.8 vs 12.7 months).
About 20% of patients with leukemia have evidence of prostatic involvement at autopsy, but fewer than 1% have clinical manifestations of prostatic symptoms as a direct result of the leukemic process. Chronic lymphocytic leukemia is the most common type. A few cases of prostatic granulocytic sarcoma have been reported.
Plasmacytoma is extremely rare in the prostate, and involvement is recognized after the diagnosis of systemic disease is made or at autopsy. In one reported case, the prostate was moderately enlarged and irregular, mimicking a primary prostatic carcinoma. On microscopic examination, sheets of neoplastic plasma cells diffusely infiltrate the prostatic stroma. Rarely mucosa-associated lymphoid tissue lymphoma ( Fig. 14A.125 ) has been reported in the prostate.
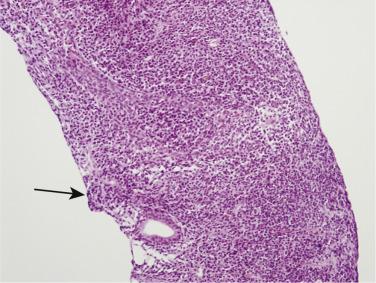
The main differential diagnoses include prostatitis and prostatic carcinoma, particularly small cell carcinoma and rhabdomyosarcoma. Clinical history and a careful microscopic evaluation with ancillary methods, including immunohistochemistry, are essential to differentiate these lesions from lymphoma or leukemia.
A few germ cell tumors involving the prostate have been reported. Some are possibly primary, but most others represent secondary involvement from the retroperitoneum or retrovesical space. The tumors occur in young to middle-aged men. The pathogenesis of the primary germ cell tumor of the prostate is unclear, but sequestration of germ cells, neoplastic transformation of uncommitted stem cells into germ cells, or metastasis from an occult testicular primary are possible explanations. The diagnosis of primary prostatic germ cell tumor can be made only after a testicular primary is excluded. Seminomas and nonseminomatous germ cell tumors, including yolk sac tumor, choriocarcinoma, teratoma, and mixed germ cell tumors, have been reported. The prognosis is favorable in patients with seminoma, but less favorable in those with nonseminomatous germ cell tumors.
Nephroblastoma, also called Wilms tumor, rarely has been described in the prostate as a primary tumor with a classic triphasic pattern. Before making a diagnosis of nephroblastoma, the possibility of teratoma should be excluded. Prostate secondarily involved by neuroblastoma has also been reported. The prognosis depends on the grade, stage, and associated genetic abnormalities.
The prostate gland is infrequently involved by metastatic tumors, if lymphoma, leukemia, and contiguous spread from the bladder and colon cancers are excluded. Johnson and colleagues found the prostate to be involved by metastatic disease in 0.5% of male patients dying of malignant tumors. In a similar study, Zein and associates reported an incidence of 2.2%. Combining these two series, the most common tumors secondarily involving the prostate are bronchogenic carcinoma and malignant melanoma. Other frequent primary sites include the gastrointestinal tract, pancreas, and kidney. Metastatic testicular germ cell tumor and metastases from carcinomas of the thyroid, penis, and larynx have also been reported.
Because of anatomic proximity, bladder and colon carcinomas frequently involve the prostate secondarily. A high-grade prostate carcinoma should be differentiated from a high-grade bladder or colorectal carcinoma. On prostate sampling, when a carcinoma exhibits “dirty” necrosis, tall columnar epithelium with mucin production, mucin-positive signet ring cells, villous architecture, or associated inflammation, a carcinoma of colorectal origin should be considered. Immunohistochemical staining with a panel of tissue-specific or tumor-specific markers, such as β-catenin, CDX2, CEA, HMWCK, PSA, PAP, P501S (prostein), and CK7/20, is helpful in making a definitive diagnosis. In addition, NKX3.1, proPSA (pPSA), and PSMA are also positive markers for prostate cancer. Urothelial markers, including HMWCK, p63, GATA3, uroplakin II and III, thrombomodulin, and S100P, can be used for differentiating high-grade prostate adenocarcinoma from high-grade urothelial carcinoma.
Cysts of the seminal vesicle are uncommon, with fewer than 100 cases reported. These lesions may be underrecognized because there is no specific clinical finding, and imaging of seminal vesicles is difficult to obtain. Cysts of seminal vesicles are frequently associated with ipsilateral renal agenesis. In some cases, postinfectious fibrosis has been suggested as the etiologic factor. Acquired cysts are usually associated with obstruction of the ejaculatory duct because of urinary tract infection, usually ascending from the lumen of prostatic urethra into the seminal vesicles. Compression of the ejaculatory duct by prostatic adenomatous nodules explains the association with BPH. The cyst is usually unilocular and can be hemorrhagic. Unilaterality is the rule, but a few cases are bilateral. Sometimes cysts of the seminal vesicles are associated with an absent testis; a few have been associated with an anomaly of the vas deferens. The differential diagnoses include abscesses, hydatid cysts, solid and cystic tumors of seminal vesicle or prostate, and diverticulum of the ejaculatory duct.
Cysts of the seminal vesicle most commonly occur in the third decade of life (range 18–59 years) and usually are asymptomatic. In symptomatic patients, the initial signs in the majority of cases are lower urinary tract symptoms, such as burning, urgency, and increased frequency of urination. Epididymitis, abdominal pain, and perineal discomfort after ejaculation are not infrequent. Most reported cases have lacked any histologic description. A few reports describe a cyst lined by a uniform layer of columnar to cuboidal epithelium, with no mesenchymal component except for a fibrous wall.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here