Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Dr. Pilitsis is a consultant for Boston Scientific, Nevro, TerSera, and Abbott and receives grant support from Medtronic, Boston Scientific, Abbott, Nevro, TerSera, GE Global Research, SBIR 1R43NS107076-01A1, NIH 2R01CA166379-06 and NIH U44NS115111. She is medical advisor for Aim Medical Robotics and Karuna and has stock equity.
The use of various neuromodulation therapies for the treatment of neurological conditions has rapidly expanded over the course of the last 20 years. Along with the increase of implantable stimulators being used for treatment, the number of device failures has also risen. Despite the large volume of research conducted on the incidence and prevalence of revision surgeries, studies detailing technical methods of how complications are resolved are lacking. Here, we describe cases for troubleshooting neuromodulatory devices.
The loss of therapy after successful treatment from a neuromodulation system can be patient dependent or due to device malfunction [ ]. In order to differentiate, a thorough history and physical is needed to assess whether the loss has been acute or developed over time. In all cases, system impedances and X-rays should be obtained to rule out lead fracture or migration. In delayed cases, other patient factors should be considered, that is, new medical problems, new medications, psychosocial factors. Loss of benefit from intrathecal drug administration devices can be due to catheter (66%) or pump malfunctions (7%) [ ]. Similarly, in SCS and DBS, the source of the problem can be narrowed down to the lead, extension (in DBS), or internal pulse generator (IPG).
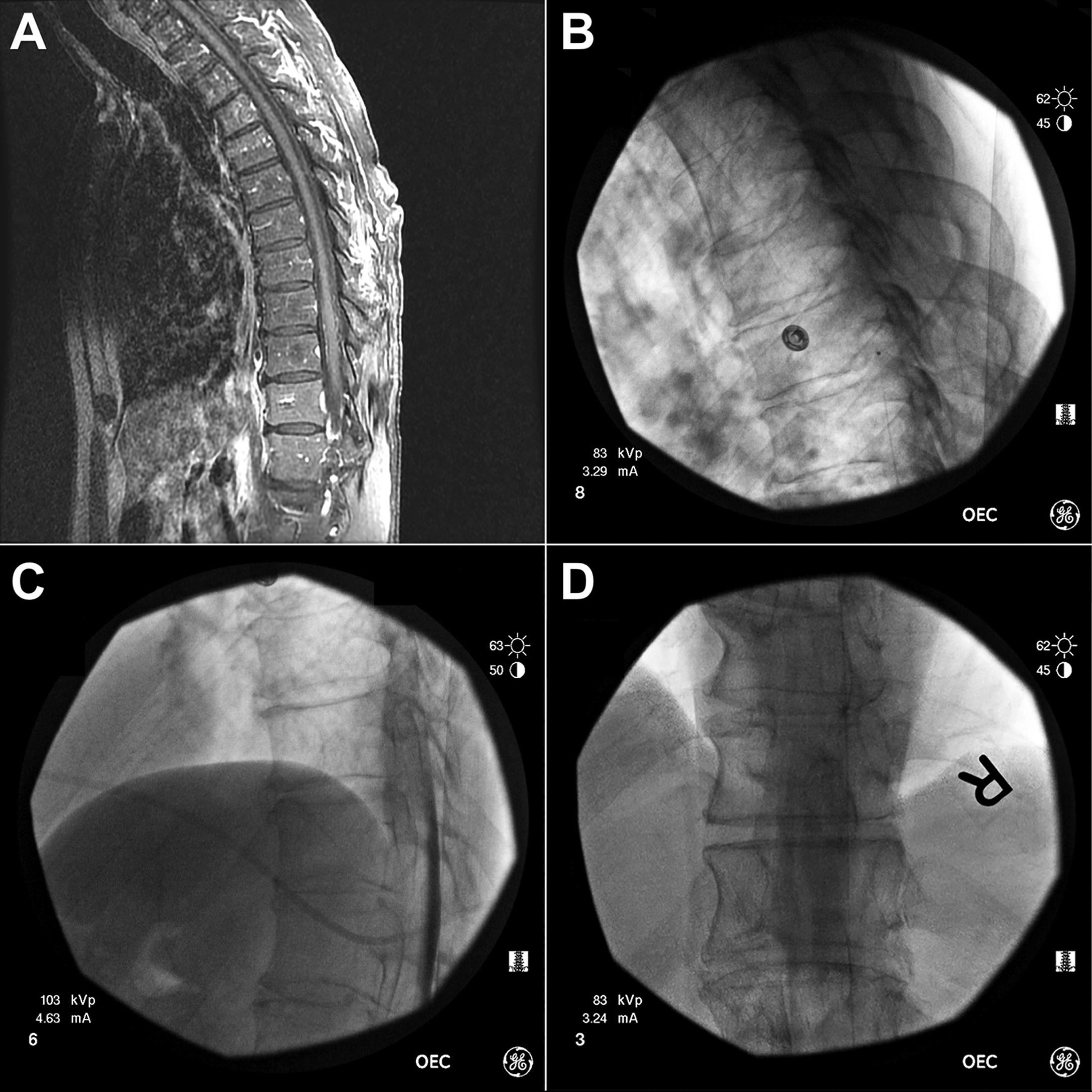
A 61-year-old male with a history of multiple sclerosis had lower extremity pain and spasticity. As increasing doses of oral antispasmodics led to somnolence, he underwent placement of an intrathecal baclofen pump. The patient did well on an intrathecal dose of baclofen of 325 mcg/day until 18 months postoperatively. At that time, he began to report increased lower extremity spasms. After attempts to regain therapeutic efficacy by increasing IT baclofen dose up to 430 mcg/day over a period of a month, X-rays of the system were performed. No abnormality was detected. Then, a dye study was performed showing no flow of baclofen past T8. The decision was made to explore the system. We obtained an MRI with and without gadolinium of his thoracic spine to rule out granuloma ( Fig. 15A.1 ). During surgery, after the pump was exposed, CSF was successfully aspirated through catheter access port. The lumbar catheter was then exposed and spontaneous CSF outflow was observed. In light of clinical and radiographic evidence that the patient was not receiving therapy, the decision was made to replace the entire system. We drew out all the drug in the existing pump and found more was present than expected indicating a pump malfunction. Based on our intraoperative findings, it was unclear how much if any baclofen the patient was receiving. As he had not gone through withdrawal and his spasticity was not as pronounced as it was pre-ITD therapy, our starting point with the new system was IT dosing at 200 mcg/day and continued oral dosing at 120 mg/day, which he was on preexploration. He was noted to have improvement in spasticity immediately postoperatively and monitored in a step-down unit for symptoms of overdose/withdrawal. Over the next few weeks, he was titrated off his oral medication and resumed 325 mcg/day.
A 31-year-old female with primary dystonia underwent bilateral DBS, which led to >80% improvement for 5 years. She then noted new symptoms including severe tingling sensations over her left scalp which precluded use of her system. Further she had worsening of her right wrist symptoms. Multiple reprogramming sessions were attempted to no avail.
We suspected lead fracture and checked impedances in the clinic at 0.7, 1.5, and 3.0 V to assess for an open or short circuit. These were essentially within normal limits with the exception of one pairing which she was not using that was low ( Fig. 15A.2A and B ). We also obtained skull, cervical spine, and chest X-rays to examine for a fracture in the lead, extension, or hardware. No abnormality was found ( Fig. 15A.2C and D ). We obtained a CT scan head with 1.5 mm fine cuts in case intracranial surgery was necessary in the future.
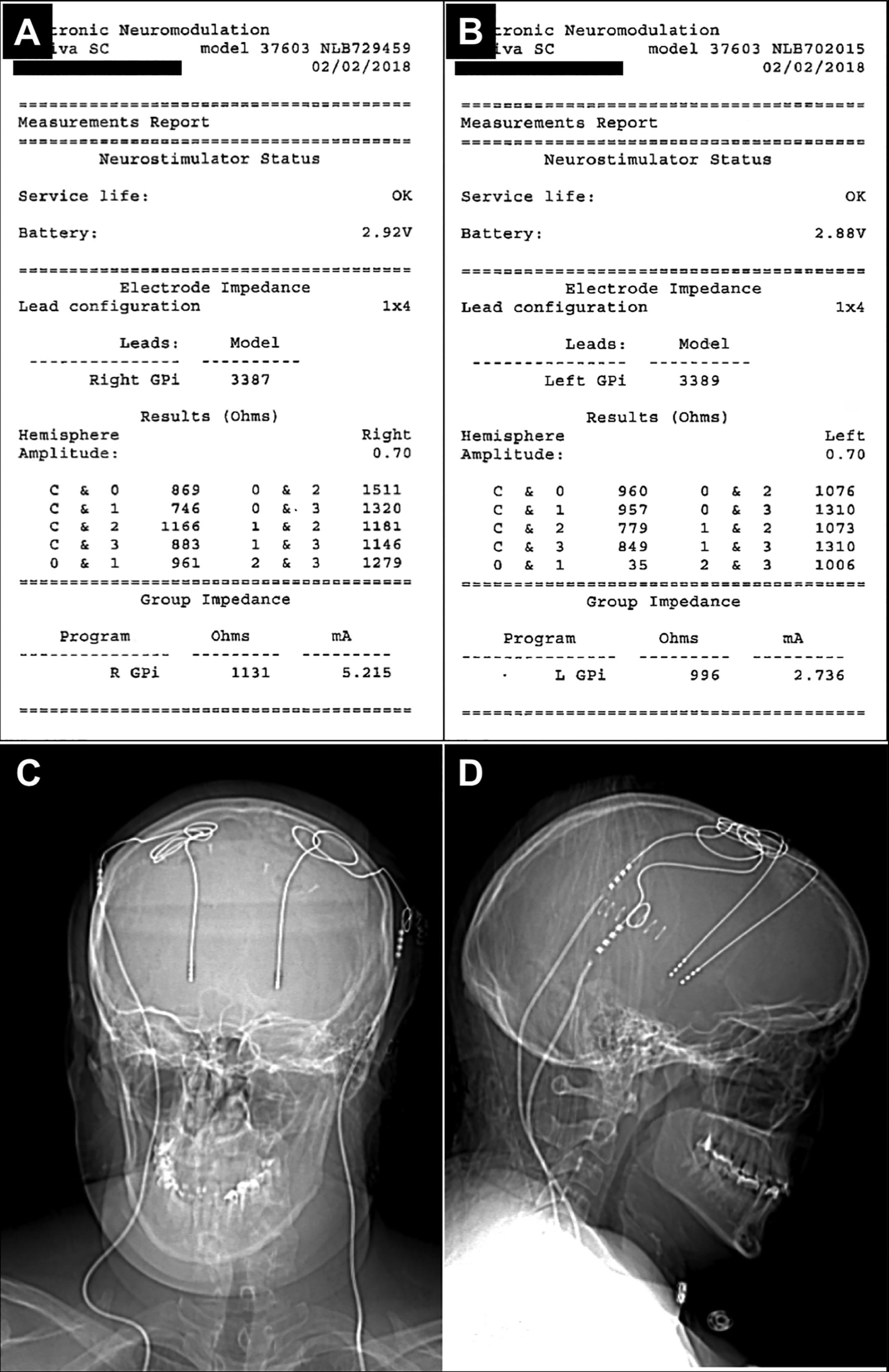
At this point, considering our high level of concern, we took the patient to the OR for exploration of the extension and generator. We first explored the region where the extension lead was secured to the brain lead. Routinely, during our stage 2 procedures, we use a second incision on the temporal region for connecting and securing our extensions. At the time of revision surgery, we opened this incision and first disconnected the brain lead from the extension, examined for fluid or breakage, reconnected, and rechecked impedances, which in this case were stable. We then decided to replace the extension and battery. Interestingly with the new extensions, the impedances were higher, with several over 2000 Ω. At this point, we reconnected the devices with the same result. In other cases, we have tested the brain lead impedances with alligator clips to examine if the problem lies in the lead. Because we were unable to safely maneuver the lead to do this, we did not in this case. This maneuver is helpful to determine the issues but should be performed with caution due to risk of causing further damage to the lead as the clips are difficult to place in a small incision. If the DBS lead itself has high impedances, the placement of a new DBS lead will be performed at a different surgery. Here, as impedances were borderline, we opted for a “watch and see” approach.
In cases where we must resort to an intracranial procedure, that is, the fracture is in the DBS lead, we confirm with the treating neurologist that the patient is receiving effective stimulation but limited therapeutic effects due to adverse effects and that he/she is unable to achieve benefit despite utilizing programming alternatives. Assuming so, we perform these cases under sedation to place the stereotactic frame and obtain a CT. We ensure that the connection site is accessible with the frame in place and include that region in our sterile field. We disconnect the extension lead and make the extension easily accessible to attach to the new lead. Then we dissect out the brain lead from the subcutaneous tissue, taking great care to prevent the lead from migrating inferiorly in the brain. We expose the area where the lead is entering the cortex and obtain a fluoroscopic image prior to removal. We quickly remove the lead to avoid CSF egress and place a “to-target” cannula to avoid a new path from being created affecting final lead position. At this point, we pull back the cannula to expose contacts and perform macrostimulation. If the lead is significantly malpositioned in the x or y axis (>3–4 mm), microelectrode recording may be beneficial. We have not found it beneficial in cases of superior migration or when revisions to the same location are undertaken.
We have also had revision cases where the traditional skull anchor has not been used and the burr hole has been filled with bone cement. In these cases, prior to the removal of the lead, we drill out the periphery of the cement along the burrhole. Once free, we carefully remove the lead and cement en bloc under direct visualization. A similar technique is necessary, albeit with less drilling, with one of the DBS skull anchoring devices currently available.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here