Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Diseases of the thoracic and thoraco-abdominal aorta are less common than infra-renal abdominal aortic aneurysms (AAAs) but their management should be familiar to vascular surgeons, as they often present acutely. This is partly because of the greater utilisation of cross-sectional imaging, which has in turn led to an apparent increase in the incidence of thoracic aortic conditions.
Over the last two decades, endovascular repair has become the first-line treatment for most distal arch, descending and thoraco-abdominal aortic pathologies. Despite the move toward minimally invasive strategies, the increasing use of endovascular repair of the thoracic aorta (thoracic endovascular aortic repair [TEVAR]) has lacked the large randomised trials that underpinned the widespread adoption of standard infra-renal repair (endovascular aortic repair [EVAR]). Registry data, institutional case series and national routine data have suggested that endovascular procedures can be offered with low mortality and morbidity rates in comparison with open repair of thoracic pathology, and appear to be equally as effective in preventing aortic-related death. , Critics of the increase in the repair of thoracic pathology draw attention to the lack of accurate contemporary natural history data for untreated pathology, and also that newer and costly technologies lack data on their long-term durability.
In the case of aortic dissection, there is continuing controversy over the role of TEVAR in uncomplicated acute and chronic dissection and the ability of this procedure to prevent aortic-related death. The advance of fenestrated and branched stent-graft technology has allowed many patients that were previously not fit enough to undergo treatment by highly invasive open surgery of the thoraco-abdominal aorta to be offered complex TEVAR, although open repair is still recommended for those with connective tissue disorders.
This chapter reviews the contemporary treatment of thoracic and thoraco-abdominal aortic disease, excluding the ascending aorta.
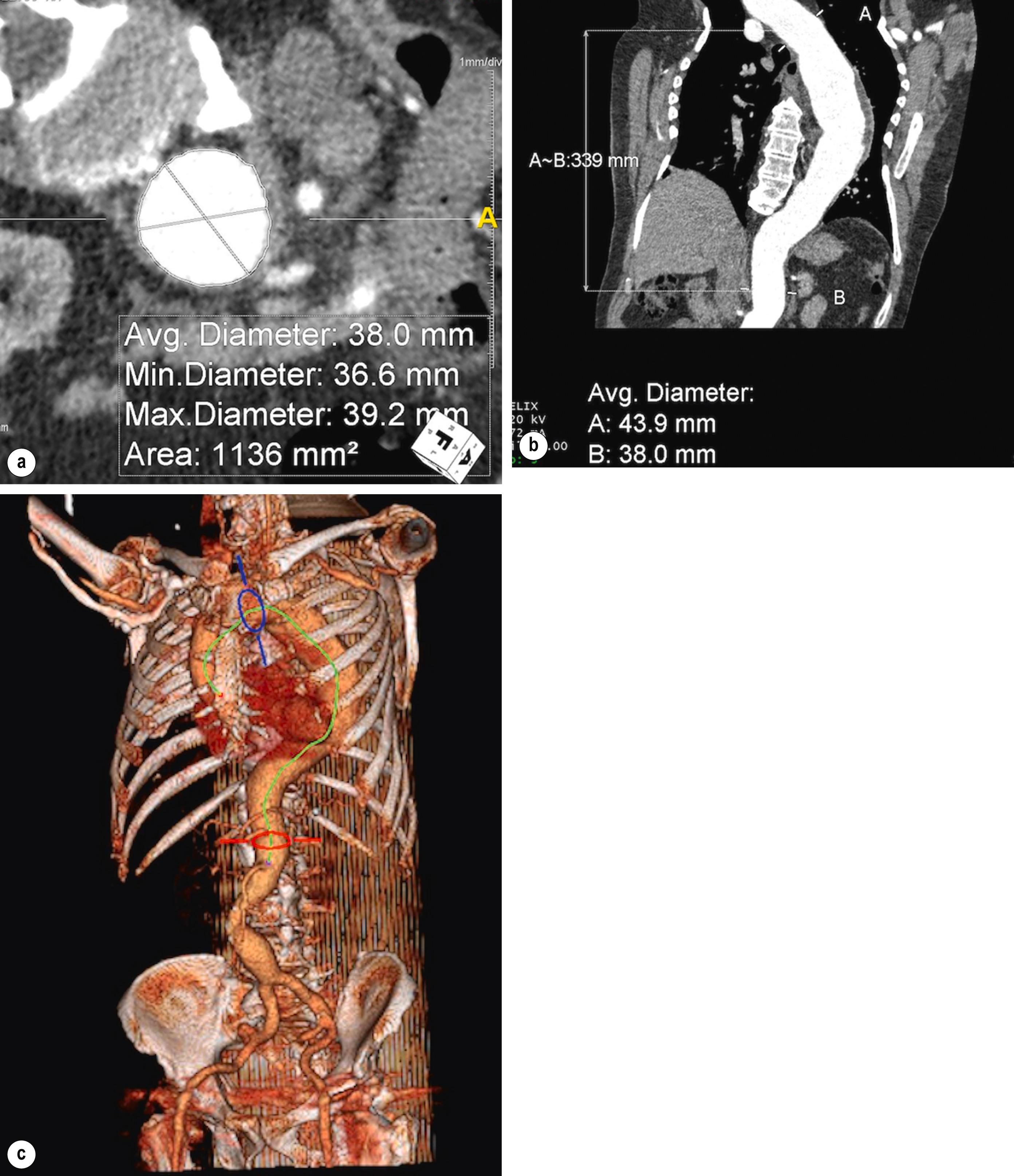
Computed tomographic angiography (CTA) is the investigation of choice to diagnose aortic pathology and plan subsequent repair. Modern workstations allow reformatting of images in multiple planes with the derivation of luminal centrelines. These techniques can be used to estimate lengths and diameters to a greater degree of accuracy than axial images alone and are essential if endovascular repair is planned ( Fig. 14.1 ). CTA is also the modality of choice when performing surveillance for patients that have had previous repair. The disadvantages of CTA are the relatively high dose of radiation required and the need for nephrotoxic intravenous contrast. , In younger patients, such as those with connective tissue disorders who are likely to require some years of surveillance, magnetic resonance angiography (MRA) may be more appropriate but offers less spatial resolution than CTA. MRA has been used in experimental applications for patients with aortic dissection to characterise flow in the false lumen and this may come to represent an important way to predict disease progression and success of treatment in the future.
The majority of thoracic aortic aneurysms (TAAs) are degenerative or atherosclerotic in nature, although approximately one-quarter are caused by a chronically dilating aortic dissection. Less frequently, aneurysms are related to connective tissue disorders such as Marfan syndrome or Ehler–Danlos syndrome. Mycotic or other inflammatory aneurysms of the thoracic aorta are relatively rare. False aneurysms can be caused by trauma and may present many years after the original precipitating event or may originate at the site of a previous surgical repair of the aorta. Aneurysms involving the descending aorta and the visceral segment are referred to as thoraco-abdominal aortic aneurysms ( TAAAs ). The majority of aneurysms are fusiform in morphology. Saccular aneurysms are less common and are usually the result of infection or previous trauma.
The incidence of aneurysms of the descending thoracic aorta appears to have increased, and in the UK has doubled from 4.4 to 9 per 100 000 patients per year over the last decade. The reason for this apparent increase is probably that thoracic aneurysms are most frequently discovered incidentally on routine chest radiography or cross-sectional imaging to investigate other conditions.
Thoracic aneurysms have a strong association with AAAs, and the conditions are concurrent in 23% of men and 48% of women with this condition. Clinical presentations include substernal, back or shoulder pain, superior vena cava syndrome, dysphagia, dyspnoea, stridor and hoarseness and rupture. The risk of rupture increases with the size of the aneurysm, but data regarding estimated annual rupture rates and aortic diameter are sparse and less robust than for the abdominal aorta.
The main indications for intervention are symptoms and size. There is controversy regarding the size criteria for treatment of TAAs, and the natural history remains poorly understood. Juvonen et al. reported that the 2-year rupture rate for TAAs was 23% in aneurysms less than 7 cm, whereas Elefteriades observed a 30% 5-year rupture rate when the aorta exceeded 6 cm. A study of 257 patients with TAAs showed that event rates at 1, 3 and 5 years were 4.3 ± 1.3%, 6.9 ± 1.9% and 9.7 ± 2.6% for definite aortic events and 6.6 ± 1.6%, 12.1 ± 2.4% and 16.5 ± 3.1% for possible events. Those with a starting aortic diameter of <50 mm experienced an event rate of less than 1%, but the rate of definite or possible event rates rose to 2.7 or 8.1% at aortic diameter between 50 mm and 60 mm and increased exponentially to 37.5 or 62.5% at >70 mm. A more recent study of 907 patients conducted by the Yale-New-Haven Aortic Institute yielded several important findings. Firstly, the majority of ruptures and aortic deaths occurred above aortic diameter of 5 cm (71%). Secondly, two key hinge-points of 6 and 6.5 cm were associated with sharp increases in the combined endpoint of aortic rupture and death. In keeping with infra-renal disease, aortic growth rates increase with aortic diameter: Descending thoracic and thoraco-abdominal aneurysms 4 cm in size grew at a rate of 0.22 cm/year, whilst 8 cm aneurysms grew at 0.42 cm/year. The American College of Cardiology/American Heart Association and European Society of Cardiology suggest considering open or endovascular repair in asymptomatic patients at a threshold of 6 cm. , Diameter thresholds should be reduced in patients with defined connective tissue disorders as rupture may occur at smaller aortic diameters.
The standard method of traditional open surgical repair of TAAs is a left thoracotomy for access, aortic clamping and inlay grafting. In cases involving the aortic arch, a two-stage approach may be used, which is known as an ‘elephant trunk’ procedure, and is usually performed in combination with cardiothoracic surgeons. This comprises open repair of the arch via a median sternotomy and is in itself comprised of two components; a proximal tube graft, which replaces the diseased arch, which is sutured to the origin of the descending aorta, and another length of unsupported graft, which is then inverted distally into the aneurysm beyond this point. In cases of more proximal disease extension, the supra-aortic trunks are managed by either incorporating them as an island or patch, which is anastomosed to the proximal component, or by “debranching”, whereby multi-branch inflow is taken from the ascending aorta and anastomosed end-to-end with the supra-aortic trunks. “Frozen elephant trunk” refers to more contemporary distal component, which is comprised of stent-supported fabric.
A second procedure can be performed through a left thoracotomy to continue the repair distally into the chest or an endovascular stent-graft can be deployed into it subsequently. In cases of more proximal disease extension, the supra-aortic trunks are managed by incorporating them as an island or patch, which is anastomosed to the tube graft used to replace the arch. The development of the more stable frozen elephant trunk has resulted in increasing numbers of second-stage procedures being completed endovascularly using TEVAR devices, with promising results. , These procedures are technically challenging and physiologically demanding for patients because of the need for aortic cross-clamping and organ ischaemia. Several adjuncts are available to achieve these aims, including the use of a Gott shunt, left heart bypass and distal aortic perfusion, selective intercostal shunting and cerebrospinal fluid (CSF) drainage.
Successful endovascular repair is dependent on the presence of suitable access vessels with adequate sealing (or ‘landing’) zones proximal and distal to the aneurysm. Pre-operative CTA is assessed to determine which endografts should be used and the mode of access. It is recommended that centreline image reconstruction is used using vascular-specific software to minimise error during planning.
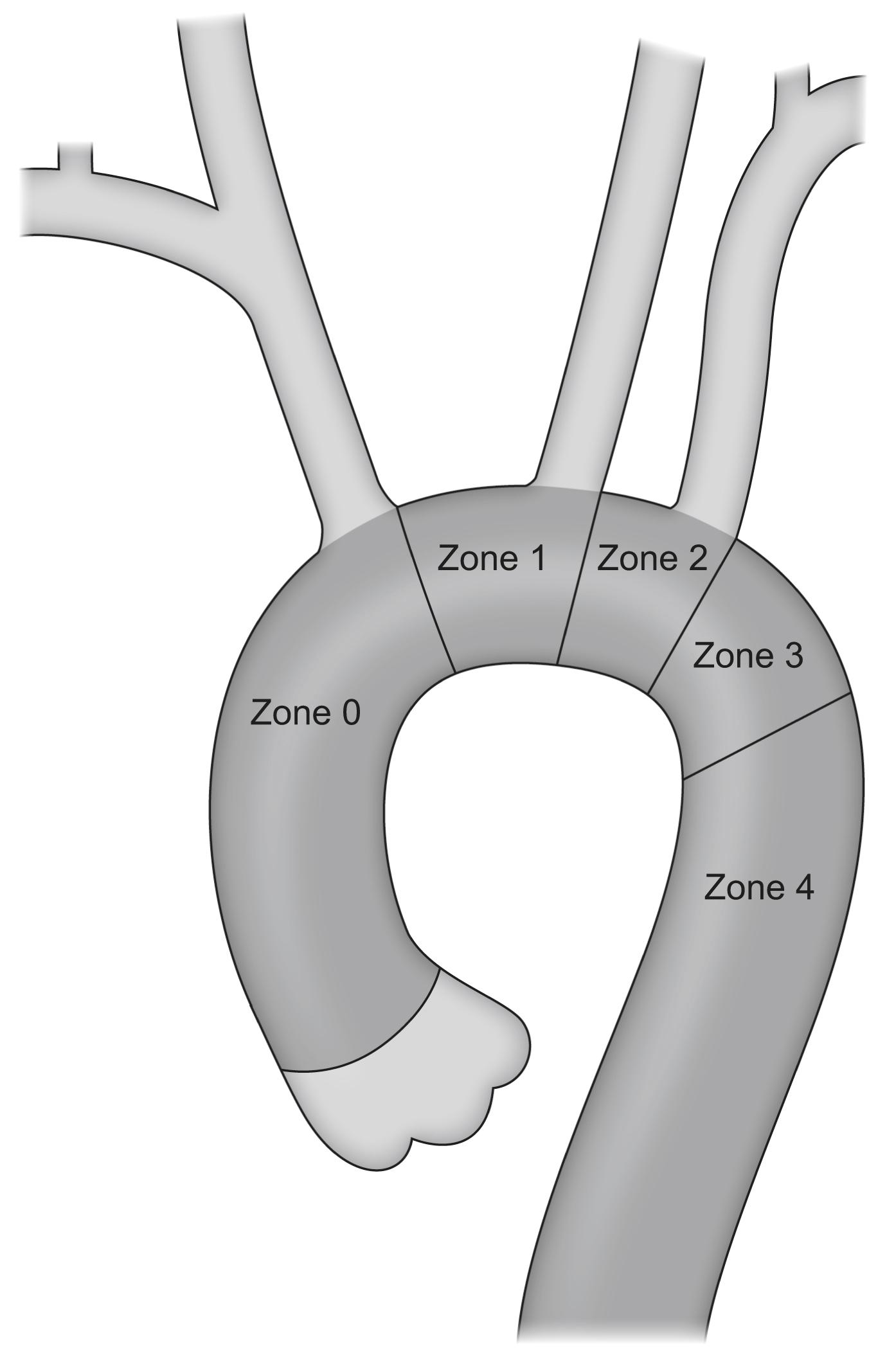
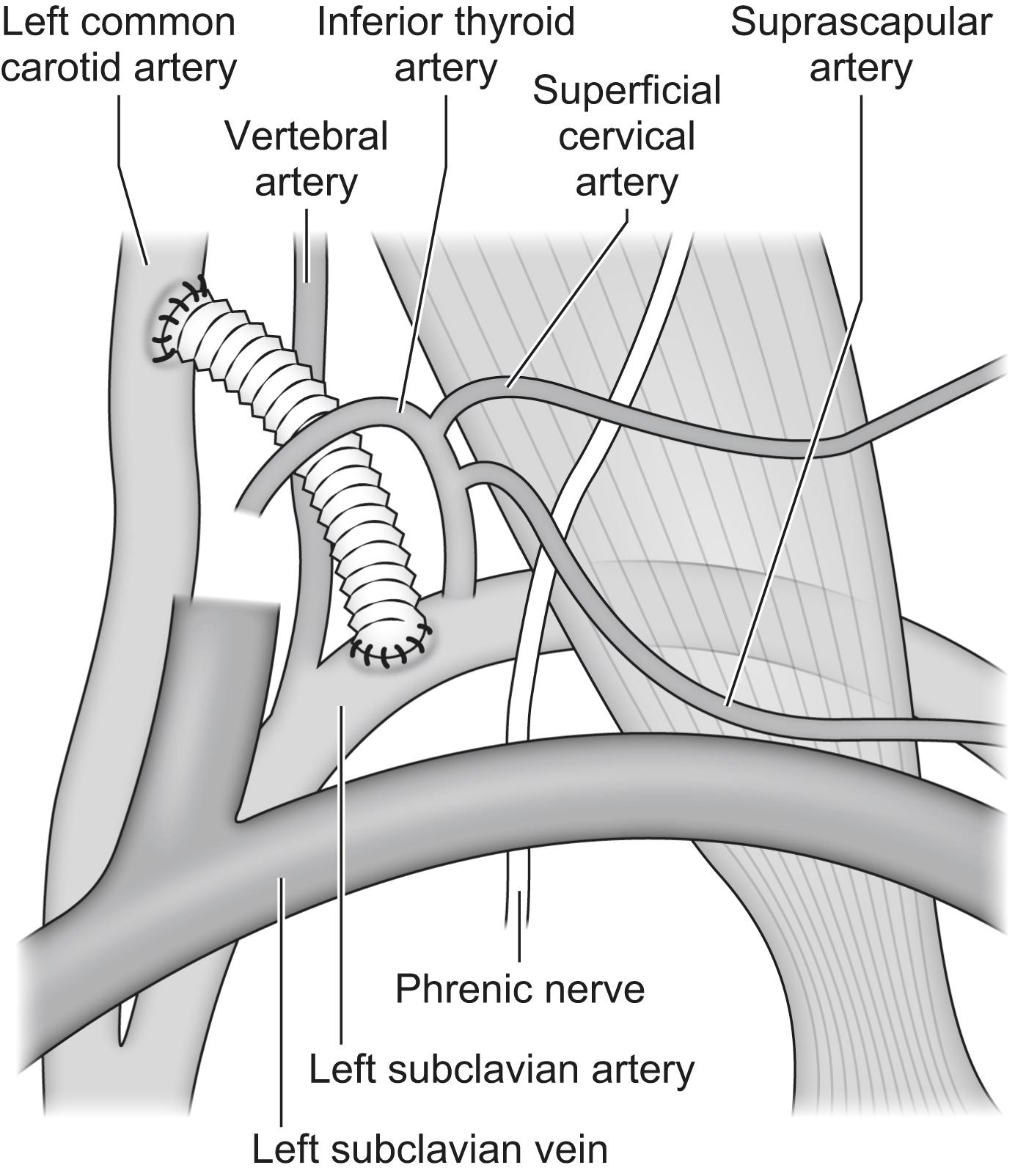
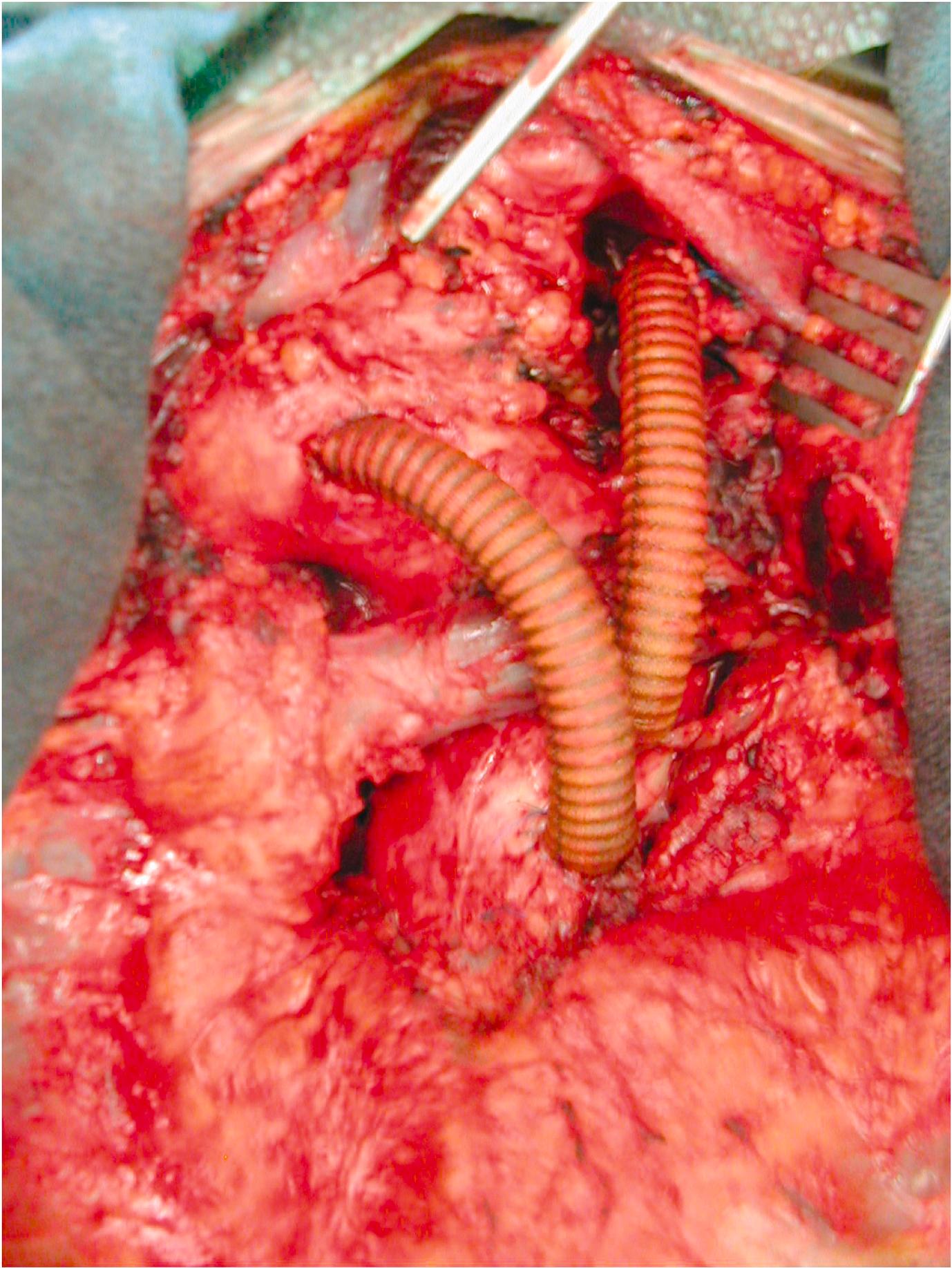
The optimal proximal landing zones should be between 15 and 20 mm along the inner curvature of the aorta, depending on the specific stent-graft manufacturer instructions for use. If the landing zone is considered to be of inadequate length, surgical bypass may be performed to lengthen the effective sealing zone. The supra-aortic trunks that would need to be covered by the endograft determine the Ishimaru zone of the repair ( Fig. 14.2 ). If left subclavian artery (LSCA) coverage for an aneurysm extending to Ishimaru zone 2 is required then a carotid subclavian bypass can be used ( Fig. 14.3 ). If Ishimaru zone 1 extent coverage is required, a right-to-left carotid-to-carotid bypass must be used with or without left subclavian revascularisation. In zone 0 coverage, ascending aorta to innominate and left common carotid bypass via a median sternotomy is performed ( Fig. 14.4 ).
Different commercially available stent-graft devices are available in different diameters, and these vary from 22–46 mm. A degree of oversizing is required with respect to the aortic diameter during device selection to ensure sufficient sealing, and it is recommended that this be 15–20%. As a result, the upper and lower limits of landing zone diameter are 40–42 mm and 18 mm, respectively. In many cases, more than one device is required, and when calculating the length of coverage then the overlap between adjacent devices recommended by manufacturers should be noted. A recent analysis has suggested that using endografts in unsuitable anatomy may significantly compromise durability.
The TEVAR procedure is usually performed under general or regional anaesthesia, although stents can be inserted under local anaesthesia. Transfemoral access is most normally obtained using percutaneous closure technique. A 300-mm length of extra-stiff guidewire (e.g., Lunderquist guidewire, Cook, UK) is advanced so that the tip is placed in the ascending aorta. A pre-curved wire can be used to enhance conformation to the aortic arch. A pigtail angiographic catheter is placed alongside this, and the first device is advanced into position along the stiff wire. Angiographic images are obtained in the correct parallax to allow visualisation of the supra-aortic branches. This usually requires a steep left anterior oblique angulation of the imaging system. The systolic blood pressure should be reduced to below 100 mmHg peak systolic pressure to allow accurate deployment of the proximal endograft ( Fig. 14.5 ). This can be achieved by pharmacological means or overdrive pacing to reduce the cardiac output and a range of opinion exists as to what is required across the range of pathologies treated. Further endografts are placed as necessary with the required overlap between endografts. Angiographic imaging is then used to ensure correct positioning and seal.
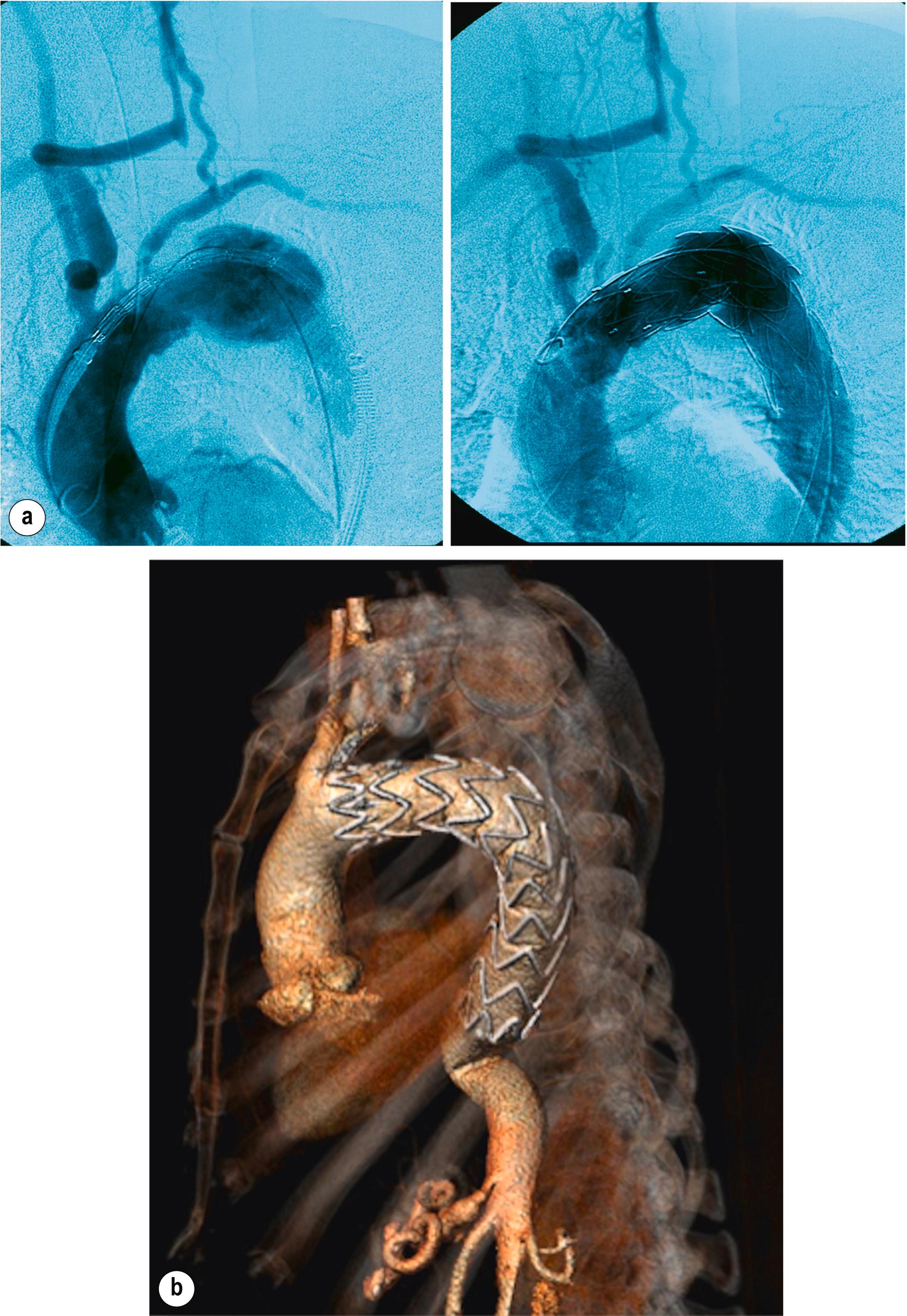
Neurological complications after TEVAR are not uncommon and are related to disturbance of flow in supra-aortic trunks or in the supply to the spinal cord. Stroke and paraplegia have devastating consequences for patients, and both are associated with a significant reduction in mid-term survival. A number of risk factors for paraplegia has been suggested, including the length of aortic coverage, previous infra-renal aortic repair, and the coverage without revascularisation of the LSCA. Emergency surgery, blood loss and renal failure have also been cited. What appears to be most important is to support and allow development of the collateral network supplying the spinal cord. To protect against this, some centres advocate prophylactic drainage of CSF whilst others adopt a selective approach. Increasingly, the safest strategy that may obviate the need for CSF drainage, appears to be staging thoraco-abdominal aortic repair into a number of smaller procedures where feasible.
If signs of spinal cord ischaemia occur then cord perfusion pressure must be increased by reducing CSF pressure via the use of a spinal drain, ensuring a mean arterial pressure of 100 mmHg with the use of fluid boluses and inotropes, and enhancing oxygen delivery maintaining PO 2 >10 and haemoglobin >10.
Regarding the management of the LSCA during thoracic endografting, consensus has determined that patients with a left internal mammary artery bypass graft or with a dominant left vertebral artery comprised a group where left subclavian revascularisation was considered mandatory. Where feasible, LSCA revascularisation is recommended in the management of atherosclerotic aneurysms, but the recommendation is weaker in the acute aortic syndromes including type B aortic dissection where paraplegia and posterior circulation stroke rates are lower. , , Normally, LSCA revascularisation is performed surgically, although a number of novel techniques have been described to undertake this endovascularly, none are yet mainstream.
Recent investigation has focused on the investigation of “silent cerebral infarction” and its implications; The STEP registry evaluated 91 patients with diffusion-weighted magnetic resonance imaging (MRI) post-operatively; whilst there were no clinically apparent strokes in this cohort, 50% demonstrated new DW-MRI lesions. Whether this translates into cognitive decline or can be effectively reduced, remains to be clarified.
Results of open repair in centres of excellence have generally been good, with less consistent results reported by smaller institutions. Focus in the last decade has been on comparing the outcome of open and endovascular repair.
The GORE TAG trial recruited 140 patients to undergo implantation of the GORE TAG (W.L. Gore & Associates, Flagstaff, Ariz, USA) device, retrospectively comparing outcomes with 94 patients undergoing repair of aneurysms in the descending or distal arch of the thoracic aorta. The 30-day mortality rate was 2.1% in the TEVAR group versus 11.7% in the open repair group. The VALOR trial recruited 195 patients who had the Talent thoracic endograft (Medtronic, Santa-Rosa, CA, USA) implanted. An observed survival rate (OSR) control arm of 189 patients were matched retrospectively as a control group. As with the TAG device, a lower 30-day mortality was noted in the TEVAR groups when compared to the OSR group (2% vs. 8%), and there were approximately half the number of major adverse events. The Cook TX2 pivotal trial recruited 160 patients to undergo treatment with the Cook TX2 thoracic endograft (Cook, Bloomington, USA), with 70 historical open surgical controls. The rate of peri-operative adverse events was low in both groups, but there was a significantly lower rate of severe morbidity in the TEVAR group. The MOTHER registry included 670 patients who underwent TEVAR for aneurysmal disease from five Medtronic device specific trials reported an early death rate of 5%.
Despite the protection that repair confers against aortic rupture, patients treated with TEVAR experience a high level of all-cause mortality that is often not related to the primary treated condition. This can be seen in comparisons with matched control groups of the same age and gender who do not have aneurysmal disease. The MOTHER registry reported a 5-year survival rate of 56%, with most patients dying of cardiovascular causes or malignancy. The Medicare and Health Episode Statistics studies study showed a similarly poor rate of survival, with many patients dying of cardiovascular or respiratory disease. , Appropriate patient selection is vital in maximising mortality benefits from TEVAR. There are no randomised trials that compare TEVAR and endovascular arch procedures to open repair, but recent meta-analysis has demonstrated reduced 30-day all-cause mortality for endovascular repair, whilst open repair showed favourable longer-term survival. In keeping with reports documenting outcomes in the visceral segment, the authors acknowledge that heterogeneous and inconsistent reporting, and differences in case selection, made meaningful data synthesis challenging.
Open surgical repair has remained the mainstay of management of more proximal lesions involving the aortic arch, given the need to preserve flow to the supra-aortic trunks, and requires cardiopulmonary bypass and deep hypothermic circulatory arrest. Even in centres of excellence, mortality rates approach 5%. The last decade, however, has seen the development of arch branch and fenestrated stent-graft designs, opening the possibility of total endovascular repair of the aortic arch. In keeping with treatment for visceral segment disease, arch devices are customised according to patient anatomy, and can incorporate scallops as needed. Proximal deployment (zones 0 and 1), variable supra-aortic trunk and access vessel anatomy, arch tortuosity and the presence dissection flaps, and atherosclerotic disease can make accurate deployment challenging. Arch manipulation and cannulation of supra-aortic predisposes to risk of stroke. The available literature reflects this and is restricted to small case-series showing a 30-day mortality of up to 10% with a contemporary meta-analysis yielding a stroke rate of 14%. Some studies of multibranched devices have reported a stroke rate of up to 40%, possibly as a result of wire manipulation in multiple supra-aortic trunks. Most of the studies report on a heterogeneous group of patients consisting of degenerative aneurysms, post-dissection aneurysms and connective tissue disorders, and this may, in part, account for the variability in outcomes.
Consistent findings are that the stroke rate generally exceeds mortality rate, and that retrograde type A dissections occur more frequently after arch repair in the post-dissection setting. Cook (Bloomington, USA) have more recently developed an inner branch device, which conceptually ought to reduce wire manipulation and shearing at the ostia of target vessels, with early reports showing promising lower stroke rates. , Whilst the mortality and stroke rates for endovascular arch treatments appear at face value to be comparable to open repair, it should be pointed out that most groups reporting this technique selected patients whose age and co-morbidities would have been prohibitive for open repair; a case-matched comparison is therefore not feasible. Going forward, total endovascular aortic repair comprised of staged fenestrated/branched arch and fenestrated/branched visceral segment intervention has been shown to be achievable, with acceptable mortality and adverse neurological event rates.
Endovascular repair of TAAs is now first-line therapy for most thoracic aneurysms. The exceptions to this would be in patients with unfavourable anatomy in whom there is a likelihood of poor long-term durability, and elderly frail patients with a poor predicted life expectancy in whom the prevention of aneurysm-related death would not improve their overall survival. , The endovascular management of patients with connective tissue disease is controversial, but increasingly used. These patients are usually younger and fitter than patients with degenerative aneurysms and often have good outcomes from conventional surgery and poorer results from TEVAR.
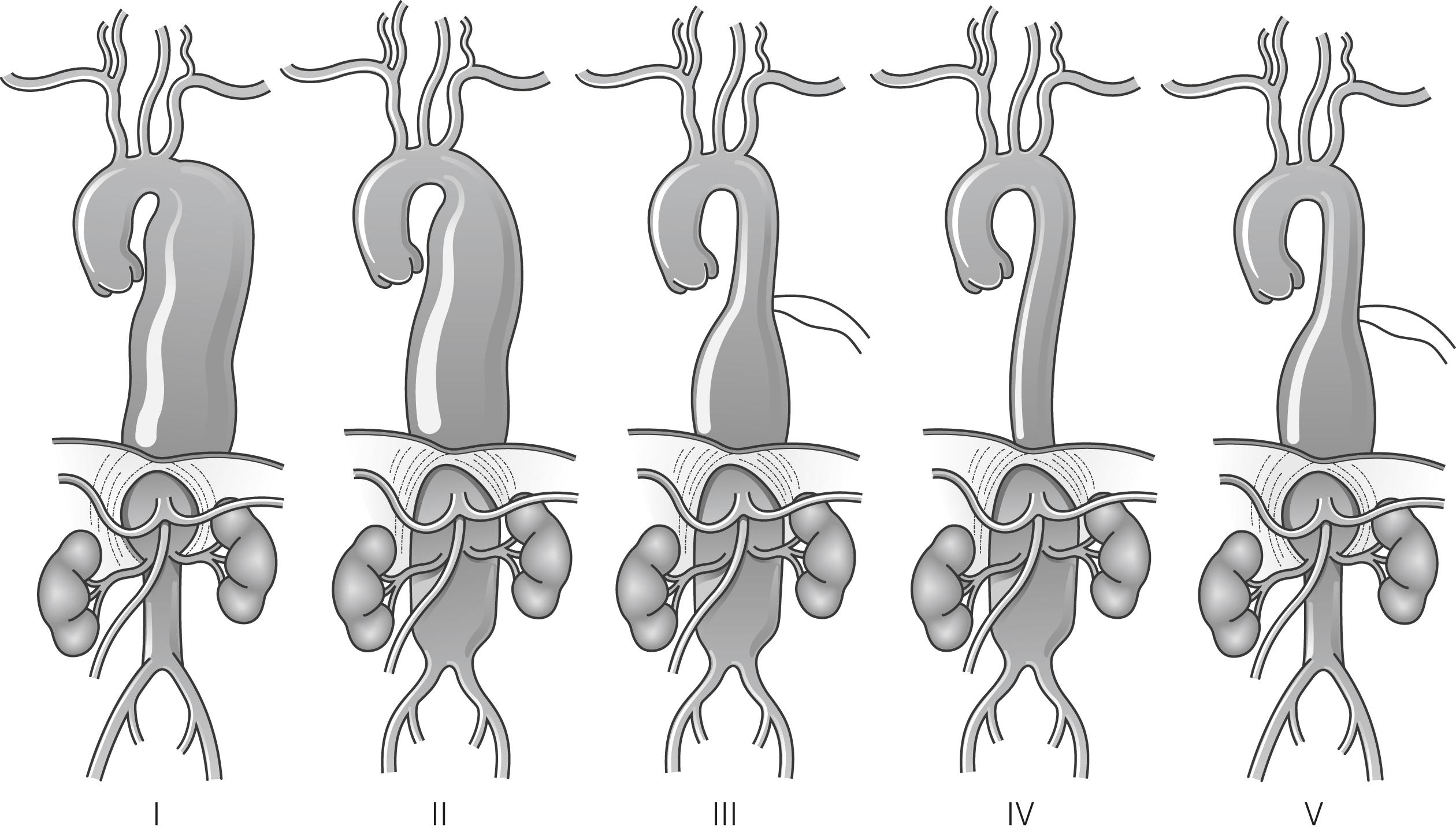
Thoracic aneurysms that involve the visceral segment of the abdominal aorta are traditionally described as thoraco-abdominal aneurysms and are classified according to the extent of the aneurysmal disease (see Fig. 14.6 for a description of the Crawford classification). The aetiology of thoraco-abdominal aneurysms differs from infra-renal aneurysms, with medial degenerative disease and chronic dissection being particularly prevalent. As with TAA, connective tissue disorders are a strong risk factor for the development of TAAAs.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here