Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The skin is supplied by small segmental arteries that may reach the skin directly or after supplying the underlying muscle and soft tissues en route. These vessels branch to supply the subcutis with a meshwork of arteries and arterioles. In the dermis, there is a deep (lower) horizontal plexus , situated near the interface with the subcutis, and a superficial (upper) horizontal plexus , at the junction of the papillary and reticular dermis. The two plexuses are joined by vertically oriented arterioles. These interconnecting arterioles also give rise to vessels that form an arborizing plexus around the hair follicles. The sweat gland plexus of vessels may also arise from the vertical arterioles or from arteries in the subcutis.
The superficial horizontal plexus is a band-like network of anastomosing small arterioles and postcapillary venules, connected by a capillary network. The bulk of the microcirculation of the skin resides in this plexus and the capillary loops that form from it and pass into the dermal papillae. The postcapillary venules are the important functional sites for disease processes in the skin. For example, they represent the site of immune complex deposition in acute vasculitis, the site of vascular permeability in urticaria, and an area for leukocyte recruitment and diapedesis in vasculitis.
The arterioles in the superficial plexus have a homogeneous basement membrane, a discontinuous subendothelial elastic lamina, and one or two layers of smooth muscle cells (in contrast to arterioles in the deep plexus, which have four or five layers). The capillary wall contains a basement membrane that changes from homogeneous to multilayered as it changes from an arteriolar capillary to a venous capillary. The postcapillary venule is surrounded by veil cells rather than the smooth muscle cells seen in arterioles. The venules drain into large veins that accompany the vertically oriented arterioles. At the dermal–subcutaneous junction, there are collecting veins with two cusped valves that are oriented to prevent the retrograde flow of blood.
In addition to the vessels listed previously, there are arteriovenous anastomoses (shunts) that bypass the capillary network. They play a role in thermoregulation. A special form of arteriovenous shunt, occurring in the periphery, is the glomus apparatus . The glomus is composed of an endothelial-lined channel surrounded by cuboidal glomus cells; it has a rich nerve supply.
The dermal lymphatic network has a similar pattern of distribution to that of the blood supply.
At a functional level, the endothelial adherens junction complex is an important mechanism in the control of leukocyte and macromolecule transmigration. The adherens junction is formed by transmembrane molecules of the cadherin family linking to catenins, which anchor the adhesion plaque to the cytoskeleton of the endothelial cell. The cadherin is cell-type specific and in the vessel is known as vascular endothelial (VE) cadherin. Interestingly, VE-cadherin knockout mice die during embryonic development, indicating the importance of this substance in organogenesis.
Diseases of cutaneous blood vessels are an important cause of morbidity. In the case of the vasculitides, mortality may occasionally result. Blood vessels have a limited number of ways in which they can react to insults of various kinds, resulting in considerable morphological overlap between the various clinical syndromes that have been described.
The broad classifications used in this chapter are morphologically rather than etiologically based because this is the most practical scheme to follow when confronted with a biopsy.
Excluding tumors and telangiectases, which are discussed in Chapter 39 , there are six major groups of vascular diseases:
Noninflammatory purpuras
Vascular occlusive diseases
Urticarias
Vasculitis
Neutrophilic dermatoses
Miscellaneous
The noninflammatory purpuras are characterized by the extravasation of erythrocytes into the dermis. There is no inflammation or occlusion of blood vessels.
The vascular occlusive diseases exhibit narrowing or obliteration of the lumina of small vessels by fibrin or platelet thrombi, cryoglobulins, cholesterol, or other material. Purpura and sometimes ulceration and necrosis may be clinical features of this group.
The urticarias involve the leakage of plasma and some cells from dermal vessels. Some of the urticarias have overlapping features with the vasculitides, further justification for their inclusion in this chapter.
In vasculitis, there is inflammation of the walls of blood vessels. In subsiding lesions, there may only be an inflammatory infiltrate in close contact with vessel walls. The vasculitides are subclassified on the basis of the inflammatory process into acute, chronic lymphocytic, and granulomatous forms. Fibrin-platelet thrombi may sometimes form, particularly in acute vasculitis, leading to some overlap with the vascular occlusive diseases.
The neutrophilic dermatoses are a group of conditions in which there is a prominent dermal infiltrate of neutrophils, but usually without the fibrinoid necrosis of vessel walls that typifies acute (leukocytoclastic) vasculitis.
The miscellaneous group of vascular disorders includes the capillary leak syndrome, vascular calcification, collagenous vasculopathy, pericapillary fibrin cuffs, vascular aneurysms, erythermalgia, and cutaneous necrosis and ulceration.
Purpura is hemorrhage into the skin. Clinically, this may take the form of small lesions less than 3 mm in diameter (petechiae) or larger areas known as ecchymoses. There is a predilection for the limbs. The numerous causes of purpura may be broadly grouped into defects of blood vessels, platelets, or coagulation factors.
At the histopathological level, purpuras are characterized by an extravasation of red blood cells into the dermis from small cutaneous vessels. If the purpura is chronic or recurrent, hemosiderin or hematoidin pigment may be present. Purpuras have traditionally been divided into an inflammatory group (vasculitis) and a noninflammatory group when there is no inflammation of vessel walls.
The noninflammatory purpuras include idiopathic thrombocytopenic purpura, senile purpura, the autoerythrocyte sensitization syndrome (psychogenic purpura), traumatic (including factitious) purpura, and drug purpuras.
The autoerythrocyte sensitization syndrome (Gardner–Diamond syndrome) is an uncommon bruising disorder affecting predominantly women with emotional instability. It is characterized by periodic, painful ecchymoses giving bruise-like areas mostly on the upper and lower limbs. Pain or a prickly sensation may precede the ecchymoses. The diagnosis can be confirmed by the intradermal injection of autologous whole blood (autoerythrocyte sensitization test), which will reproduce the tender bruises in this syndrome. Painful bruising in a bizarre, almost factitious arrangement was reported in a male traveler. He did not appear to have this syndrome.
Sickle-cell disease is an unusual cause of purpura. Only senile purpura is considered in further detail in this volume.
Senile purpura is a common form of noninflammatory purpura that occurs on the extensor surfaces of the forearms and hands of elderly individuals. Usually, large ecchymoses are present. It has been suggested that the bleeding results from minor shearing injuries to poorly supported cutaneous vessels. Senile purpura tends to persist longer than other forms of purpura, indicating slower removal or breakdown of the erythrocytes. Furthermore, senile purpura does not usually show the color changes of bruising, as seen in purpura of other causes.
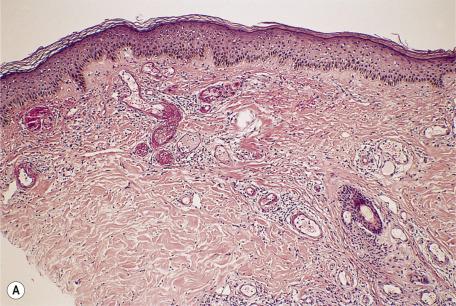
Senile purpura is characterized by extravasation of red blood cells into the dermis ( Fig. 9.1 ). This is most marked in the upper dermis and in a perivascular location. There is also marked solar elastosis and often some thinning of the dermis with atrophy of collagen bundles.

Occlusion of cutaneous blood vessels is quite uncommon. The clinical picture that results is varied. It may include purpura, livedo reticularis, erythromelalgia, ulceration, or infarction. Cutaneous infarction only occurs when numerous vessels in the lower dermis and subcutis are occluded. Because livedo reticularis is a clinical feature of some of the diseases discussed in this section, it is considered first.
Livedo reticularis consists of macular, violaceous, connecting rings that form a net-like pattern. In most cases, it is a completely benign finding related to cold exposure. It results from increased visibility of the venous plexus that can result from venodilation often caused by deoxygenation of blood in the venous plexus. Cold results in deoxygenation by vasospasm leading to venodilation. Most of the diseases discussed as venocclusive diseases can produce secondary livedo reticularis.
In addition other vascular diseases, drugs, infections, and neurological and metabolic diseases can be associated with livedo reticularis. The drugs that can result in livedo reticularis include amantadine, minocycline, gemcitabine, heparin, and interferon (IFN) therapy. Laser-assisted hair removal may result in a reticulate erythema resembling livedo reticularis. Almost 100 different diseases and drugs that can cause livedo reticularis are listed in the excellent review of this subject by Gibbs et al. in 2005.
Nicolau's syndrome (livedo-like dermatitis, embolia cutis medicamentosa) has painful areas resembling livedo reticularis at the edge of hemorrhagic aseptic necrosis. It has followed the intramuscular injection of bismuth salts, nonsteroidal antiinflammatory drugs (NSAIDs), penicillin, cyanacobalamin, piroxicam, thiocolchicoside, and diphtheria–tetanus–pertussis (DTP) vaccine. Retiform purpura in plaques (RPP) is a morphological sign that differs from livedo reticularis by the presence in RPP of ischemia-related hemorrhage around a vessel, before its complete occlusion.
Complete or partial occlusion of cutaneous vessels usually results from the lodgment of fibrin-platelet thrombi. Other causes include platelet-rich thrombi in thrombocythemia and thrombotic thrombocytopenic purpura, cryoglobulins in cryoglobulinemia, cholesterol in atheromatous emboli, swollen endothelial cells containing numerous acid-fast bacilli in Lucio's phenomenon of leprosy, fungi in mucormycosis, and fibrous tissue producing intimal thickening in endarteritis obliterans.
Excluding vasculitis, which is considered later, vascular occlusion may be seen in the following circumstances:
Protein C and protein S deficiencies
Prothrombin gene mutations
Warfarin necrosis
Atrophie blanche (livedoid vasculopathy)
Disseminated intravascular coagulation
Purpura fulminans
Thrombotic thrombocytopenic purpura
Thrombocythemia
Cryoglobulinemia
Cholesterol and other types of embolism
Antiphospholipid syndrome
Factor V Leiden mutation
Sneddon's syndrome
CADASIL
Miscellaneous conditions.
Because deficiencies, congenital or acquired, of protein C or protein S underlie some of the diseases considered in this section, they are considered first. Because protein S is a cofactor of activated protein C, they are discussed together.
Protein C, a vitamin K–dependent plasma glycoprotein, has, when activated, an important anticoagulant role by destroying factors Va and VIII. Congenital protein C deficiency (OMIM 176860 ) is usually inherited as an autosomal dominant trait, although a few autosomal recessive cases have been described. It results from mutations in the protein C (PROC) gene on chromosome 2q13–q14. Patients with a homozygous deficiency usually present with purpura fulminans (see p. 245 ) in childhood. Heterozygous deficiency predisposes to venous thrombosis in adulthood or warfarin necrosis (see p. 244 ). Skin necrosis is another manifestation of this deficiency. Acquired deficiencies have resulted from infections, chronic ulcerative colitis, and anticoagulant therapy. Acquired deficiencies can also predispose to purpura fulminans and to warfarin necrosis.
Protein S is another vitamin K–dependent protein that serves as a cofactor for activated protein C. Protein S deficiency (OMIM 176880 ) can predispose to the same conditions as protein C deficiency, including warfarin necrosis. It is caused by a mutation in the protein S (PROS1) gene on 3p11.1–q11.2. Protein S appears also to have functions outside the coagulation pathway.
The 20210G-A mutation in the prothrombin gene (coagulation factor II; OMIM *176930) is found in 0.7% to 2.6% of the population and in 4% to 8% of patients presenting with first-episode venous thrombosis. It leads to hyperprothrombinemia. It may occur in association with other genetic disorders of the coagulation pathway, particularly the factor V Leiden mutation, which is also very common in the community. Both mutations probably occurred more than 20,000 years ago, and both appear to have provided selective evolutionary advantages. It has been reported in association with atrophie blanche (livedoid vasculopathy).
Cutaneous infarction is a rare, unpredictable complication of anticoagulant therapy with the coumarin derivative warfarin sodium. It has a predilection for fatty areas such as the thighs, buttocks, and breasts of obese, middle-aged women. Lesions usually develop several days after the commencement of therapy. Late-onset lesions can be difficult to diagnose. There are well-defined ecchymotic changes that rapidly progress to blistering and necrosis. Purpuric and linear lesions have been reported.
Warfarin necrosis is related to low levels of protein C, a vitamin K–dependent plasma protein with potent anticoagulant properties. This deficiency in protein C may be induced by the anticoagulant therapy or preexist in those who are heterozygous for protein C deficiency. It can also result from protein S deficiency and a mutation in the methylenetetrahydrofate reductase ( MTHFR ) gene (OMIM *607093). Warfarin necrosis has also been associated with the factor V Leiden mutation, indicating that several closely related factors may contribute to warfarin-induced necrosis. Acute renal failure was thought to be the cause in one case. Rarely, widespread disseminated intravascular coagulation is associated with warfarin therapy. Warfarin necrosis has been successfully treated with human protein C concentrate.
Various reactions, including hemorrhagic bullae, urticaria, necrosis caused by thrombosis of dermal capillary vessels, and eczematous lesions, may occur at sites of subcutaneous heparin injections or at sites distant from the injection site. The mechanism of formation of the hemorrhagic bullae is unknown. It is not related to warfarin necrosis in any way, although in a given case heparin-induced thrombocytopenia can be associated with warfarin necrosis. Widespread skin necrosis may also occur as a result of thrombosis of dermal vessels in patients receiving heparin therapy. The incidence of this complication varies between 1% and 3% for a 1-week course of intravenous heparin therapy. It is often a result of the presence of antibodies directed against heparin-platelet factor 4.
Fibrin-platelet thrombi are present in venules and arterioles in the deep dermis and subcutis. There is variable hemorrhage and subsequently the development of infarction. Large areas of the skin may be involved.
Although previously considered to be caused by a vasculitis, atrophie blanche (white atrophy) is best regarded as a manifestation of a thrombogenic vasculopathy in which occlusion of small dermal vessels by fibrin thrombi is the primary event. It appears to result from decreased fibrinolytic activity of the blood with defective release of tissue plasminogen activator from vessel walls. The platelets usually show an increased tendency to aggregate. An elevated level of fibrinopeptide A, suggestive of a thrombogenic state, has been found in patients with this condition. Several cases with a lupus-type anticoagulant and an increased level of anticardiolipin antibodies have been reported. This abnormality can alter fibrinolytic activity. Other associations of atrophie blanche include protein C deficiency, factor V Leiden mutation, prothrombin mutations, IgM anti–phosphatidylserine–prothombin complex antibody, plasminogen activator inhibitor-1 promoter mutations (OMIM *173360), antithrombin III deficiency, methylenetetrahydrofolate reductase mutations ( MTHFR ; OMIM *607093), homocysteinemia (OMIM 603174 ), and essential cryoglobulinemia, all of which can contribute to a hypercoagulable condition.
Atrophie blanche (synonyms: livedoid vasculopathy, livedo vasculitis, segmental hyalinizing vasculitis, and painful purpuric ulcers with reticular patterning on the lower extremities [PURPLE]) is characterized by the development of telangiectatic, purpuric papules and plaques leading to the formation of small crusted ulcers, which heal after many months to leave white atrophic stellate scars. The ulcers are painful and recurrent. Sometimes they are large and slow to heal. The lower parts of the legs, especially the ankles and the dorsum of the feet, are usually involved, although rarely the extensor surfaces of the arms below the elbows can be affected. Many patients also have livedo reticularis, and some may have systemic diseases such as scleroderma, systemic lupus erythematosus (SLE), and cryoglobulinemia. The disorder has a predilection for middle-aged women, but all ages may be affected.
Atrophie blanche–like scarring can occur uncommonly after pulsed dye laser treatment of vascular malformations.
Therapies that have been used with varying degrees of success include antiplatelet drugs and antithrombotic drugs, dapsone, nicotinic acid, intravenous immunoglobulin, psoralen-UV-A (PUVA) therapy, danazol, hyperbaric oxygen, warfarin therapy, and amelogenin (enamel matrix protein) extracellular matrix (ECM) protein. Callen published a therapeutic “ladder” for the management of this condition.
The changes will depend on the age of the lesion that is biopsied. The primary event is the formation of hyaline thrombi in the lumen of small vessels in the upper and mid dermis. Rarely, deeper vessels are involved. Polyarteritis nodosa is rarely present. Fibrinoid material is also present in the walls of these blood vessels and in perivascular stroma ( Fig. 9.2 ). This material is periodic acid–Schiff (PAS) positive and diastase resistant. There is usually infarction of the superficial dermis, often with a small area of ulceration. Sometimes a thin parakeratotic layer is present overlying infarcted or atrophic epidermis. The epidermis adjacent to the ulceration may be spongiotic. A sparse perivascular lymphocytic infiltrate may be present, but there is no vasculitis. Neutrophils, if present, are usually sparse and confined to the infarcted upper dermis and ulcer base. There are often extravasated red cells in the upper dermis. Small blood vessels are often increased in the adjacent papillary dermis, but this is a common feature in biopsies from the lower parts of the legs and is therefore of no diagnostic value.
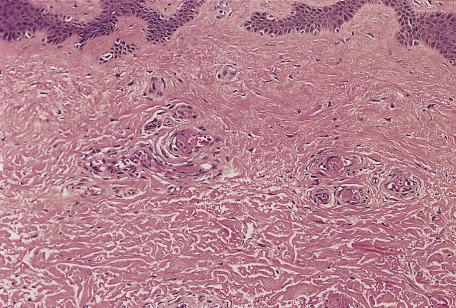
In older lesions, there is thickening and hyalinization of vessels in the dermis with some endothelial cell edema and proliferation. Fibrinoid material may also be present in vessel walls. Note that fibrinoid material is almost invariably present in blood vessels in the base of ulcers, of many different causes, on the lower legs. In atrophie blanche, the involved vessels are not only in the base of any ulcer but also may be found at a distance beyond this. In even later lesions, there are dermal sclerosis and scarring with some dilated lymphatics and epidermal atrophy. There may be a small amount of hemosiderin in the upper dermis. Because these areas may become involved again, it is possible to find dermal sclerosis in some early lesions.
Immunofluorescence will demonstrate fibrin in vessel walls in early lesions, whereas in later stages there are also immunoglobulins and complement components in broad bands about vessel walls.
This has confirmed the presence of luminal fibrin deposition with subsequent endothelial damage.
Disseminated intravascular coagulation (DIC) is an acquired disorder in which activation of the coagulation system leads to the formation of thrombi in the microcirculation of many tissues and organs. As a consequence of the consumption of platelets and of fibrin and other factors during the coagulation process, hemorrhagic manifestations also occur. DIC may complicate infections, various neoplasms, certain obstetric incidents, massive tissue injury such as burns, and miscellaneous conditions such as liver disease, snake bite, and vasculitis.
Cutaneous changes are present in approximately 70% of cases, and these may be the initial manifestations of DIC. Petechiae, ecchymoses, hemorrhagic bullae, purpura fulminans, bleeding from wounds, acral cyanosis, and frank gangrene have all been recorded.
The formation of thrombi appears to be a consequence of the release of thromboplastins into the circulation and/or widespread injury to endothelial cells. Decreased levels of protein C have been reported; the level returns to normal with clinical recovery.
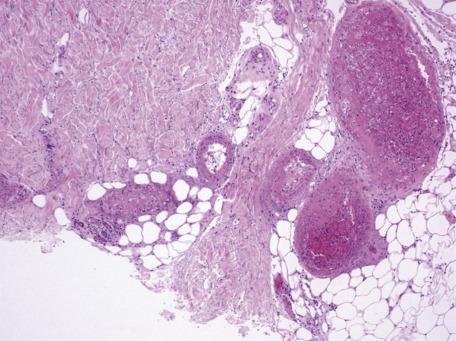
In early lesions, fibrin thrombi are present in capillaries and venules of the papillary dermis and occasionally in vessels in the reticular dermis and subcutis ( Fig. 9.3 ). Hemorrhage is also present, but there is no vasculitis or inflammation.
In older lesions, of 2 to 3 days’ duration, there may be epidermal necrosis, subepidermal bullae, extensive hemorrhage, and patchy necrosis of eccrine glands, the pilosebaceous apparatus, and the papillary dermis. Nearby blood vessels are thrombosed, but there is only mild inflammation in the dermis. In chronic states, some vascular proliferation and ectasia may occur.
Purpura fulminans is a rare clinical manifestation of DIC in which there are large cutaneous ecchymoses and hemorrhagic necrosis of the skin resulting from thrombosis of the cutaneous microvasculature. It is not known why only certain patients with DIC develop the full picture of purpura fulminans. Hypotension and fever are also present, but visceral manifestations are uncommon. Only a few cases have been reported in adults ; the majority arise in infancy and early childhood some days after an infectious illness, usually streptococcal, meningococcal, pneumococcal, or viral. A number of cases associated with varicella infection have been reported in recent years. An association with malaria has been reported. Rarely, purpura fulminans occurs in the neonatal period associated with severe congenital deficiency of protein C or protein S. As previously noted, protein S is a cofactor for protein C. Acquired deficiencies of these proteins, probably because of the primary infectious process, appear to be responsible for cases in later life, but they are also responsible for some cases of purpura fulminans in childhood. The factor V Leiden mutation has been incriminated in a small number of adult and childhood cases.
The cutaneous lesions can commence as erythematous macules that rapidly enlarge and develop central purpura. The central zone becomes necrotic, and the eventual removal of the resulting eschar leads to an area of ulceration. There is a predilection for the lower extremities and the lateral aspect of the buttocks and thighs. Peripheral gangrene may sometimes develop. The use of fresh frozen plasma has considerably improved the prognosis of this disease. Administration of human protein C concentrate has been beneficial in some cases.
Fibrin thrombi fill most of the venules and capillaries in the skin. There is a mild perivascular infiltrate in some areas, but no vasculitis. Extensive hemorrhage is present with the subsequent development of epidermal necrosis. Occasionally, a subepidermal bulla develops.
Thrombotic thrombocytopenic purpura is a rare syndrome characterized by the clinical picture of microangiopathic hemolytic anemia, thrombocytopenia, neurological symptoms, renal disease, and fever. Cutaneous hemorrhages in the form of petechiae and ecchymoses are quite common. Before the introduction of plasmapheresis and antiplatelet agents, the disease was almost invariably fatal.
It appears to result from prostacyclin inhibition and impaired fibrinolysis. Although drugs, infectious agents, and obstetrical incidents have been implicated in triggering this syndrome, in the majority of individuals there is no apparent causal event or underlying disease process. Drugs incriminated include the antiplatelet drugs ticlopidine and clopidogrel. Coagulation studies fail to show evidence of DIC.
Platelet-rich thrombi admixed with a small amount of fibrin deposit in vessels at the level of the arteriolocapillary junction. They may also involve small arteries. There may be slight dilatation of vessels proximal to the thrombi. The material is PAS positive. There is no evidence of any associated vasculitis. Extravasation of red blood cells also occurs. In severe cases, necrosis of the epidermis may ensue.
Thrombocythemia is a rare, chronic myeloproliferative disorder characterized by a significant increase in the platelet count. Cutaneous manifestations, which include livedo reticularis, erythromelalgia, and ischemic manifestations, occur in approximately 20% of patients. Platelet plugging, leading to erythematous plaques, may develop in other myeloproliferative disorders.
If biopsies are taken from areas of erythromelalgia, there is fibromuscular intimal proliferation involving arterioles and small arteries similar to the changes seen in Sneddon's syndrome.
In a case associated with both erythromelalgia and livedo reticularis, biopsy of the latter showed hyalinized, thrombosed deep dermal vessels with perivascular lymphohistiocytic infiltrates. If ischemic areas are biopsied, there is vascular thrombosis, which may involve vessels of all sizes. Infarction of the dermis and/or epidermis may accompany these thrombosed vessels.
Cryoglobulins are immunoglobulins that reversibly precipitate from the serum or plasma on cooling. There are two distinct types of cryoglobulinemia, monoclonal (type I) and mixed (type II), which reflect the composition of the cryoglobulins involved. In the monoclonal type, intravascular deposits of cryoglobulins can be seen in biopsy specimens, whereas the mixed variant is a vasculitis. They are considered together for convenience.
The monoclonal variant, which accounts for approximately 25% of cases of cryoglobulinemia, is associated with the presence of immunoglobulin G (IgG) or IgM cryoglobulin or, rarely, of a cryoprecipitable light chain. Monoclonal cryoglobulins, also known as type I cryoglobulins, are usually seen in association with multiple myeloma, Waldenström's macroglobulinemia, and chronic lymphatic leukemia. Sometimes, no underlying disease is present (essential cryoglobulinemia). The condition may be asymptomatic or result in purpura, acral cyanosis, or focal ulceration, which is usually limited to the lower extremities. Generalized livedo reticularis is another manifestation. In a study of 15 patients with leg ulcers associated with cryoglobulinemia, 8 patients had type I cryoglobulins and 7 patients had type II. Plasma exchange combined with immunosuppressive drugs usually cures the ulcers. Thalidomide, rituximab, and bortezomib have also been used.
In the mixed variant, the cryoglobulins are composed of either a monoclonal immunoglobulin that possesses antibody activity toward, and is attached to, polyclonal IgG (type II) or two or more polyclonal immunoglobulins (type III). Mixed cryoglobulins usually take the form of immune complexes and interreact with complement. They are seen in autoimmune diseases such as rheumatoid arthritis, SLE, and Sjögren's syndrome; in lymphoproliferative diseases; as well as in various chronic infections resulting from the hepatitis and Epstein–Barr viruses. Numerous cases associated with infection with the hepatitis C virus have been reported in recent years. Vasculitis can also occur in patients with hepatitis C virus who do not have mixed cryoglobulinemia; clinical differences are slight, except for a higher incidence of arthralgias among those who do have mixed cryoglobulinemia. A mixed cryoglobulinemia may be the cause of some cases of the “red finger syndrome” seen in patients with HIV or hepatitis infection. Association with parvovirus B19 infection has been reported on several occasions. In other patients, no etiology or pathogenesis is apparent. In approximately 50% of cases, no detectable autoimmune, lymphoproliferative, or active infective disease is present. These cases of essential mixed cryoglobulinemia are characterized by a chronic course with intermittent palpable purpura, polyarthralgia, Raynaud's phenomenon, and occasionally glomerulonephritis. In a recent survey of mixed cryoglobulinemia, 13 of 33 patients had essential mixed cryoglobulinemia; these individuals tended to have more severe disease than those with secondary forms of the disease, and they more often developed renal and peripheral nerve involvement. Other cutaneous manifestations that may be present include ulcers, urticaria, digital necrosis, and, rarely, pustular purpura.
A localized cryoglobulinemic vasculitis can be seen in tick bite reactions, particularly when tick mouthparts are retained in the tissue.
Purpuric lesions will show extravasation of red blood cells into the dermis. Small vessels in the upper dermis may be filled with homogeneous, eosinophilic material that is also PAS positive. These intravascular deposits are seen more commonly beneath areas of ulceration. Although there is generally no vasculitis, there may be a perivascular infiltrate of predominantly mononuclear cells ( Fig. 9.4 ). Microscopic changes of leukocytoclastic vasculitis have been reported in one case of monoclonal cryoglobulinemia (associated with Waldenström's macroglobulinemia). Vacuole-like cytoplasmic inclusions were found in peripheral blood neutrophils, monocytes, lymphocytes, and platelets (but not in bone marrow cells) in cryoglobulinemia associated with IgG-κ monoclonal gammopathy; this was of undetermined significance.
The histological features are those of an acute vasculitis. There may be some variation from case to case in the extent of the infiltrate and the degree of leukocytoclasis, probably reflecting the stage of the lesion. There are usually some extravasated red blood cells; in cases of long standing, hemosiderin may be present. Intravascular hyaline deposits are the exception, but they may be found beneath areas of ulceration. A septal panniculitis is a rare association. Immunoglobulins and complement are often found in vessel walls by immunofluorescence.
The ultrastructural features depend on the involved immunoglobulins and on their respective quantities. There may be tubular microcrystals, filaments, or cylindrical and annular bodies. In a recently reported case of monoclonal cryoglobulinemia, immunoglobulin crystalloid structures were present within the small cutaneous vessels, admixed with fibrin and red blood cells.
Despite the possible clinical resemblance between lesions of perniosis and cryoglobulinemia, these diseases are not associated in adult patients. The microscopic findings are quite different, with perniosis showing perivascular lymphocytic infiltrates with evidence for lymphocytic vasculitis.
Embolism to the skin is no longer restricted to cholesterol crystals. It may involve exogenous material introduced during a procedure or consist of detached tumor fragments. Cutaneous involvement occurs in 35% of patients with cholesterol crystal embolization. The incidence was much higher in a study of cholesterol embolism. The source of the emboli is atheromatous plaques in major blood vessels, particularly the abdominal aorta. This material may dislodge spontaneously or after vascular procedures or anticoagulant therapy. There is a high mortality. Cutaneous lesions are found particularly on the lower limbs and include livedo reticularis, gangrene, ulceration, cyanosis, purpura, and cutaneous nodules. Rare manifestations have included erythematous and hyperpigmented papules and plaques on the trunk, a hemorrhagic panniculitis on the chest, and an eschar on the ear.
Multiple sections are sometimes required to find the diagnostic acicular clefts indicating the site of cholesterol crystals in arterioles and small arteries in the lower dermis or subcutis. A fibrin thrombus often surrounds the cholesterol material. Foreign body giant cells and a few inflammatory cells may also be present. Rarely, there are numerous eosinophils. Cutaneous infarction and associated inflammatory changes sometimes develop. Cholesterol emboli may be an incidental finding.
Deposits of atrial myxoma in peripheral arterioles and small arteries may at first glance be mistaken for cholesterol emboli. However, a loose myxoid stroma and lack of cholesterol clefting should allow a confident diagnosis of “metastatic” atrial myxoma.
Deposits of catheter tip can also form cutaneous emboli if fragments break off during a procedure. In one such case, purpuric lesions developed on the palm and fingers following percutaneous cardiac catheterization through the brachial artery.
Arterial embolization may follow the injection of hyaluronic acid used for tissue augmentation, and follow the use of acrylic cement during vertebroplasty.
The antiphospholipid syndrome is characterized by the presence of autoantibodies directed against phospholipids and associated with repeated episodes of thrombosis, fetal loss, and thrombocytopenia. These antibodies can be detected as a lupus anticoagulant, as anticardiolipin, or as anti-β 2 glycoprotein I–dependent antibodies. A seronegative category of antiphospholipid syndrome is now being recognized. Antibodies can be found in 20% to 50% of patients with SLE, but only approximately half of these patients have the antiphospholipid syndrome. The syndrome may also occur in association with rheumatoid arthritis, infections, dermographism, urticaria, certain drugs, and malignant disorders. The term primary antiphospholipid syndrome is used for patients who do not have an associated disease. Revised criteria for the diagnosis of antiphospholipid syndrome have been evaluated with particular application to patients presenting with dermatological symptoms. It has been suggested that anetoderma is a cutaneous marker for the antiphospholipid syndrome.
Patients may be asymptomatic or develop thrombosis with systemic and/or cutaneous disease. Skin lesions include livedo reticularis, Raynaud's phenomenon, thrombophlebitis, cutaneous infarction and gangrene of the digits, ulceration, subungual splinter hemorrhages, and painful skin nodules resembling vasculitis (but without vasculitis on biopsy). Antiphospholipid antibodies can be associated with Sneddon's syndrome (see later). Cutaneous necrosis can sometimes be quite extensive. Gangrene of the digits in a newborn was attributed to maternal antiphospholipid syndrome. There are isolated reports of its association with atrophie blanche, malignant atrophic papulosis (Degos), hyperhomocysteinemia (OMIM 603174 ), and factor V Leiden mutation.
It is not known why only a minority of patients with the autoantibodies develop thrombotic complications. Furthermore, the mechanisms by which the thromboses occur are poorly understood.
Thrombotic events should be treated with anticoagulants, initially heparin, followed by long-term warfarin titrated to an international normalized ratio (INR) of 2 to 3.
In early cutaneous lesions, there is prominent dermal edema and hemorrhage with thrombi in both arteries and veins. Some endothelial cells may have pyknotic nuclei, whereas others are swollen and disrupted. There is usually a mild lymphocytic infiltrate around involved vessels, and sometimes there are plasma cells as well. There is no vasculitis. In livedo reticularis lesions, thrombosis is not generally identified, except in catastrophic disease; instead, one may see vascular proliferation or endarteritis obliterans. Painful, small ulcers show fibrin deposition in and around dermal vessels and hyalinization of vessel walls.
In later lesions, there is organization of thrombi and subsequent recanalization. Reactive vascular proliferation develops. Some endothelial cells may be prominent and contain eosinophilic globules similar to those seen in Kaposi's sarcoma. Hemosiderin pigment is also present in late lesions. Some vessels may show intimal hyperplasia. This may reflect organization of earlier mural thrombi. A panniculitis has been described, manifesting as tender nodules on the legs. Microscopic features included a mixed septal and lobular panniculitis, with septal fibrosis, fat necrosis, mucin deposition, and an infiltrate composed of histiocytes, neutrophils, and eosinophils.
Resistance to activated protein C (APC), leading to a hypercoagulable state, occurs in 2% to 5% of the U.S. population. It is more common in some areas of Europe. In most patients, this APC resistance is caused by a single point mutation (Leiden mutation) involving a substitution of glutamine for arginine in the gene (F5) encoding factor V, situated at 1q23 (OMIM 227400 ). This mutation makes the activated form of factor V relatively resistant to degradation by APC. Other mutations causing APC resistance have been described.
The factor V Leiden mutation increases the risk of venous thrombosis up to 10-fold. It is an important cause of venous leg ulcers. The condition may enhance other pro-clotting defects or drugs to produce vascular thrombosis. In the presence of the mutation, trauma may produce leg ulcers. It has also been found that this mutation is increased in patients who present with leg ulcers caused by leukocytoclastic vasculitis. It has been suggested that the gene confers selective evolutionary advantages in those individuals who carry it—a likely explanation for its high prevalence in the community. In previous times, people did not live long enough for its adverse thrombotic effects to become manifest.
Sneddon's syndrome (OMIM 182410 ) is a rare neurocutaneous disorder of uncertain etiology, characterized by widespread livedo reticularis and ischemic cerebrovascular manifestations resulting from damage to small and medium-sized arteries. It may present with ulcers on the lower legs. It most commonly affects young and middle-aged women; it is rare in children. It has a chronic progressive course and a mortality rate of approximately 10%.
Detailed pathological studies by Zelger et al. have shown an “endothelitis” in affected vessels, followed by a subendothelial proliferation of smooth muscle cells and fibrosis. Vascular occlusion may occur (see later). Antiendothelial cell antibodies have been demonstrated in 35% of patients with Sneddon's syndrome, but their significance in the pathogenesis is uncertain. Possibly of greater relevance is the finding of similar intimal changes in thrombocythemia, suggesting that platelet-derived factors may also play a role in the pathogenesis of Sneddon's syndrome. Other factors appear to be important because Sneddon's syndrome can occur in association with the antiphospholipid syndrome (discussed previously) and with SLE. In one case, dysfibrinogenemia was present.
Treatment is often unsatisfactory. Rheological agents (heparin, warfarin, and coumarin) are usually tried first. These may be taken with immunosuppressive agents such as corticosteroids, azathioprine, or cyclophosphamide. Intravenous immunoglobulin (IVIG) has also been used. Intravenous recombinant tissue plasminogen activator has been used in one patient with Sneddon's syndrome and acute ischemic stroke, with subsequent improvement in neurological status.
The sensitivity of skin biopsies in Sneddon's syndrome has been evaluated. The white areas, rather than the red areas, should be biopsied. The sensitivity of one 4-mm biopsy is only 27%, but if three biopsies are taken, it increases to 80%. In the skin, small to medium-sized arteries near the dermal–subcutaneous junction are affected in a stage-specific sequence. The initial phase involves attachment of lymphohistiocytic cells to the endothelium with detachment of the endothelium (“endothelitis”). This is followed by partial or complete occlusion of the lumen by a plug of fibrin admixed with the inflammatory cells. This plug is replaced by proliferating subendothelial cells, which have the markers of smooth muscle cells. A corona of dilated small capillaries usually develops in the adventitia. Fibrosis of the intima or the plug may occur. Shrinkage and atrophy of these vessels then occurs.
CADASIL is the acronym used in preference to c erebral a utosomal d ominant a rteriopathy with s ubcortical i nfarcts and l eukoencephalopathy (OMIM 125310 ), a vascular disorder associated with migraines, recurrent ischemic strokes, and early-onset dementia. There is luminal obliteration of small leptomeningeal and intracerebral arteries but no vascular occlusion in the skin. Clinically apparent skin involvement is usually absent, but one case presented with hemorrhagic macules and patches. The defect involves a mutation in the NOTCH3 gene on chromosome 19q12, which encodes a transmembrane receptor protein. A commercial laboratory test for this gene is available.
Electron microscopic examination of skin biopsies is diagnostic. A granular, electron-dense, osmiophilic material is present in the basement membrane of vascular smooth muscle cells. Because the deposits may be patchy in superficial dermal vessels, several sections should be searched to avoid a false-negative result. Light microscopy is usually normal, but one case showed thickened and dilated vessels in the papillary dermis as a consequence of deposits of fibrin, complement, and immunoglobulins that were confirmed by immunofluorescence. A few perivascular lymphocytes were also present.
Rare causes of cutaneous microthrombi include the hypereosinophilic syndrome ; paroxysmal nocturnal hemoglobinuria ; renal failure with hyperparathyroidism (the vascular calcification–cutaneous necrosis syndrome) ; protein C and/or protein S deficiency ; increased activity of factor VIII coagulant ; cryofibrinogenemia ; dopamine, vasopressin, Depo-Provera, and heparin therapy (embolia cutis medicamentosa) ; the use of intravenous immunoglobulin, temazepam, or flunitrazepam ; the treatment of afibrinogenemia ; ulcerative colitis ; and embolic processes, including bacterial endocarditis. The eschar found in some rickettsial infections is a cutaneous infarct with fibrin-platelet thrombi in marginal vessels. A lymphocytic vasculitis is usually present as well.
Cutaneous microthrombi have also been associated with the intravascular injection of large amounts of cocaine. Moreover, there have been numerous recent descriptions of a cutaneous vasculopathy associated with levamisole-adulterated cocaine. Levamisole is a white powder used as a cutting agent in cocaine. In addition to expanding the volume of the material and thereby increasing profits, levamisole is said to increase the stimulatory and euphoric effects of this drug. Skin lesions include tender purpura, with necrosis and eschar formation, involving the ears, and a retiform purpura of the legs and elsewhere. Laboratory abnormalities include leukopenia and a subset of antineutrophil cytoplasmic antibodies (cytoplasmic ANCA; c-ANCA) directed toward human neutrophil elastase (HNE). Microscopic examination shows thrombosis and/or vasculitis ; the small vessel thrombi have a fibrinous appearance and may demonstrate cribriform recanalization. Direct immunofluorescence may show changes of vasculitis.
Vascular occlusion, either partial or complete, may occur in endarteritis obliterans as a result of fibrous thickening of the intima. It may be seen in a range of clinicopathological settings that include peripheral atherosclerosis, Raynaud's phenomenon, scleroderma, diabetes, hypertension, and healed vasculitis. Painful digital infarction may occur in patients with abnormalities in their coagulation factors that may mimic Buerger's disease, suggesting that this disease needs to be reappraised in light of our new knowledge of these various coagulation disorders.
Obstruction of the vena cava may produce cutaneous symptoms. The superior vena cava syndrome, which occurs when extrinsic compression or intraluminal occlusion impedes blood flow through the vessels, may present as persistent erythematous edema of the face. Obstruction of the inferior vena cava usually results from pelvic tumors, but a case related to factor V Leiden mutation has been reported. The patient presented with varicose and thrombosed veins on her abdominal wall.
Acrocyanosis of uncertain pathogenesis has been reported after the inhalation of butyl nitrite.
Although the differential diagnosis among possible causes of leukocytoclastic vasculitis or small vessel thrombosis is broad, the combination of these two changes raises three major considerations: levamisole-adulterated cocaine, septic vasculitis, and cryoglobulinemia.
Urticaria is a cutaneous reaction pattern characterized clinically by transient, often pruritic, edematous, erythematous papules or wheals that may show central clearing. Angioedema is a related process in which the edema involves the subcutaneous tissues and/or mucous membranes. It may coexist with urticaria. Both urticaria and angioedema may be a manifestation of anaphylaxis, a potentially life-threatening condition that may present with flushing and systemic symptoms. Both allergic and some physical urticarias may progress to anaphylaxis.
Urticaria is a common affliction, affecting 15% or more of the population on at least one occasion in their lifetime. Most cases are transient (acute), and the cause is usually detected. Infection was the most commonly documented cause of acute urticaria in one study. The use of antibiotics for viral illnesses is another cause. In one series, food allergies accounted for only 2.7% of acute urticarias in childhood. A variant of acute urticaria in infants and young children is the annular form that often results from the use of furazolidone in the treatment of diarrhea. On the other hand, chronic urticaria, which is less common than acute urticaria, is arbitrarily defined as urticaria persisting for longer than 6 weeks. It is idiopathic in approximately 75% of cases. It tends to involve middle-aged people, in contrast to acute urticaria, which occurs more commonly in children and young adults. When chronic urticaria does occur in children, the causal factors are more readily identifiable than in adults. In children, physical factors account for half of the cases of chronic urticaria. Chronic urticaria may be aggravated by salicylates, food additives, and the like. This has led to the suggestion that chronic urticaria results from an occult allergy to some everyday substance. An unusual variant of acute urticaria thought to be due to the sexual transfer of ampicillin from one person to his or her hypersensitive partner has been reported.
Papular urticaria is a clinical variant of urticaria in which the lesions are more persistent than the usual urticarial wheal. It may result from a hypersensitivity reaction to the bites of arthropods such as fleas, lice, mites, and bed bugs. It is more common in young children. It usually occurs in crops on exposed skin. Erosions are common. Rare clinical variants of urticaria include bullous and purpuric forms, urticaria with anaphylaxis, and recurrent urticaria with fever and eosinophilia.
Most of the literature on urticaria refers to the chronic form of the disease because this is often an important clinical problem. The remainder of this discussion refers to chronic urticaria.
Chronic urticaria is urticaria persisting longer than 6 weeks. There are many variants of chronic urticaria, although they share in common the presence of erythematous papules or wheals. Pruritus is often present; it is worse at night and in the evening. These variants are usually classified etiopathogenetically, as follows :
Physical urticarias
Cholinergic urticaria
Angioedema
Urticarias caused by histamine-releasing agents
IgE-mediated urticarias
Immune complex–mediated urticarias
Urticarial dermatitis
Schnizler's syndrome
Autoinflammatory urticarial syndromes
Idiopathic urticarias
Autoimmune chronic urticaria
Miscellaneous urticarias.
Some overlap exists between these various categories. For example, drugs and foodstuffs may produce chronic urticaria through more than one of the previous mechanisms. Patients with a physical urticaria may also develop an idiopathic urticaria. A good clinical history is essential in the categorization of some cases.
Physical stimuli such as heat, cold, pressure, light, water, and vibration are the most commonly identified causes of chronic urticaria, accounting for approximately 15% of all cases. The incidence was 33.2% in one series and 14% in another. The wheals are usually of short duration and limited to the area of the physical stimulus. Accordingly, the physical urticarias tend to occur on exposed areas. Angioedema may coexist. More than one of the physical agents listed previously may induce urticaria in some individuals. Some of the physical urticarias can be transferred passively. The physical urticarias undergo spontaneous remission in less than 20% of cases. They have the lowest remission rate of any of the chronic urticarias.
Cold urticaria, which accounts for 1% to 3% of all cases of urticaria, is produced at sites of localized cooling of the skin. It is usually acquired, although a rare familial form has been reported. It has now been identified as the familial cold, autoinflammatory syndrome (OMIM 120100 ) caused by a mutation in the CIAS1 gene on chromosome 1q44 (see p. 269 ). A variant with localized perifollicular lesions has been reported. Cold urticaria may follow a viral illness or the use of drugs such as penicillin, an angiotensin-converting enzyme (ACE) inhibitor, or griseofulvin. It has been reported in association with mycosis fungoides. A few cases are associated with the presence of cryoproteins. In most cases, no underlying cause is found. A history of atopy is sometimes present. Antihistamine receptor blockers show the best effect for treating this distressing condition. Patients should be advised that avoidance of cold exposure is the best prophylaxis. Coronary artery bypass grafting has been successfully performed in patients with cold urticaria using isothermic/normothermic blood cardioplegia.
Heat urticaria , by contrast, is exceedingly rare. There are now more than 50 reported cases of this disease. It is more common in women. It presents with an eruption of wheals at sites of exposure to heat. One patient developed wheals and loss of consciousness after taking a hot bath. H1 blockers are the treatment of choice.
Solar urticaria is another rare physical urticaria that results from exposure to sun and light. The prevalence of idiopathic solar urticaria in Tayside, Scotland, was estimated to be 3.1 per 100,000. Action spectra are typically broad, with 63% in one series ( n = 87) reacting to more than one wave band, and the most common provoking wavelengths were the longer UV-A and the shorter visible ones. The majority of patients are still affected after 10 years. Radiation transmitted through glass affects the majority of patients with this condition. One study showed a predominance of UV-A sensitization compared with visible light. An inhibition spectrum may be present, whereby activation occurs after exposure to a certain range of wavelengths, but not if reirradiation with certain other wavelengths is immediately carried out. Solar urticaria has a faster onset and shorter duration than polymorphic light eruption. Progression to or coexisting polymorphic light eruption is not uncommon. A rare variant, limited to fixed skin sites, has been reported in three patients. A similar fixed urticaria may rarely occur as a manifestation of a drug reaction. Solar urticaria has been induced by tetracycline therapy. It has been reported as a manifestation of Churg–Strauss syndrome. There is one reported case in which solar urticaria was limited to areas of bruising; evidence indicated that a serum factor was responsible.
Aquagenic urticaria follows contact with water of any temperature. Sometimes there is an underlying disorder, the expression of which may coincide with the onset of the urticaria. Examples include the myelodysplastic syndrome, the hypereosinophilic syndrome, HIV infection, and an underlying cancer. One case of aquagenic urticaria associated with occult papillary thyroid carcinoma improved after total thyroidectomy. A rare familial form (OMIM 191850 ) has been identified, and the condition has been described in monozygotic twins. Much more common is aquagenic pruritus, in which prickly discomfort occurs in the absence of any cutaneous lesion. It appears to be an entity distinct from aquagenic urticaria and may follow increased degranulation of a normal number of mast cells.
In pressure urticaria, deep wheals develop after a delay of several hours after the application of pressure. It is probably more common than generally realized, being present in some patients who do not report pressure-related wheals. It can affect quality of life. Systemic symptoms, which include an influenza-like illness, may also develop. A bullous form with intraepidermal bullae filled with eosinophils has been reported. Many eosinophils were also in the dermis. Eosinophil granule major basic protein and neutrophil granule elastase are significantly increased in delayed pressure urticaria, in a similar manner to the IgE-mediated late phase reaction. There is no increase of these products in dermographism. Other abnormalities include elevated interleukin-6 (IL-6), C-reactive protein, and plasma D-dimer concentration, the latter possibly a result of the systemic inflammatory response. It usually responds poorly to treatment, but tranexamic acid produced resolution in one reported case. Vibratory urticaria is a related disorder resulting from vibratory stimuli.
Dermographism, which is the production of a linear wheal in response to a scratch, is an accentuation of the physiological whealing of the Lewis triple response. It has followed trauma from a coral reef and has been reported in patients with adult-onset Still's disease. Minor forms of dermographism are quite common, but only a small percentage of affected persons are symptomatic with pruritus. A delayed form, which is an entity similar to delayed pressure urticaria, also occurs. Galvanic urticaria follows the administration of an electrical current given during iontophoresis.
The physical urticaria that is least well understood is contact urticaria . In this variant, a wheal and flare response is usually elicited 30 to 60 minutes after skin contact with various chemicals in medicaments, cosmetics, foods, and industrial agents. Contact with cosmetics appears to be responsible for the persistent erythema and edema of the mid-third and upper aspect of the face, called morbus morbihan by French dermatologists. It may follow exposure to the common stinging nettle (Urtica dioica) and a long list of other substances, including latex, flavored toothpaste, insect repellent, formaldehyde, cornstarch powder, apples, tofu, topical immunotherapy with diphenylcyclopropenone, petrol, and kerosene. Contact urticaria may follow the application of p -chloro- m -cresol, a preservative used in a number of topical preparations. Distinction from a contact irritant dermatitis may sometimes be difficult. An IgE-mediated form and a nonimmunological form have been delineated. Contact urticaria may be superimposed on an eczematous dermatitis of different types. A recurrent urticaria has been reported at the previous injection sites of insulin. Contact sensitization appears to play an important role in other types of chronic urticaria. In a study of 121 patients with chronic urticaria, 50 (41%) tested positive to contact allergens. In all patients, avoidance measures led to a complete remission within 1 month. In an Australian study of occupational contact urticaria, natural rubber latex accounted for the majority of cases. Other common causes were foodstuffs and ammonium persulfate utilized as hairdressing bleach. Atopy was a significant risk factor.
Cholinergic urticaria is produced by exercise, heat, and emotion, with general overheating of the body as the final common pathway. Accordingly, it is sometimes included with the physical urticarias, with which it may coexist. Young adults are mainly affected, but the condition is usually mild, not requiring medical attention. Adrenergic urticaria is a related condition that is also caused by stress. It is distinguishable from cholinergic urticaria because each papule in adrenergic urticaria is surrounded by a striking white halo of blanched vasconstricted skin.
We have seen several cases of cholinergic urticaria in medical practitioners, and others, after a game of golf. Marathon runners may also experience a cholinergic urticaria. Both groups, as well as patients participating in exercise, may experience a vasculitis instead of urticaria. Angioedema and anaphylaxis are other manifestations of exercise.
An attempt has been made to classify cholinergic urticaria into four subtypes: with poral occlusion, with acquired generalized hypohidrosis, with sweat allergy, and idiopathic. A subtype of generalized anhidrosis—idiopathic pure sudomotor failure—begins at an early age, shows sudden onset, and is accompanied by generalized pain or cholinergic urticaria and an elevated serum IgE level. The lesions of cholinergic urticaria are distinctive and consist of 2- to 3-mm wheals surrounded by large erythematous flares. There is a predilection for blush areas. Increased sympathetic activity may result in the release of acetylcholine at nerve endings, causing mast cells to degranulate.
Angioedema refers to abrupt and short-lived swelling of the skin, mucous membranes, or both, including the upper respiratory and intestinal epithelial linings. It shows a predilection for areas where the skin is lax, especially the face and genitalia. It should be distinguished from lymphedema. Angioedema results from an increase in permeability of capillaries and postcapillary venules, resulting from either mast cell degranulation or activation of kinin formation. The former group (mast cell degranulation), which includes those caused by NSAIDs and autoimmune mechanisms, is usually accompanied by urticaria, but the “kinin group” caused by ACE inhibition of bradykinin degradation is not accompanied by hives (see later). In most cases, the NSAID-induced urticaria/angioedema is pharmacological, involving inhibition of cyclooxygenase (COX), a rate-limiting enzyme in the biosynthesis of prostaglandins. COX-2 inhibitors do not induce urticaria in these patients.
Angioedema is often seen at the injection site of granulocyte-monocyte colony-stimulating factor (GM-CSF). It may also be associated with the use of ACE inhibitors, the ingestion of certain foods, and intrauterine infection with parvovirus B19. Angioedema induced by ACE inhibitors is due to a mutation in the XPNPEP2 gene, which encodes aminopeptidase P. The gene is on chromosome Xq25 (OMIM 300145 ).
There is a special form of angioedema (hereditary angioedema) that may be associated with swelling of the face and limbs or involve the larynx with potentially life-threatening consequences. It results from an absolute or functional deficiency of C1 esterase inhibitor. There are three types of hereditary angioedema. Type I (OMIM 106100 ) accounts for 85% of cases. It is caused by mutations in the C1 inhibitor gene C1NH (also known as SERPING 1 ) on chromosome 11q11–q13.1. Serum levels of the gene product C1NA are less than 35% of normal. In type II, the serum levels are normal or elevated but the protein is nonfunctional. The two types are clinically indistinguishable. The third variant (type III; OMIM 610618 ) is caused by a mutation in the gene encoding coagulation factor XII (Hageman factor), which is at 5q35.2–q35.3. Misdiagnosis of hereditary angioedema is not uncommon. It results from unchecked bradykinin formation. Acquired deficiencies of C1 esterase inhibitor may be seen in lymphoma and certain autoimmune connective tissue diseases. In one case, there was an associated leukocytoclastic vasculitis. Treatment with danazol has been effective in many cases of hereditary angioedema.
Histamine may be released by mast cells in response to certain drugs, such as opiates, and some foodstuffs, including strawberries, egg white, and lobster. Familial aggregation of aspirin/NSAID-induced urticaria has been reported. Mutations in the leukotriene C4 synthase ( LTC4S ) gene (OMIM *246530) and the glutathione S-transferase M1 (GSTM1) gene (OMIM *138350) have been found in these patients.
Antigen-specific IgE may be responsible for some of the urticarias caused by certain foods, drugs, and pollens in addition to the urticaria related to parasitic infestations and stings. More than 300 causes have been identified. Mast cell degranulation is the final common pathway. Total serum IgE levels are often elevated in patients with chronic urticaria, correlating with severity and duration of the condition.
Immune complexes may be involved in the pathogenesis of the chronic urticaria seen in infectious hepatitis, infectious mononucleosis, SLE, and serum sickness–like illnesses. They have also been implicated in the urticarial reactions that are an uncommon manifestation of various internal cancers. In some instances, fever, purpura, and joint pains are also present. The urticaria is usually more persistent, particularly in those cases with an accompanying vasculitis. A serum sickness–like reaction has been seen as a complication of cefaclor, an oral cephalosporin.
The term urticarial dermatitis was introduced by Kossard and colleagues for a clinicopathological entity with urticarial and eczematous features that presented with erythematous and pruritic lesions. Some cases resembled a fixed urticaria. Similar cases have been reported as urticarial papulosis. Kossard believes that urticarial dermatitis is a subset of dermal hypersensitivity reaction, a term that has been disputed by many but to which there have been a number of converts (see p. 1174 ). It has also been regarded as a group of skin diseases that share clinical and histopathological manifestations. Patients tend to be late middle-aged or older (mean age 70 years in the study by Garcia del Pozo et al. ; 60 years in that of Hannon et al. ). The pruritus has been considered significant and, in some cases, intolerable. In the Hannon study, 4 of 40 patients developed a malignancy within 4 months of the onset of urticarial dermatitis. The usual treatments for this and similar conditions (antihistamines, topical and systemic corticosteroids, narrowband UV-B, topical calcineurin inhibitors) are often unsatisfactory. However, other agents that have been used include cyclosporine, mycophenolate mofetil, azathioprine, dapsone, and hydroxyurea. Microscopic findings include mild spongiosis and a perivascular papillary dermal infiltrate containing lymphocytes and variable numbers of eosinophils.
Schnitzler's syndrome combines nonpruriginous chronic urticaria, monoclonal IgM gammopathy, fever, arthralgias, and disabling bone pain. Monoclonal IgG has been present in other cases. An association with cold urticaria has been reported. The link between these disparate disorders is unknown, but there are some similarities with the autoinflammatory syndromes (see later). Treatment has usually been with systemic corticosteroids, with cyclosporine (ciclosporin), chlorambucil, or cyclophosphamide used in an attempt to avoid the overuse of corticosteroids. Antihistamines and NSAIDs have also been used. Pefloxacin, given for a concurrent urinary tract infection, produced dramatic and sustained improvement. Its use in other patients with the syndrome produced improvement in most cases. The anti-CD20 monoclonal antibody rituximab has also controlled symptoms. This treatment failed in another patient who subsequently had a response to anakinra, an IL-1 receptor antagonist. Response to this agent supports the concept that dysregulation of the IL-1 pathway is involved in the pathogenesis of the disease. Other successful reports have followed.
There are several familial urticarial disorders of early onset associated with mutations in the CIAS1 gene on chromosome 1q44, controlling the formation of cryopyrin. They are included in the broader group of autoinflammatory diseases, also known as the periodic fever syndromes (see p. 269 ). The three autoinflammatory diseases in which urticaria is a major feature are familial cold autoinflammatory syndrome (OMIM 120100 ); Muckle–Wells syndrome (OMIM 191900 ), in which progressive deafness is also present, as well as amyloidosis in approximately one-third of affected patients; and neonatal-onset multisystem inflammatory disorder (NOMID, CINCA (OMIM 607115 )). Hyperpigmented sclerodermoid lesions have been reported in Muckle–Wells syndrome.
There are some similarities between these syndromes and cases reported as familial cold urticaria; it was described before the recognition of these cryopyrin-associated syndromes, and they may be the same disorder. The histology is variable, but there may be a heavy interstitial, perivascular, and periadnexal infiltrate of neutrophils that is closer to a neutrophilic dermatosis than a neutrophilic urticaria.
Up to 75% of all chronic urticarias fall into the idiopathic category. It accounted for 36% of cases in a series of 220 patients reported in 2001. Nearly half of the cases experienced spontaneous remission of their urticaria. Up to 50% of patients with idiopathic urticaria have histamine-releasing autoantibodies in their blood that act against the IgE and FcεRIα receptors. Such cases should be recategorized as autoimmune chronic urticaria (see later). Angioedema is present in approximately 40% of cases.
There is now strong evidence for an autoimmune basis for a group of patients with chronic urticaria, previously classified as idiopathic urticaria. These patients have functional autoantibodies against the α chain of high-affinity IgE receptor (FcεRIα). An additional 10% to 15% of patients with idiopathic urticaria have antibodies against the IgE molecule. It is difficult to distinguish the autoimmune group from the idiopathic group, although the autologous serum skin test (ASST) is a useful screening test for “autoreactive” diseases, not autoantibodies. The CD63 expression assay (a modified serum-induced basophil activation test) seems to be a reliable test in the diagnosis of autoimmune chronic urticaria. There is an association with autoimmune thyroid disease in this form of urticaria.
An urticarial reaction is sometimes seen in individuals who are infected with Candida albicans ; the role of this organism in the causation of urticarias has been overstated in the past. Recently, Helicobacter pylori gastritis has been reported in some patients with chronic urticaria, but its etiological significance has since been challenged. Urticaria may be a manifestation of autoimmune progesterone dermatitis (see p. 149 ), a disorder that is usually manifest some days before the menses. The mechanism is probably not an autoimmune reaction, despite the title. An estrogen-induced urticaria (see p. 150 ) also occurs.
Other published causes of urticaria include hepatitis C infection ; dental infection ; IL-3 therapy ; gelatin in a vaccine ; hepatitis B immunization ; rabies vaccine ; ethanol ; drugs such as nonsteroidal antiinflammatory agents, bupropion, povidone in a paracetamol preparation, methylprednisolone (intraarticular), bleomycin, polidocanol, cetirizine, alendronate, and minocycline ; nicotine in tobacco smoke ; kava ; ovarian cancer, multiple piloleiomyomas, lymphoma, colonic adenocarcinoma, and carcinoid tumor ; and the parasites Anisakis sp., Blastocystis hominis, and Hymenolepis nana. It has also followed contact with hedgehogs, ants, and caterpillars. It has been associated with thyroid autoimmunity, adult Still's disease, and the chronic, infantile, neurological, cutaneous and articular syndrome (CINCA). Common variable immunodeficiency may present as a chronic urticaria. Angioedema and urticaria have been associated with the use of isotretinoin, topical imiquimod, and sodium benzoate. Urticaria may develop at a site distal to the application of diphencyprone for resistant viral warts. Acute urticaria has followed the use of “gomutra” (cow's urine) gargles.
Recall urticaria consists of urticaria in a previously injected site when antigens enter from another source such as orally.
Urticaria results from vasodilatation and increased vascular permeability associated with the extravasation of protein and fluids into the dermis. Angioedema results when a similar process occurs in the deep dermis and subcutis. Histamine has generally been regarded as the mediator of these changes, although other mediators such as prostaglandin D 2 and IL-1 are possibly involved in some circumstances. IL-1 and other cytokines can induce the expression of endothelial adhesion molecules, which is upgraded in urticarial reactions. Both immunological (type I and type III) and nonimmunological mechanisms can cause mast cells and basophils to degranulate, liberating histamine and other substances. Although immediate (type I), IgE-mediated mast cell release is the classic underlying mechanism of urticarial reactions, there is evidence that delayed-type hypersensitivity reactions are often involved as well. For example, the cytokine IL-4 can upregulate IgE secretion, whereas the opposite occurs with IFN. There is also an increase in IL-10-secreting T cells in chronic idiopathic urticaria. IL-18 is not increased compared with controls. The neuropeptide substance P, derived from unmyelinated sensory nerve endings, can also evoke the release of histamine, but not prostaglandin D 2 , from cutaneous mast cells. ACE is one of the major peptidases for the degradation of substance P, but polymorphisms in the ACE gene do not appear to increase the risk of developing chronic urticaria, although they can be a contributing factor to susceptibility of angioedema accompanying chronic urticaria. Eosinophil degranulation occurs in most urticarias. The number of free eosinophil granules correlates with the duration of the wheal; granules are numerous in wheals of long duration. The granules may provoke the persistent activation of the mast cells in the wheal.
Other research has been directed at aspects of urticaria not necessarily involving mast cells in the first instance. It has been found that oxidative stress is common to the physical urticarias. Basophils have become the center of attention once again in chronic urticaria. In addition to the basopenia, intrinsic defects of the anti-IgE cross-linking signaling pathway of basophils have been described. Basophils in these patients have an activated profile, possibly because of an in vivo priming by IL-3.
Various studies have shown that approximately one-third or more of patients with chronic urticaria have circulating autoantibodies to FcεRIα (the high-affinity IgE receptor), resulting in the release of histamine from mast cells. This subset of patients (autoimmune chronic urticaria) has more severe disease. An association with HLA class II has been reported. The beneficial effect of cyclosporine and IVIG on chronic urticaria provides further support for the role of histamine-releasing autoantibodies in its pathogenesis. Extensive laboratory investigations do not contribute substantially to the diagnosis of chronic urticaria or the detection of underlying disorders.
In summary, several different mechanisms have been elucidated that are capable of stimulating the release of histamine from mast cells and/or basophils.
An excellent evidence-based protocol on the management of urticaria has been published by the British Association of Dermatologists. Practitioners should be aware of dosing, side effects, and potential interactions of the drugs mentioned. A nonsedating H1 antihistamine is the treatment of choice, although not all patients respond. A sedating antihistamine is sometimes used at bedtime. One study demonstrated the effectiveness of the monoclonal antibody, omalizumab, for physical urticarias. This agent selectively binds to free IgE. Rapid desensitization to autologous sweat has been used in the treatment of cholinergic urticaria. Other agents used in varying circumstances include corticosteroids, cyclosporine, intravenous immunoglobulin, mycophenolate mofetil, doxepin, scopolamine (in cholinergic urticaria), methotrexate, sulphasalazine, antibiotics, and anticoagulants (for the prevention of NSAID-induced urticaria.
The cell type and the intensity of the inflammatory response in urticaria are quite variable. There is increasing evidence that the age of the lesion biopsied and the nature of the evoking stimulus may both influence the type and the intensity of the inflammatory response.
Dermal edema, which is recognized by separation of the collagen bundles, is an important feature of urticaria. Mild degrees may be difficult to detect. Urticarial edema differs from the mucinoses by the absence of granular, stringy, basophilic material in the widened interstitium. There is also dilatation of small blood vessels and lymphatics, and swelling of their endothelium is often present ( Fig. 9.5 ). The histopathological changes in urticaria are most marked in the upper dermis, but involvement of the deep dermis may be present, particularly in those with coexisting angioedema.

The cellular infiltrate in urticaria is usually mild and perivascular in location. It consists of lymphocytes and, in most cases, a few eosinophils. Occasional interstitial eosinophils and mast cells are also present. Eosinophil granule major basic protein has been identified in the dermis in several types of urticaria. Neutrophils are often noted in early lesions, but in most cases they are relatively sparse; there is evidence that they are more prominent in the physical urticarias, particularly delayed pressure urticaria. A recent study of this form of urticaria, elicited by a pressure-challenge test, emphasized the prominence of eosinophils. Eosinophil extracellular traps have been identified in bullous delayed pressure urticaria using confocal scanning laser microscopy. These structures contain mitochondrial DNA and eosinophil cationic protein and can be found using dual-labeling fluorescent methods. They are believed to constitute a primitive antibacterial defense mechanism. An important diagnostic feature of many early urticarias is the presence of neutrophils and sometimes eosinophils in the lumen of small vessels in the upper dermis. The presence of neutrophils in other circumstances is discussed later. Although mast cells are often increased in early lesions and even in nonstimulated skin of patients with chronic urticaria, they appear to be decreased in late lesions, apparently as a result of the failure of histochemical methods to detect degranulated mast cells.
Neutrophils have been described in urticaria in several different circumstances. As previously noted, a few intravascular and perivascular neutrophils are a common feature in early urticaria. At times, there may be sufficient transmigration of neutrophils through vessel walls to give a superficial resemblance to vasculitis, but there is no fibrinoid change, hemorrhage, or leukocytoclasis. A more diffuse dermal neutrophilia has also been described in nearly 10% of urticarias. The term neutrophilic urticaria is used in these circumstances. In these cases, neutrophils are scattered among the collagen bundles, usually in the upper dermis but sometimes throughout its thickness. The infiltrate is usually mild in intensity. Rare nuclear “dusting” may also be present. Interstitial eosinophils and perivascular eosinophils and lymphocytes are usually noted as well. Neutrophils are also present in the leukocytoclastic vasculitis that sometimes accompanies chronic urticaria. The term urticarial vasculitis has been applied to cases of this type in which the clinical picture is that of an urticaria and the histopathology is a vasculitis. In urticarial vasculitis (also see p. 260 ) the urticarial lesions are usually of longer duration than in the usual chronic urticaria and may be accompanied by systemic symptoms. The incidence of vasculitis in chronic urticaria varies with the strictness of the criteria used for defining vasculitis. There appears to be a continuum of changes, with a few intravascular and perivascular neutrophils at one end of the spectrum and an established leukocytoclastic vasculitis at the other. In a large series of patients with urticaria subjected to biopsy, 57% were found to have “neutrophilic urticaria.” This high percentage was attributed to the tendency to biopsy lesions of new onset. There was no difference in prevalence of rheumatic disease among those with neutrophilic compared with conventional urticaria.
Zembowicz and colleagues examined 16 skin biopsies from patients with aspirin-induced chronic idiopathic urticaria. A classic urticarial pattern occurred in the majority of cases (12 of 16). Two biopsies showed an interstitial fibrohistiocytic (granuloma annulare–like) pattern, one showed a sparse perivascular lymphocytic infiltrate, and the other showed a paucicellular dermal mucinosis.
Other histopathological features have been described in several of the specific types of urticaria. In dermographism, perivascular mononuclear cells are increased, even before the initiation of a wheal. In contact urticaria, subcorneal vesiculation and spongiosis have been reported, but this may simply reflect a concomitant allergic or irritant contact dermatitis. In papular urticaria, the inflammatory cell infiltrate is usually heavier than in other chronic urticarias and consists of a superficial and deep infiltrate of lymphocytes and eosinophils in a perivascular location. The infiltrate is often wedge shaped. Interstitial eosinophils may also be present, and in lesions of less than 24 hours’ duration there are also some neutrophils. There is variable subepidermal edema. In angioedema , the edema and vascular dilatation involve the deep dermis and/or subcutis, although in the hereditary form there is usually no infiltrate of inflammatory cells. It is also quite mild in other forms of the disease. In Schnitzler's syndrome, there are often neutrophils in the dermal infiltrate in a perivascular location mimicking a neutrophilic urticaria. Monoclonal IgM and its autoantibody deposit in the epidermis and at the dermoepidermal junction in the region of the anchoring fibrils. One case of cholinergic urticaria associated with hypohidrosis showed partial occlusion of the intraepidermal portion of an eccrine sweat duct by keratinous material, with proximal ductal dilatation and periductal lymphocytic infiltration.
There are platelets and other cells in the lumen of mildly dilated vessels in the upper dermis. Lymphocytes and dendritic cells are close to the vessels. Mast cells may be normal or degranulated. Using immunoelectron microscopy, tryptase and factor XIIIa have been found in the superficial nerves of patients with drug-induced acute urticaria.
When urticarial lesions show perivascular lymphocytic infiltrates, the differential diagnosis can be quite broad and includes polymorphic light eruption, drug-induced exanthem, arthropod reaction, viral exanthem, and id reaction. Although the presence of eosinophils often narrows the focus somewhat to drug or arthropod reaction, clearly eosinophils appear in urticaria due to a variety of factors, including (as noted previously) delayed pressure, and they may be entirely absent in reactions to arthropods or drugs. The finding of diffuse neutrophils with mild perivascular inflammation also raises the possibility of cellulitis or erysipelas; bacteria are typically difficult to identify in tissue sections of those lesions. Neutrophilic urticarias usually lack the other features commonly associated with true vasculitis: endothelial swelling, leukocytoclasis, extravasated erythrocytes, and fibrin deposition. If those features are found in varying combinations, one should suspect urticarial vasculitis. Direct immunofluorescence may then be helpful in that vasculitic lesions show immunoglobulin and complement deposition in vessel walls. A recently described group of disorders termed neutrophilic urticarial dermatosis can also display leukocytoclasia; disorders showing these changes include SLE, Still's disease (juvenile rheumatoid arthritis), and Schnitzler's syndrome. Spongiotic changes should ordinarily argue against a diagnosis of urticaria, unless another process can be invoked (e.g., superimposed allergic contact dermatitis or secondary eczematization of an urticarial lesion). In addition, both papular urticaria and urticarial dermatitis may show spongiosis. A variety of other dermatoses may well have an urticaria-like dermal component and yet not represent a primary form of urticaria. A variant of follicular mucinosis has been described that has clinical resemblances to urticaria but characteristic features of follicular mucinosis on biopsy.
The term vasculitis refers to a heterogeneous group of disorders in which there is inflammation and damage of blood vessel walls. It may be limited to the skin or some other organ or be a multisystem disorder with protean manifestations. Numerous classifications of vasculitis have been proposed, but there is still no universally acceptable one. Even the Chapel Hill Consensus Conference has failed to come up with a problem-free classification. Several reviews of cutaneous vasculitis have been published.
Vasculitis can be classified on an etiological basis, although in approximately 50% of cases there is no discernible cause. Furthermore, a single causal agent can result in several clinical expressions of vasculitis. A modified etiological classification, based in part on pathogenesis but taking into account unique clinical and microscopic features, is shown in Table 9.1 . The major shortcoming of this type of classification is that it ignores the close interrelationship of the various components of the immune system, more than one of which is likely to be involved in some of the conditions. Another approach to the classification of the vasculitides has been on the basis of the size and type of blood vessel involved because this correlates to some extent with the cutaneous manifestations. For example, small or medium-sized arteries are involved in polyarteritis nodosa, Kawasaki disease, and nodular vasculitis, whereas large arteries are involved in giant cell (temporal) arteritis and Takayasu arteries and large veins are involved in thrombophlebitis. Both small and large vessels are involved in rheumatoid arthritis (RA) and certain “collagen diseases.” However, most cases of cutaneous vasculitis involve small vessels, particularly venules. The classification to be adopted here is based on the nature of the inflammatory response, with three major categories: acute (neutrophilic) vasculitis, chronic lymphocytic vasculitis, and vasculitis with granulomatosis ( Fig. 9.6 ). A fourth category, the neutrophilic dermatoses, is included, although most of the diseases in this group have been regarded at various times as instances of acute (neutrophilic) vasculitis. There is an infiltration of neutrophils in the dermis, but there is usually no fibrin extravasation into the vessel wall, a cardinal feature of acute vasculitis.
| Type of vasculitis | Causes/comments |
|---|---|
| Leukocytoclastic vasculitis | Generally neutrophilic; immune complex mediated |
| Henoch–Schönlein purpura | |
| Drug | |
| Infectious diseases | |
| Connective tissue diseases | |
| Malignancy | |
| Acute hemorrhagic edema | Infants; follows infections or vaccination; does not show IgA deposition |
| Microscopic polyangiitis | p-ANCA |
| Purpura hyperglobulinemica (Waldenström) | Associations with Sjögren's syndrome, lupus erythematosus; antibodies to Ro/SSA |
| Urticarial vasculitis | Causes: drug, connective tissue disease, viral |
| Leukocytoclastic vasculitis with thrombosis | |
| Septic vasculitis (gonococcemia, meningococcemia) | |
| Cryoglobulinemia | |
| Use of levamisole-adulterated cocaine | |
| Chronic forms of leukocytoclastic vasculitis | |
| Granuloma faciale | |
| Erythema elevatum diutinum | |
| Large vessel vasculitis | |
| Buerger's disease (thromboangiitis obliterans) | Involves small and medium arteries and veins |
| Nodular vasculitis | Involves subcutaneous arteries and veins |
| Polyarteritis nodosa | Cutaneous and systemic |
| Superficial thrombophlebitis | |
| Mondor's disease | |
| Eosinophilic vasculitis | With hypereosinophilic syndrome or connective tissue disease |
| Lymphocytic vasculitis | |
| Connective tissue diseases (e.g., lupus erythematosus) | |
| Pityriasis lichenoides | |
| Pigmented purpuric dermatosis | |
| Perniosis | |
| Infectious exanthems | For example, Rocky Mountain spotted fever |
| Granulomatous vasculitis | |
| Wegener's granulomatosis | c-ANCA |
| Churg–Strauss syndrome (allergic granulomatosis) | p-ANCA; occasionally c-ANCA |
| Giant cell (temporal) arteritis |
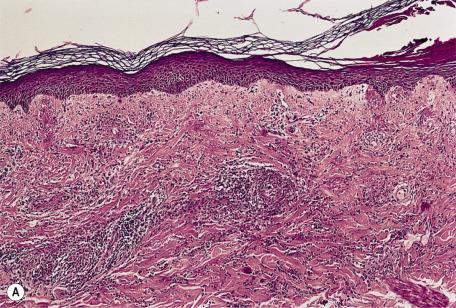
The following classification has some shortcomings because vasculitis is a dynamic process, with the evolution of some acute lesions into a chronic stage. Furthermore, lesions are sometimes seen in which there are features of both acute and chronic vasculitis. Whether this represents a stage in the evolution of the disease or a change de novo is debatable. Each category of vasculitis can be further subdivided into a number of clinicopathological entities. In the case of acute vasculitis, they are as follows:
Leukocytoclastic (hypersensitivity) vasculitis
Henoch–Schönlein purpura
Eosinophilic vasculitis
Rheumatoid vasculitis
Urticarial vasculitis
Mixed cryoglobulinemia
Hypergammaglobulinemic purpura
Hyperimmunoglobulinemia D syndrome
Septic vasculitis
Erythema elevatum diutinum
Granuloma faciale
Localized chronic fibrosing vasculitis
Microscopic polyangiitis (polyarteritis)
Polyarteritis nodosa
Kawasaki syndrome
Superficial thrombophlebitis
Sclerosing lymphangitis of the penis
Miscellaneous associations.
The term leukocytoclastic vasculitis has been chosen for this group of vasculitides in place of the equally unsatisfactory term hypersensitivity vasculitis . Common usage has influenced the decision to make this change. To be cynical, if dermatomyositis can be diagnosed in the absence of myositis, then perhaps leukocytoclastic vasculitis can be diagnosed in the absence of leukocytoclasis, this being the situation in at least 20% of biopsies of otherwise typical cases. Other diagnoses that have been applied to this condition include allergic vasculitis, hypersensitivity angiitis, and necrotizing vasculitis. Necrotizing is a particularly inappropriate group designation. Like leukocytoclasis, necrosis is not invariably present and is usually applied indiscriminately to cases with minimal fibrin extravasation.
Leukocytoclastic vasculitis usually presents with erythematous macules or palpable purpura, with a predilection for dependent parts, particularly the lower parts of the legs. Other lesions that may be present include hemorrhagic vesicles and bullae, nodules, crusted ulcers, and, less commonly, livedo reticularis, pustules, or annular lesions. Segmental leukocytoclastic vasculitis has accompanied herpes zoster. Nodular angiomatoid lesions have been described in a patient with IgA-λ myeloma. An erythema gyratum repens–like eruption occurred in one patient. Cases with urticarial lesions are classified separately as urticarial vasculitis (see later). Individual lesions vary from 1 mm to several centimeters in diameter. Large lesions are sometimes painful. There may be a single crop of lesions that subside spontaneously after a few weeks or crops of lesions at different stages of evolution that may recur intermittently. Unilateral and localized lesions are rare. Koebnerization is also surprisingly uncommon. Interestingly, a “reverse” Koebner phenomenon has also been described: disappearance of vasculitic lesions where a pressure bandage had been applied to a skin biopsy site.
Extracutaneous manifestations occur in approximately 20% of affected individuals and include arthralgia, myositis, low-grade fever, and malaise. In a retrospective series of 93 adult patients reported in 2006, extracutaneous involvement was found in 39.8% of patients. Less commonly, there are renal, gastrointestinal, pulmonary, or neurological manifestations. Bilateral acute angle closure glaucoma was the presenting feature of a case of systemic leukocytoclastic vasculitis. The severity of the histopathological changes is not predictive of extracutaneous involvement. Many of these cases with systemic manifestations would now be classified as microscopic polyangiitis (polyarteritis) (see p. 265 ), although renal involvement is still associated with cutaneous small vessel vasculitis. There is a low mortality in leukocytoclastic vasculitis; death, when caused by the vasculitis, is related to involvement of systemic vessels. In one study of 160 patients with leukocytoclastic vasculitis, the mortality rate was 1.9%.
There are several different etiological groups, although in approximately 40% of cases there is no apparent cause. The recognized groups include infections, drugs and chemicals, cancers, and systemic diseases. In a retrospective study of 93 adult patients (referred to previously), no obvious cause was found in 44.1%. Drugs and infections were the most common cause. Miscellaneous causes include certain arthropod bites, severe atherosclerosis, gallbladder vasculitis, prolonged exercise, some coral ulcers, and coronary artery bypass surgery. Exercise-induced vasculitis can occur in marathon runners, in women after long walks, and in golfers. Sometimes only an urticaria is present. A deep leukocytoclastic vasculitis has been found in cases of lower extremity inflammatory lymphedema, an exquisitely tender condition occurring in otherwise healthy adult military trainees during the first 72 hours of basic training and associated with prolonged standing at attention.
Streptococcal infection of the upper respiratory tract is the most commonly implicated infection. A streptococcal bullous erysipelas has also been associated with a vasculitis. Other infections include influenza, Mycobacterium tuberculosis , M. haemophilum , hepatitis B, herpes simplex, herpes zoster/varicella, human herpesvirus 6 (HHV-6), cytomegalovirus, parvovirus infection, HIV infection, malaria, and scrub typhus, with disseminated intravascular coagulation, caused by Orientia tsutsugamushi . It has followed influenza immunization and envenomation of the brown recluse spider (Loxosceles reclusa).
The drugs that cause vasculitis are listed in Table 9.2 . Chemicals used in industry, drug and food additives, and hyposensitizing antigens are also linked to leukocytoclastic vasculitis. The cancers associated with leukocytoclastic vasculitis include lymphomas (e.g., angioimmunoblastic T-cell lymphoma), diffuse large B-cell lymphoma, myelodysplastic syndrome, mycosis fungoides, hairy cell leukemia, chronic lymphocytic leukemia, multiple myeloma, IgA-λ monoclinal gammopathy, and visceral tumors such as gastric and colonic adenocarcinoma, pleural mesothelioma, squamous cell carcinoma of the lung, and hypernephroma.
| Adalimumab | Intravenous drug abuse |
| Allopurinol | Letrozole |
| Alprenolol | Levamisole |
| Amiodarone | Mefloquine |
| Ampicillin | Metformin |
| Aspirin | Methimazole |
| Atenolol | Naproxen |
| Black cohosh | Nicotine patch |
| Bortezomib | Orlistat |
| Cannabis | Oxprenolol |
| Carvedilol | Paroxetine |
| Cefazolin | Penicillin |
| Celecoxib | Phenothiazines |
| Cephalosporins | Phenylbutazone |
| Chlorzoxazone | Phenytoin |
| Cimetidine | Potassium iodide |
| Clindamycin | Practolol |
| Cytarabine | Procainamide |
| Disulfiram | Propranolol |
| Dronedarone | Propylthiouracil (some cases are ANCA positive) |
| Erlotinib | Quinidine |
| Erythromycin | Rifampicin |
| Escitalopram | Rituximab |
| Etanercept | Rivaroxaban |
| Etoposide | Rofecoxib |
| Famciclovir | Sertraline |
| Fluoxetine | Sibutramine |
| Furosemide (frusemide) | Sorafenib |
| G-CSF | Sotalol |
| Gefitinib | Sulfonamides |
| Gold | Sunitinib |
| Griseofulvin | Tamoxifen |
| Haloperidol | Tattoo pigments |
| Indinavir | Thiazides |
| Indomethacin | Thiouracil |
| Infliximab | Valproic acid |
| Insulin | Vancomycin |
| Interferon-α (injection site) | Warfarin |
The systemic diseases include SLE, dermatomyositis, generalized morphea, mixed connective tissue disease, Behçet's disease, celiac disease, inflammatory bowel disease, cystic fibrosis, Sjögren's disease, subcorneal pustular dermatosis, pyoderma gangrenosum, sarcoidosis, the Wiskott–Aldrich syndrome, and α 1 -antitrypsin deficiency.
The pathogenesis of leukocytoclastic vasculitis involves the deposition of immune complexes in vessel walls with activation of the complement system, particularly the terminal components. This contrasts with microscopic polyangiitis in which immune complexes are often absent. Chemotaxis of neutrophils and injury to vessel walls with exudation of serum, erythrocytes, and fibrin result. Cell adhesion molecules play a critical role in the interaction between the vascular endothelium and leukocytes. These adhesion molecules are likely to be involved in the recruitment of the neutrophils. Mast cells may also play a role. The size of the immune complexes, which depends on the valence of the respective antigen and antibody and their relative concentrations (which is in part determined by the number of binding sites on the antigen), determines the likelihood of their deposition. The complexes most likely to precipitate are those with antigens bearing two to four binding sites and in a concentration approximately equivalent with antibody. These are the largest immune complexes that ordinarily remain soluble.
Various antibodies have been detected in patients with acute vasculitis, including ANCA and IgA class anticardiolipin antibodies. ANCA-positive vasculitis is known to be associated with the antithyroid drug propylthiouracil; both myeloperoxidase ANCA and proteinase-3 ANCA have been reported. In one study, cryoglobulins were found in 25% of cases.
Lymphokines also play an important role in the evolution of the vascular lesions. Interleukins (IL-1, IL-6, and IL-8) are increased in the circulation, as is tumor necrosis factor (TNF). Both TNF and IL-1 may stimulate the endothelium to activate the intrinsic and extrinsic coagulation pathways and reduce its fibrinolytic activity. This may be the explanation for the thrombosis that occurs in vasculitis.
The formation of platelet aggregates appears to play a thus far unrecognized role in cutaneous small vessel vasculitis. In all cases of vasculitis, platelet clumps are associated with diffuse immunostaining of the perivascular stroma with the initiator of platelet aggregation, anti–von Willebrand's factor.
Hemodynamic factors such as turbulence and increased venous pressure, as well as the reduced fibrinolytic activity that occurs in the legs, may explain why localization of lesions to this site is common. Rarely, lesions may develop in areas of previously traumatized skin—the Koebner phenomenon. The size and configuration of the complexes may also determine the class of vessel affected. The size of the affected vessels correlates with the clinical features. For example, involvement of small dermal vessels results in erythema or palpable purpura, whereas lesions in larger arteries may lead to nodules, ulcers, or livedo reticularis. Deep venous involvement results in nodules without ulceration.
Drug- and infection-related vasculitis usually respond to the withdrawal of the drug and the treatment of the infection, respectively. Systemic corticosteroids can be used in severe drug-related cases to hasten the resolution of the disease. In more chronic cases, as seen with some of the tumor-associated and idiopathic vasculitides, other therapy is used, such as dapsone, methotrexate, mycophenolate mofetil, azathioprine, cyclosporine, cyclophosphamide, and intravenous immunoglobulin. Rituximab has been used for small vessel vasculitis, particularly the type II mixed cryoglobulinemic vasculitis. Hyperbaric oxygen therapy has been used as a successful adjuvant in vasculitides with nonhealing skin ulcers.
Acute vasculitis is a dynamic process. Accordingly, not all the features described here will necessarily be present at a particular stage in the evolution of the disease. It is best to perform a biopsy on a lesion of 18 to 24 hours’ duration because this will show the most diagnostic features. The changes to be described usually involve the small venules (postcapillary venules) in the dermis, although in severe cases arterioles may also be affected. In the cases associated with lower extremity inflammatory lymphedema in military basic trainees, leukocytoclastic vasculitis involved the deep dermal vascular plexus. Some of the cases involving arterioles would now be classified as microscopic polyangiitis (see p. 265 ). There is infiltration of vessel walls with neutrophils that also extend into the perivascular zone and beyond. These neutrophils undergo degeneration (leukocytoclasis) with the formation of nuclear dust ( Fig. 9.7 ). Nuclear dust is not synonymous with this condition. LeBoit has reviewed the other circumstances in which it can be found. The vessel walls are thickened by the exudate of inflammatory cells and edema fluid. There is also exudation of fibrin (“fibrinoid necrosis”) that often extends into the adjacent perivascular connective tissue. Endothelial cells are usually swollen and some are degenerate. Thrombosis of vessels is sometimes present. The dermis shows variable edema and extravasation of red blood cells.
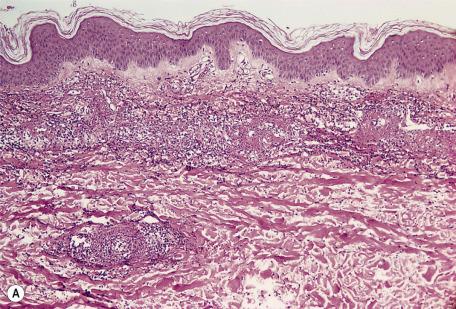
In some lesions, particularly those of longer duration, eosinophils and lymphocytes are also present, particularly in a perivascular location. Macrophages, which are scattered in the interstitium even in the early stages, show a time-dependent increase. In vasculitis caused by drugs, a mixed inflammatory cell infiltrate of eosinophils, lymphocytes, and occasional neutrophils is commonly seen. The mean eosinophil count per high power field was 5.20 in a series of drug-induced cases and 1.05 in non–drug-induced cases. The vasculitis induced by tamoxifen (discussed previously) was unusual in being localized to the mastectomy scar and right thorax.
In resolving lesions, there is usually only a mild perivascular infiltrate of lymphocytes and some eosinophils. A rare plasma cell may also be present. Perivascular hemophagocytosis is another late feature. A striking feature of late resolving lesions is hypercellularity of the dermis with an increased number of interstitial fibroblasts and histiocytes, giving the appearance of a “busy dermis” ( Fig. 9.8 ). There is sometimes a mild increase in acid mucopolysaccharides imparting a vague “necrobiotic” appearance. These findings on the evolutionary changes in vasculitis are based on a study by the author of five cases of leukocytoclastic vasculitis, biopsied several times over the course of a 2-week period. It was never published.

Uncommonly, the subepidermal edema is so pronounced that vesiculobullous lesions result. Cutaneous infarction, usually involving only the epidermis and upper third of the dermis, may follow thrombosis of affected vessels. Presumptive ischemic changes are not uncommon in sweat glands, even in the absence of infarction. They include apoptosis, necrosis, and basal cell hyperplasia. Rare changes reported in leukocytoclastic vasculitis include epidermal lichenoid changes, subepidermal microabscess formation resembling dermatitis herpetiformis, and intraepidermal vesiculation with acantholytic cells containing vacuolated nuclei. Cytomegalovirus-associated vasculitis can sometimes occur without inclusion body changes.
Although previous studies suggested that direct immunofluorescence is not particularly useful unless performed on lesions less than 6 hours old, studies have found fibrinogen, C3, and IgM in early lesions; albumin, fibrinogen, and IgG in fully developed lesions; and mainly fibrinogen, with some C3, in late lesions. Lesional skin is more often positive than nonlesional skin. Another study found vascular deposits of immunoglobulin in 81% of cases. The presence of immunoglobulin was seen more often in patients who had at least one extracutaneous manifestation. The presence of IgA was a strong predictor of renal disease. E-selectin (ELAM-1) is present early in the course of vasculitis but decreases as the lesion evolves.
The main differential diagnostic problem is the recognition of leukocytoclastic vasculitis when changes are subtle, obscured by dense inflammation, or mimicked (to an extent) by neutrophilic perivascular infiltrates. Diagnosis therefore requires careful inspection of vessel changes, facilitated at times by multiple levels and direct immunofluorescence study. One must also be aware of the phenomenon of “secondary vasculitis” as seen, for example, at the base of ulcers or in heavy neutrophilic dermal infiltrates. In such circumstances, it is worthwhile to evaluate those vessels that are not in the immediate vicinity of an ulcer and to examine for evidence of true vessel injury. Some examples of neutrophilic urticaria, a potential clinical mimic of urticarial vasculitis, can create considerable diagnostic difficulty. This form of urticaria consists of long-standing lesions that are accompanied by pain rather than pruritus. Included are a number of the “physical urticarias”—cholinergic, cold, and delayed pressure urticarias. Neutrophils aggregate around dermal vessels, but leukocytoclasis is not observed, nor is there evidence for endothelial injury or fibrin deposition.
The Henoch–Schönlein variant of leukocytoclastic vasculitis (HSP), which represents approximately 10% of all cases of cutaneous vasculitis, is characterized by a purpuric rash, usually on the lower parts of the legs, which is often accompanied by one or more of the following: arthritis, abdominal pain, hematuria, and, rarely, cardiac or neurological manifestations. Intussusception is an uncommon disease complication that has been reported with increasing frequency in recent years. Pancreatitis is another uncommon complication. In adult cases, there is a relatively high incidence of renal involvement. The European League Against Rheumatism/Pediatric Rheumatology International Trials Organization/Pediatric Rheumatology European Society (EULAR/PRINTO/PRES) validated classification for HSP includes palpable purpura (mandatory) and at least one of the following: leukocytoclastic vasculitis with predominant IgA deposits on skin biopsy, diffuse abdominal pain, acute arthritis or arthralgia, or renal involvement (proteinuria and/or hematuria). In doubtful cases with atypical distribution of purpuric lesions, the previously described cutaneous histopathological and immunohistochemical findings or proliferative glomerulonephritis with predominant IgA deposits are recommended for diagnosis. The rash may also be macular, papular, urticarial, and, rarely, vesiculobullous. Vesiculobullous lesions may occur on the lips and buccal mucosa. Redness, swelling, and limb pain accompanied by Raynaud's syndrome have been reported in a recent case. The bullae may be hemorrhagic. It is usually preceded by an upper respiratory tract infection or the ingestion of certain drugs or foods, with the formation of IgA-containing complexes, which may precipitate in vessels in the skin and certain other organs. The alternate complement pathway is thereby activated. Anticardiolipin antibodies and ANCA, both of IgA class, have been reported in HSP. However, IgA ANCA are present in only 10% of cases. They can be detected in a wide variety of other cutaneous vasculitides. Specific associations of HSP have included pregnancy ; alcohol ; α 1 -antitrypsin deficiency ; H. pylori infection of the stomach; Proteus mirabilis urinary tract infection ; Mycoplasma pneumoniae infection ; other bacterial and viral infections, including parvovirus ; vaccinations ; and drugs such as penicillin, ampicillin, vancomycin, clarithromycin, erythromycin, chlorpromazine, losartan, aspirin (acetylsalicylic acid), erlotinib, isotretinoin, and adalimumab. It has been reported in association with the periodic fever syndromes, including hyperimmunoglobulinemia D, and familial Mediterranean fever. Of interest is the finding of mutations in the MEFV gene, which causes familial Mediterranean fever, in some children in Israel with HSP. It has been associated with malignancy in adults. In one case, HSP was mimicked by brucellosis. HSP has a predilection for children, although occurrence in adults is known. In a Mayo Clinic study of 56 children, 48% were due to IgA vasculitis (HSP); the other cases were due to cutaneous small vessel vasculitis (34%), urticarial vasculitis (9%), ANCA-associated vasculitis (7%), and acute hemorrhagic edema of infancy (2%—one patient). HSP has been reported in two siblings who had simultaneous onset but no obvious underlying infection.
The disease usually runs a self-limited course, although in a small percentage of patients persistent renal involvement occurs. Pulmonary involvement is a rare complication of HSP.
The role of corticosteroids in the treatment of this disease remains controversial. Although they do not appear to alter the course of the disease, they alleviate symptoms and may be of benefit in cases of ulcerative and extensive disease. They appear to offer a protective effect for nephritis, and they may modify accompanying arthritis and gastrointestinal disease.
The condition reported as infantile acute hemorrhagic edema of the skin (Finkelstein's disease) is a closely related condition. Infants present with rounded, red to purpuric plaques, sometimes with a targetoid appearance, over the cheeks, ears, and extremities, associated with nonpitting acral edema. Low-grade fever may be present, but involvement of organ systems is minimal and laboratory studies are often unrevealing; recovery typically occurs within 1 to 3 weeks. Approximately 300 children (median age, 11 months) have been reported with this condition. Rare cases have been reported in adults. It has followed cytomegalovirus infection, herpes simplex stomatitis, rotavirus infection, and measles and H1N1 vaccination. In one report, infantile acute hemorrhagic edema was a presenting feature of the Wiskott–Aldrich syndrome. The histopathology is the same, although IgA deposition is not a predictable feature. It has a benign course, leading to the suggestion that it be considered a separate entity. Both systemic corticosteroids and antihistamines have been used to treat this condition, but there is no evidence that they do any good, although a rapid response has been achieved in a few patients.
The appearances are usually indistinguishable from those seen in leukocytoclastic vasculitis. IgA, predominantly of the IgA1 subclass, is demonstrable in vessel walls in both involved and uninvolved skin in most cases, provided that the biopsy is taken early in the course of the disease. It has been suggested that the punch biopsy should be taken from the edge of a fresh lesion to maximize the chances of finding IgA deposits. Lesions should be less than 48 hours old. Apoptotic dendrocytes (factor XIIIa positive) were noted around vessels in one case. Johnson et al. found that renal involvement is significantly associated with papillary dermal edema on histopathology and with perivascular C3 deposition on direct immunofluorescence. The latter finding has been supported in another large study, in which it was also found that IgM deposition has an association with articular involvement.
Henoch–Schönlein purpura is not synonymous with IgA-associated vasculitis. Rather, it is one subcategory of it. An IgA vasculitis has been seen, without the usual clinical features of Henoch–Schönlein purpura, in a setting of prior infection, usually of the upper respiratory tract, in granulomatosis with polyangiitis (formerly Wegener's granulomatosis), inflammatory bowel disease, and malignancies. IgA vasculitis and nephropathy have followed Bartonella henselae infection. IgA deposits in superficial dermal vessels have also been reported in patients with evidence of alcohol abuse; the frequency of these deposits apparently does not differ when comparing patients with and without IgA nephritis.
Eosinophilic vasculitis is a recently identified form characterized by an eosinophil-predominant necrotizing vasculitis affecting small dermal vessels. It presents with pruritic, erythematous, and purpuric papules and plaques. It may be idiopathic or associated with connective tissue diseases, the hypereosinophilic syndrome, or HIV infection.
However, recurrent cutaneous eosinophilic vasculitis appears to be a separate entity from these or other conditions with systemic aspects. A disorder that is more common in females, peripheral eosinophilia may be absent, but if it is present tends to be not as pronounced is it would be, for example, in hypereosinophilic syndrome or eosinophilic angioedema. Its pathogenesis is unclear, but an abnormal eosinophilic response to some unidentified antigen has been suspected. Treatment with systemic corticosteroids is usually effective, but recurrences are common. Tacrolimus can improve the clinical manifestations and decrease the corticosteroid dosage needed to control the disease. In one case, the addition of suplatast tonsilate, a dimethylsulfonium agent, was required to produce resolution.
There is a necrotizing, eosinophil-rich vasculitis involving small vessels in the dermis and, sometimes, the subcutis ( Fig. 9.9 ). It involves a smaller class of vessel than Churg–Strauss syndrome. There is marked deposition of eosinophil granule major basic protein in the vessel walls. Dermal infiltration by eosinophils is prominent, but there is little or no leukocytoclasis. The overlying epidermis is often normal, but there may be minor eosinophilic spongiosis.
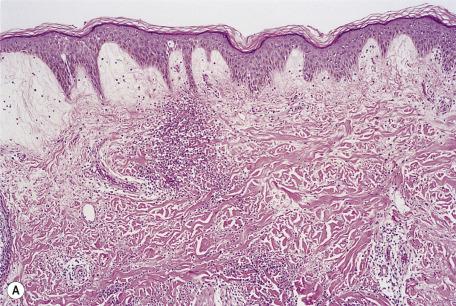
Dermoscopy has shown spots and purpuric globules (representing damaged superficial dermal vessels with erythrocyte extravasation), with an irregular red-orange background. Such changes can also be seen in other forms of vasculitis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here