Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Poor bone health can lead to failure of the primary spinal procedure and precipitate the need for a revision spine surgery. Postoperative complications related to poor bone health include failure of fixation, fracture, kyphosis, implant subsidence, pseudarthrosis, and worsening sagittal and/or coronal deformity. Elderly patients and those with comorbidities are at an increased risk of having poor bone health. Thus the risk of failure following spine surgery is higher in these patients. Secondary fracture prevention programs have proven effective to address osteoporosis after fracture, and can be applied before spinal surgery, especially in those undergoing revision surgery.
Poor bone health includes deficits in bone mineral density (known as osteopenia and osteoporosis), vitamin D deficiency leading to a failure in mineralization (known as osteomalacia), and a breakdown in bone microarchitecture (owing to deficits in bone quality). In addition, osteoporosis affects the number, quality, and responsiveness of mesenchymal stem cells needed to differentiate into osteoblastic lineages. Low bone mass is an encompassing term for these conditions but has also recently been defined as osteopenia based on bone mineral density (BMD).
The purpose of this chapter is to review the diagnosis and evaluation of the bone health of patients undergoing revision spine surgery. The rationale for this review is the direct relationship between the need for revision surgery and poor bone health. A systematic approach to assessing and optimizing bone health before revision surgery will be discussed.
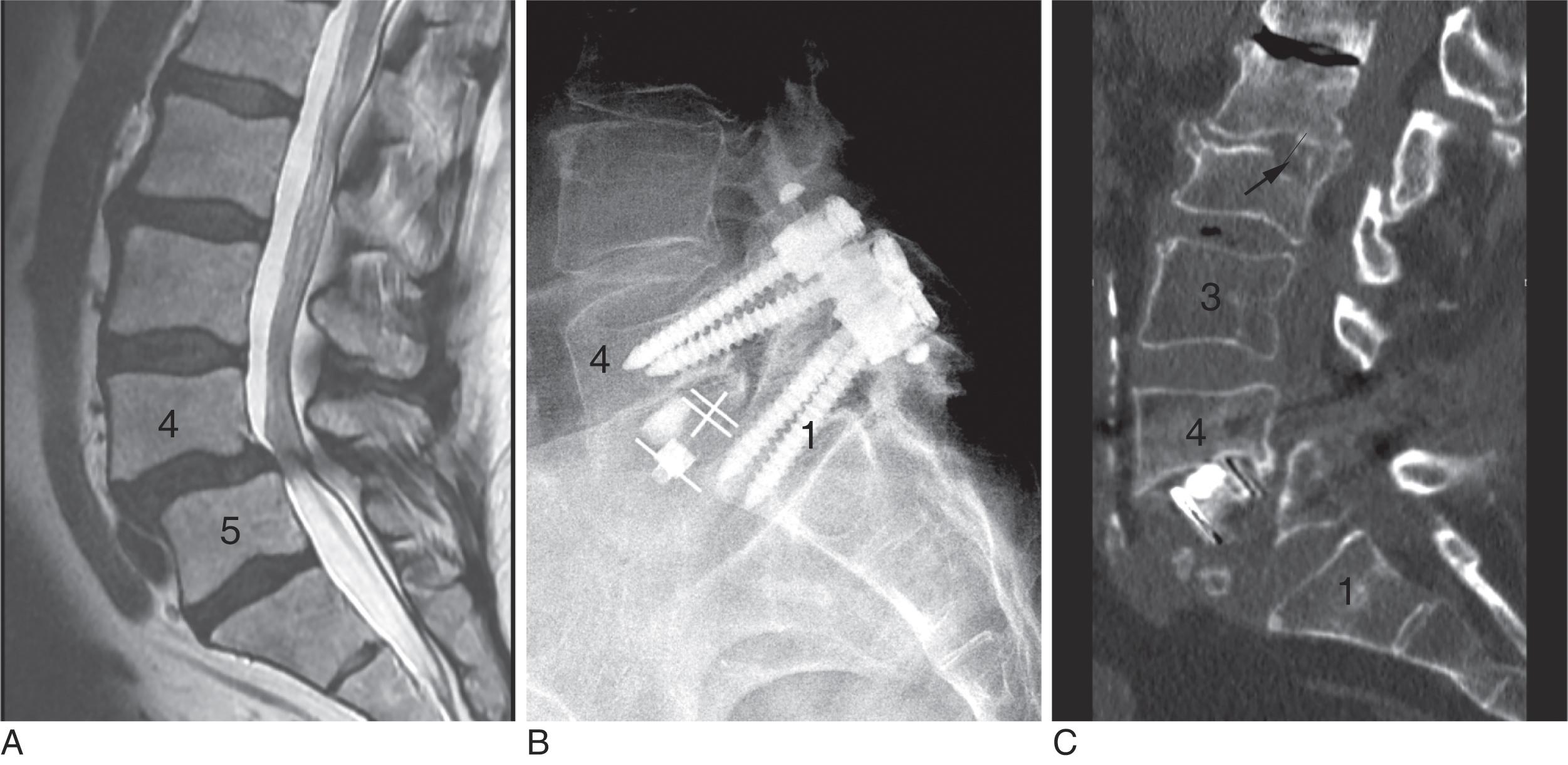
Adverse consequences of poor bone health include poorer surgical outcomes and greater complications, often leading to increased rates of revision surgery ( Table 2.1 ). Bjerke et al. reported fusion success rates and bone-related complications in 140 patients undergoing lumbar spinal fusion. They found that osteoporotic patients had a 50% rate of nonunion compared with 18% of those with normal bone density. Osteoporosis-related complications including hardware failure, compression fracture, kyphosis, and nonunion occurred in 19% of normal, 28% of osteopenic, and 67% of osteoporotic patients ( Figs. 2.1 and 2.2 ). Other complications that have been reported relating to osteoporosis include screw loosening, cage subsidence, increased spondylolisthesis, sacral and pelvic insufficiency fracture, durotomy, and revision surgery (see Table 2.1 ).
| Nonunion | Bjerke et al., Cho et al. |
| Screw loosening | Bjerke et al., Sakai et al., Bredow et al. |
| Hardware failure | Bjerke et al., Bernstein et al. |
| Interbody cage subsidence | Formby et al., Tempel et al. |
| Proximal junctional fracture | Uei et al., Meredith et al. |
| Proximal junction al kyphosis | Yagi et al. |
| Increase spondylolisthesis | Wang et al., Andersen et al. |
| Increased scoliosis | Yu et al. |
| Revision surgery | Bjerke et al., Sheu et al., Puvanesarajah et al. |
| Pelvic and sacral insufficiency fractures posterior fusion | Meredith et al., Papadopoulos et al., Odate et al., Klineberg et al. |
| Compression fracture | Formby et al. |
In patients undergoing primary spine surgery, low bone mass and vitamin D deficiency are commonly observed. In patients undergoing revision spine surgery, these factors are likely even more prevalent. Vitamin D deficiency (defined as 25(OH) vitamin D <20 ng/mL) occurs in 30% to 65% of patients whereas vitamin D insufficiency (defined as 25(OH) vitamin D 20–30 ng/mL) occurs in 27% to 40% of patients. Osteopenia and osteoporosis are present in 30% to 50% and 10% to 20% of patients, respectively.
Hills et al. retrospectively reviewed the role of endocrine disorders in 169 patients with pseudarthrosis after spinal fusion. Overall, endocrine disorders were present in 82% of patients, and endocrinology referrals were made in 59 patients (34.9%). Osteopenia and osteoporosis were the most commonly observed endocrine disorders. Before referral, these were diagnosed in 18.9% of patients. After endocrinology assessment, however, this number increased to 45%. Vitamin D deficiency was also highly prevalent, occurring in 38% of patients. Other endocrine disorders likely affecting skeletal function included diabetes (27%), hyperparathyroidism (5%), and sex hormone deficiency (18%).
An essential component of the evaluation of a postoperative patient is understanding the original indications for the surgery, patient comorbidities, postoperative course, and any structural changes (seen on postoperative imaging) since surgery. Bone health is likely to be a factor and therefore should be completely evaluated before further surgery is considered. Bone health evaluation consists of screening to determine if bone densitometry is warranted, identification and treatment of vitamin D and calcium deficits, assessment of nutritional status, and analysis of falls and generalized muscle weakness. Unfortunately, the spine surgeon’s attitude toward screening for osteoporosis is relatively poor, with only 19% of surgeons reporting that they check dual energy x-ray absorptiometry (DXA) when evaluating patients with pseudarthrosis.
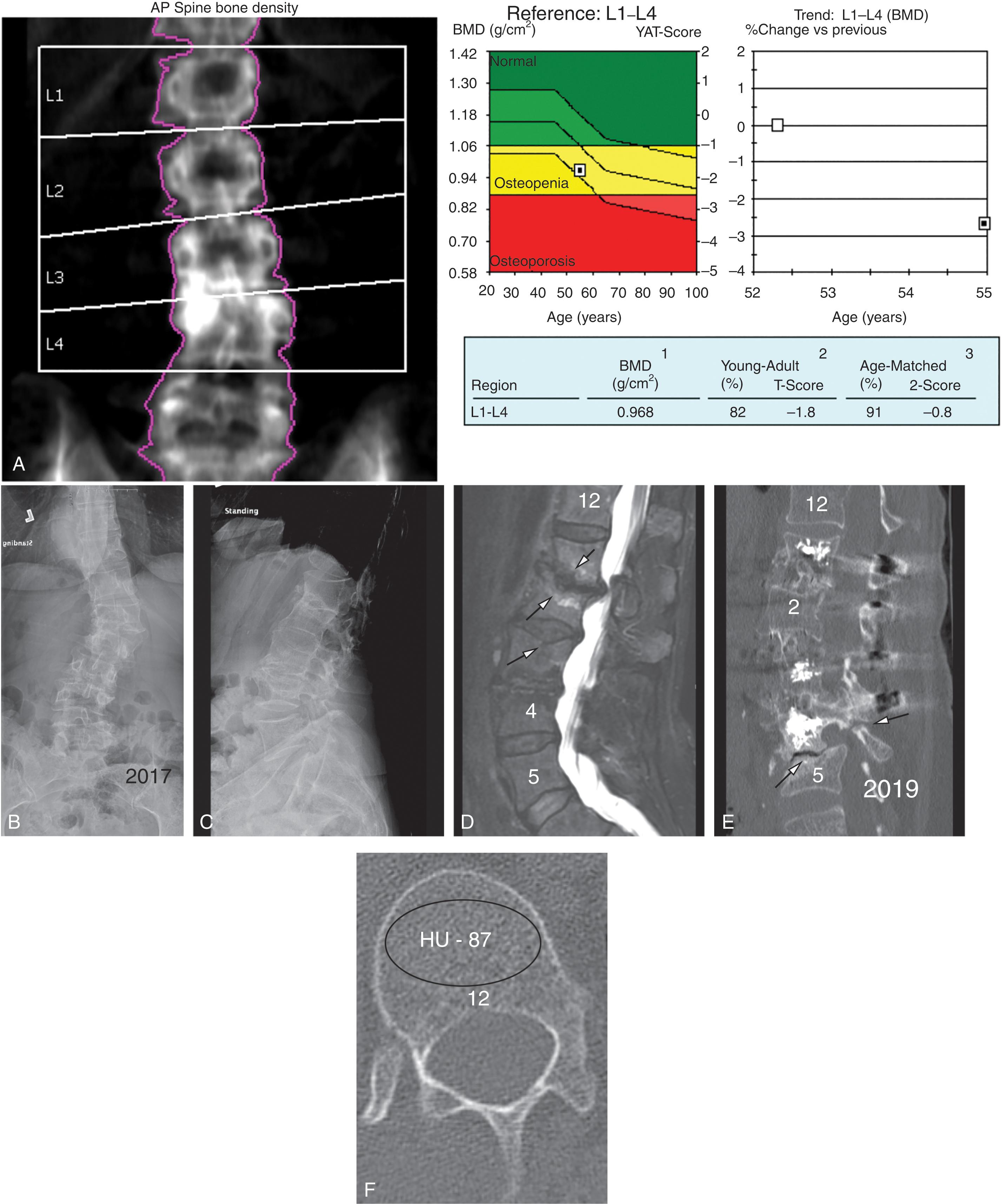
DXA is the gold standard to measure BMD. Results are reported in gm/cm 2 in regions of interest (ROI). The BMD is used to determine statistical results (referred to as T-scores and Z-scores). These indicate how many standard deviations away a patient’s BMD is from a reference standard. T-scores are most commonly used based on healthy young females as the reference standard. Z-scores are used for premenopausal females and men younger than 50 years and are compared with age-gender matched controls. If possible, the total hip and total femoral neck T-scores are used. If unavailable, then lumbar spine T-scores (L1–L4) are used. Spine DXA is often unreliable in spine patients with degenerative changes or deformities; it is unreliable in those who have previously undergone surgery. In patients for whom hip and spine scores are not available, the one-third radius BMD is used. To evaluate longitudinal changes, BMDs rather than T-scores are used.
The World Health Organization classifies bone density based on DXA T-scores ( Table 2.2 ):
A T-score less than or equal to −2.5 indicates osteoporosis
A T-score between −2.4 and −1.0 indicates osteopenia
A T-score greater than −1.0 signifies normal bone.
| World Health Organization | ||
| Classification | T-score | |
| Normal | > −1.0 | |
| Osteopenia (low bone mass) | Between −1.0 and −2.4 | |
| Osteoporosis | < −2.5 | |
| National Bone Health Alliance (NBHA) Recommendations for Diagnosis of Osteoporosis | ||
| T-score ≤ −2.5 of hip, spine, or one-third radius | ||
| Hip and spine fracture (low energy) | ||
| Osteopenia T-score and fragility fracture of wrist, pelvis, or proximal humerus | ||
| Osteopenia and high fracture risk assessment tool (FRAX) score | ||
This definition has poor sensitivity as less than 50% of patients who have had fragility fractures are classified as osteoporotic; additionally, this classification system does not aid in making treatment decisions. A more clinical definition of osteoporosis has been proposed by the National Bone Health Alliance (NBHA). This definition includes any of the following as indicating osteoporosis: T-score less than −2.5; the presence of a hip and spine fracture; osteopenia and wrist, pelvis, or proximal humerus fracture; and having a high 10-year hip and major fracture risk based on the fracture risk assessment tool (FRAX; Table 2.3 ).
| Risk Factors | Results | |
|---|---|---|
| Age | Current smoker | 10-year hip fracture risk (%) |
| Gender | Glucocorticoid use | 10-year major fracture risk (%) |
| Height | Rheumatoid arthritis | |
| Weight | Secondary osteoporosis | |
| Prior fracture | Alcohol ≥3 units/day | |
| Parent with hip fracture | Femoral neck BMD (gm/cc) or T-score (FRAX may be calculated without BMD data) | |
| Treatment Criteria | ||
| Hip fracture risk >3% or major fracture risk >20% | ||
| Screening criterion to determine who should have DXA based on FRAX with BMD | ||
| Major fracture risk >8.4% | ||
The FRAX calculator is a 10-year fracture prediction tool based on known risk factors. 25a The risk factors used in calculating the FRAX have been established based on longitudinal studies of large populations. The FRAX can be calculated with or without the BMD. When used for treatment decisions, a patient with a FRAX 10-year risk of hip or major fracture of 3% and 20%, respectively, is high risk and should be evaluated for pharmaceutical treatment. In the case of screening to determine if a patient needs a DXA, a lower threshold of 8.4% for a 10-year risk of major fracture is used.
The International Society of Clinical Densitometry (ISCD) and National Osteoporosis Foundation (NOF) have developed guidelines to determine which patients should undergo bone densitometry testing ( Table 2.4 ). Any of the “adverse outcomes” following spine surgery described in Table 2.1 should prompt the need for bone densitometry testing. In addition, reviewing relevant imaging studies such as plain radiographs and computed tomography (CT) scans (described later) to evaluate bone quality may identify patients who should undergo DXA testing.
| Women age >65 | |
| Men age >70 | |
| History of fracture age >50 | |
| FRAX risk of major osteoporotic fracture >8.4% | |
| High-risk medication use (i.e., corticosteroids) | |
| Low body weight | |
| Revision spine surgery age >50 a |
New techniques in bone densitometry can aid in identifying those patients with occult vertebral fractures and can provide an improved understanding of bone microarchitecture. Vertebral fracture assessment (VFA) is obtained during DXA examination by placing the patient in the decubitus position and scanning from T3 to L5. This produces an excellent lateral image of the spine and has the advantage that the x-ray is parallel to each disc space (eliminating parallax). Vertebral fractures are found in up 30% of patients having DXA, the majority of which are occult. The presence of fractures indicates the diagnosis of osteoporosis and should be taken into consideration when planning revision surgery.
The trabecular bone score (TBS) of the lumbar spine (L1–L4) vertebral bodies is obtained using software and standard DXA data. The TBS provides a measure of bone microarchitecture that is independent of BMD. The microarchitecture is assessed by textural analysis where large variation between adjacent pixels of cross-sections indicate degraded microarchitecture compared with those without variations. A TBS score is calculated and graded as degraded, partially degraded, or not degraded bone. The TBS can be substituted for BMD in FRAX, and has been correlated with greater pedicle screw insertion strength when compared with BMD.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here