Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Porphyrias are metabolic diseases resulting from altered activities of specific enzymes of the heme biosynthetic pathway. These enzymes are most active in bone marrow and liver. Erythropoietic porphyrias , in which overproduction of heme pathway intermediates occurs primarily in bone marrow erythroid cells, usually present at birth or in early childhood with cutaneous photosensitivity, or in the case of congenital erythropoietic porphyria, even in utero as nonimmune hydrops. Erythropoietic protoporphyria is the most common porphyria in children and of most interest to pediatricians. Most porphyrias are hepatic, with overproduction and initial accumulation of porphyrin precursors or porphyrins in the liver. Activation of hepatic porphyrias is very rare during childhood, reflecting the distinct hepatic regulatory mechanisms for heme biosynthesis that are influenced by pubertal development. Homozygous forms of the hepatic porphyrias may manifest clinically before puberty. Children who are heterozygous for inherited hepatic porphyrias may present with nonspecific and unrelated symptoms, and parents often request advice about long-term prognosis and express concerns about drugs that may exacerbate these conditions.
The DNA sequences and chromosomal locations are established for the genes of the enzymes in this pathway, and multiple disease-related mutations have been found for each porphyria. However, benign variants identified by gene sequencing can be misleading. The inherited porphyrias display autosomal dominant, autosomal recessive, or X-linked inheritance. Although initial diagnosis of porphyria by biochemical methods remains essential, it is especially important to confirm the diagnosis by demonstrating a specific pathogenic gene mutation(s).
Heme is required for a variety of hemoproteins, such as hemoglobin, myoglobin, respiratory cytochromes, and cytochrome P450 enzymes (CYPs). It is believed that the 8 enzymes in the pathway for heme biosynthesis are active in all tissues. Hemoglobin synthesis in erythroid precursor cells accounts for approximately 85% of daily heme synthesis in humans. Hepatocytes account for most of the rest, primarily for synthesis of CYPs, which are especially abundant in the liver endoplasmic reticulum, and turn over more rapidly than many other hemoproteins, such as the mitochondrial respiratory cytochromes. Pathway intermediates are the porphyrin precursors δ-aminolevulinic acid ( ALA , also known as 5-aminolevulinic acid) and porphobilinogen (PBG) , as well as porphyrins (mostly in their reduced forms, known as porphyrinogens ) ( Fig. 110.1 ). These intermediates do not accumulate in significant amounts under normal conditions or have important physiologic functions.
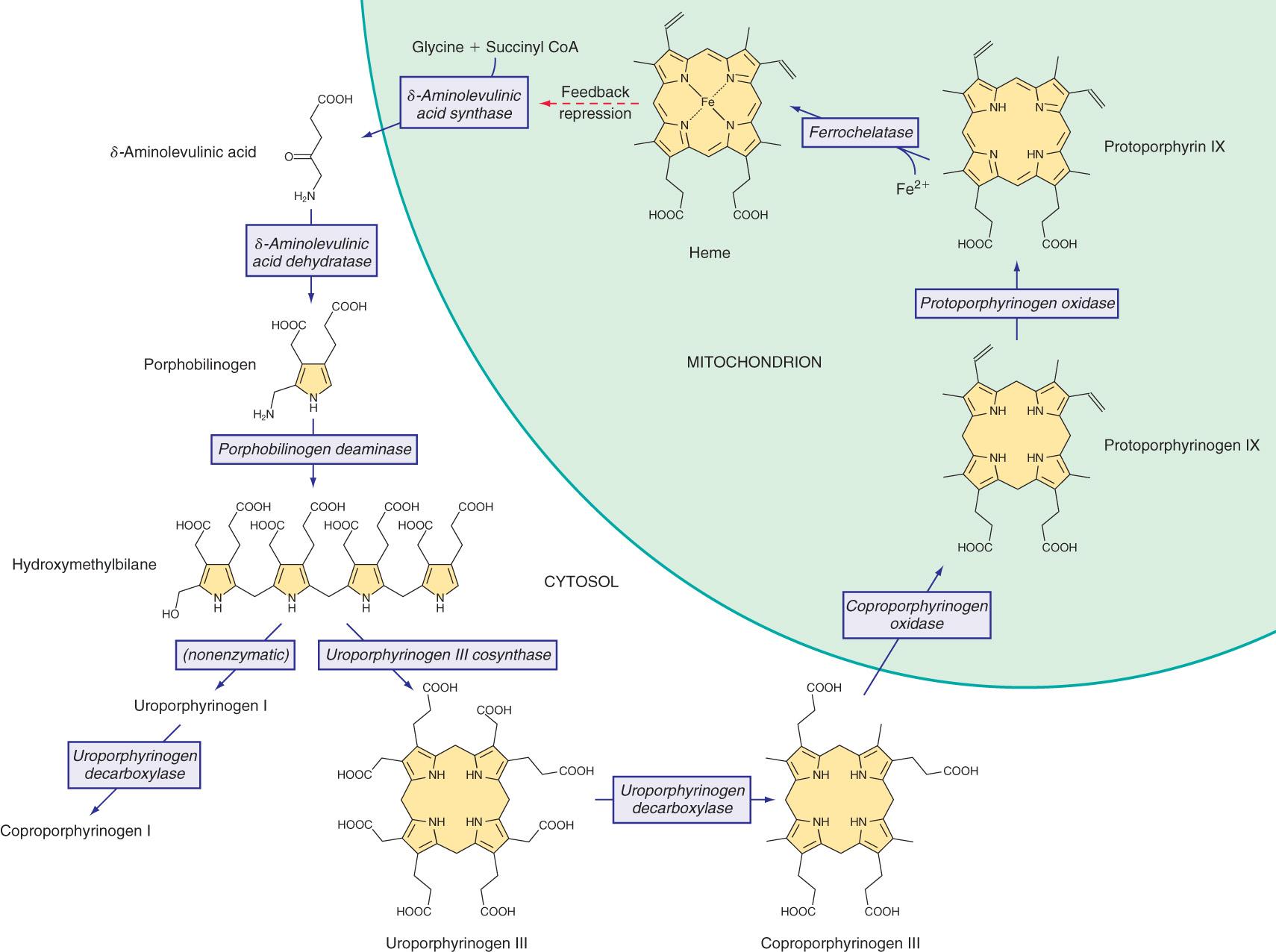
Altered activity of each enzyme in the pathway has been associated with a specific type of porphyria ( Table 110.1 ). The first enzyme, ALA synthase (ALAS), occurs in 2 forms. An erythroid specific form, ALAS2, is deficient in X-linked sideroblastic anemia, as a result of mutations of the ALAS2 gene on chromosome Xp11.2. Gain-of-function mutations of ALAS2 caused by deletions in the last exon cause X-linked protoporphyria ( XLP ), which is phenotypically identical to erythropoietic protoporphyria.
| DISEASE | ENZYME | INHERITANCE | PRESENTATION | Classification * | |||
|---|---|---|---|---|---|---|---|
| H | E | A/N | C | ||||
| X-Linked protoporphyria (XLP) | δ-Aminolevulinate synthase 2 (ALAS2) | X-linked | Childhood | X | X | ||
| δ-Aminolevulinic acid dehydratase porphyria (ADP) | δ-Aminolevulinic acid dehydratase (ALAD) | Autosomal recessive | Mostly post puberty | X | X * | X | |
| Acute intermittent porphyria (AIP) | Porphobilinogen deaminase (PBGD) | Autosomal dominant | Post puberty | X | X | ||
| Homozygous AIP | Homozygous dominant | Childhood | X | X | X | ||
| Congenital erythropoietic porphyria (CEP) | Uroporphyrinogen III synthase (UROS) | Autosomal recessive | In utero or infancy | X | X | ||
| Porphyria cutanea tarda (PCT) type 1 | Uroporphyrinogen decarboxylase (UROD) | Sporadic | Adults | X | X | ||
| PCT type 2 † | Autosomal dominant | Adults | X | X | |||
| PCT type 3 | Unknown | Adults | X | X | |||
| Hepatoerythropoietic porphyria (HEP) | Homozygous dominant | Childhood | X | X * | X | ||
| Hereditary coproporphyria (HCP) | Coproporphyrinogen oxidase (CPOX) | Autosomal dominant | Post puberty | X | X | X | |
| Homozygous HCP | Homozygous dominant | Childhood | X | X | X | X | |
| Variegate porphyria (VP) | Protoporphyrinogen oxidase (PPOX) | Autosomal dominant | Post puberty | X | X | X | |
| Homozygous VP | Homozygous dominant | Childhood | X | X | X | X | |
| Erythropoietic protoporphyria (EPP) | Ferrochelatase (FECH) | Autosomal recessive (most commonly heteroallelic with hypomorphic allele) | Childhood | X | X | ||
* Classification abbreviations: H, Hepatic; E, Erythropoietic; A/N, Acute/Neurologic; C, Cutaneous.
† PCT is a result of inhibition of hepatic UROD. Autosomal dominant inheritance of a partial deficiency of UROD is a predisposing factor in cases defined as familial (type 2) PCT.
Regulation of heme synthesis differs in the 2 major heme-forming tissues. Liver heme biosynthesis is primary controlled by the ubiquitous form of ALAS (ALAS1). Synthesis of ALAS1 in liver is regulated by a “free” heme pool (see Fig. 110.1 ), which can be augmented by newly synthesized heme or by existing heme released from hemoproteins and destined for breakdown to biliverdin by heme oxygenase.
In the erythron, novel regulatory mechanisms allow for the production of the very large amounts of heme needed for hemoglobin synthesis. The response to stimuli for hemoglobin synthesis occurs during cell differentiation, leading to an increase in cell number. Also, unlike the liver, heme has a stimulatory role in hemoglobin formation, and the stimulation of heme synthesis in erythroid cells is accompanied by increases not only in ALAS2, but also by sequential induction of other heme biosynthetic enzymes. Separate erythroid-specific and nonerythroid or “housekeeping” transcripts are known for the first 4 enzymes in the pathway. The separate forms of ALAS are encoded by genes on different chromosomes, but for each of the other three, erythroid and nonerythroid transcripts are transcribed by alternative promoters in the same gene. Heme also regulates the rate of its synthesis in erythroid cells by controlling the transport of iron into reticulocytes.
Intermediates of the heme biosynthetic pathway are efficiently converted to heme and, normally, only small amounts of the intermediates are excreted. Some may undergo chemical modifications before excretion. Whereas the porphyrin precursors ALA and PBG are colorless, nonfluorescent, and largely excreted unchanged in urine, PBG may degrade to colored products such as the brownish pigment called porphobilin or spontaneously polymerize to uroporphyrins. Porphyrins are red in color and display bright-red fluorescence when exposed to long-wavelength ultraviolet (UV) light. Porphyrinogens are the reduced form of porphyrins, and are colorless and nonfluorescent, but are readily autoxidized to the corresponding porphyrins when they accumulate or are outside the cell. Only the type III isomers of uroporphyrinogen and coproporphyrinogen are converted to heme (see Fig. 110.1 ).
ALA and PBG are excreted in urine. Excretion of porphyrins and porphyrinogens in urine or bile is determined by the number of carboxyl groups. Those with many carboxyl groups, such as uroporphyrin (octacarboxyl porphyrin) and heptacarboxyl porphyrin, are water soluble and readily excreted in urine. Those with fewer carboxyl groups, such as protoporphyrin (dicarboxyl porphyrin), are not water soluble and are excreted in bile and feces. Coproporphyrin (tetracarboxyl porphyrin) is excreted partly in urine and partly in bile. Because coproporphyrin I is more readily excreted in bile than coproporphyrin III, impaired hepatobiliary function may increase total urinary coproporphyrin excretion and the ratio of these isomers.
Two useful classification schemes reflect either the underlying pathophysiology or the clinical features of porphyrias (see Table 110.1 ). In hepatic porphyrias and erythropoietic porphyrias the source of excess production of porphyrin precursors and porphyrins is the liver and bone marrow, respectively. Acute porphyrias cause neurologic symptoms that are associated with increases of one or both of the porphyrin precursors, ALA and PBG. In the cutaneous porphyrias, photosensitivity results from transport of porphyrins in blood from the liver or bone marrow to the skin. Dual porphyria refers to the very rare cases of porphyria with deficiencies of 2 different heme pathway enzymes.
Porphyria cutanea tarda ( PCT ), acute intermittent porphyria ( AIP ), and erythropoietic protoporphyria ( EPP ) are the 3 most common porphyrias, in that order, considering all age-groups, and are very different in clinical presentation, precipitating factors, methods of diagnosis, and effective therapy ( Table 110.2 ). Two less common acute porphyrias, hereditary coproporphyria ( HCP ) and variegate porphyria ( VP ), can also cause blistering photosensitivity (see Table 110.1 ). Congenital erythropoietic porphyria ( CEP ) causes more severe blistering lesions, often with secondary infection and mutilation. EPP and XLP have the same phenotype and are distinct from the other cutaneous porphyrias in causing nonblistering photosensitivity that occurs acutely after sun exposure. EPP is also the most common porphyria to become manifest before puberty.
| PRESENTING SYMPTOMS | EXACERBATING FACTORS | MOST IMPORTANT SCREENING TESTS | TREATMENT | |
|---|---|---|---|---|
| Acute intermittent porphyria | Neurologic, adult onset | Drugs (mostly P450 inducers), progesterone, dietary restriction | Urinary porphobilinogen | Hemin, glucose |
| Porphyria cutanea tarda | Skin blistering and fragility (chronic), adult onset | Iron, alcohol, smoking, estrogens, hepatitis C, HIV, halogenated hydrocarbons | Plasma (or urine) porphyrins | Phlebotomy, low-dose hydroxychloroquine |
| Erythropoietic protoporphyria | Phototoxic pain and swelling (mostly acute), childhood onset | Total erythrocyte protoporphyrin with metal-free and zinc protoporphyrin | Sun protection |
A few sensitive and specific first-line laboratory tests should be obtained whenever symptoms or signs suggest the diagnosis of porphyria. If a first-line or screening test is significantly abnormal, more comprehensive testing should follow to establish the type of porphyria. Overuse of lab tests for screening can lead to unnecessary expense and even delay in diagnosis. In patients who present with a past diagnosis of porphyria, lab reports that were the basis for the original diagnosis must be reviewed, and if these were inadequate, further testing considered.
Acute porphyria should be suspected in patients with neurovisceral symptoms such as abdominal pain after puberty, when initial clinical evaluation does not suggest another cause. Urinary PBG and total porphyrins should be measured. Urinary PBG is virtually always increased during acute attacks of AIP, HCP, and VP and is not substantially increased in any other medical conditions. Therefore this measurement is both sensitive and specific. Results from spot (single void) urine specimens are highly informative because very substantial increases are expected during acute attacks of porphyria. A 24 hr collection can unnecessarily delay diagnosis. The same spot urine specimen should be saved for quantitative determination of PBG and total porphyrins (both expressed relative to creatinine) to confirm the qualitative PBG result. ALA is often measured as well, but is usually less elevated than PBG in AIP, HCP, and VP. In ALA dehydratase porphyria, urinary ALA and porphyrins, but not PBG, are greatly elevated. Urinary porphyrins may remain increased longer than porphyrin precursors in some cases of HCP and VP. Measurement of urinary porphyrins alone should be avoided for screening, however, because they are often increased in many disorders other than porphyrias, such as liver diseases, and misdiagnoses of porphyria can result from minimal increases in urinary porphyrins that have no diagnostic significance.
Blistering skin lesions caused by porphyria are virtually always accompanied by increases in total plasma and urinary porphyrins. Porphyrins in plasma in VP are mostly covalently linked to plasma proteins and readily detected by a diagnostic peak in a fluorescence scanning method. The normal range for plasma porphyrins is somewhat increased in patients with end-stage renal disease.
Measurement of total erythrocyte protoporphyrin and, if the total amount is elevated, fractionation of protoporphyrin into its metal-free and zinc-chelated forms, is essential for diagnosis of EPP and XLP. Unfortunately, this is not offered by some major commercial laboratories. Results of zinc protoporphyrin measurements are often recorded (even in the same report) as both protoporphyrin and free erythrocyte protoporphyrin , with each calculated differently, based on past practices for screening for lead poisoning (which only increases zinc protoporphyrin). Thus the obsolete term free protoporphyrin does not mean metal-free protoporphyrin, because it was defined as iron-free protoporphyrin, and dates from before it was known that (except in protoporphyrias) protoporphyrin in erythrocytes is mostly zinc chelated. This unnecessary confusion makes diagnosis and reliable exclusion of protoporphyrias difficult. Total plasma porphyrins are elevated in most but not all cases of protoporphyria, so a normal level should not be relied on to exclude protoporphyria when total erythrocyte protoporphyrin is elevated.
Increases in erythrocyte total and zinc-chelated protoporphyrin occur in many other conditions, including iron deficiency, lead poisoning, hemolysis, anemia of chronic disease, and other erythrocyte disorders. Therefore the diagnosis of EPP must be confirmed by showing a predominant increase in metal-free protoporphyrin. In XLP, both free and zinc protoporphyrin are elevated.
More extensive testing is well justified when a first-line test is positive. For example, a substantial increase in PBG may be caused by AIP, HCP, or VP, and these can be distinguished by measuring erythrocyte porphobilinogen deaminase, urinary porphyrins (using the same spot urine sample), fecal porphyrins, and plasma porphyrins. The various porphyrias that cause blistering skin lesions are differentiated by measuring porphyrins in urine, feces, and plasma. Confirmation at the gene level is important once the diagnosis is established by biochemical testing.
It is often difficult to diagnose or rule out porphyria in patients who had suggestive symptoms months or years in the past, and in relatives of patients with acute porphyrias, because porphyrin precursors and porphyrins may be normal. More extensive testing and consultation with a specialist laboratory and physician may be needed. Before evaluating relatives, the diagnosis of porphyria should be firmly established in an index case, and the lab results reviewed to guide the choice of tests for the family members. The index case or another family member with confirmed porphyria should be retested if necessary. Identification of a disease-causing mutation in an index case greatly facilitates detection of additional gene carriers, because biochemical tests in latent carriers may be normal.
ALA dehydratase deficient porphyria ( ADP ) is sometimes termed Doss porphyria after the investigator who described the first cases. The term plumboporphyria emphasizes the similarity of this condition to lead poisoning, but incorrectly implies that it is caused by lead exposure.
This porphyria results from a deficiency of ALA dehydratase (ALAD), which is inherited as an autosomal recessive trait. Only six cases have been confirmed by mutation analysis. The prevalence of heterozygous ALAD deficiency was estimated to be <1% in Germany and approximately 2% in Sweden.
ALAD catalyzes the condensation of 2 molecules of ALA to form the pyrrole PBG (see Fig. 110.1 ). The enzyme is subject to inhibition by a number of exogenous and endogenous chemicals. ALAD is the principal lead-binding protein in erythrocytes, and lead can displace the zinc atoms of the enzyme. Inhibition of erythrocyte ALAD activity is a sensitive index of lead exposure.
Eleven abnormal ALAD alleles, most with point mutations, have been identified, some expressing partial activity, such that heme synthesis is partially preserved. The amount of residual enzyme activity may predict the phenotypic severity of this disease.
ADP is often classified as a hepatic porphyria, although the site of overproduction of ALA is not established. A patient with severe, early-onset disease underwent liver transplantation, without significant clinical or biochemical improvement, which might suggest that the excess intermediates did not originate in the liver. Excess urinary coproporphyrin III in ADP might originate from metabolism of ALA to porphyrinogens in a tissue other than the site of ALA overproduction. Administration of large doses of ALA to normal individuals also leads to substantial coproporphyrinuria. Increased erythrocyte protoporphyrin, as in all other homozygous porphyrias, may be explained by accumulation of earlier pathway intermediates in bone marrow erythroid cells during hemoglobin synthesis, followed by their transformation to protoporphyrin after hemoglobin synthesis is complete. Neurologic symptoms are attributed to neurotoxic effects of ALA, but this is unproven.
In most cases, symptoms resemble other acute porphyrias, including acute attacks of abdominal pain and peripheral neuropathy. Precipitating factors, such as exposure to harmful drugs, have not been evident in most cases. Four of the reported cases were adolescent males. A Swedish infant had more severe disease, with neurologic impairment and failure to thrive. A 63 yr old man in Belgium developed an acute motor polyneuropathy concurrently with a myeloproliferative disorder.
Urinary ALA, coproporphyrin III, and erythrocyte zinc protoporphyrin are substantially increased. Urinary PBG is normal or slightly increased. Erythrocyte ALAD activity is markedly reduced, and both parents have approximately half-normal activity of this enzyme and normal urinary ALA.
The other 3 acute porphyrias are characterized by substantial increases in both ALA and PBG. In contrast, ALA but not PBG is substantially increased in ADP. A marked deficiency of erythrocyte ALAD and half-normal activity in the parents support the diagnosis. Other causes of ALAD deficiency, such as lead poisoning , must be excluded. Succinylacetone accumulates in hereditary tyrosinemia type 1 and is structurally similar to ALA, inhibits ALAD, and can cause increased urinary excretion of ALA and clinical manifestations that resemble acute porphyria. Idiopathic acquired ALAD deficiency has been reported. Unlike lead poisoning, the deficient ALAD activity in ADP is not restored by the in vitro addition of sulfhydryl reagents such as dithiothreitol. Even if no other cause of ALAD deficiency is found, it is essential to confirm the diagnosis of ADP by molecular studies.
Treatment experience with ADP is limited but is similar to other acute porphyrias. Glucose seems to have minimal effectiveness but may be tried for mild symptoms. Hemin therapy was apparently effective for acute attacks in male adolescents, and weekly infusions prevented attacks in 2 of these patients. Hemin was not effective either biochemically or clinically in the Swedish child with severe disease, and it produced a biochemical response but no clinical improvement in the Belgian man with a late-onset form, who had a peripheral neuropathy but no acute attacks. Hemin is also effective in treating porphyria-like symptoms associated with hereditary tyrosinemia and can significantly reduce urinary ALA and coproporphyrin in lead poisoning. Avoidance of drugs that are harmful in other acute porphyrias is advisable. Liver transplantation was not effective in the child with severe disease.
The outlook is generally good in typical ADP cases, although recurrent attacks may occur. The course was unfavorable in the Swedish child with more severe disease and is uncertain in adults with late-onset disease associated with myeloproliferative disorders.
Heterozygous parents should be aware that subsequent children are at risk for ADP, as in any autosomal recessive disorder. Prenatal diagnosis is possible but has not been reported.
AIP is also termed pyrroloporphyria, Swedish porphyria, and intermittent acute porphyria and is the most common type of acute porphyria in most countries.
AIP results from the deficient activity of the housekeeping form of porphobilinogen deaminase (PBGD) . This enzyme is also known as hydroxymethylbilane (HMB) synthase (the prior term, uroporphyrinogen I synthase, is obsolete). PBGD catalyzes the deamination and head-to-tail condensation of 4 PBG molecules to form the linear tetrapyrrole, HMB (also known as preuroporphyrinogen; see Fig. 110.1 ). A unique dipyrromethane cofactor binds the pyrrole intermediates at the catalytic site until 6 pyrroles (including the dipyrrole cofactor) are assembled in a linear fashion, after which the tetrapyrrole HMB is released. The apo-deaminase generates the dipyrrole cofactor to form the holodeaminase, and this occurs more readily from HMB than from PBG. Indeed, high concentrations of PBG may inhibit formation of the holodeaminase. The product HMB can cyclize nonenzymatically to form nonphysiologic uroporphyrinogen I, but in the presence of the next enzyme in the pathway is more rapidly cyclized to form uroporphyrinogen III.
Erythroid and housekeeping forms of the enzyme are encoded by a single gene on human chromosome 11 (11q24.1→q24.2), which contains 15 exons. The 2 isoenzymes are both monomeric proteins and differ only slightly in molecular weight (approximately 40 and 42 kDa), and result from alternative splicing of 2 distinct messenger RNA (mRNA) transcripts arising from 2 promoters. The housekeeping promoter functions in all cell types, including erythroid cells.
The pattern of inheritance of AIP is autosomal dominant, with very rare homozygous cases that present in childhood. More than 400 PBGD mutations, including missense, nonsense, and splicing mutations, and insertions and deletions have been identified in AIP and in many population groups, including blacks. Most mutations are found in only one or a few families. Because of founder effects, however, some are more common in certain geographic areas, such as northern Sweden (W198X), Holland (R116W), Argentina (G116R), Nova Scotia (R173W), and Switzerland (W283X). De novo mutations may be found in approximately 3% of cases. The nature of the PBGD mutation does not account for the severity of the clinical presentation, which varies greatly within families. Chester porphyria was initially described as a variant form of acute porphyria in a large English family but was found to be caused by a PBGD mutation.
Most mutations lead to approximately half-normal activity of the housekeeping and erythroid isozymes and half-normal amounts of their respective enzyme proteins in all tissues of heterozygotes. In approximately 5% of unrelated AIP patients, the housekeeping isozyme is deficient, but the erythroid-specific isozyme is normal. Mutations causing this variant are usually found within exon 1 or its 5′ splice donor site or initiation of translation codon.
Induction of the rate-limiting hepatic enzyme ALAS1 is thought to underlie acute exacerbations of this and the other acute porphyrias. AIP remains latent (or asymptomatic) in the great majority of those who are heterozygous carriers of PBGD mutations, and this is almost always the case before puberty. In those with no history of acute symptoms, porphyrin precursor excretion is usually normal, suggesting that half-normal hepatic PBGD activity is sufficient unless hepatic ALAS1 activity is increased. Patients can also be asymptomatic with elevated levels of porphyrin precursors and are classified as asymptomatic high excretors. These patients may have a remote history of symptoms. Many factors that lead to clinical expression of AIP, including certain drugs and steroid hormones, have the capacity to induce hepatic ALAS1 and CYPs. When hepatic heme synthesis is increased, half-normal PBGD activity may become limiting, and ALA, PBG, and other heme pathway intermediates may accumulate. In addition, heme synthesis becomes impaired, and heme-mediated repression of hepatic ALAS1 is less effective.
It is not proved, however, that hepatic PBGD remains constant at approximately 50% of normal activity during exacerbations and remission of AIP, as in erythrocytes. An early report suggested that the enzyme activity is considerably less than half-normal in the liver during an acute attack. Hepatic PBGD activity might be reduced further once AIP becomes activated if, as suggested, excess PBG interferes with assembly of the dipyrromethane cofactor for this enzyme. It also seems likely that currently unknown genetic factors play a contributing role in, for example, patients who continue to have attacks even when known precipitants are avoided.
AIP is almost always latent before puberty and becomes active mostly in adult women, which suggests that endocrine factors, and especially adult levels of female steroid hormones, are important for clinical expression. Premenstrual attacks are probably the result of endogenous progesterone. Acute porphyrias are sometimes exacerbated by exogenous steroids, including oral contraceptive preparations containing progestins. Surprisingly, pregnancy is usually well tolerated, suggesting that beneficial metabolic changes may ameliorate the effects of high levels of progesterone.
Drugs that are unsafe in acute porphyrias ( Table 110.3 ) include those having the capacity to induce hepatic ALAS1, which is closely associated with induction of CYPs. Some chemicals (e.g., griseofulvin) can increase heme turnover by promoting the destruction of specific CYPs to form an inhibitor (e.g., N -methyl protoporphyrin) of ferrochelatase (FECH, the final enzyme in the pathway). Sulfonamide antibiotics are harmful but apparently not inducers of hepatic heme synthesis. Ethanol and other alcohols are inducers of ALAS1 and some CYPs.
| UNSAFE | SAFE |
|---|---|
| Barbiturates (all) | Narcotic analgesics |
| Sulfonamide antibiotics * | Aspirin |
| Meprobamate * (also mebutamate, * tybutamate * ) | Acetaminophen (paracetamol) |
| Carisoprodol * | Phenothiazines |
| Glutethimide * | Penicillin and derivatives |
| Methyprylon | Streptomycin |
| Ethchlorvynol * | Glucocorticoids |
| Mephenytoin | Bromides |
| Phenytoin * | Insulin |
| Succinimides | Atropine |
| Carbamazepine * | Cimetidine |
| Clonazepam ‡ | Ranitidine † |
| Primidone * | Acetazolamide |
| Valproic acid * | Allopurinol |
| Pyrazolones (aminopyrine, antipyrine) | Amiloride |
| Griseofulvin * | Bethanidine |
| Ergots | Bumetanide |
| Metoclopramide * ‡ | Coumarins |
| Rifampin * | Fluoxetine |
| Pyrazinamide * ‡ | Gabapentin |
| Diclofenac * ‡ | Gentamicin |
| Fluconazole * | Guanethidine |
| Oral contraceptives | Ofloxacin |
| Progesterone and synthetic progestins * | Propranolol |
| Danazol * | Succinylcholine |
| Alcohol | Tetracycline |
| ACEIs (especially enalapril) ‡ | |
| Spironolactone | |
| CCBs (especially nifedipine) ‡ | |
| Ketoconazole | |
| Ketamine * |
* Porphyria has been listed as a contraindication, warning, precaution, or adverse effect in U.S. labeling for these drugs. Estrogens are also listed as harmful in porphyria but have been implicated as harmful in acute porphyrias, mostly based only on experience with estrogen-progestin combinations. Although estrogens can exacerbate porphyria cutanea tarda, there is little evidence they are harmful in the acute porphyrias.
† Porphyria has been listed as a precaution in U.S. labeling for this drug. However, this drug is regarded as safe by other sources.
‡ These drugs have been classified as probably safe by some sources, but this is controversial, and they should be avoided.
Nutritional factors , principally reduced intake of calories and carbohydrates, as may occur with illness or attempts to lose weight, can increase porphyrin precursor excretion and induce attacks of porphyria. Increased carbohydrate intake may ameliorate attacks. Hepatic ALAS1 is modulated by the peroxisome proliferator-activated receptor-γ coactivator-1α, which is an important link between nutritional status and exacerbations of acute porphyria.
Other factors have been implicated. Chemicals in cigarette smoke, such as polycyclic aromatic hydrocarbons, can induce hepatic CYPs and heme synthesis. A survey of AIP patients found an association between smoking and repeated porphyric attacks. Attacks may result from metabolic stress and impaired nutrition associated with major illness, infection, or surgery. Clinical observations suggest an additive effect of multiple predisposing factors, including drugs, endogenous hormones, nutritional factors, and smoking, are common.
The mechanism of neural damage in acute porphyrias is poorly understood. The most favored hypothesis at present is that 1 or more heme precursors, or perhaps a derivative, are neurotoxic. Increased ALA in AIP, HCP, VP, ADP, plumbism, and hereditary tyrosinemia type 1, which have similar neurologic manifestations, suggests that this substance or a derivative may be neuropathic. Porphyrins derived from ALA after its uptake into cells may have toxic potential. ALA can also interact with γ-aminobutyric acid (GABA) receptors. Severe AIP greatly improves after allogeneic liver transplantation. This experience and the demonstration that recipients of AIP livers develop porphyria support the hypothesis that heme precursors from the liver cause the neurologic manifestations.
AIP occurs in all races and is the most common acute porphyria, with an estimated prevalence in most countries of 5 in 100,000. In Sweden, prevalence was estimated to be 7.7 in 100,000, including latent cases with normal porphyrin precursors. A much higher prevalence of 60-100 in 100,000 in northern Sweden is the result of a founder effect. The combined prevalence of AIP and VP in Finland is approximately 3.4 in 100,000. A survey of chronic psychiatric patients in the United States using an erythrocyte PBGD determination found a high prevalence (210 in 100,000) of PBGD deficiency, but a study in Mexico found a similar prevalence in psychiatric patients and controls. Population screening by erythrocyte PBGD activity or DNA analysis revealed a prevalence of 200 heterozygotes per 100,000 people in Finland, and 1 in approximately 1,675 (60 in 100,000) in France. Studies using exomic/genomic databases show that the estimated frequency of pathogenic mutations in the HMBS gene is 0.00056 (56 in 100,000) suggesting that the penetrance of this disorder may be as low as 1%, and that carriers of PBGD mutations that can cause AIP are much more common than previously believed.
Neurovisceral manifestations of acute porphyrias may appear any time after puberty, but rarely before ( Table 110.4 ). Symptomatic childhood cases have been reported, but most were not adequately documented biochemically and confirmed by genetic testing. Abdominal pain is the most common presenting symptom in such cases, but seizures are common and may precede the diagnosis of AIP. Other manifestations reported in children include peripheral neuropathy, myalgias, hypertension, irritability, lethargy, and behavioral abnormalities. A population-based study in Sweden indicated that symptoms suggestive of porphyria may occur in heterozygotes during childhood, even, in contrast to adults, when urinary porphyria precursors are not elevated. This study did not compare the frequency of such nonspecific symptoms in a control group of children. Very rare cases of homozygous AIP present differently, with severe neurologic manifestations early in childhood.
| SYMPTOMS AND SIGNS | FREQUENCY (%) | COMMENT |
|---|---|---|
| GASTROINTESTINAL | ||
| Abdominal pain | 85–95 | Usually unremitting (for hours or longer) and poorly localized but can be cramping. |
| Vomiting | 43–88 | Neurologic in origin and rarely accompanied by peritoneal signs, fever, or leukocytosis. |
| Constipation | 48–84 | Nausea and vomiting often accompany abdominal pain. May be accompanied by bladder paresis. |
| Diarrhea | 5–12 | |
| NEUROLOGIC | ||
| Pain in extremities, back | 50–70 | Pain may begin in the chest or back and move to the abdomen. Extremity pain chest, neck, or head indicates involvement of sensory nerves; objective sensory loss reported in 10–40% of cases. |
| Paresis | 42–68 | May occur early or late during a severe attack. Muscle weakness usually begins proximally rather than distally and more often in the upper than lower extremities. |
| Respiratory paralysis | 9–20 | Preceded by progressive peripheral motor neuropathy and paresis. |
| Mental symptoms | 40–58 | May range from minor behavioral changes to agitation, confusion, hallucinations, and depression. |
| Convulsions | 10–20 | A central neurologic manifestation of porphyria or caused by hyponatremia, which often results from syndrome of inappropriate antidiuretic hormone secretion or sodium depletion. |
| CARDIOVASCULAR | ||
| Tachycardia | 64–85 | May warrant treatment to control rate, if symptomatic |
| Systemic arterial hypertension | 36–55 | May require treatment during acute attacks, and sometimes becomes chronic. |
Acute attacks in adults are characterized by a constellation of nonspecific symptoms, which may become severe and life threatening. Abdominal pain occurs in 85–95% of AIP patients; is usually severe, steady, and poorly localized, but is sometimes cramping; and accompanied by signs of ileus, including abdominal distention and decreased bowel sounds. Nausea, vomiting, and constipation are common, but increased bowel sounds and diarrhea may occur. Bladder dysfunction may cause hesitancy and dysuria. Tachycardia , the most common physical sign, occurs in up to 80% of attacks. This is often accompanied by hypertension , restlessness, coarse or fine tremors, and excess sweating, which are attributed to sympathetic overactivity and increased catecholamines. Other common manifestations include mental symptoms; pain in the extremities, head, neck, or chest; muscle weakness; and sensory loss. Because all these manifestations are neurologic rather than inflammatory, there is little or no abdominal tenderness, fever, or leukocytosis.
Porphyric neuropathy is primarily motor and appears to result from axonal degeneration rather than demyelinization. Sensory involvement is indicated by pain in the extremities, which may be described as muscle or bone pain, and by numbness, paresthesias, and dysesthesias. Paresis may occur early in an attack but is more often a late manifestation in an attack that is not recognized and adequately treated. Rarely, severe neuropathy develops when there is little or no abdominal pain. Motor weakness most commonly begins in the proximal muscles of the upper extremities and then progresses to the lower extremities and the periphery. It is usually symmetric, but occasionally asymmetric or focal. Initially, tendon reflexes may be little affected or hyperactive and become decreased or absent. Cranial nerves, most often X and VII, may be affected, and blindness from involvement of the optic nerves or occipital lobes has been reported. More common central nervous system (CNS) manifestations include seizures, anxiety, insomnia, depression, disorientation, hallucinations, and paranoia. Seizures may result from hyponatremia, porphyria itself, or an unrelated cause. Chronic depression and other mental symptoms occur in some patients, but attribution to porphyria is often difficult.
Hyponatremia is common during acute attacks. Inappropriate antidiuretic hormone (ADH) secretion is often the most likely mechanism, but salt depletion from excess renal sodium loss, gastrointestinal (GI) loss, and poor intake have been suggested as causes of hyponatremia in some patients. Unexplained reductions in total blood and red blood cell volumes are sometimes found and increased ADH secretion might then be an appropriate physiologic response. Other electrolyte abnormalities may include hypomagnesemia and hypercalcemia.
The attack usually resolves within several days, unless treatment is delayed. Abdominal pain may resolve within a few hours and paresis within a few days. Even severe motor neuropathy can improve over months or several years, but may leave some residual weakness. Progression of neuropathy to respiratory paralysis and death seldom occurs with appropriate treatment and removal of harmful drugs. Sudden death may result from cardiac arrhythmia.
Levels of ALA and PBG are substantially increased during acute attacks. These levels may decrease after an attack but usually remain increased unless the disease becomes asymptomatic for a prolonged period.
Porphyrins are also markedly increased, which accounts for reddish urine in AIP. These are predominantly uroporphyrins, which can form nonenzymatically from PBG. Because the increased urinary porphyrins in AIP are predominantly isomer III, however, their formation is likely to be largely enzymatic, which might occur if excess ALA produced in the liver enters cells in other tissues and is then converted to porphyrins by the heme biosynthetic pathway. Porphobilin, a degradation product of PBG, and dipyrrylmethenes appear to account for brownish urinary discoloration. Total fecal porphyrins and plasma porphyrins are normal or slightly increased in AIP. Erythrocyte protoporphyrin may be somewhat increased in patients with manifest AIP.
Erythrocyte PBGD activity is approximately half-normal in most patients with AIP. The normal range is wide and overlaps with the range for AIP heterozygotes. Some PBGD gene mutations cause the enzyme to be deficient only in nonerythroid tissues. PBGD activity is also highly dependent on erythrocyte age, and an increase in erythropoiesis from concurrent illness in an AIP patient may raise the activity into the normal range. Thus, PBGD activity alone is insufficient to make the diagnosis of AIP.
An increased urinary PBG level establishes that a patient has 1 of the 3 most common acute porphyrias (see Table 110.2 ). Measuring PBG in serum is preferred when there is coexistent severe renal disease but is less sensitive when renal function is normal. Measurement of urinary ALA is less sensitive than PBG and also less specific, but will detect ADP, the 4th type of acute porphyria. Erythrocyte PBGD activity is decreased in most AIP patients and helps confirm the diagnosis in a patient with high PBG. A normal enzyme activity in erythrocytes does not exclude AIP.
Knowledge of the PBGD mutation in a family enables reliable identification of other gene carriers. Prenatal diagnosis can be performed by amniocentesis or chorionic villus sampling (CVP) in a fetus with a known PBGD mutation in the family. Prenatal diagnosis is typically not performed due to the low penetrance of the disorder and favorable prognosis with treatment.
AIP and other acute porphyrias are typically associated with mild abnormalities in liver function tests; some patients develop chronic liver disease. The risk of hepatocellular carcinoma is also increased, perhaps 60-70–fold after age 50, even in asymptomatic individuals who have increased porphyrins or porphyrin precursors. Few patients who developed this neoplasm had increases in serum α-fetoprotein. Patients with acute porphyrias, especially >50 yr old, should be screened at least yearly by ultrasound or an alternative imaging method.
The risk of chronic hypertension and impaired renal function is increased in these patients, most often with evidence of interstitial nephritis. A nephrotoxic effect of ALA may contribute. This may progress to severe renal failure and require renal transplantation.
Patients with recurrent attacks may develop chronic neuropathic pain , although this has not been well characterized. Referral to a neurologist is recommended for any patient with ongoing or residual neurologic symptoms. In addition, depression and anxiety are common in these patients.
Intravenous (IV) hemin is the treatment of choice for most acute attacks of porphyria. There is a favorable biochemical and clinical response to early treatment with hemin, but less rapid clinical improvement if treatment is delayed. It is no longer recommended that therapy with hemin for a severe attack be started only after an unsuccessful trial of IV glucose for several days. Mild attacks without severe manifestations, such as paresis, seizures, hyponatremia, or pain requiring opioids, may be treated with IV glucose. After IV administration, hemin binds to hemopexin and albumin in plasma and is taken up primarily in hepatocytes, where it augments the regulatory heme pool in hepatocytes, represses the synthesis of hepatic ALAS1, and dramatically reduces porphyrin precursor overproduction.
Hemin *
* Hemin is the generic name for all heme preparations used for intravenous administration. Hemin is also a chemical term that refers to the oxidized (ferric) form of heme (iron protoporphyrin IX) and is usually isolated as hemin chloride. In alkaline solution, the chloride is replaced by the hydroxyl ion, forming hydroxyheme, or hematin.
is available for IV administration in the United States as lyophilized hematin (Panhematin, Recordati). Degradation products begin to form as soon as the lyophilized product is reconstituted with sterile water, and these are responsible for phlebitis at the site of infusion and a transient anticoagulant effect. Loss of venous access due to phlebitis is common after repeated administration. Stabilization of lyophilized hematin by reconstitution with 30% human albumin can prevent these adverse effects; this is recommended especially if a peripheral vein is used for the infusion. Uncommon side effects of hemin include fever, aching, malaise, hemolysis, anaphylaxis, and circulatory collapse. Heme arginate, a more stable hemin preparation, is available in Europe and South Africa.
Hemin treatment should be instituted only after a diagnosis of acute porphyria has been initially confirmed by a marked increase in urinary PBG. When prior documentation of the diagnosis is available for review, it is not essential to confirm an increase in PBG with every recurrent attack, if other causes of the symptoms are excluded clinically. The standard regimen of hemin for treatment of acute porphyric attacks is 3-4 mg/kg/day for 4 days. Lower doses have less effect on porphyrin precursor excretion and probably less clinical benefit.
Drugs that may exacerbate porphyrias (see Table 110.3 ) should be discontinued whenever possible, and other precipitating factors identified. Hospitalization is warranted, except for mild attacks; for treatment of severe pain, nausea, and vomiting; for administration of hemin and fluids; and for monitoring vital capacity, nutritional status, neurologic function, and electrolytes. Pain usually requires an opioid; there is low risk for addiction after recovery from the acute attack. Ondansetron or a phenothiazine such as chlorpromazine is needed for nausea, vomiting, anxiety, and restlessness. Low doses of short-acting benzodiazepines can be given for restlessness or insomnia. β-Adrenergic blocking agents may be useful during acute attacks to control tachycardia and hypertension but may be hazardous in patients with hypovolemia and incipient cardiac failure.
The effects of carbohydrates on repressing hepatic ALAS1 and reducing porphyrin precursor excretion are weak compared to those of hemin. Therefore, carbohydrate loading is seldom beneficial except in mild attacks. Glucose polymer solutions by mouth are sometimes tolerated. At least 300 g of IV glucose, usually given as a 10% solution, has been recommended for adults hospitalized with attacks of porphyria. Amounts up to 500 g daily may be more effective, but large volumes may favor development of hyponatremia.
Liver transplantation was effective in several patients with severe AIP. A group from the United Kingdom reported their experience with liver transplantation in 10 AIP patients with significantly impaired quality of life and recurrent attacks refractory to medical management. Patients had a complete biochemical and symptomatic resolution after transplantation; 2 patients succumbed to multiorgan failure. Liver transplantation was also successful in a U.S. patient with AIP and intractable symptoms who became unresponsive to hemin therapy; liver transplantation normalized porphyrin precursor excretion, and symptoms resolved. However, liver transplantation is a high-risk procedure and should be considered only as a last resort. Hepatocyte-targeted RNA interference ( RNAi ) therapy is being developed to reverse directly the extremely elevated hepatic ALAS1 mRNA in this disease. Preliminary results from clinical trials are promising.
Seizures caused by hyponatremia or other electrolyte imbalances may not require prolonged treatment with anticonvulsant drugs, most of which have at least some potential for exacerbating acute porphyrias. Bromides, gabapentin, and probably vigabatrin are safe. Clonazepam may be less harmful than phenytoin or barbiturates. Control of hypertension is important and may help prevent chronic renal impairment, which can progress and require renal transplantation.
Patients often do well with avoidance of harmful drugs. Table 110.3 lists some drugs known or strongly suspected to be harmful or safe in the acute porphyrias. More extensive listings are available from the European Porphyria Network ( www.porphyria-europe.com ) and American Porphyria Foundation ( www.porphyriafoundation.com ), but some listings are controversial. Information regarding safety is lacking for many drugs, especially for those recently introduced.
Exogenous progestins, usually in combination with estrogens, can induce attacks of porphyria. Estrogens are seldom reported to be harmful when given alone. Synthetic steroids with an ethynyl substituent can cause a mechanism-based destruction of hepatic CYPs and should probably be avoided in patients with acute porphyria. Danazol is especially contraindicated.
Major surgery can be carried out safely in patients with acute porphyria, especially if barbiturates are avoided. Halothane has been recommended as an inhalation agent and propofol and midazolam as IV induction agents.
Pregnancy is usually well tolerated, which is surprising, because levels of progesterone, a potent inducer of hepatic ALAS1, are considerably increased during pregnancy. Some women do experience continuing attacks during pregnancy. This has sometimes been attributed to reduced caloric intake or metoclopramide, a drug sometimes used to treat hyperemesis gravidarum and considered harmful in acute porphyrias.
Diabetes mellitus and other endocrine conditions are not known to precipitate attacks of porphyria. In fact, the onset of diabetes mellitus and resulting high circulating glucose levels may decrease the frequency of attacks and lower porphyrin precursor levels in AIP.
The outlook for patients with acute porphyrias has improved greatly in the past several decades. In Finland, for example, 74% of patients with AIP or VP reported that they led normal lives, and <30% had recurrent attacks during several years of follow-up. In those presenting with acute symptoms, recurrent attacks were most likely within the next 1-3 yr. Moreover, only 6% of gene carriers who had never had attacks developed symptoms. The improved outlook may result from earlier detection, better treatment of acute attacks, and replacement of harmful drugs such as barbiturates and sulfonamides with safer drugs. However, some patients continue to have recurrent attacks, chronic pain, and other symptoms, even after avoiding known exacerbating factors.
For prevention of attacks, it is important to identify multiple inciting factors and remove as many as possible. Drugs for concurrent medical conditions should be reviewed. Because dietary factors are often unapparent, consultation with a dietitian may be useful. A well-balanced diet that is somewhat high in carbohydrate (60–70% of total calories) and sufficient to maintain weight is recommended. There is little evidence that additional dietary carbohydrate helps further in preventing attacks, and it may lead to weight gain. Patients who wish to lose excess weight should do so gradually and when they are clinically stable. Rapid weight loss after bariatric surgery may exacerbate acute porphyrias. Iron deficiency, which can be detected by a low serum ferritin level, should be corrected.
Gonadotropin-releasing hormone (GnRH) analogs, which reversibly suppress ovulation, can be dramatically effective for preventing frequently recurring luteal phase attacks, but baseline and continuing gynecologic evaluation and bone mineral density measurements are important; transdermal estrogen or a bisphosphonate may be added to prevent bone loss. Hemin administered once or twice weekly can prevent frequent, noncyclic attacks of porphyria in some patients. Alternatively, single-dose hemin can be administered “on demand” at an outpatient infusion center to abort an attack and prevent hospitalization, if a patient can recognize early “prodromal” symptoms. Inpatient management is warranted, however, if advanced manifestations such as vomiting, paresis, or other neuropsychiatric symptoms have developed.
A mutation identified in the index case can be sought in the child. Counseling should emphasize that the great majority of those who inherit a PBGD mutation never develop symptoms, and the prognosis of those who do is favorable. Therefore a normal, healthy life is expected, especially with avoidance of harmful drugs and other factors and prompt recognition and treatment of symptoms should they occur. Given the favorable outlook for most mutation carriers, even during pregnancy, having children is not precluded, and prenatal diagnosis of acute porphyrias is less important than it is for many other inherited diseases.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here