Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Everything that surrounds individual neurons can be considered part of the neuronal microenvironment. Technically, therefore, the neuronal microenvironment includes the extracellular fluid (ECF), capillaries, glial cells, and adjacent neurons. Although the term often is restricted to just the immediate ECF, the ECF cannot be meaningfully discussed in isolation because of its extensive interaction with brain capillaries, glial cells, and cerebrospinal fluid (CSF). How the microenvironment interacts with neurons and how the brain (used here synonymously with central nervous system, or CNS ) stabilizes it to provide constancy for neuronal function are the subjects of this discussion.
The concentrations of solutes in brain extracellular fluid (BECF) fluctuate with neural activity, and conversely, changes in ECF composition can influence nerve cell behavior. Not surprisingly, therefore, the brain carefully controls the composition of this important compartment. It does so in three major ways: First, the brain uses the blood-brain barrier (BBB) to protect the BECF from fluctuations in blood composition. Second, the CSF, produced by choroid plexus epithelial cells, strongly influences the composition of the BECF. Third, the surrounding glial cells “condition” the BECF.
The ratio of brain weight to body weight in humans is the highest in the animal kingdom. The average adult brain weight is ~1400 g in men and ~1300 g in women—approximately the same weight as the liver (see p. 944 ). This large and vital structure, which has the consistency of thick pudding, is protected from mechanical injury by a surrounding layer of bone and by the CSF in which it floats.
The brain is also metabolically fragile. This fragility arises from its high rate of energy consumption, absence of significant stored fuel in the form of glycogen (~5% of the amount in the liver), and rapid development of cellular damage when ATP is depleted. However, the brain is not the greediest of the body's organs; both the heart and kidney cortex have higher metabolic rates. Nevertheless, although it constitutes only 2% of the body by weight, the brain receives ~15% of resting blood flow and accounts for ~20% and 50% of total resting oxygen and glucose utilization, respectively. The brain's high metabolic demands arise from the need of its neurons to maintain the steep ion gradients on which neuronal excitability depends. In addition, neurons rapidly turn over their actin cytoskeleton. Neuroglial cells, the other major cells in the brain, also maintain steep transmembrane ion gradients. More than half of the energy consumed by the brain is directed to maintain ion gradients, primarily through operation of the Na-K pump (see pp. 115–117 ). An interruption of the continuous supply of oxygen or glucose to the brain results in rapid depletion of energy stores and disruption of ion gradients. Because of falling ATP levels in the brain, consciousness is lost within 10 seconds of a blockade in cerebral blood flow. Irreversible nerve cell injury can occur after only 5 to 10 minutes of interrupted blood flow.
CSF is a colorless, watery liquid. It fills the ventricles of the brain and forms a thin layer around the outside of the brain and spinal cord in the subarachnoid space. CSF is secreted within the brain by a highly vascularized epithelial structure called the choroid plexus and circulates to sites in the subarachnoid space, where it enters the venous blood system. The composition of CSF is highly regulated, and because it directly mixes with BECF, it helps regulate the composition of BECF. The choroid plexus can be thought of as the brain's “kidney” in that it stabilizes the composition of CSF, just as the kidney stabilizes the composition of blood plasma.
The ventricles of the brain are four small compartments located within the brain ( Fig. 11-1 A ). Each ventricle contains a choroid plexus and is filled with CSF. The ventricles are linked together by channels, or foramina, that allow CSF to move easily between them. The two lateral ventricles are the largest and are symmetrically located within the cerebral hemispheres. The choroid plexus of each lateral ventricle is located along the inner radius of this horseshoe-shaped structure (see Fig. 11-1 B ). The two lateral ventricles each communicate with the third ventricle, which is located in the midline between the thalami, through the two interventricular foramina of Monro. The choroid plexus of the third ventricle lies along the ventricle roof. The third ventricle communicates with the fourth ventricle by the cerebral aqueduct of Sylvius. The fourth ventricle is the most caudal ventricle and is located in the brainstem. It is bounded by the cerebellum superiorly and by the pons and medulla inferiorly. The choroid plexus of the fourth ventricle lies along only a portion of this ventricle's tent-shaped roof. The fourth ventricle is continuous with the central canal of the spinal cord. CSF escapes from the fourth ventricle and flows into the subarachnoid space through three foramina: the two laterally placed foramina of Luschka and the midline opening in the roof of the fourth ventricle, called the foramen of Magendie. We shall see below how CSF circulates throughout the subarachnoid space of the brain and spinal cord, and how it moves through brain tissue itself.
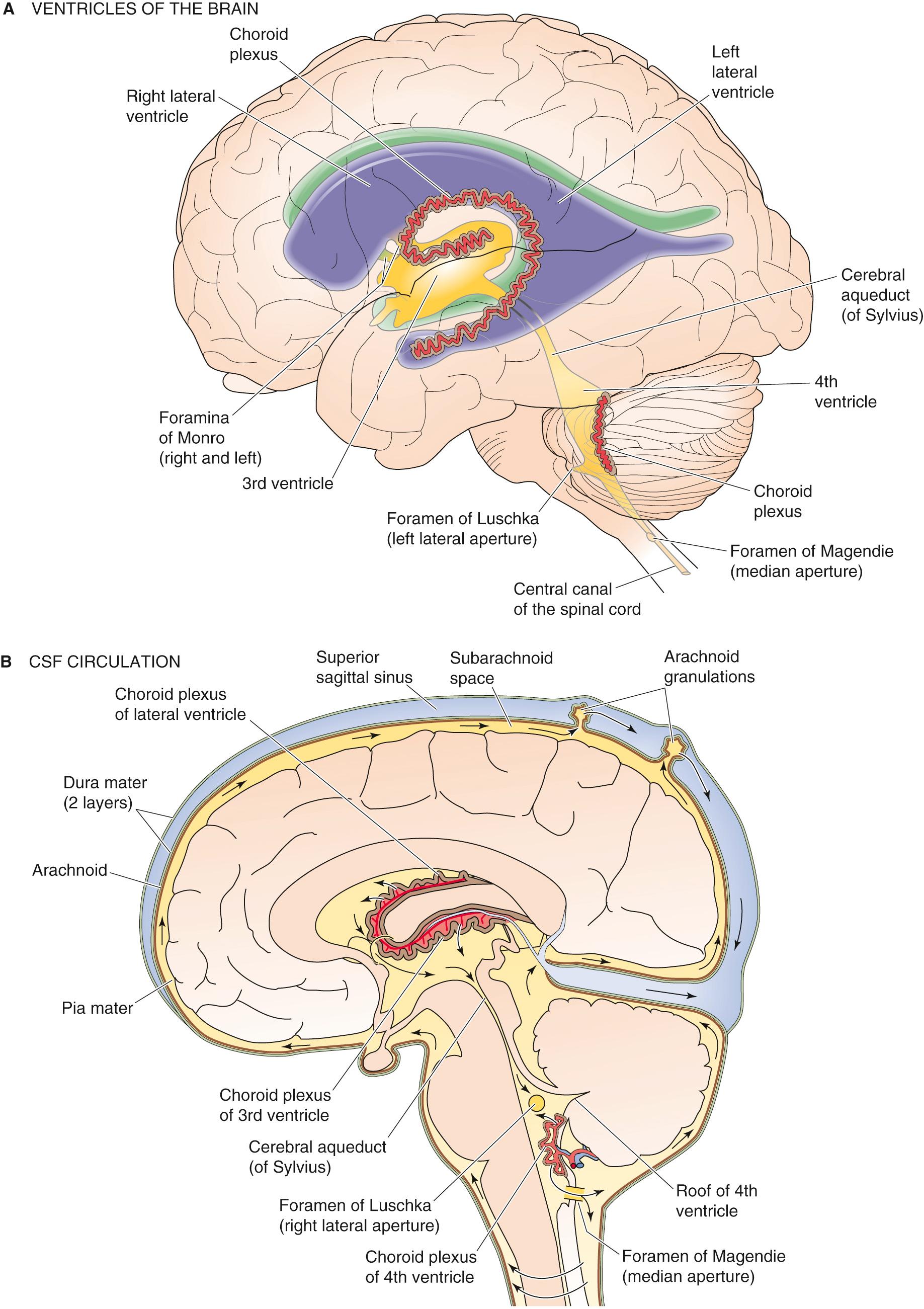
The brain and spinal cord are covered by two membranous tissue layers called the leptomeninges, which are in turn surrounded by a third, tougher layer. The innermost of these three layers is the pia mater; the middle is the arachnoid mater (or arachnoid membrane); and the outermost layer is the dura mater ( Fig. 11-2 ). Between the arachnoid mater and pia mater (i.e., the leptomeninges) is the subarachnoid space, which is filled with CSF that flows from the fourth ventricle. The CSF in the subarachnoid space completely surrounds the brain and spinal cord. In adults, the subarachnoid space and the ventricles with which they are continuous contain ~150 mL of CSF, 30 mL in the ventricles and 120 mL in the subarachnoid spaces of the brain and spinal cord.
The pia mater (Latin for “tender mother”) is a thin layer of connective tissue cells that is very closely applied to the surface of the brain and covers blood vessels as they plunge through the arachnoid into the brain. A nearly complete layer of astrocytic endfeet (see p. 286 )—the glia limitans —abuts the pia from the brain side and is separated from the pia by a basement membrane. The pia adheres so tightly to the associated glia limitans in some areas that the two seem to be continuous with each other; this combined structure is sometimes called the pial-glial membrane or layer. This layer does not restrict diffusion of substances between the BECF and the CSF.
The arachnoid membrane (from the Greek arachnoeides [cobweb-like]) is composed of layers of cells, resembling those that make up the pia, linked together by tight junctions. The arachnoid isolates the CSF in the subarachnoid space from blood in the overlying vessels of the dura mater. The cells that constitute the arachnoid and the pia are continuous in the trabeculae that span the subarachnoid space. These arachnoid and pial layers are relatively avascular; thus, the leptomeningeal cells that form them probably derive nutrition from the CSF that they enclose as well as from the ECF that surrounds them. The leptomeningeal cells can phagocytose foreign material in the subarachnoid space.
The dura mater is a thick, inelastic membrane that forms an outer protective envelope around the brain. The dura has two layers that split to form the intracranial venous sinuses. Blood vessels in the dura mater are outside the BBB (see below), and substances could easily diffuse from dural capillaries into the nearby CSF if it were not for the blood-CSF barrier created by the arachnoid.
An important function of CSF is to buffer the brain from mechanical injury. The CSF that surrounds the brain reduces the effective weight of the brain from ~1400 g to <50 g. This buoyancy is a consequence of the difference in the specific gravities of brain tissue (1.040) and CSF (1.007). The mechanical buffering that the CSF provides greatly diminishes the risk of acceleration-deceleration injuries in the same way that wearing a bicycle helmet reduces the risk of head injury. As you strike a tree, the foam insulation of the helmet gradually compresses and reduces the velocity of your head. Thus, the deceleration of your head is not nearly as severe as the deceleration of the outer shell of your helmet. The importance of this fluid suspension system is underscored by the consequences of reduced CSF pressure, which sometimes happens transiently after the diagnostic procedure of removal of CSF from the spinal subarachnoid space ( Box 11-1 ). Patients with reduced CSF pressure experience severe pain when they try to sit up or stand because the brain is no longer cushioned by shock-absorbing fluid and small gravity-induced movements put strain on pain-sensitive structures. Fortunately, the CSF leak that can result from lumbar puncture is only temporary; the puncture hole easily heals itself, with prompt resolution of all symptoms.
One of the most important diagnostic tests in neurology is the sampling of CSF by lumbar puncture. Critical information about the composition of CSF and about intracranial pressure can be obtained from this procedure. The anatomist Vesalius noted in 1543 that the ventricles are filled with a clear fluid, but the diagnostic technique of placing a needle into the lumbar subarachnoid space to obtain CSF was not introduced until 1891 by the neurologist Heinrich Quincke. ![]() N11-7 The method of lumbar puncture is dictated by spine anatomy. In adults, the spinal cord ends at the interspace between L1 and L2 (see p. 271 ). A hollow needle for sampling of CSF can be safely inserted into the subarachnoid space at the level of the L3-L4 interspace, well below the end of the spinal cord.
N11-7 The method of lumbar puncture is dictated by spine anatomy. In adults, the spinal cord ends at the interspace between L1 and L2 (see p. 271 ). A hollow needle for sampling of CSF can be safely inserted into the subarachnoid space at the level of the L3-L4 interspace, well below the end of the spinal cord.
Once the needle is in the subarachnoid space, the physician attaches it to a manometer to measure pressure. With the patient lying on the side, normal pressure varies from 100 to 180 mm H 2 O, or 7 to 13 mm Hg. With the subject in this position, and in the absence of a block to the free circulation of CSF, lumbar CSF pressure roughly corresponds to intracranial pressure. The physician can demonstrate direct communication of the pressure in the intracranial compartment to the lumbar subarachnoid space by gently compressing the external jugular veins in the neck for 10 seconds. This maneuver, called the Queckenstedt test, rapidly increases intracranial pressure because it increases the volume of intracranial venous blood. It quickly leads to an increase in lumbar pressure, which just as rapidly dissipates when the jugular pressure is removed.
CSF pressure can become elevated because of a pathological mass within the cranium, such as a tumor or collection of blood, or because the brain is swollen as a result of injury or infection (see Box 11-3 ). If a “mass lesion” (i.e., any pathological process that occupies intracranial space) is large or critically placed, it can displace the brain and cause interference with the free circulation of CSF. For example, an expanding mass in the cerebellum can force the inferior part of the cerebellum into the foramen magnum and block flow of CSF into the spinal subarachnoid space. Under these conditions, performance of lumbar puncture can precipitate a neurological catastrophe. If a needle is placed in the lumbar subarachnoid space and fluid is removed for diagnostic examination or leaks out after the needle is removed, the ensuing decrease in pressure in the lumbar space creates a pressure gradient across the foramen magnum and potentially forces the brain down into the spinal canal. This disaster is called herniation. For this reason, a computed tomographic scan or magnetic resonance image of the head is usually obtained before a lumbar puncture is attempted; the imaging study can rule out the possibility of a large intracranial lesion that might raise intracranial pressure and increase the risk of herniation when the subarachnoid space is punctured and CSF withdrawn. The Queckenstedt test must also be avoided when an intracranial mass is suspected because it could enhance the pressure gradient and hasten herniation.
For more information about Heinrich Quincke, visit the following websites:
Total CSF production is ~500 mL/day. Because the entire volume of CSF is ~150 mL, the CSF “turns over” about three times each day. Most of the CSF is produced by the choroid plexuses, which are present in four locations (see Fig. 11-1 ): the two lateral ventricles, the third ventricle, and the fourth ventricle. The capillaries within the brain appear to form as much as 30% of the CSF.
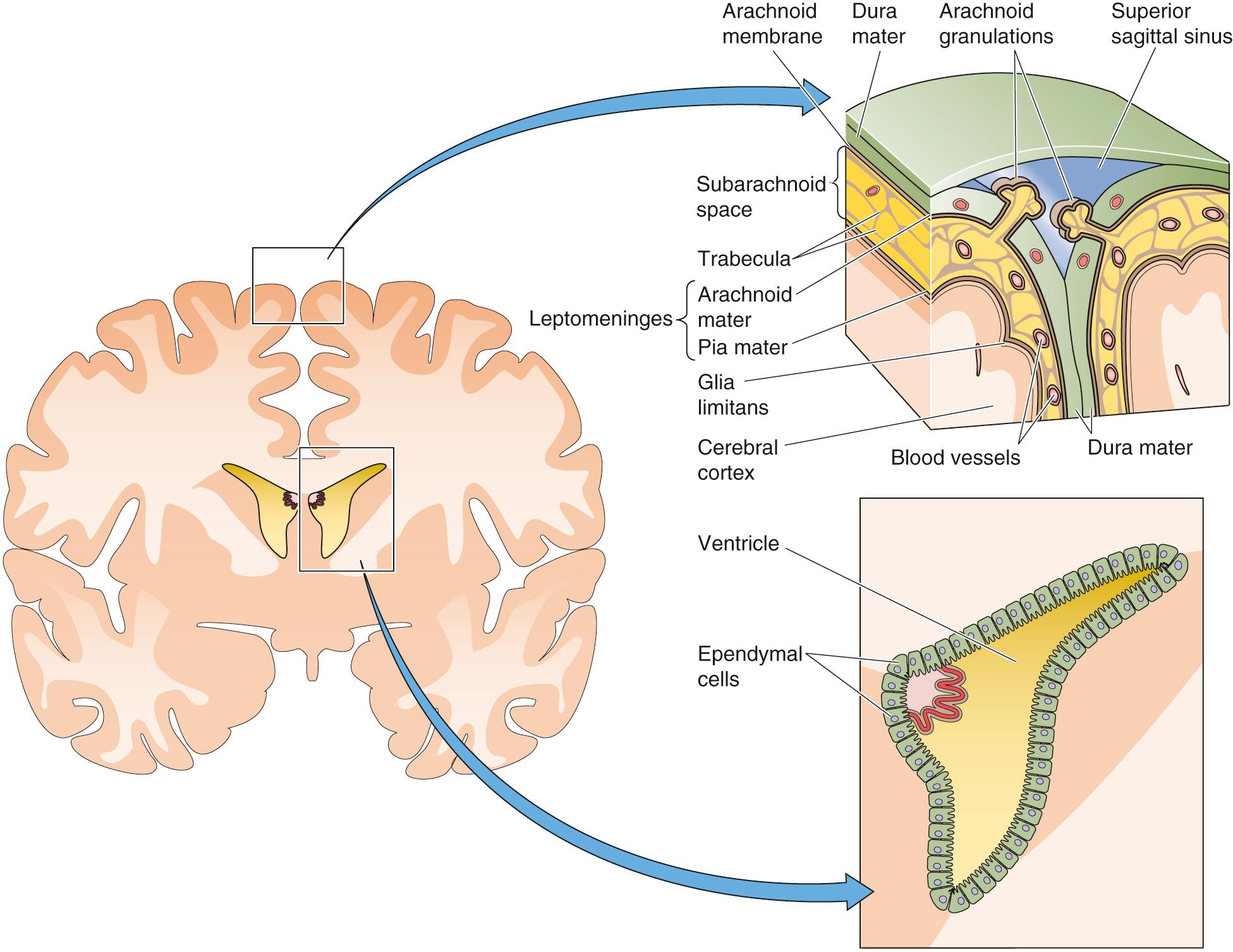
Secretion of new CSF creates a slight pressure gradient, which drives the circulation of CSF from its ventricular sites of origin into the subarachnoid space through three openings in the fourth ventricle, as discussed above. CSF percolates throughout the subarachnoid space and is finally absorbed into venous blood in the superior sagittal sinus, which lies between the two cerebral hemispheres (see Fig. 11-2 ). The sites of absorption are specialized evaginations of the arachnoid membrane into the venous sinus ( Fig. 11-3 A ). These absorptive sites are called pacchionian granulations ![]() N11-1 or simply arachnoid granulations when they are large (up to 1 cm in diameter) and arachnoid villi if their size is microscopic. These structures act as pressure-sensitive one-way valves for bulk CSF clearance; CSF can cross into venous blood but venous blood cannot enter CSF. The actual mechanism of CSF absorption may involve transcytosis (see pp. 477–487 ), or the formation of giant fluid-containing vacuoles that cross from the CSF side of the arachnoid epithelial cells to the blood side (see Fig. 11-3 A ). CSF may also be absorbed into spinal veins from herniations of arachnoid cells into these venous structures.
N11-1 or simply arachnoid granulations when they are large (up to 1 cm in diameter) and arachnoid villi if their size is microscopic. These structures act as pressure-sensitive one-way valves for bulk CSF clearance; CSF can cross into venous blood but venous blood cannot enter CSF. The actual mechanism of CSF absorption may involve transcytosis (see pp. 477–487 ), or the formation of giant fluid-containing vacuoles that cross from the CSF side of the arachnoid epithelial cells to the blood side (see Fig. 11-3 A ). CSF may also be absorbed into spinal veins from herniations of arachnoid cells into these venous structures.
Occasionally, arachnoid granulations are referred to by their old names: Pacchioni's granulations, pacchionian bodies, or pacchionian granulations— named after Italian anatomist Antonio Pacchioni.
The pressure of the CSF, which is higher than that of the venous blood, promotes net CSF movement into venous blood. When intracranial pressure (equivalent to CSF pressure) exceeds ~70 mm H 2 O, absorption commences and increases in a graded fashion with further intracranial pressure (see Fig. 11-3 B ). In contrast to CSF absorption, CSF formation is not sensitive to intracranial pressure. This arrangement helps stabilize intracranial pressure. Thus, if intracranial pressure increases, CSF absorption selectively increases as well, so that absorption exceeds formation. This response lowers CSF volume and tends to counteract the increased intracranial pressure. However, if absorption of CSF is impaired even at an initially normal intracranial pressure ( Box 11-2 ), CSF volume increases and causes an increase in intracranial pressure, which can lead to a disturbance in brain function.
Impaired CSF absorption is one mechanism proposed to explain a clinical form of ventricular enlargement called normal-pressure hydrocephalus. This condition is somewhat misnamed because the intracranial pressure is often intermittently elevated. Damage to the arachnoid villi can occur most commonly from infection or inflammation of the meninges or from the presence of an irritating substance, such as blood in the CSF after a subarachnoid hemorrhage. A spinal tap reveals normal pressure readings, but computed tomography or magnetic resonance imaging of the head shows enlargement of all four ventricles. Patients with normal-pressure hydrocephalus typically have progressive dementia, urinary incontinence, and gait disturbance, probably caused by stretching of axon pathways that course around the enlarged ventricles. A flexible plastic tube can be placed in one of the lateral ventricles to shunt CSF to venous blood or to the peritoneal cavity and thereby reduce CSF pressure. This procedure may reduce ventricular size and decrease neurological symptoms. The shunting procedure is also used for patients with obstructive hydrocephalus. In this condition, CSF outflow from the ventricles is blocked, typically at the aqueduct of Sylvius.
Some subarachnoid CSF flows into “sleeves” around arteries that dive from the subarachnoid space (see Fig. 11-2 ) deeply into the substance of the brain. This CSF appears to exit the sleeve by flowing across the pia/glia limitans and into brain extracellular space (BECS), where it mixes with BECF. Moving by convection, the BECF eventually appears to cross the pia/glia limitans that surrounds veins, enter via perivenous sleeves, and return to the subarachnoid space. This parenchymal CSF circuit—termed the glymphatic system ![]() N11-2 —may be an efficient pathway to rid the brain of extracellular debris, including glutamate and potentially dangerous peptides such as amyloid beta.
N11-2 —may be an efficient pathway to rid the brain of extracellular debris, including glutamate and potentially dangerous peptides such as amyloid beta.
The parenchymal CSF flow out of the periarterial sleeves, through the parenchyma of the brain, and into the perivenous sleeves is surprisingly brisk. Fluorescent dye experimentally placed in the subarachnoid space enters the BECS within minutes. Evidence suggests that arterial pulsations drives this parenchymal CSF circulation. Curiously, most of this CSF exchange with BECF occurs during sleep (or, artificially, during anesthesia) when BECS volume expands, thus enhancing transit of BECF through the tortuous extracellular spaces. This recently discovered CSF circulation pattern through brain tissue has been called the glymphatic system.
The cellular and molecular mechanisms by which fluid would cross the pia/glia limitans to exit periarterial sleeves, or to enter perivenous sleeves, is not understood. The entire glymphatic process appears to require the water channel AQP4, which is expressed at low density in the plasma membrane over the soma of astrocytes and at extremely high density in the plasma membrane of astrocytic endfeet that abuts either CNS capillaries or the pia mater (i.e., the glia limitans). AQP4 could play a role in the shrinkage of astrocytes during sleep (thereby promoting the expansion of the BECS) or in promotion of water movement through the astrocytic endfeet.
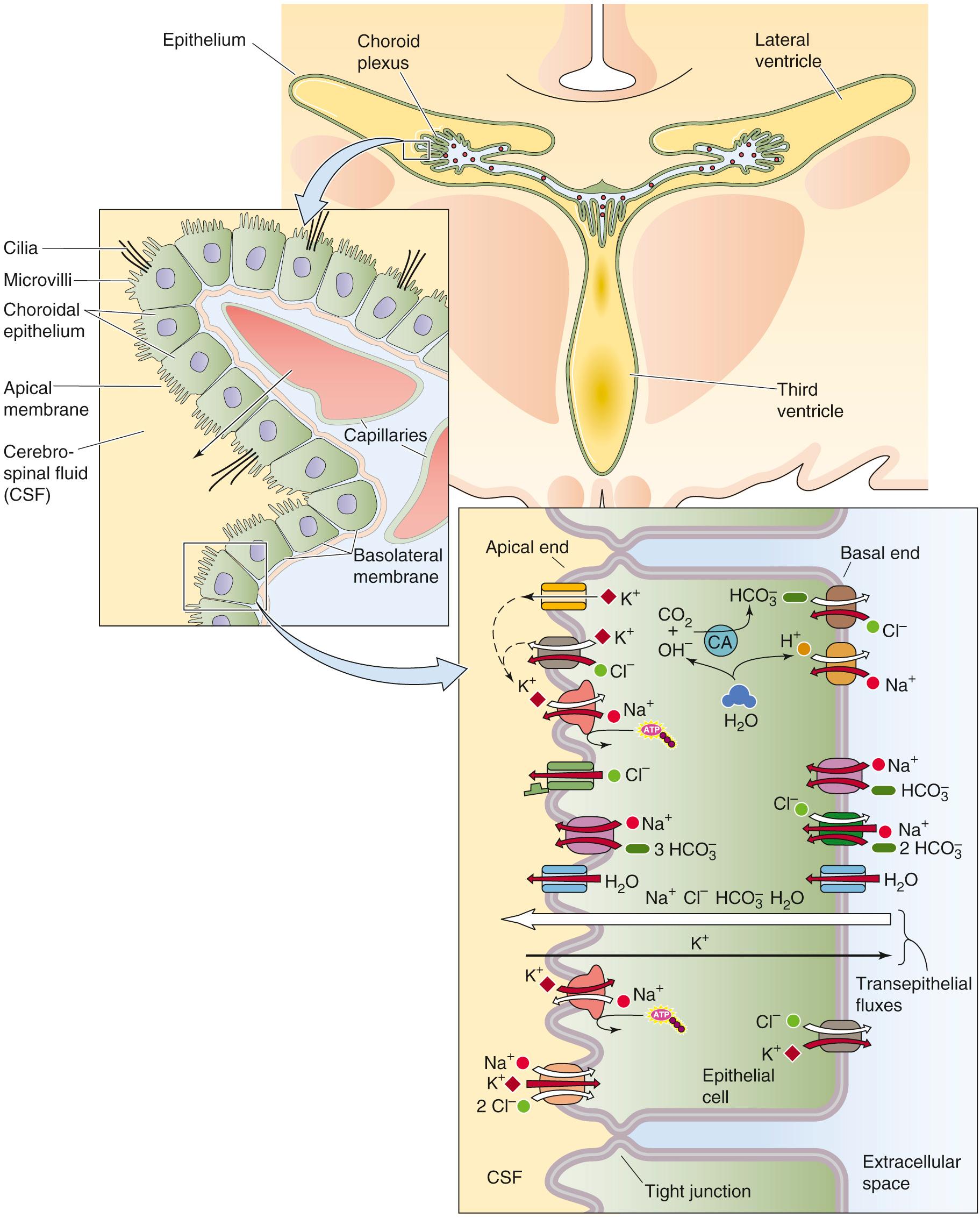
Each of the four choroid plexuses is formed during embryological development by invagination of the tela choroidea into the ventricular cavity ( Fig. 11-4 ). The tela choroidea consists of a layer of ependymal cells covered by the pia mater and its associated blood vessels. The choroid epithelial cells (see Fig. 11-4 , first inset) are specialized ependymal cells and therefore contiguous with the ependymal lining of the ventricles at the margins of the choroid plexus. Choroid epithelial cells are cuboidal and have an apical border with microvilli and cilia that project into the ventricle (i.e., into the CSF). The plexus receives its blood supply from the anterior and posterior choroidal arteries; blood flow to the plexuses—per unit mass of tissue—is ~10-fold greater than the average cerebral blood flow. Sympathetic and parasympathetic nerves innervate each plexus, and sympathetic input appears to inhibit CSF formation. A high density of relatively leaky capillaries is present within each plexus; as discussed below, these capillaries are outside the BBB. The choroid epithelial cells are bound to one another by tight junctions that completely encircle each cell, an arrangement that makes the epithelium an effective barrier to free diffusion. Thus, although the choroid capillaries are outside the BBB, the choroid epithelium insulates the ECF around these capillaries (which has a composition more similar to that of arterial blood) from the CSF. Moreover, the thin neck that connects the choroid plexus to the rest of the brain isolates the ECF near the leaky choroidal capillaries from the highly protected BECF in the rest of the brain.
The composition of CSF differs considerably from that of plasma; thus, CSF is not just an ultrafiltrate of plasma ( Table 11-1 ). For example, CSF has lower concentrations of K + and amino acids than plasma does, and it contains almost no protein. Moreover, the choroid plexuses rigidly maintain the concentration of ions in CSF in the face of large swings in ion concentration in plasma. This ion homeostasis includes K + ,  , Mg 2+ , Ca 2+ , and, to a lesser extent, Na + and Cl − . All these ions can affect neural function, hence the need for tight homeostatic control. The neuronal microenvironment is so well protected from the blood by the choroid plexuses and the rest of the BBB that essential micronutrients, such as vitamins and trace elements that are needed in very small amounts, must be selectively transported into the brain. Some of these micronutrients are transported into the brain primarily by the choroid plexus and others primarily by the endothelial cells of the blood vessels. In comparison, the brain continuously metabolizes relatively large amounts of “macronutrients,” such as glucose and some amino acids.
, Mg 2+ , Ca 2+ , and, to a lesser extent, Na + and Cl − . All these ions can affect neural function, hence the need for tight homeostatic control. The neuronal microenvironment is so well protected from the blood by the choroid plexuses and the rest of the BBB that essential micronutrients, such as vitamins and trace elements that are needed in very small amounts, must be selectively transported into the brain. Some of these micronutrients are transported into the brain primarily by the choroid plexus and others primarily by the endothelial cells of the blood vessels. In comparison, the brain continuously metabolizes relatively large amounts of “macronutrients,” such as glucose and some amino acids.
| SOLUTE | PLASMA (mM OF PROTEIN-FREE PLASMA) | CSF (mM) | CSF/PLASMA RATIO |
|---|---|---|---|
| Na + | 153 | 147 | 0.96 |
| K + | 4.7 | 2.9 | 0.62 |
| Ca 2+ | 1.3 (ionized) | 1.1 (ionized) | 0.85 |
| Mg 2+ | 0.6 (ionized) | 1.1 (ionized) | 1.8 |
| Cl − | 110 | 113 | 1.03 |
 |
24 | 22 | 0.92 |
 and and  |
0.75 (ionized) | 0.9 | 1.2 |
| pH | 7.40 | 7.33 | |
| Amino acids | 2.6 | 0.7 | 0.27 |
| Proteins | 7 g/dL | 0.03 g/dL | 0.004 |
| Osmolality | 290 mOsm | 290 mOsm | 1.00 |
CSF forms in two sequential stages. First, ultrafiltration of plasma occurs across the fenestrated capillary wall (see p. 462 ) into the ECF beneath the basolateral membrane of the choroid epithelial cell. Second, choroid epithelial cells secrete fluid into the ventricle. CSF production occurs with a net transfer of NaCl and NaHCO 3 that drives water movement isosmotically (see Fig. 11-4 , right inset, large white arrow labeled “Transepithelial fluxes”). The renal proximal tubule (see p. 761 ) and small intestine (see p. 903 ) also perform near-isosmotic transport, but in the direction of absorption rather than secretion. In addition, the choroid plexus conditions CSF by absorbing K + (see Fig. 11-4 , right inset, thin black arrow labeled “Transepithelial fluxes”) and certain other substances (e.g., 5-hydroxyindoleacetic acid, a metabolite of serotonin).
The upper portion of the right inset of Figure 11-4 summarizes the ion transport processes that mediate CSF secretion. The net secretion of Na + from plasma to CSF is a two-step process. The Na-K pump in the choroid plexus, unlike that in other epithelia (see pp. 137–138 ), is unusual in being located on the apical membrane, where it moves Na + out of the cell into the CSF—the first step. This active movement of Na + out of the cell generates an inward Na + gradient across the basolateral membrane, energizing basolateral Na + entry—the second step—through Na-H exchange and Na + -coupled  transport. In the case of Na-H exchange, the limiting factor is the availability of intracellular H + , which carbonic anhydrase generates, along with
transport. In the case of Na-H exchange, the limiting factor is the availability of intracellular H + , which carbonic anhydrase generates, along with  , from CO 2 and H 2 O. Thus, blocking of the Na-K pump with ouabain halts CSF formation, whereas blocking of carbonic anhydrase with acetazolamide slows CSF formation.
, from CO 2 and H 2 O. Thus, blocking of the Na-K pump with ouabain halts CSF formation, whereas blocking of carbonic anhydrase with acetazolamide slows CSF formation.
The net secretion of Cl − , like that of Na + , is a two-step process. The first step is the intracellular accumulation of Cl − by the basolateral Cl-HCO 3 exchanger. Note that the net effect of parallel Cl-HCO 3 exchange and Na-H exchange is NaCl uptake. The second step is efflux of Cl − across the apical border into the CSF through either a Cl − channel or a K/Cl cotransporter.
 secretion into CSF is important for neutralizing acid produced by CNS cells. At the basolateral membrane, the epithelial cell probably takes up
secretion into CSF is important for neutralizing acid produced by CNS cells. At the basolateral membrane, the epithelial cell probably takes up  directly from the plasma filtrate through electroneutral Na/HCO 3 cotransporters (see Fig. 5-11 F ) and the Na + -driven Cl-HCO 3 exchanger (see Fig. 5-13 C ). As noted before,
directly from the plasma filtrate through electroneutral Na/HCO 3 cotransporters (see Fig. 5-11 F ) and the Na + -driven Cl-HCO 3 exchanger (see Fig. 5-13 C ). As noted before,  can also accumulate inside the cell after CO 2 entry. The apical step, movement of intracellular
can also accumulate inside the cell after CO 2 entry. The apical step, movement of intracellular  into the CSF, probably occurs by an electrogenic Na/HCO 3 cotransporter (see Fig. 5-11 D ) and Cl − channels (which may be permeable to
into the CSF, probably occurs by an electrogenic Na/HCO 3 cotransporter (see Fig. 5-11 D ) and Cl − channels (which may be permeable to  ).
).
The lower portion of the right inset of Figure 11-4 summarizes K + absorption from the CSF. The epithelial cell takes up K + by the Na-K pump and the Na/K/Cl cotransporter at the apical membrane (see Fig. 5-11 G ). Most of the K + recycles back to the CSF, but a small amount exits across the basolateral membrane and enters the blood. The concentration of K + in freshly secreted CSF is ~3.3 mM. Even with very large changes in plasma [K + ], the [K + ] in CSF changes very little. The value of [K + ] in CSF is significantly lower in the subarachnoid space than in choroid secretions, which suggests that brain capillary endothelial cells remove extracellular K + from the brain.
Water transport across the choroid epithelium is driven by a small osmotic gradient favoring CSF formation. This water movement is facilitated by the water channel aquaporin 1 (AQP1; see p. 110 ) on both the apical and basal membranes as in the renal proximal tubule (see pp. 761–762 ).
The average width of the space between brain cells is ~20 nm, which is about three orders of magnitude smaller than the diameter of either a neuron or a glial cell body ( Fig. 11-5 ). However, because the surface membranes of neurons and glial cells are highly folded (i.e., have a large surface-to-volume ratio), the BECF in toto has a sizable volume fraction, ~20%, of total brain volume. The fraction of the brain occupied by BECF varies somewhat in different areas of the CNS and increases during sleep. Moreover, because brain cells can increase volume rapidly during intense neural activity, the BECF fraction can reversibly decrease within seconds from ~20% to ~17% of brain volume.
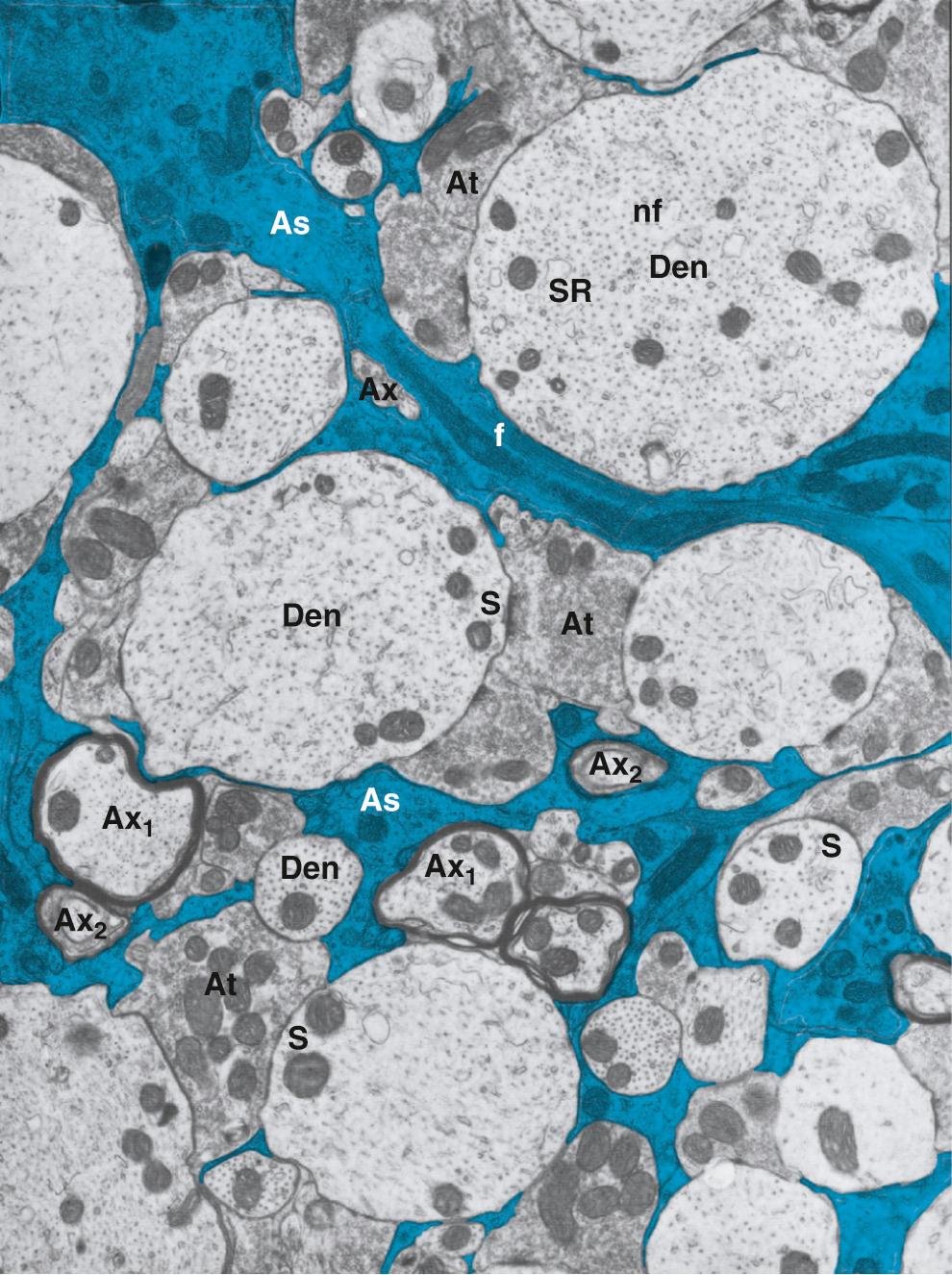
Even though the space between brain cells is extremely small, diffusion of ions and other solutes within this thin BECF space is reasonably high. However, a particle that diffuses through the BECF from one side of a neuron to the other must take a circuitous route that is described by a parameter called tortuosity. For a normal width of the cell-to-cell spacing, this tortuosity reduces the rate of diffusion by ~60% compared with movement in free solution. Decreases in cell-to-cell spacing can further slow diffusion. For example, brain cells, especially glial cells, swell under certain pathological conditions and sometimes with intense neural activity. Cell swelling is associated with a reduction in BECF because water moves from the BECF into cells. The intense cell swelling associated with acute anoxia, for example, can reduce BECF volume from ~20% to ~5% of total brain volume. By definition, this reduced extracellular volume translates to reduced cell-to-cell spacing, which further slows the extracellular movement of solutes between the blood and brain cells ( Box 11-3 ).
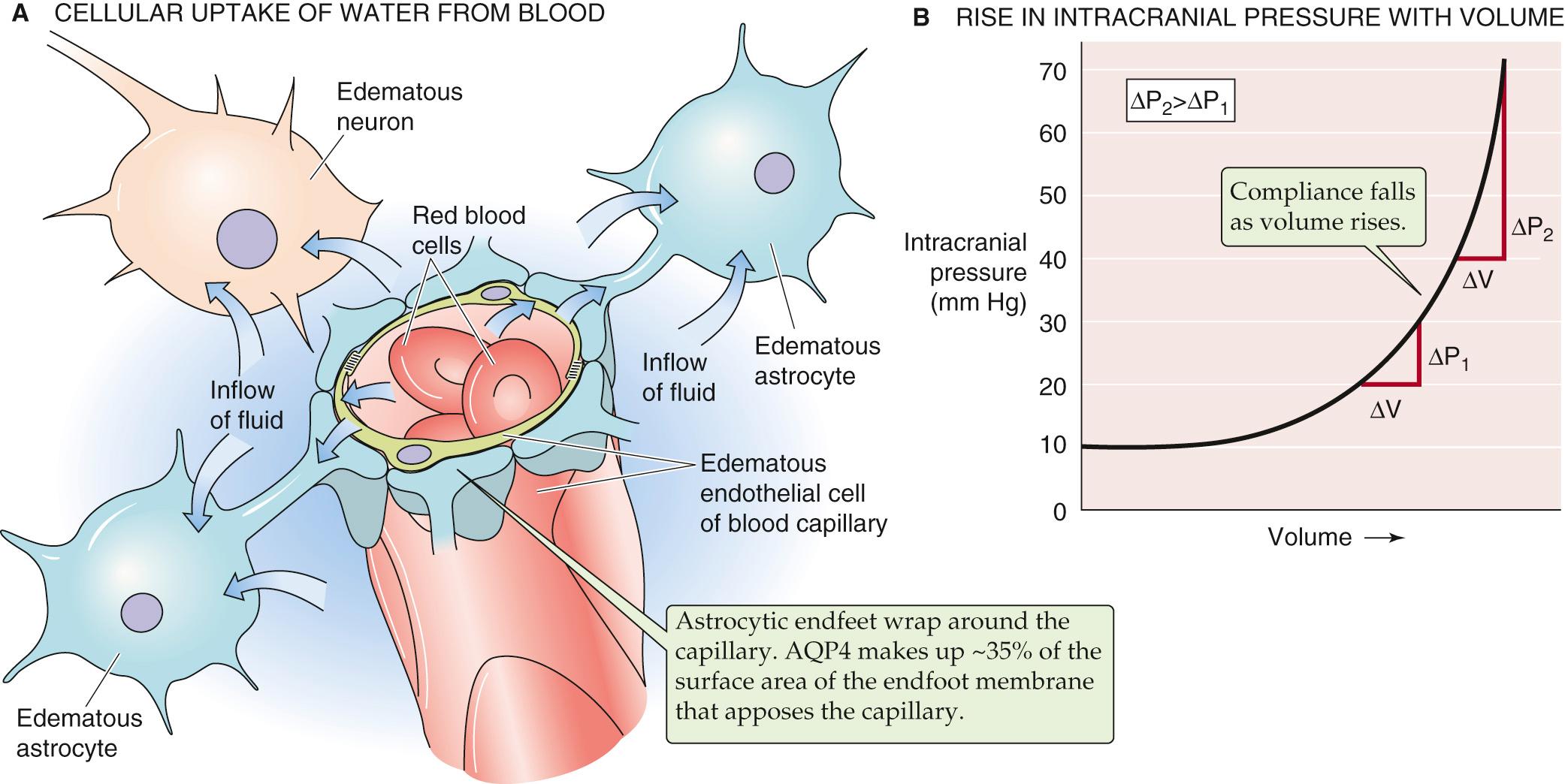
Almost any type of insult to the brain causes cell swelling, which is frequently accompanied by a net accumulation of water within the brain that is referred to as cerebral edema. Cell swelling in the absence of net water accumulation in the brain does not constitute cerebral edema. For example, intense neural activity causes a rapid shift of fluid from the BECF to the intracellular space, with no net change in brain water content. In cerebral edema, the extra water comes from the blood, as shown in Figure 11-6 .
The mechanisms by which glial cells and neurons swell are not completely understood. Neuron cell bodies and dendrites, but not axons, swell when exposed to high concentrations of the neurotransmitter glutamate. This transmitter, along with others, is released to the BECF in an uncontrolled fashion with brain injury. Activation of ionotropic glutamate receptors (see pp. 323–324 ) allows Na + to enter neurons, and water and Cl − follow passively. Glial cells, both astrocytes and oligodendrocytes, swell vigorously under pathological conditions. One mechanism of glial swelling is an increase in [K + ] o , which is a common ionic disturbance in a variety of brain pathological processes. This elevated [K + ] o causes a net uptake of K + , accompanied by the passive influx of Cl − and water.
Cerebral edema can be life threatening when it is severe. The problem is a mechanical one. The skull is an inelastic container housing three relatively noncompressible substances: brain, CSF, and blood. A significant increase in the volume of CSF, blood, or brain rapidly causes increased pressure within the skull (see Fig. 11-6 B ). If the cerebral edema is generalized, it can be tolerated until intracerebral pressure exceeds arterial blood pressure, at which point blood flow to the brain stops, with disastrous consequences. Fortunately, sensors in the medulla detect the increased intracerebral pressure and can partially compensate by increasing arterial pressure (Cushing reflex; see p. 559 ). Focal cerebral edema (i.e., edema involving an isolated portion of the brain) causes problems by displacing nearby brain tissue. This abnormality may result in distortion of normal anatomical relationships, with selective pressure on critical structures such as the brainstem.
Clinical evidence of cerebral edema results directly from the increased intracranial pressure and includes headache, vomiting, altered consciousness, and focal neurological problems. Because of its long intracranial course, the sixth cranial nerve is especially vulnerable to stretching and dysfunction.
Hyperventilation is the most effective means of combating the acute increase in intracranial pressure associated with severe cerebral edema. Hyperventilation causes a prompt respiratory alkalosis (see p. 634 ) that is rapidly translated to an increase in the pH surrounding vascular smooth muscle, thereby triggering vasoconstriction and reduced cerebral blood flow (see p. 559 ). Thus, total intracranial blood content falls, with a rapid subsequent drop in intracranial pressure. Alternatively, the brain can be partially dehydrated by adding osmoles to the blood in the form of intravenously administered mannitol (see p. 131 ).
The BECF is the route by which important molecules such as oxygen, glucose, and amino acids reach brain cells and by which the products of metabolism, including CO 2 and catabolized neurotransmitters, leave the brain. The BECF also permits molecules that are released by brain cells to diffuse to adjacent cells. Neurotransmitter molecules released at synaptic sites, for example, can spill over from the synaptic cleft and contact nearby glial cells and neurons, in addition to their target postsynaptic cell. Glial cells express neurotransmitter receptors, and neurons have extrajunctional receptors; therefore, these cells are capable of receiving “messages” sent through the BECF. Numerous trophic molecules (see p. 292 ) secreted by brain cells diffuse in the BECF to their targets. Intercellular communication by way of the BECF is especially well suited for the transmission of tonic signals that are ideal for longer-term modulation of the behavior of aggregates of neurons and glial cells. The chronic presence of variable amounts of neurotransmitters in the BECF supports this idea.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here