Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The primordial lymphatic system is first seen during the sixth week of development in the form of lymph sacs located next to the jugular veins. During the eighth week, the cisterna chyli forms just dorsal to the aorta, and, at the same time, two additional lymphatic sacs corresponding to the iliofemoral vascular pedicles begin forming. Communicating channels connecting the lymph sacs, which will become the thoracic duct, develop during the ninth week.
From this primordial lymphatic system sprout endothelial buds that grow with the venous system to form the peripheral lymphatic plexus ( Fig. 66.1 ). Failure of one of the initial jugular lymphatic sacs to develop proper connections and drainage with the lymphatic system and, subsequently, the venous system may produce focal lymph cysts (cavernous lymphangiomas), also known as cystic hygromas. Similarly, failure of embryologic remnants of lymphatic tissues to connect to efferent channels leads to the development of cystic lymphatic formations (simple capillary lymphangiomas) that, depending on their location, are classified as truncal, mesenteric, intestinal, and retroperitoneal lymphangiomas. Hypoplasia or failure of development of drainage channels connecting the lymphatic systems of extremities to the main primordial lymphatic system of the torso may result in primary lymphedema of the extremities.
Lymphangiogenesis appears to be regulated by the vascular endothelial growth factors C and D (VEGF-C, VEGF-D); their receptor, VEGFR-3; and their binding protein, neuropilin 2 (Nrp2). Consistent with these findings, Nrp2-deficient mice have lymphatic hypoplasia, and heterozygous inactivating mutation of VEGFR-3 is found in Chy mice, an animal model of primary lymphedema, which appears to be the underlying problem in patients with Milroy disease (congenital familial lymphedema). A number of additional genes have recently been found to be related to lymphatic disorders. The best studied at this point are the gene for the forkhead family transcription factor FOXC2 (responsible for the hereditary lymphedema-distichiasis syndrome) and the gene for the transcription factor SOX18 (related to recessive and dominant forms of hypotrichosis-lymphedema-telangiectasia). As more causal genes are identified, the possibility arises for a classification built on patient phenotypes for which the gene is known. Receptor activity-modifying protein 1 (RAMP1) has also been identified as an important factor for lymphangiogenesis. RAMP1 knockout mice have greater amounts of surgery-associated lymphedema compared to controls. This is thought to occur from a lack of attenuation of proinflammatory macrophage recruitment in the RAMP1 knockout mice.
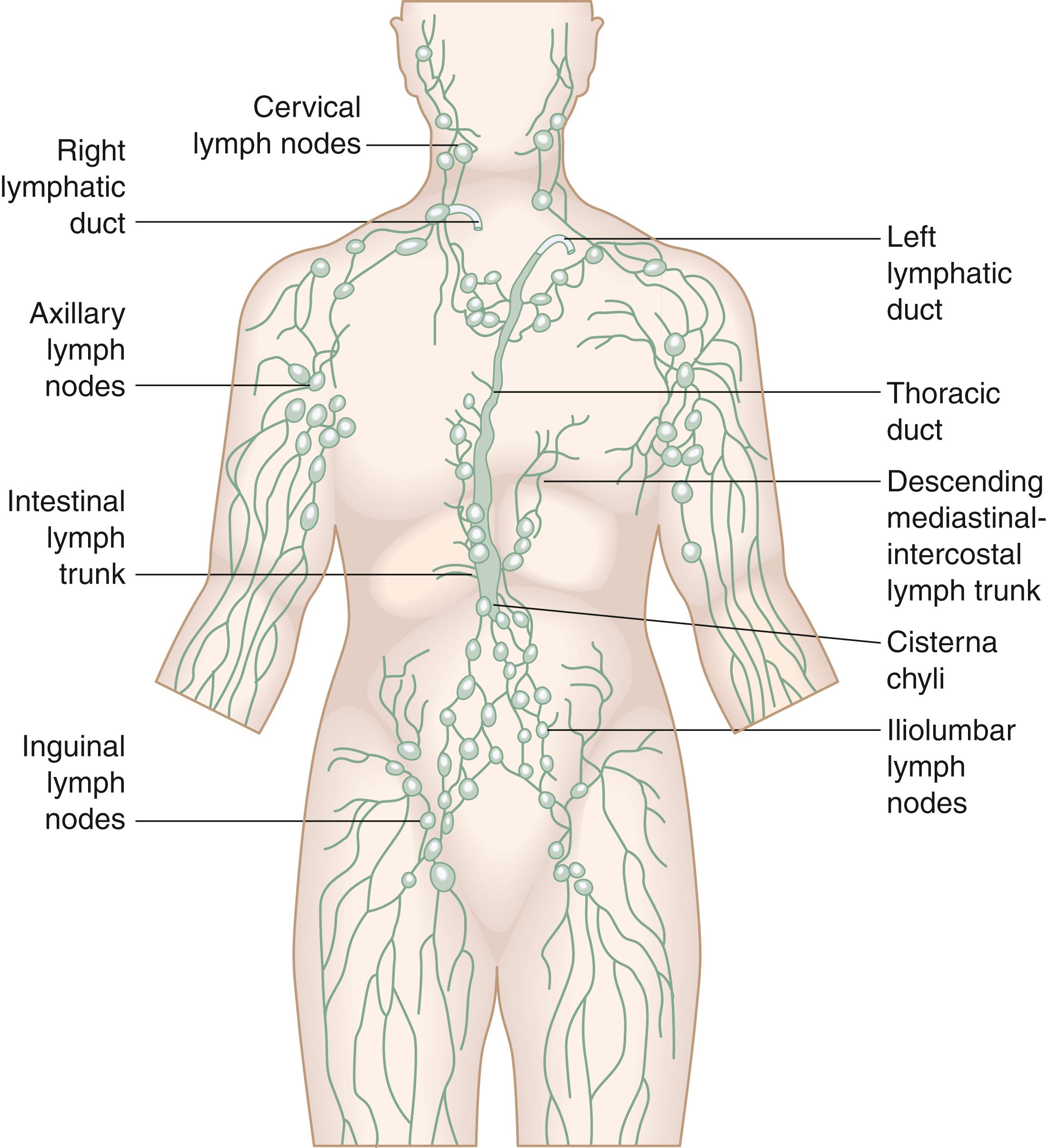
The lymphatic system is composed of three elements: (1) the initial or terminal lymphatic capillaries, which absorb lymph; (2) the collecting vessels, which serve primarily as conduits for lymph transport; and (3) the lymph nodes, which are interposed in the pathway of the conducting vessels, filtering the lymph and serving a primary immunologic role.
The terminal lymphatics have special structural characteristics that allow entry not only of large macromolecules but even of cells and microbes. Their most important structural feature is a high porosity resulting from a very small number of tight junctions between endothelial cells, a limited and incomplete basement membrane, and anchoring filaments (4–10 nm) tethering the interstitial matrix to the endothelial cells. These filaments, once the turgor of the tissue increases, are able to pull on the endothelial cells and essentially introduce large gaps between them, which then allow very low resistance influx of interstitial fluid and macromolecules in the lymphatic channels. The collecting vessels ascend alongside the primary blood vessels of the organ or limb, pass through the regional lymph nodes, and drain into the main lymph channels of the torso. These channels eventually empty into the venous system through the thoracic duct. There are additional communications between the lymphatic and the venous systems. These smaller lymphovenous shunts mostly occur at the level of lymph nodes and around major venous structures, such as the jugular, subclavian, and iliac veins. Several structures in the body contain no lymphatics. Specifically, lymphatics have not been found in the epidermis, cornea, central nervous system, cartilage, tendon, and muscle.
The lymphatic system has three main functions. First, tissue fluid and macromolecules that undergo ultrafiltration at the level of the arterial capillaries are reabsorbed and returned to the circulation through the lymphatic system. Every day, 50% to 100% of the intravascular proteins are filtered this way in the interstitial space. Normally, they then enter the terminal lymphatics and are transported through the collecting lymphatics back into the venous circulation. Second, antigens, immune cells, microbes, and mutant cells arriving in the interstitial space enter the lymphatic system and are presented to the lymph nodes, which represent the first line of the immune system. Last, at the level of the gastrointestinal tract, lymph vessels are responsible for the uptake and transport of most of the fat absorbed from the bowel. Recent data suggest that a relationship between fat and lymphatics may exist well beyond the gut alone. It appears that peripheral tissue lipid transport and homeostasis may be, in part, determined by lymphatic function, hence the increased fat deposition seen in lymphedema. ,
In contrast to what happens with venous forward flow, lymph’s centripetal transport occurs mainly through intrinsic contractility of the individual lymphatic vessels, which in concert with competent valvular mechanisms is effective in establishing constant forward flow of lymph. In addition to the intrinsic contractility, other factors, such as surrounding muscle activity, negative pressure secondary to breathing, and transmitted arterial pulsations, have a lesser role in the forward lymph flow. These secondary factors appear to become more important under conditions of lymph stasis and congestion of the lymphatic vessels.
Lymphedema is the result of an inability of the existing lymphatic system to accommodate the protein and fluid entering the interstitial compartment at the tissue level. Impaired lymphatic vasculature can be either aplastic, hypoplastic, or hyperplastic. All of these patterns can lead to clinical lymphedema. In aplasia and hypoplasia an absent or diminished number of lymphatics are seen. In hyperplasia, the vessels are incompetent and tortuous. Hyperplasia is only a cause in 8% of patients with lymphedema.
An international working group developed a clinical staging for lymphedema. In the latent phase, excess fluid accumulates and the lymphatics become fibrosed. In the latent phase, there is no clinical evidence of edema. In the first stage of lymphedema, impaired lymphatic drainage results in protein-rich fluid accumulation in the interstitial compartment. Clinically, this is manifested as soft pitting edema. In the second stage of lymphedema, the clinical condition is further exacerbated by accumulation of fibroblasts, adipocytes, and, perhaps most important, macrophages in the affected tissues, which culminates in a local inflammatory response. This results in important structural changes from the deposition of connective tissue and adipose elements at the skin and subcutaneous level. In the second stage of lymphedema, tissue edema is more pronounced, is nonpitting, and has a spongy consistency. Elevation does not reduce the edema and skin fibrosis is more apparent. In the third and most advanced stage of lymphedema, the affected tissues sustain further injury as a result of both the local inflammatory response and recurrent infectious episodes that typically result from minimal subclinical breaks in the skin. Such repeated episodes injure the incompetent, remaining lymphatic channels, progressively worsening the underlying insufficiency of the lymphatic system. This eventually results in excessive subcutaneous fibrosis and scarring with associated severe skin changes characteristic of lymphostatic elephantiasis. Edema is irreversible at this stage.
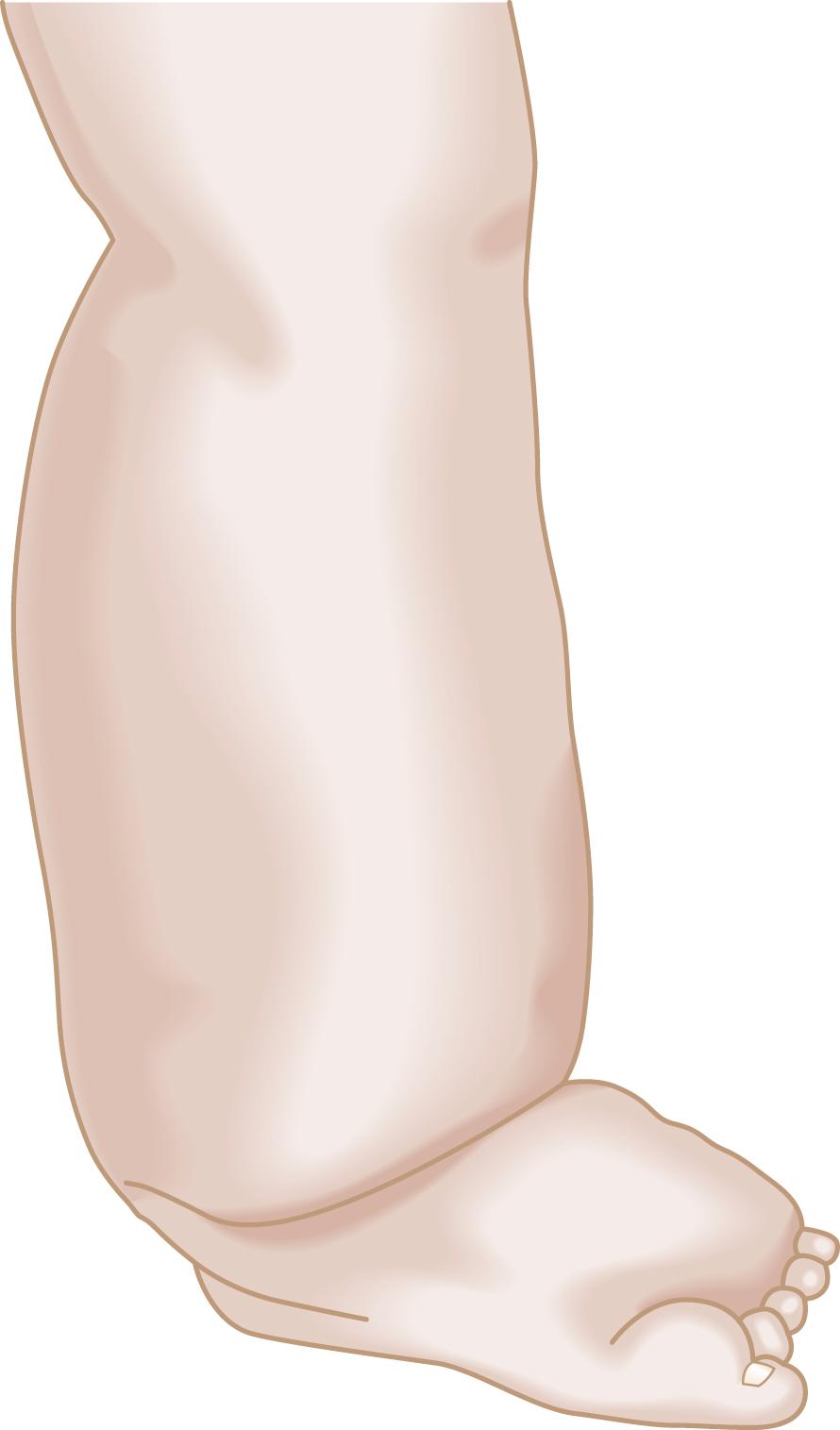
In most patients with second- or third-stage lymphedema, the characteristic findings on physical examination can usually establish the diagnosis. The edematous limb has a firm and hardened consistency. There is loss of the normal perimalleolar shape, resulting in a “tree trunk” pattern. The dorsum of the foot is characteristically swollen, resulting in the appearance of the “buffalo hump,” and the toes become thick and squared known as Stemmer sign ( Fig. 66.2 ). In advanced lymphedema, the skin undergoes characteristic changes, such as lichenification, development of peau d’orange, and hyperkeratosis. In addition, the patients give a history of recurrent episodes of cellulitis and lymphangitis after trivial trauma and frequently present with fungal infections affecting the forefoot and toes. Patients with isolated lymphedema usually do not have the hyperpigmentation or ulceration one typically sees in patients with chronic venous insufficiency. Lymphedema does not respond significantly to overnight elevation, whereas edema secondary to central organ failure or venous insufficiency does.
The evaluation of a swollen extremity should start with a detailed history and physical examination. The most common causes of bilateral extremity edema are of systemic origin. The most common cause is cardiac failure followed by renal failure. Hypoproteinemia secondary to cirrhosis, nephrotic syndrome, and malnutrition can also produce bilateral lower extremity edema. Another important cause to consider with bilateral leg enlargement is lipedema. Lipedema is not true edema but rather excessive subcutaneous fat typically found in obese women. It is bilateral, nonpitting, and greatest at the ankle and legs, with characteristic sparing of the feet. There are no skin changes, and the enlargement is not affected by elevation. The history usually indicates that this has been a lifelong problem that “runs in the family.”
Once the systemic causes of edema are excluded in the patient with unilateral extremity involvement, edema secondary to venous and lymphatic disease should be entertained. Venous disease is overwhelmingly the most common cause of unilateral leg edema. Leg edema secondary to venous disease is usually pitting and is greatest at the legs and ankles with a sparing of the feet. The edema responds promptly to overnight leg elevation and is controlled with regular compression. In the later stages, the skin is atrophic with brawny pigmentation from hemosiderin deposition. Ulceration associated with venous insufficiency occurs above or posterior to and beneath the malleoli.
Lymphedema is generally classified as primary when there is no known cause and secondary when its cause is a known disease or disorder. Primary lymphedema has generally been classified on the basis of the age at onset and presence of familial clustering. Primary lymphedema with onset before the first year of life is called congenital lymphedema. The familial version of congenital lymphedema is known as Milroy disease and is inherited as a dominant trait. Primary lymphedema with onset between the ages of 1 and 35 years is called lymphedema praecox. The familial version of lymphedema praecox is known as Meige disease with an onset around puberty. Finally, primary lymphedema with onset after the age of 35 years is called lymphedema tarda. The primary lymphedemas are relatively uncommon, occurring in one out of every 10,000 individuals. The most common form of primary lymphedema is praecox, which accounts for approximately 80% of the patients. Congenital and tarda lymphedemas each account for 10%. Worldwide, the most common cause of secondary lymphedema is infestation of the lymph nodes by the parasite Wuchereria bancrofti in the disease state called filariasis. In the developed countries, the most common causes of secondary lymphedema involve resection or ablation of regional lymph nodes by surgery, radiation therapy, tumor invasion, direct trauma, or, less commonly, an infectious process.
The diagnosis of lymphedema is relatively easy in the patient who presents in the second and third stages of the disease. It can, however, be a difficult diagnosis to make in the first stage, particularly when the edema is mild, pitting, and relieved with simple maneuvers such as elevation. , For patients with suspected secondary forms of lymphedema, computed tomography and magnetic resonance imaging are valuable and indeed essential for exclusion of underlying oncologic disease states. In patients with known lymph node excision and radiation treatment as the underlying problem of their lymphedema, additional diagnostic studies are rarely needed except as these studies relate to follow-up of an underlying malignant disease. For patients with edema of unknown cause and a suspicion for lymphedema, lymphoscintigraphy is the diagnostic test of choice. When lymphoscintigraphy confirms that lymphatic drainage is delayed, the diagnosis of primary lymphedema should never be made until neoplasia involving the regional and central lymphatic drainage of the limb has been excluded through computed tomography or magnetic resonance imaging. If a more detailed diagnostic interpretation of lymphatic channels is needed for operative planning, contrast lymphangiography may be considered.
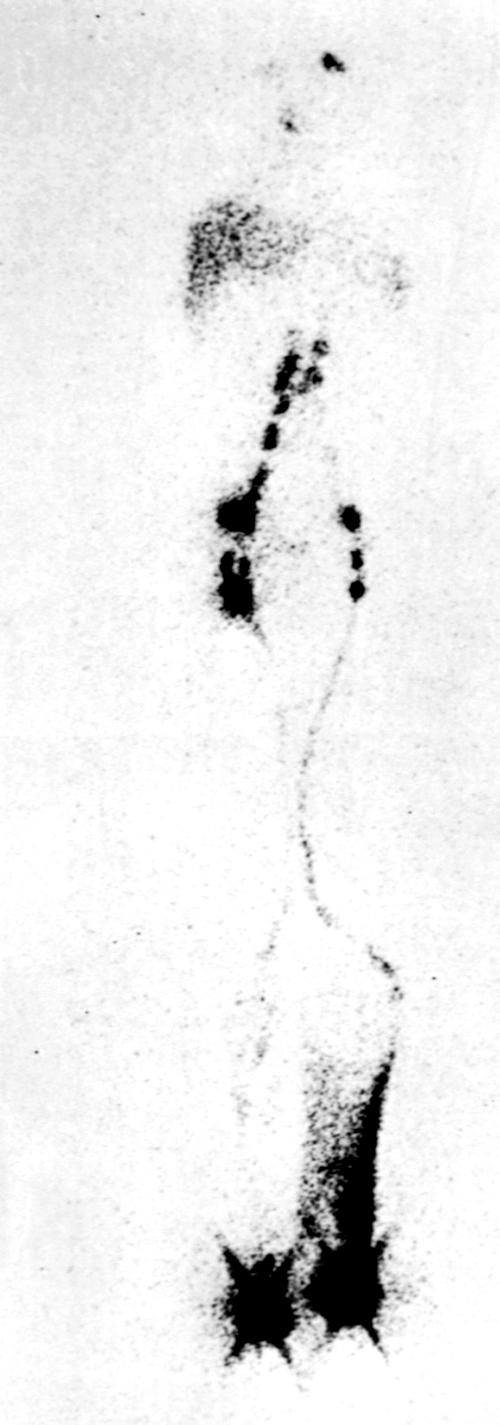
Lymphoscintigraphy (or isotope lymphography) has emerged as the test of choice in patients with suspected lymphedema. , It cannot differentiate between primary and secondary lymphedemas; however, it has a sensitivity of 70% to 90% and a specificity of nearly 100% in differentiating lymphedema from other causes of limb swelling. The test assesses lymphatic function by quantitating the rate of clearance of a radiolabeled macromolecular tracer ( Fig. 66.3 ). The advantages of the technique are that it is simple, safe, and reproducible, with small exposure to radioactivity (approximately 5 mCi). It involves the injection of a small amount of radioiodinated human albumin or technetium-99m–labeled sulfur colloid into the first interdigital space of the foot or hand. Migration of the radiotracer within the skin and subcutaneous lymphatics is easily monitored with a whole body gamma camera, thus producing clear images of the major lymphatic channels in the leg as well as measuring the amount of radioactivity at the inguinal nodes 30 and 60 minutes after injection of the radiolabeled substance in the feet. An uptake value that is less than 0.3% of the total injected dose at 30 minutes is diagnostic of lymphedema. The normal range of uptake is between 0.6% and 1.6%. In patients with edema secondary to venous disease, isotope clearance is usually abnormally rapid, resulting in more than 2% ilioinguinal uptake. Importantly, variation in the degree of edema involving the lower extremity does not appear to significantly change the rate of clearance of the isotope.
Direct contrast lymphangiography provides the finest details of the lymphatic anatomy. However, it is an invasive study that involves exposure and cannulation of lymphatics at the dorsum of the forefoot, followed by slow injection of contrast medium (ethiodized oil). The procedure is tedious, the cannulation often necessitates aid of magnification optics (frequently an operating microscope is needed), and the dissection requires some form of anesthetic. After cannulation of a superficial lymph vessel, contrast material is slowly injected into the lymphatic system. A total of 7 to 10 mL of contrast material is ideal for lower extremity evaluation and 4 to 5 mL for upper extremity evaluation. Potential complications include damage of the visualized lymphatics, allergic reactions, and pulmonary embolism if the oil-based contrast agent enters the venous system through lymphovenous anastomoses. Lymphangiography in present surgical practice is used infrequently and solely reserved for the preoperative evaluation of selected patients who are candidates for direct operations on their lymphatic vessels.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here