Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Takayasu’s arteritis is an immune arteritis causing inflammation of the aorta, its major branches, and pulmonary arteries. It is predominantly a disease of young women, with onset typically in the second or third decade of life. The disease is more common among Asians, although all ethnicities can be affected. The annual incidence in North America is approximately 2.6 per million people.
The first description of a patient with Takayasu arteritis (TA) may have occurred as early as the 18th century. Morgagni found large-vessel aneurysms and stenoses at autopsy in a 40-year-old woman in 1761. In 1830 Yamamoto described a 45-year-old man who originally presented with complaints of fever and developed pulselessness in the radial arteries 1 year later. TA is named after Japanese ophthalmologist Mikito Takayasu, who presented his findings of arteriovenous anastomoses of the ocular papilla in a 21-year-old woman with sudden vision loss in 1908. Two other ophthalmologists at the same meeting presented similar findings in patients with absent radial pulses. The term Takayasu arteritis was first coined in 1939 by Yasuzo Shinmi when reporting on a patient with comparable symptoms and physical findings. In 1975 the research committee of the Japanese Department of Health and Welfare officially proposed adopting the term Takayasu arteritis for the disease.
Although TA is a chronic disease, it is characterized by a waxing and waning course. Symptoms range from fever, myalgias, and loss of appetite to cerebral, visceral, and extremity ischemia. This variation in presentation frequently leads to a delay in diagnosis.
TA most commonly affects patients from East Asia, and the incidence varies by geographic location. The estimated incidence in Japan is approximately 150 new cases per year, and postmortem studies reveal a prevalence of 1 in 3000 patients. The incidence in India is 200 to 300 per million, whereas the incidence in Sweden is 0.8 per million, with a prevalence of 0.64 per 100,000. In Birmingham, England, the incidence is 0.2 per million. Finally, a population-based study in Olmstead County, Minnesota, found an incidence of 2.6 per million.
TA affects women 6 to 8 times more frequently than men, although this distribution may depend on the region being studied. Authors from India have found female to male ratios of 4:1, whereas others from China have reported ratios of 3:1. The median age at diagnosis in a cohort of 60 patients from the National Institutes of Health (NIH) was 25 years. Although the detection of TA in some patients after age 40 years may be related to delay in diagnosis, it is now understood that a significant number of patients do not develop symptoms until later in life. When the results from most major studies are examined, the majority of patients develop symptoms between the ages of 20 and 40 years, with no trend by region.
A genetic predisposition to the disease has been suggested because of the predilection for patients of Asian descent, especially those with a Japanese background, and the occasional familial clustering of the disease. HLA-Bw52 occurs in approximately 44% of Japanese patients with TA, compared with 13% in the general Japanese population. Furthermore, the haplotype of Bw52-Dw12 confers a greater likelihood of an active inflammatory state and rapid disease progression. Other studies have found increased susceptibility in patients with gene mutations in the HLA-A, -D, and -DR regions. , In a group of North American patients with TA, there was no significant association between any HLA antigen and disease severity or complications. In fact, there was a negative association between TA and the HLA-DR1 antigen, suggesting a possible protection against disease development. Further studies examining the genetic factors associated with TA must be performed because consistent associations are lacking.
Immune-mediated mechanisms, including both antibody- and cell-mediated mechanisms, have also been suggested as a cause of TA. TA has been associated with rheumatoid arthritis, systemic lupus erythematosus, inflammatory bowel disease, and glomerulonephritis. All these conditions have autoimmune mechanisms associated with their pathology, which suggests a common mechanism with TA. Numerous studies have suggested a link between TA and Mycobacterium tuberculosis (TB) due to the granulomatous nature of the two disease processes. Despite the high prevalence of TB in TA patients, a definitive causal relationship has not been identified.
Cell-mediated mechanisms of endothelial damage have been implicated in TA. Gamma/delta T cells in patients with TA are reactive to Hsp60 and exhibit cytotoxicity to aortic endothelial cells, suggesting a role in the pathogenesis of TA. This mechanism occurs via increased production of interferon gamma and is distinct from other similar rheumatologic disorders.
Antibody-mediated mechanisms of TA have been advocated based on elevated levels of antiaortic endothelial cell antibodies. These lead to the induction of endothelial adhesion molecules, cytokines, and apoptosis. Levels of antiaortic endothelial cell antibodies are 20 times greater in patients with TA compared with healthy controls and with patients with other autoimmune disorders.
The fact that TA is commonly found in women of childbearing age suggests that sex hormones may play a role in its development. Studies of patients with TA compared with healthy controls have shown elevated urinary estrogens. Furthermore, the association of Klinefelter syndrome in patients with TA may implicate elevated estrogens in the disease.
Many causative factors have been implicated in TA, but none is found consistently. The cause of TA is likely multifactorial, and further studies are necessary to elucidate the associations among the various suggested mechanisms.
Histologic examination of inflammatory lesions of TA reveals a panarteritis of all three layers of the vessel wall, with skip areas along the length of the vessel ( Fig. 140.1 ). These lesions can range from acute exudative inflammation to chronic, nonspecific productive inflammation to various types of granulomatous inflammation. Active inflammatory lesions begin in the vasa vasorum, extend through the media and adventitia, and terminate in a diffuse or nodular fibrosis. Smooth muscle cells and fibroblasts invade the intima and produce excessive ground substance. Occasionally, intimal thickening of the peripheral branches of inflamed arteries may lead to end-organ ischemia.
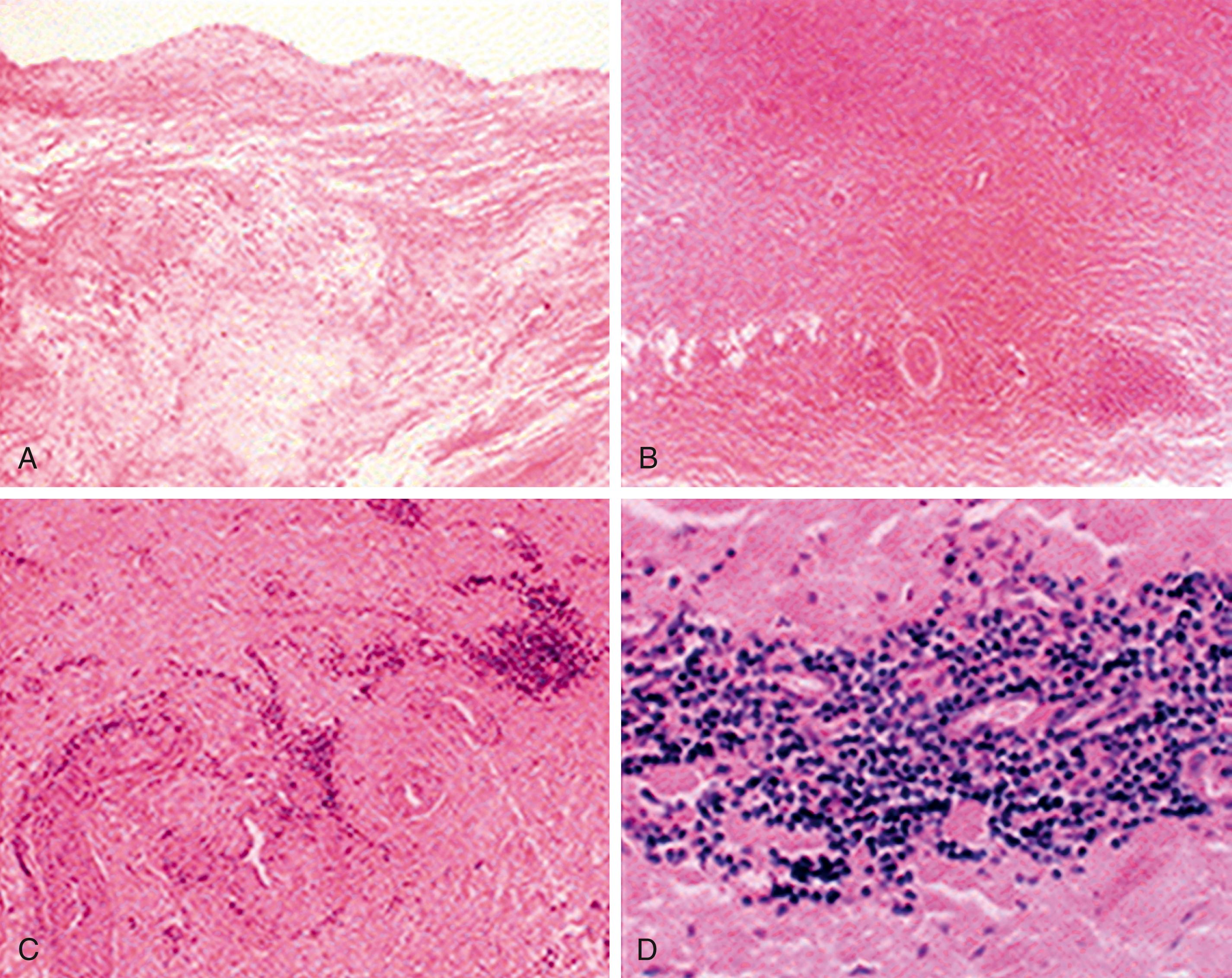
Immunohistochemical examination of postoperative aortic specimens with active inflammation has shown replacement of the muscular and elastic layers of the media and adventitia by dense fibrous tissue. In certain patients, inflammatory nodules of numerous T cells and B cells colocalized with dendritic cells have been observed in the deep portion of the intima and adventitia. This colocalization suggests that interactions between dendritic cells and lymphocytes may be important in the pathogenesis of TA.
In the chronic phase of TA, inflammation leads to thickening of the entire vessel wall. The vessel lumen narrows owing to proliferation of the intima, destruction of the elastic fibers of the media, and fibrosis of the adventitia.
Aneurysmal disease may also be associated with TA, although it is less frequent than stenotic disease. It has been hypothesized that aneurysmal disease occurs when destruction of the elastic component of the media occurs before fibrosis of the adventitia, leading to a weakened vessel wall.
TA was first thought to progress in three phases. The first phase was characterized by an inflammatory period with constitutional symptoms such as fever, headache, weight loss, myalgia, and arthralgia. The second phase was characterized by vessel inflammation with symptoms of vessel pain and tenderness, or carotidynia. The third and final phase, called the “burned-out” phase, was characterized by vessel fibrosis or aneurysmal degeneration. In this phase, patients presented with the classic signs of ischemia or aneurysm. It is now understood that although most patients with TA experience these three phases, many do not; and patients with TA can endure very diverse courses. In a group of North American patients, 10% were asymptomatic at presentation, 57% never had any constitutional symptoms, and only 33% developed systemic symptoms. Some TA patients present with inflammatory and fibrotic changes simultaneously, further emphasizing the diverse nature of this disease.
It is likely that the nonspecific nature of constitutional complaints leads to the delay in diagnosis that is common in patients with TA. A median delay of 1 year before diagnosis in a group of North American patients has been reported, similar to the 10 to 15.5 months reported in other studies. , Approximately 90% of patients see more than one medical doctor, with a mean of five doctors, before obtaining the correct diagnosis of TA.
Physical examination may help in diagnosis but is not particularly sensitive. However, normal physical exam findings should not be thought to exclude the presence of arterial disease.
The clinical presentation of patients with TA varies by geographic region ( Table 140.1 ). The most frequent finding in patients from North America and Italy were carotid bruits, , versus absent pulses in Mexico and hypertension in India.
| Major Clinical Feature | Percentage of Patients (%) | ||||
|---|---|---|---|---|---|
| United States | Mexico | India | Korea | Japan | |
| Bruit | 80 | 94 | 71 | 37 | – |
| Claudication | 70 | 29 | 20 | 21 | 13 |
| Diminished or absent pulses | 60 | 96 | – | 55 | 62 |
| Musculoskeletal symptoms | 53 | 53 | 31 | – | – |
| Asymmetrical blood pressure | 47 | – | – | – | – |
| Constitutional symptoms | 43 | 78 | 16 | 34 | 27 |
| Headache | 42 | 57 | 22 | 60 | 31 |
| Lightheadedness or dizziness | 35 | – | 44 | 36 | 40 |
| Hypertension | 33 | 72 | 76 | 40 | 33 |
| Carotidynia | 32 | – | 4 | 2 | 21 |
Overall, arterial involvement is often symmetric in paired arteries and contiguous in the aorta. Cerebrovascular signs and symptoms are common because of the frequency of aortic arch and branch involvement. Approximately 32% of patients present with carotidynia. Many patients report syncope or presyncope at some point during the disease course. Ischemic symptoms of stroke, transient ischemic attack, or amaurosis fugax occur in 5% to 20% of patients. Visual disturbances occur in up to 30% of patients and are statistically associated with vertebral and common carotid artery involvement ( Fig. 140.2 ). The retinal disease pattern described by Takayasu also leads to visual disturbances due to central retinal hypoperfusion.
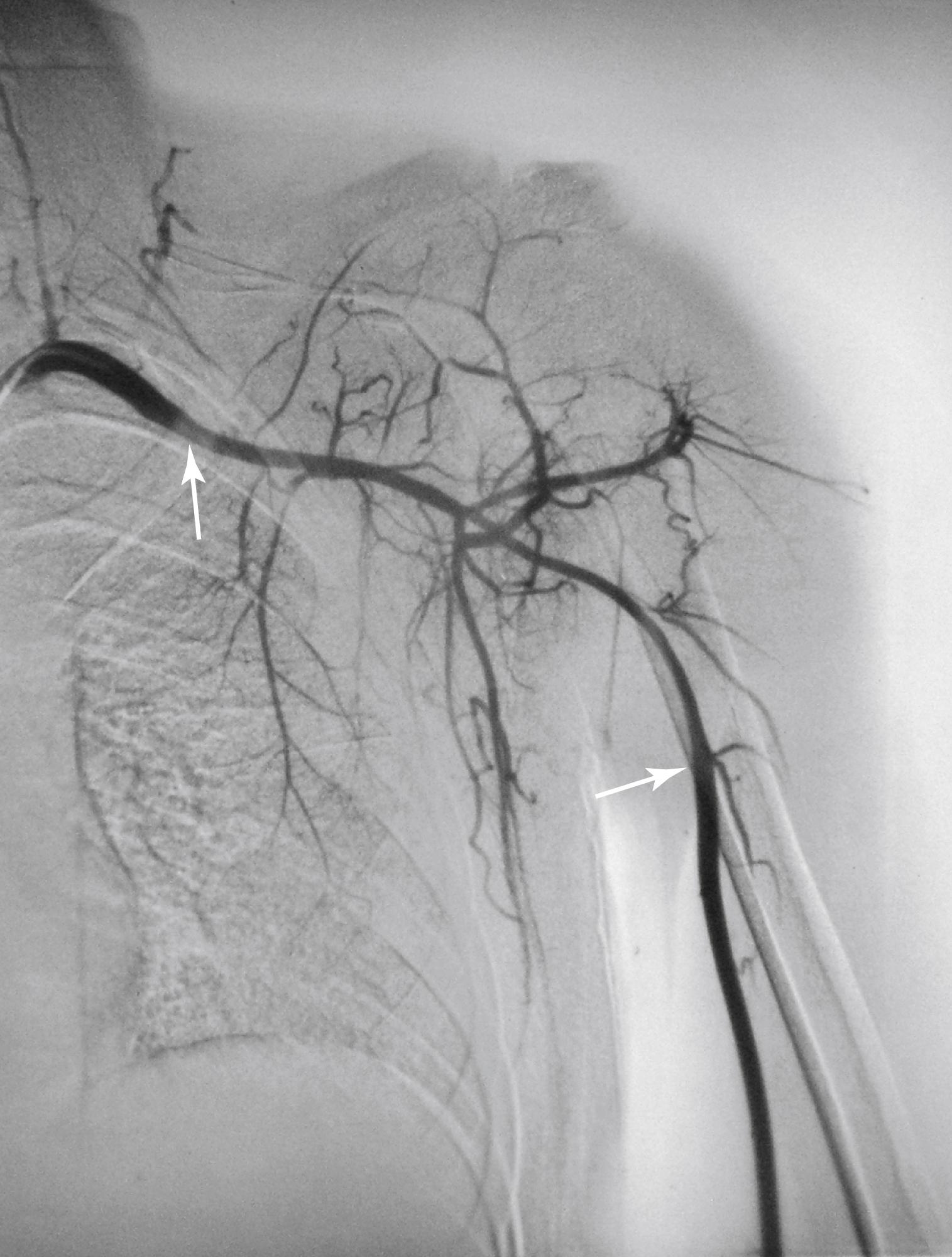
Upper extremity involvement is common in patients with TA. Diminished or absent pulses occur in 53% to 98% of patients. , Claudication is more frequent in the upper extremities than the lower extremities and occurs in approximately 62% of patients. This may be because the left subclavian artery is more commonly affected by TA than other arteries ( Fig. 140.3 ). , It has been hypothesized that subclavian steal syndrome is rare in patients with TA because the subclavian artery is involved both proximal and distal to the origin of the vertebral artery. More recent studies have shown that this may not always be true, with angiography demonstrating isolated disease in the proximal subclavian artery 80% of the time.
Cardiac manifestations of TA are common. Coronary artery involvement occurs in 6% to 16% of patients with TA and may lead to ischemic symptoms or congestive heart failure. Aortic valve regurgitation occurs in 12% to 24% of patients and is generally caused by dilation of the aortic root. Aortic valve regurgitation seems to be more common in TA patients from India. Mitral valve regurgitation occurs in 3% to 11% of patients. , The cause of mitral valve regurgitation is unclear but is thought to be independent of aortic regurgitation. Pulmonary artery involvement with TA may be underdiagnosed. In studies in which pulmonary angiography was performed, up to 70% of patients had evidence of disease. , These patients most often present with symptoms of pulmonary hypertension. Finally, diffuse vascular involvement of the myocardium has been reported in the absence of coronary stenosis or aortic valve regurgitation. Patients present with symptoms of congestive heart failure, and biopsies of the myocardium show diffuse myocarditis.
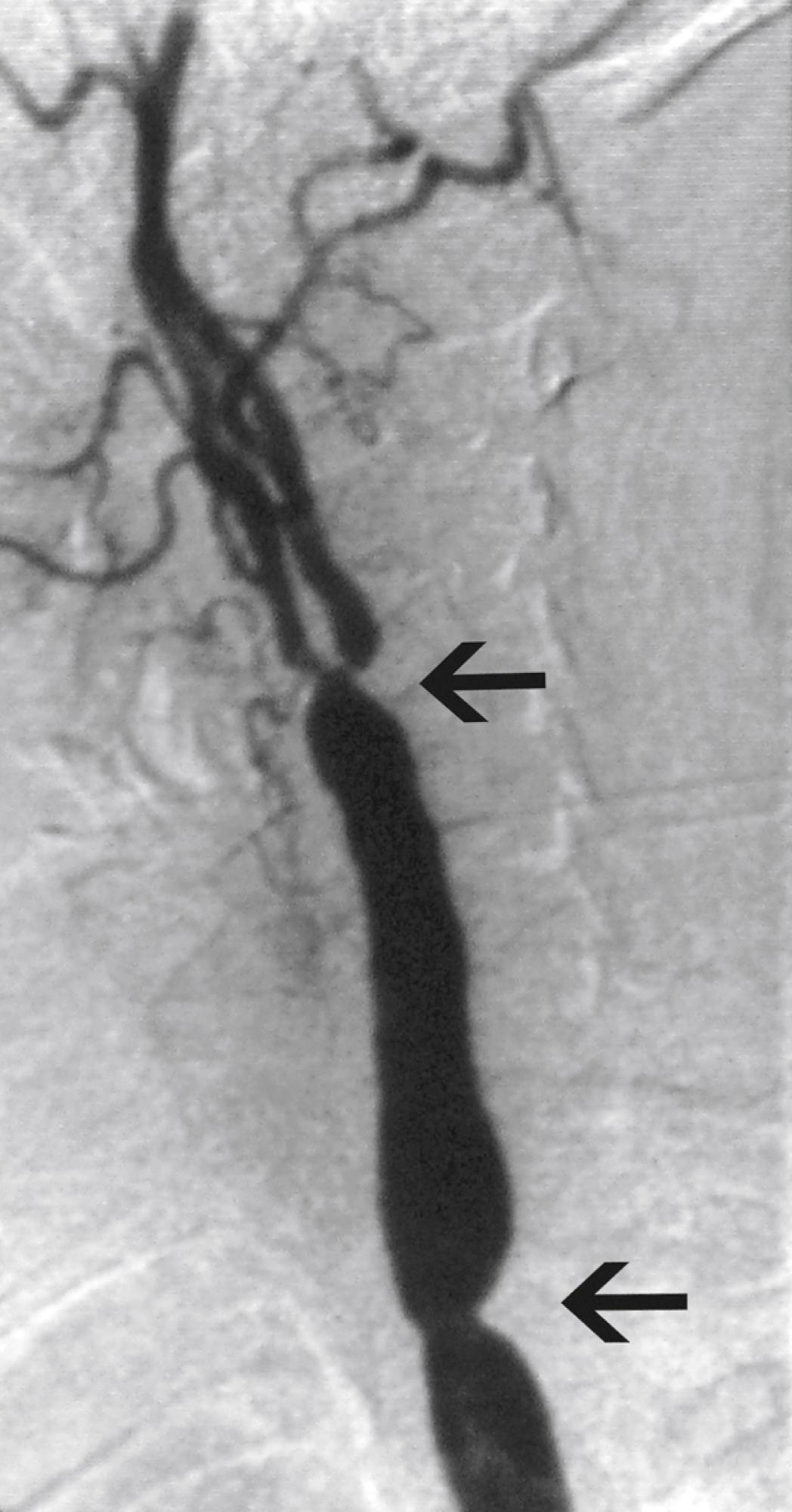
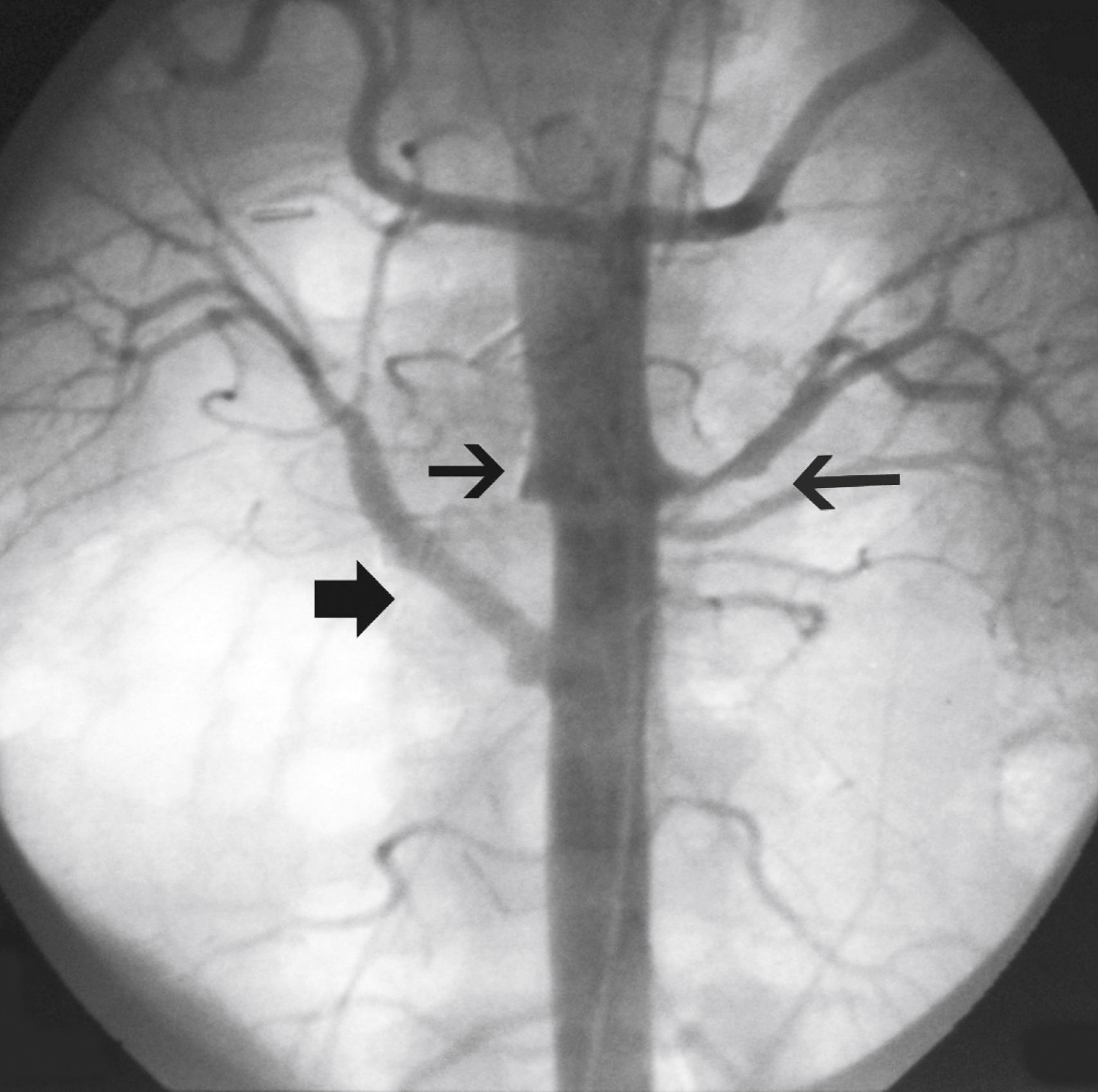
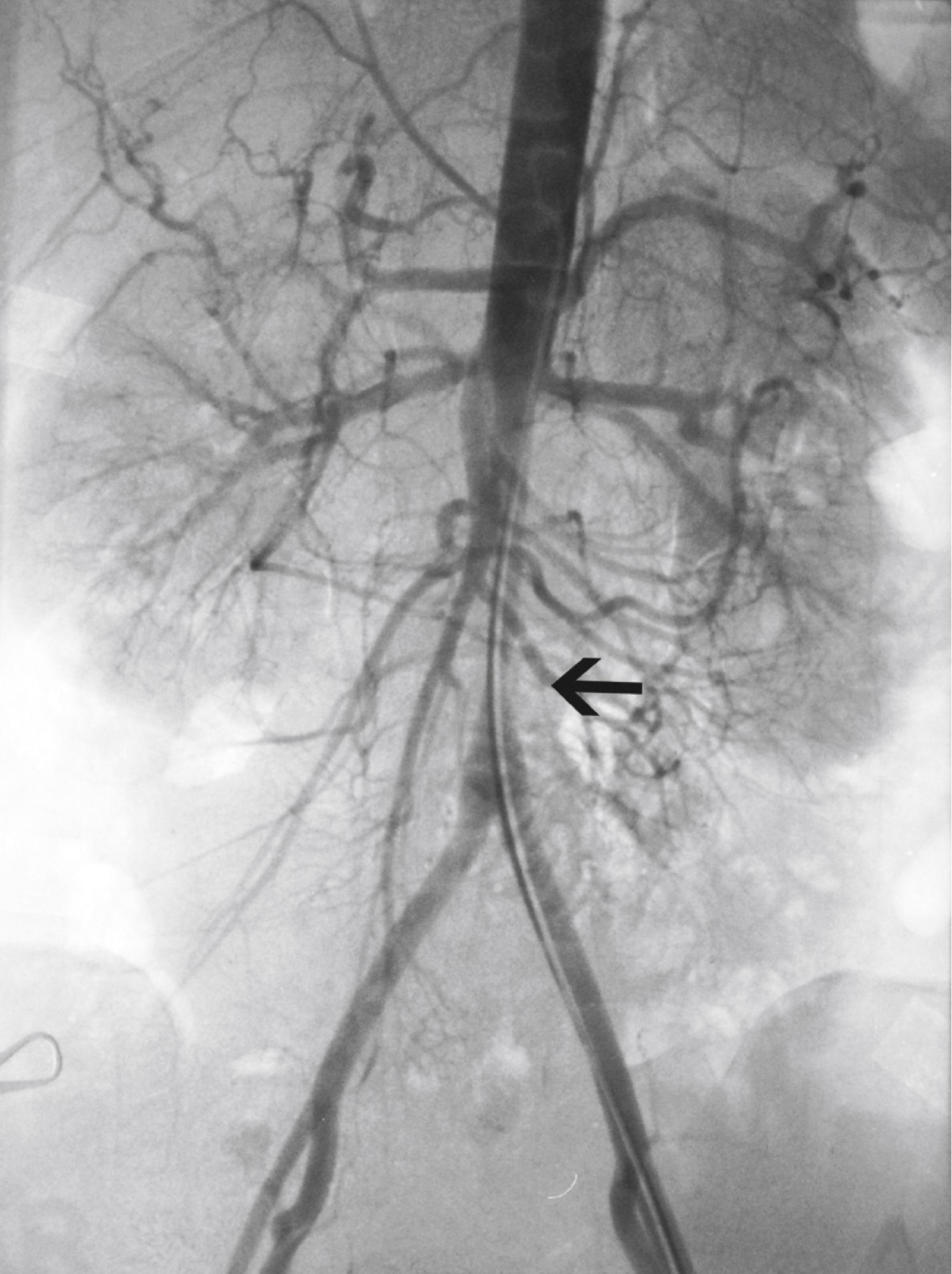
Hypertension is one of the most common presenting signs in patients with TA and occurs in 33% to 60% of patients. , Bilateral stenosis of the subclavian arteries occurs in up to 92% of patients with TA and can mask hypertension. Therefore blood pressure measurements of both upper and lower extremities should be performed in any patient suspected of having TA. Renal artery stenosis is a significant cause of hypertension in patients with TA. Involvement of the renal arteries occurs in approximately 20% to 50% of patients. , The stenoses are frequently bilateral and involve the ostia of the vessels ( Fig. 140.4 ). Hypertension in the absence of renal artery stenosis occurs in 5% to 30% of patients. , Causes of hypertension in this group of patients include middle aortic syndrome, reduced compliance of the aorta, and dysfunction of the carotid baroceptors ( Fig. 140.5 ). , Sen and colleagues first reported on 16 patients with middle aortic syndrome in 1963. All patients presented with hypertension, and more than half of them had decreased or absent lower extremity pulses. In a 1991 report from the Japanese Ministry of Health and Welfare, middle aortic syndrome occurred in 16% of surgically treated patients with TA. Long-segment narrowing of the descending thoracic or abdominal aorta is characteristic, and interscapular or abdominal bruits are frequently heard on auscultation. According to one report, the majority of patients with middle aortic syndrome die by age 35 if left untreated. Middle aortic syndrome is not unique to TA and may be due to congenital hypoplasia, von Recklinghausen disease, fibromuscular dysplasia, and tuberculosis.
Visceral arterial involvement occurs in 3% to 31% of patients with TA. , Despite these findings, symptoms of mesenteric ischemia are rare. Aortic endarterectomy for visceral ischemia has been performed, with mixed results. It is controversial in patients with TA because the inflammatory lesions typically involve all three layers of the arterial wall, making the procedure difficult. ,
Lower extremity ischemic symptoms occur in 24% to 32% of patients and, as stated previously, are less common than upper extremity symptoms. , Iliac artery disease is seen in approximately 20% of patients undergoing angiography. , Angiographic data suggest that claudication of the lower extremities may be related to abdominal aortic involvement because stenotic disease of the femoral, popliteal, and tibial arteries is rare.
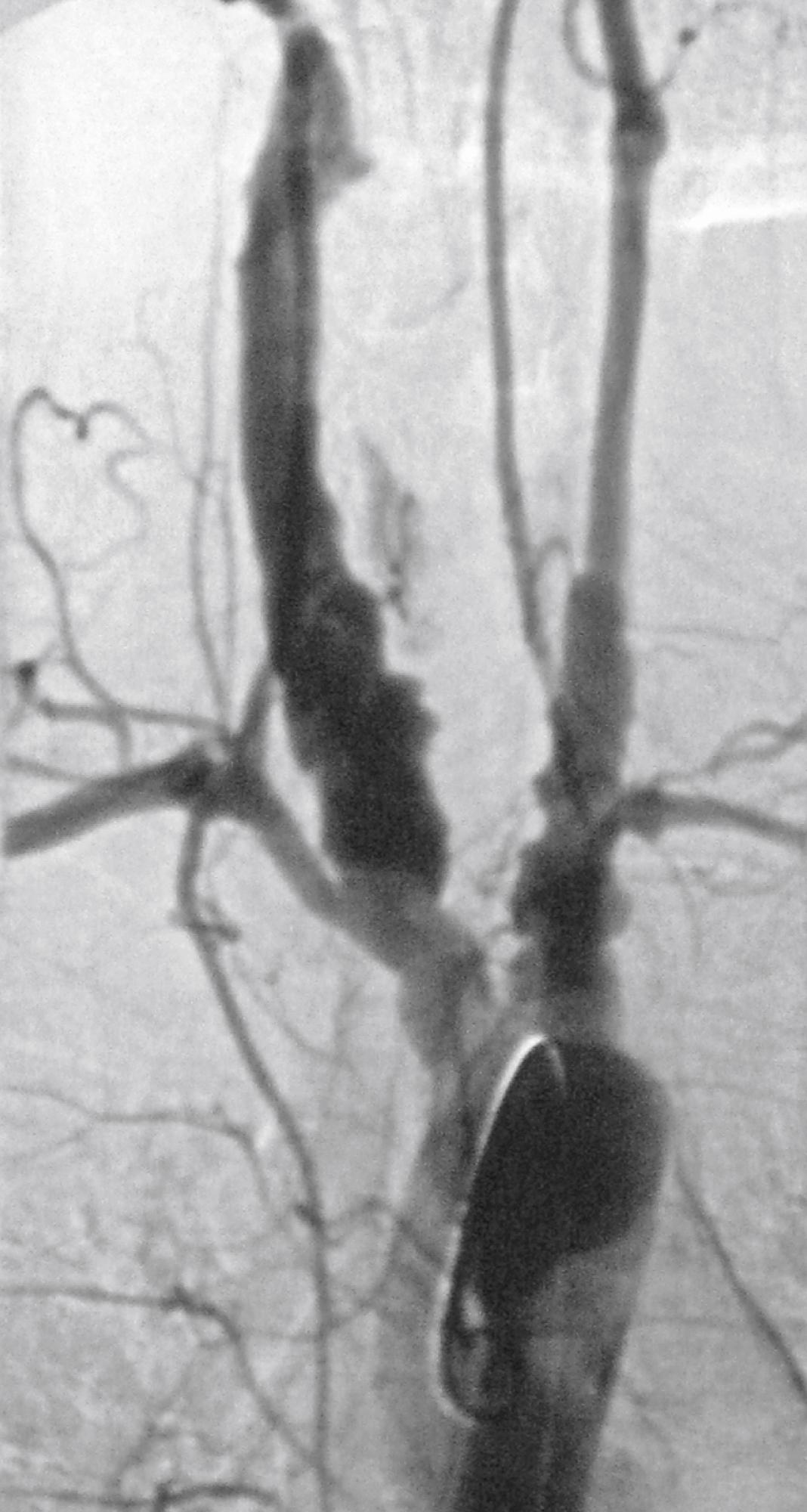
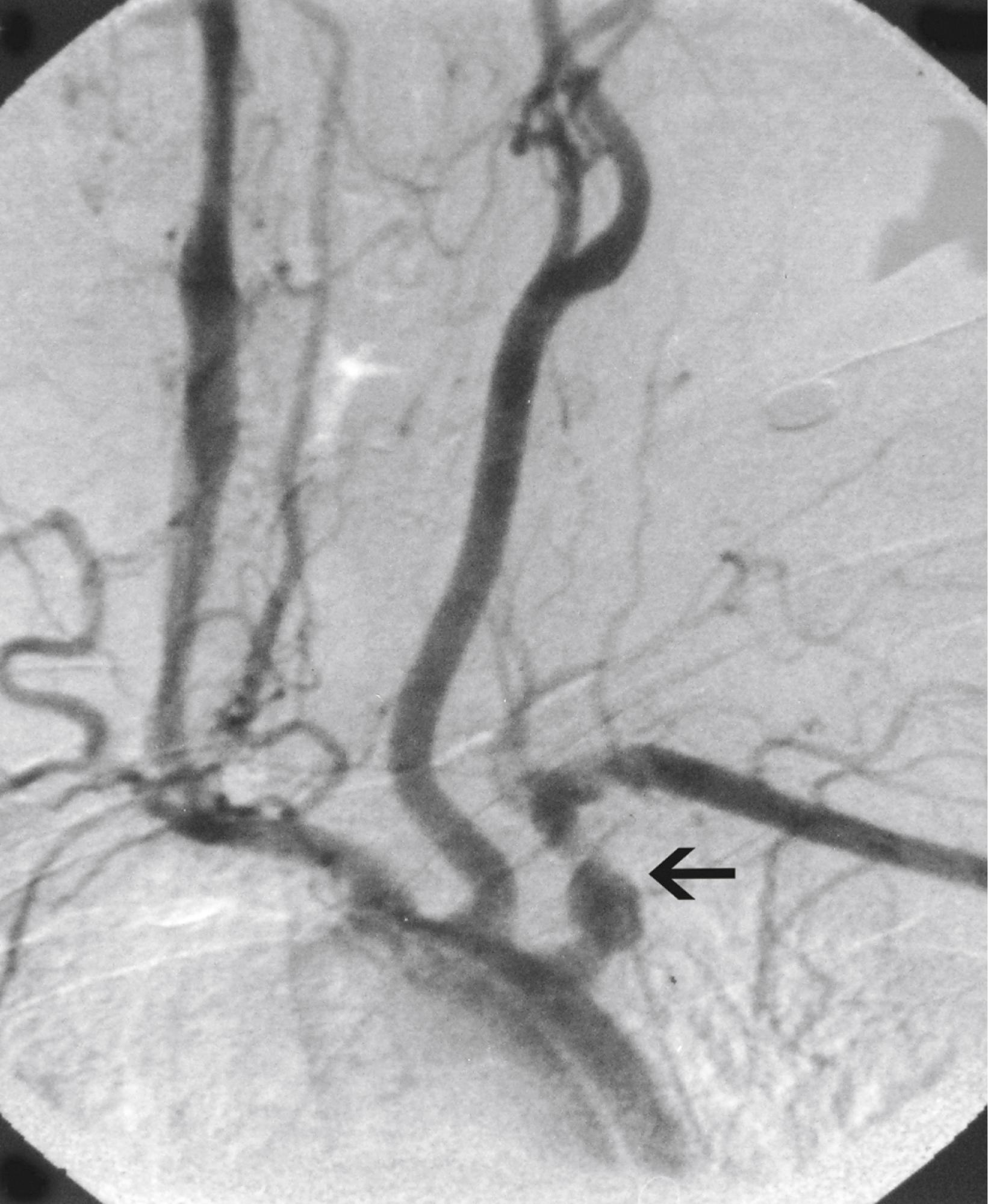
Aneurysmal disease in patients with TA varies by geographic location. In South Africa, up to 71% of patients underwent surgery for aneurysmal disease. In a study of Thai patients, 60% of patients had aneurysms, with the abdominal aorta being the most common location, followed by the subclavian artery and the thoracic aorta. In North America, 27% of patients had aneurysms, with the aortic arch being the most common site, followed by the abdominal aorta ( Fig. 140.6 ). Elsewhere, 13.7% of patients in India and 7% of patients in Italy had aneurysms on angiography. , A study from Japan reported 32% of patients with aneurysmal disease and noted that the aneurysms were frequently multiple and associated with stenotic lesions ( Fig. 140.7 ). In most series the incidence of aneurysm rupture is low compared with that of noninflammatory aneurysms.
Patients with TA may present with nonischemic symptoms. Raynaud phenomenon has been reported in approximately 8% to 14% of patients. Skin changes have been reported in 8% to 25% of patients. , These cutaneous manifestations of TA range from erythema nodosum to pyoderma gangrenosum to a lupus-like malar flush. ,
Because TA affects patients in myriad ways, there have been many proposed diagnostic criteria. The first diagnostic criteria were set forth by Ishikawa, based on a cohort of 96 Japanese patients with TA and 12 patients with other diseases of the aorta. These criteria included an obligatory age less than 40 years. The Ishikawa criteria identified 96% of young patients and 80% of older patients with active disease but, unfortunately, only 67% of young patients and 64% of older patients with inactive disease.
In 1990 the American College of Rheumatology created new diagnostic criteria based on a North American group of patients with TA ( Table 140.2 ). It removed Ishikawa age criterion because 13% of patients developed the disease after age 40. The presence of at least three of the six criteria from the traditional classification demonstrated a sensitivity of 90.5% and a specificity of 97.8%. The same group also created a classification tree with five of these six criteria, omitting claudication of an extremity. The classification tree demonstrated a sensitivity of 92.1% and a specificity of 97.0%.
| Criteria | Definition |
|---|---|
| Age at disease onset in years | Symptoms or findings of Takayasu arteritis at <40 years |
| Extremity claudication | Lower or upper extremity muscle fatigue during exercise |
| Diminished brachial artery pulse | Unilateral or bilateral |
| Blood pressure difference >10 mmHg | Measured systolic blood pressure between upper extremities |
| Bruit over subclavian arteries or aorta | One or both subclavian arteries or aorta |
| Angiographic abnormalities | Narrowing or occlusion of the aorta, primary branches, or large arteries in proximal extremities; must not be secondary to atherosclerosis, fibromuscular dysplasia, or other causes; usually focal or segmental |
In 1996 Sharma and associates devised a third set of diagnostic criteria. The Ishikawa criteria were modified in an effort to improve early diagnosis and to take into account geographic and racial variations ( Table 140.3 ). , The presence of two major criteria, one major criterion and two minor criteria, or four minor criteria was found to have a sensitivity of 92.5% and a specificity of 95% in a group of Indian patients and 96% sensitivity and 96% specificity in a group of Japanese patients.
| Criteria | Definition |
|---|---|
| Three Major Criteria | |
| Left mid-subclavian artery lesion | Severe stenosis or occlusion present in midportion from 1 cm proximal to the left vertebral artery origin to a point 3 cm distal to the origin |
| Right mid-subclavian artery lesion | Severe stenosis or occlusion in midportion from the right vertebral artery origin to a point 3 cm distal to the origin |
| Characteristic signs and symptoms of at least 1 month in duration | Includes limb claudication, pulselessness or pulse difference in the limbs, unobtainable or significant blood pressure difference (>10 mmHg systolic difference in limb), fever, neck pain, transient amaurosis, blurred vision, syncope, dyspnea, or palpitations |
| Ten Minor Criteria | |
| Elevated erythrocyte sedimentation rate | Unexplained persistent erythrocyte sedimentation rate >20 mm/h at diagnosis or presence of evidence in patient history |
| Carotidynia | Unilateral or bilateral tenderness of the common carotid artery on palpation; must differentiate from neck muscle tenderness |
| Hypertension | Persistent elevation of blood pressure above 140/90 mmHg (brachial) or 160/90 mmHg (popliteal) |
| Aortic regurgitation or annulo-aortic ectasia | By auscultation, echocardiography, or angiography |
| Pulmonary artery disease | Lobar or segmental artery occlusion or equivalent; stenosis, aneurysm, luminal irregularity, or any combination of these findings in pulmonary trunk or pulmonary arteries |
| Left mid-common carotid artery lesion | Severe stenosis or occlusion in midportion, 5 cm in length from a point 2 cm distal to the origin |
| Distal brachiocephalic trunk lesion | Severe stenosis or occlusion in distal third |
| Descending aortic lesion | Narrowing, dilatation or aneurysm, luminal irregularity, or combination of these findings involving the thoracic aorta; tortuosity alone is insufficient |
| Abdominal aortic lesion | Narrowing, dilatation or aneurysm, luminal irregularity, or combination of these findings involving the abdominal aorta; tortuosity alone is insufficient |
| Coronary artery lesion | Narrowing, dilatation or aneurysm, luminal irregularity, or combination of these findings before the age of 30 years in the absence of atherosclerotic risk factors, such as hyperlipidemia or diabetes mellitus |
Although there is no global consensus on the best diagnostic criteria, it is generally agreed that the diagnosis of TA should not be made without ruling out all other vasculitides, vascular infections, fibromuscular dysplasia, and idiopathic inflammatory syndromes.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here