Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease that can affect multiple organs, including the skin, joints, brain, peripheral nervous system, heart, gastrointestinal tract, and kidneys. Kidney involvement in SLE, generally termed lupus nephritis (LN) , is a major contributor to SLE-associated morbidity and mortality. Up to 50% of SLE patients have clinically evident kidney disease at presentation, and, during follow-up, kidney involvement occurs in up to 75% of patients, with an even greater representation among children and young adults. LN impacts clinical outcomes in SLE both directly via target organ damage and indirectly through complications of therapy.
Most patients with SLE will develop laboratory evidence of kidney involvement at some point during the course of their disease. In about one-third of SLE patients, kidney involvement first manifests with proteinuria and/or microscopic hematuria; this eventually progresses to reduction in kidney function. However, early in the course of disease, it is unusual for patients to present with a low glomerular filtration rate (GFR), except in very aggressive cases of LN, some of which present as rapidly progressive glomerulonephritis. Instead, patients often present initially with evidence of non-kidney organ involvement, such as malar rash, arthritis, and oral ulcers. After a diagnosis of SLE is confirmed with appropriate laboratory tests, evidence of kidney disease, if present, usually emerges within the first 3 years of diagnosis.
Signs of kidney involvement tend to correlate with laboratory abnormalities. For example, patients with nephrotic-range proteinuria often present with edema of the lower extremities and, if proteinuria is severe, periorbital edema in the morning. When GFR falls, as is the case with progressive forms of LN, elevated blood pressure is common. The rare development of dark or tea-colored urine is a sign of gross hematuria. A number of tools, such as the SLE Disease Activity Index (SLEDAI) and the British Isles Lupus Assessment Group (BILAG) Index, have been developed to assess the systemic severity of lupus symptoms. Although these questionnaires are primarily used to codify symptoms for clinical trial settings, they also can be helpful to elicit a detailed history from a patient with SLE.
The American College of Rheumatology (ACR) lists 11 diagnostic criteria for SLE: antinuclear antibodies (ANA), arthritis, immunologic disorders (including anti-double-stranded DNA [dsDNA] antibody, antiphospholipid antibody, or anti-Smith antibody), malar rash, discoid rash, photosensitivity, oral ulcers, serositis, hematologic disorder, neurologic disorder, and kidney disorder ( Table 25.1 ). Ideally, four or more of these criteria should be present to diagnose SLE, including laboratory findings of a positive ANA and/or anti-dsDNA antibody. In addition to the ANA and dsDNA antibody, serum complement (C3, C4, CH50) should be checked whenever kidney involvement is suspected, because these are often low when disease is active, as is usually the case with any severe proliferative LN. Antiphospholipid and anticardiolipin antibodies are useful in gauging the risk for clotting abnormalities that can accompany SLE.
| Criteria | Description |
| Malar rash | Flat or raised erythematous rash over the malar eminences |
| Discoid rash | Erythematous raised patches, usually circular, with adherent keratotic scaling; atrophic scarring may occur |
| Photosensitivity | Rash upon exposure to ultraviolet light |
| Oral ulcers | Oral and/or nasopharyngeal ulcerations |
| Arthritis | Nonerosive arthritis of at least two peripheral joints, with tenderness and/or swelling |
| Serositis | Pleuritis or pericarditis |
| Kidney disorder | Proteinuria, hematuria, and/or elevated creatinine |
| Neurologic disorder | Seizures or psychosis without other etiologies |
| Hematologic disorder | Anemia (hemolytic), leukopenia, or thrombocytopenia without other etiologies |
| Immunologic disorder | Anti-dsDNA, anti-Sm, and/or antiphospholipid antibodies |
| ANA | An abnormal ANA titer in the absence of drugs known to induce ANAs |
Laboratory testing is used both to diagnose kidney involvement and to assess response to therapy in patients with SLE. Traditional parameters, such as serum creatinine and urinary protein excretion (quantified by either 24-hour collection, urine protein-to-creatinine ratio, or urine albumin-to-creatinine ratio), are supplemented by serial review of microscopic urinary sediment, changes in serum complement levels, and titers of ANA and dsDNA antibodies. Because cytopenias are often seen in active SLE, complete blood counts should be checked regularly. A number of urine and serologic tests have been studied as biomarkers for SLE and, specifically, LN disease activity. These include molecules specific to lupus (e.g., anti-C1q antibodies), mediators of chronic inflammation (e.g., TNF-like weak inducer of apoptosis [TWEAK]), and generalized markers of kidney injury (urinary neutrophil gelatinase-associated lipocalin [uNGAL]). However, the clinical utility of this approach remains unproven, and no serum or urine disease markers are able to provide as much information as a kidney biopsy. Hence, virtually all patients with SLE with suspected kidney involvement undergo one or more kidney biopsies at some point during their care.
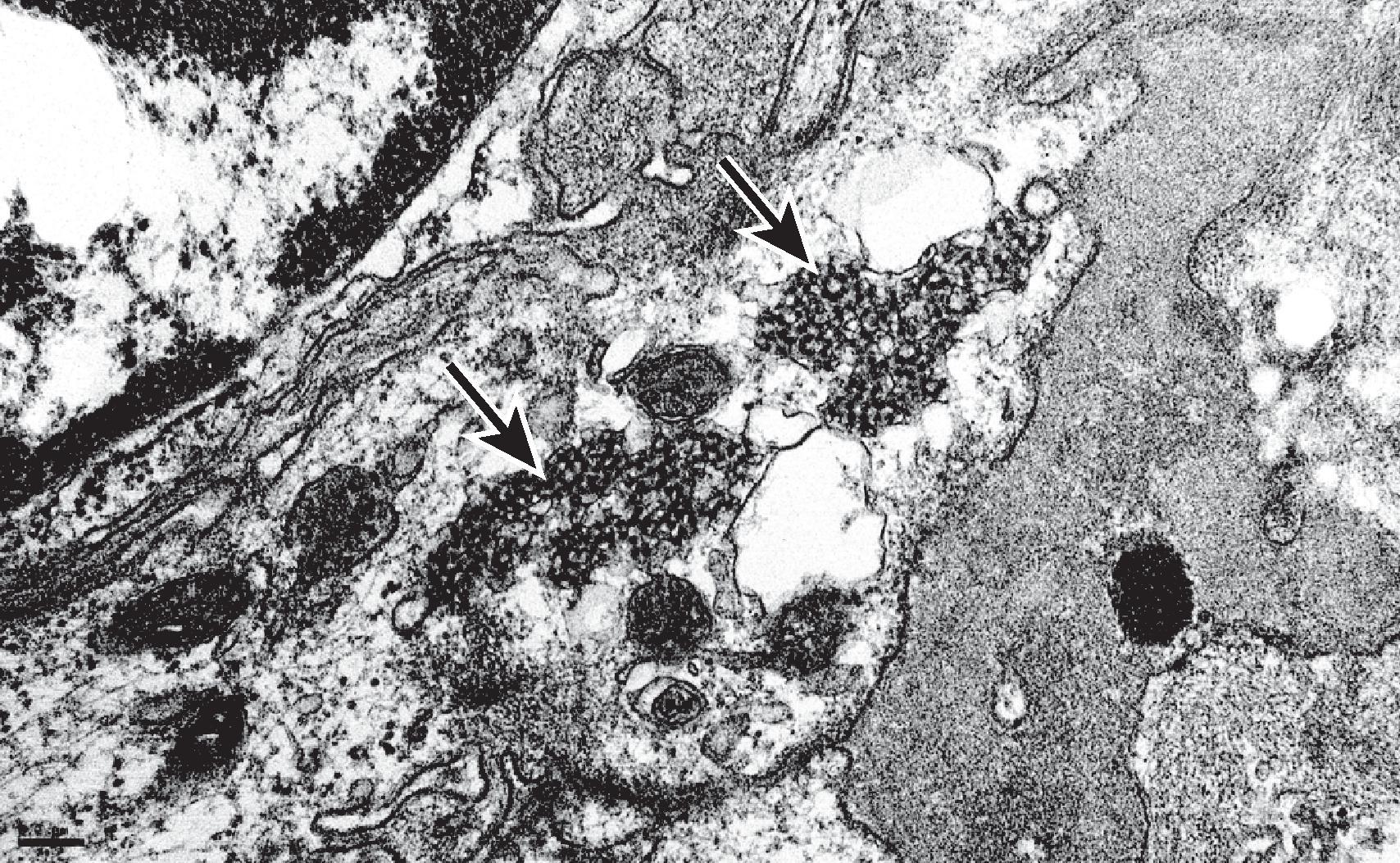
The classic pattern of LN is an immune complex–mediated glomerulonephritis; however, the pathology of LN can be varied and at times can cause confusion with other immune complex–mediated glomerulonephritides. Particular biopsy findings highly characteristic of LN include (1) glomerular deposits that stain dominantly for IgG with co-deposits of IgA, IgM, C3, and C1q, the so-called full house immunofluorescence (IF) pattern ; (2) intense staining for C1q; (3) extraglomerular immune-type deposits within tubular basement membranes, the interstitium, and/or blood vessels; (4) the ultrastructural finding of coexistent mesangial, subendothelial, and subepithelial electron-dense deposits; and (5) the ultrastructural finding of tubuloreticular inclusions, which represent “interferon footprints” in the glomerular endothelial cell cytoplasm ( Fig. 25.1 ).
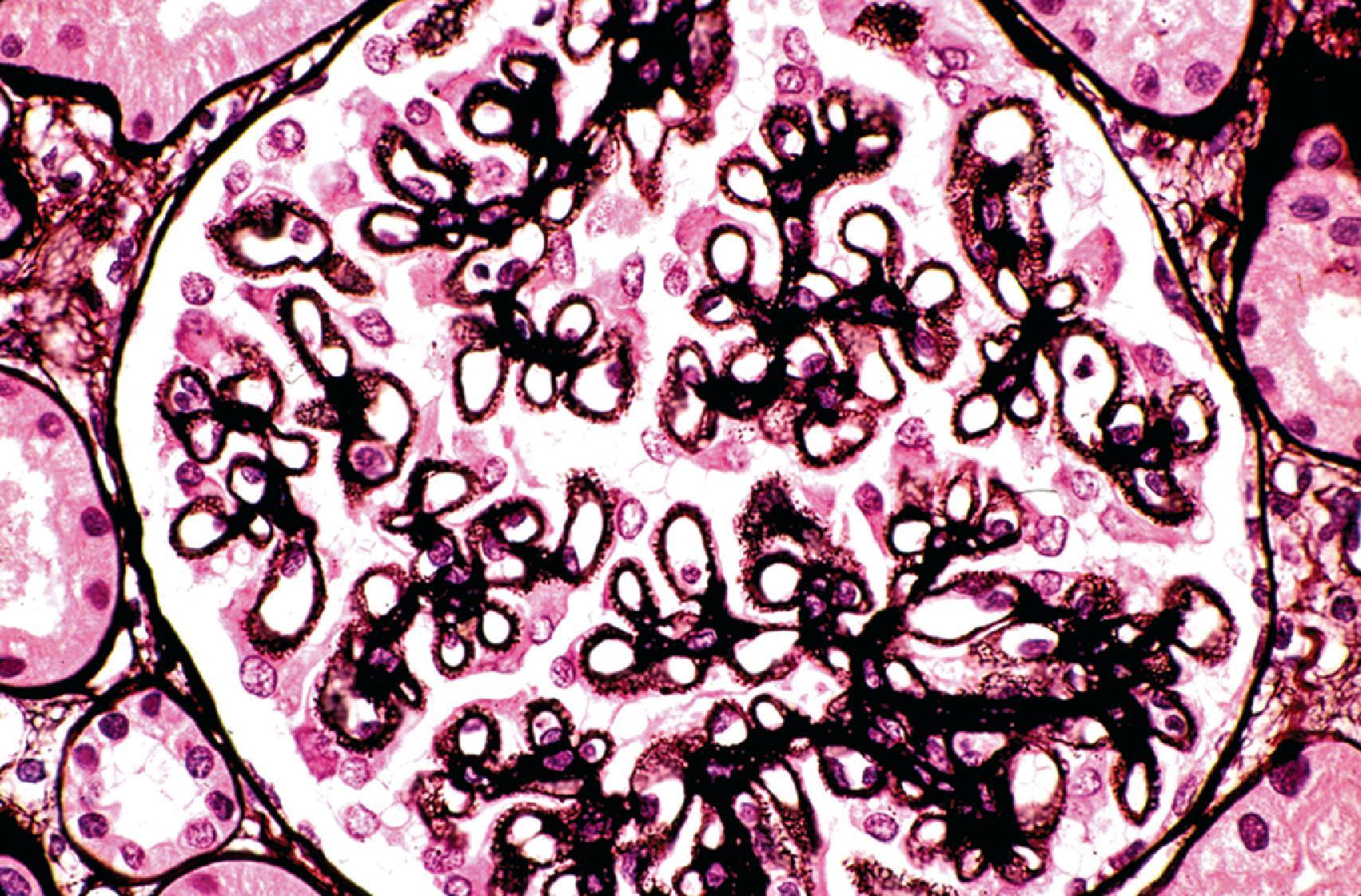
Although lupus may affect all compartments of the kidney, including glomeruli, tubules, interstitium, and blood vessels, glomerular involvement is the best studied component and correlates well with presentation, course, and treatment response. Accordingly, disease classification is based largely on the glomerular alterations, as assessed by the combined modalities of light microscopy, IF, and electron microscopy (EM). Over the past 5 decades, there have been attempts by different societies, particularly the World Health Organization (WHO), to classify the diverse glomerulopathies associated with SLE. Based on clinicopathologic correlations, a revised classification system of LN was developed by a working group of kidney pathologists, nephrologists, and rheumatologists under the joint auspices of the International Society of Nephrology (ISN) and the Renal Pathology Society (RPS) and was published in 2004 as the ISN/RPS classification. A minor revision of this classification was published in 2018. By refining and clarifying many of the deficiencies of the older WHO classification, this revised schema has eliminated ambiguities and has achieved greater reproducibility. The ISN/RPS classification recognizes six different classes of immune complex–mediated lupus glomerulonephritis based on biopsy findings ( Table 25.2 ). These classes are not static entities but may transform from one class to another, both spontaneously and after therapy.
| Designation | Description | Characteristic Clinical Features |
| Class I: minimal mesangial lupus nephritis | No LM abnormalities; isolated mesangial IC deposits on IF and/or EM | Normal urine or microscopic hematuria |
| Class II: mesangial proliferative lupus nephritis | Mesangial hypercellularity or matrix expansion with mesangial IC deposits on IF and/or EM | Microscopic hematuria and/or low-grade proteinuria |
| Class III: focal lupus nephritis a | <50% of glomeruli on LM display segmental (<50% of glomerular tuft) or global (>50% of glomerular tuft) endocapillary and/or extracapillary proliferation or sclerosis; mesangial and focal subendothelial IC deposits on IF and EM | Nephritic urine sediment and subnephrotic proteinuria |
| Class IV: diffuse lupus nephritis a | ≥50% of glomeruli on LM display segmental or global endocapillary and/or extracapillary proliferation or sclerosis; mesangial and diffuse subendothelial IC deposits on IF and EM | Nephritic and nephrotic syndromes, hypertension, reduced kidney function |
| Class V: membranous lupus nephritis b | Diffuse thickening of the glomerular capillary walls on LM with subepithelial IC deposits on IF and EM, with or without mesangial IC deposits | Nephrotic syndrome |
| Class VI: advanced sclerosing lupus nephritis | >90% of glomeruli on LM are globally sclerosed with no residual activity | Markedly reduced kidney function, hypertension |
a Both class III and class IV may have active (proliferative), chronic, inactive (sclerosing), or combined active and chronic lesions, which should be graded using modified NIH activity and chronicity indices.
b Class V may coexist with class III or class IV, in which case both classes are diagnosed. EM, Electron microscopy; IC, immune complex; IF, immunofluorescence; LM, light microscopy.
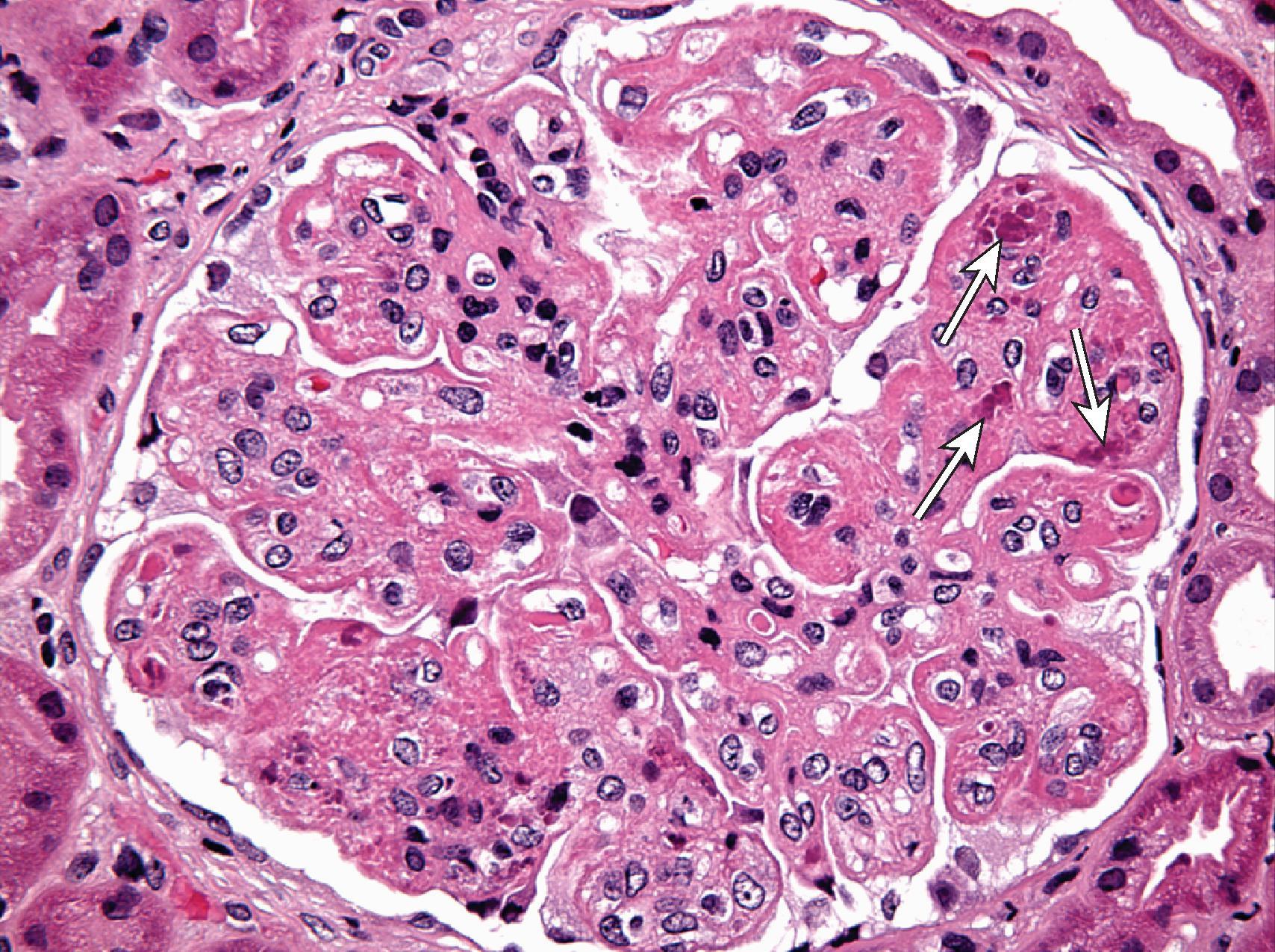
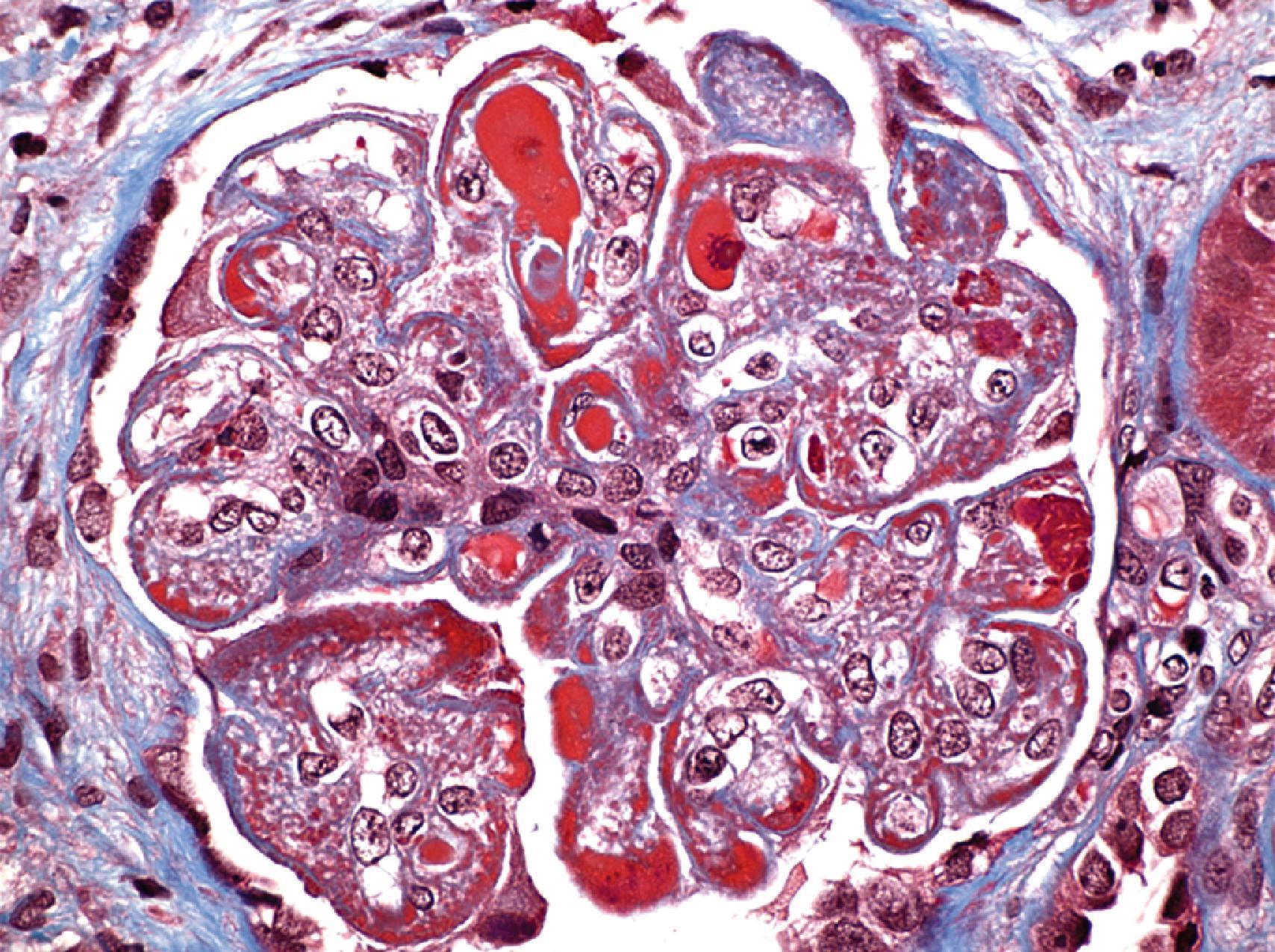
ISN/RPS class I represents the mildest possible glomerular lesion—immune deposits limited to the mesangium, without associated mesangial hypercellularity. In class II, the mesangial deposits detected by IF and/or EM are accompanied by mesangial hypercellularity of any degree. In class III, there is focal and predominantly segmental endocapillary proliferation and/or sclerosis, affecting less than 50% of glomeruli sampled. The active endocapillary lesions typically include infiltrating monocytes and neutrophils and may exhibit necrotizing features, including fibrinoid necrosis, rupture of glomerular basement membrane, and nuclear apoptosis, forming pyknotic or karyorrhectic debris. These segmental lesions often arise on a background of mesangial proliferation and immune deposits. In class IV, the endocapillary lesions involve ≥50% of glomeruli sampled, typically in a diffuse and global distribution ( Fig. 25.2 ). Subendothelial immune deposits are a feature of the endocapillary lesion in class III and class IV, where they vary from focal and segmental (class III) to more diffuse and global (class IV; Fig. 25.3 ). Both class III and class IV may exhibit extracapillary proliferation in the form of cellular crescents, a feature that correlates best with a rapidly progressive clinical course. Subendothelial deposits that are large enough to be visible by light microscopy may form “wire loops,” or intracapillary “hyaline thrombi” ( Fig. 25.4 ). A pathognomonic, but uncommon, feature of active LN is glomerular “hematoxylin bodies,” which consist of extruded nuclei from dying cells that have bound to ambient ANA to form basophilic rounded bodies (“LE bodies”; see Fig. 25.2 ). Class V denotes membranous LN. Subepithelial deposits are the defining feature, usually superimposed on a base of mesangial hypercellularity and/or mesangial immune deposits. Well-developed examples of class V typically exhibit glomerular basement membrane spikes between the subepithelial deposits ( Fig. 25.5 ). Class V may progress to glomerulosclerosis without the development of a superimposed proliferative lesion. In those patients with combined membranous and endocapillary lesions, a diagnosis of both class V and class III or IV is made. These mixed classes carry a worse prognosis than pure class V LN. Class VI identifies advanced chronic disease exhibiting greater than 90% sclerotic glomeruli, without residual activity.
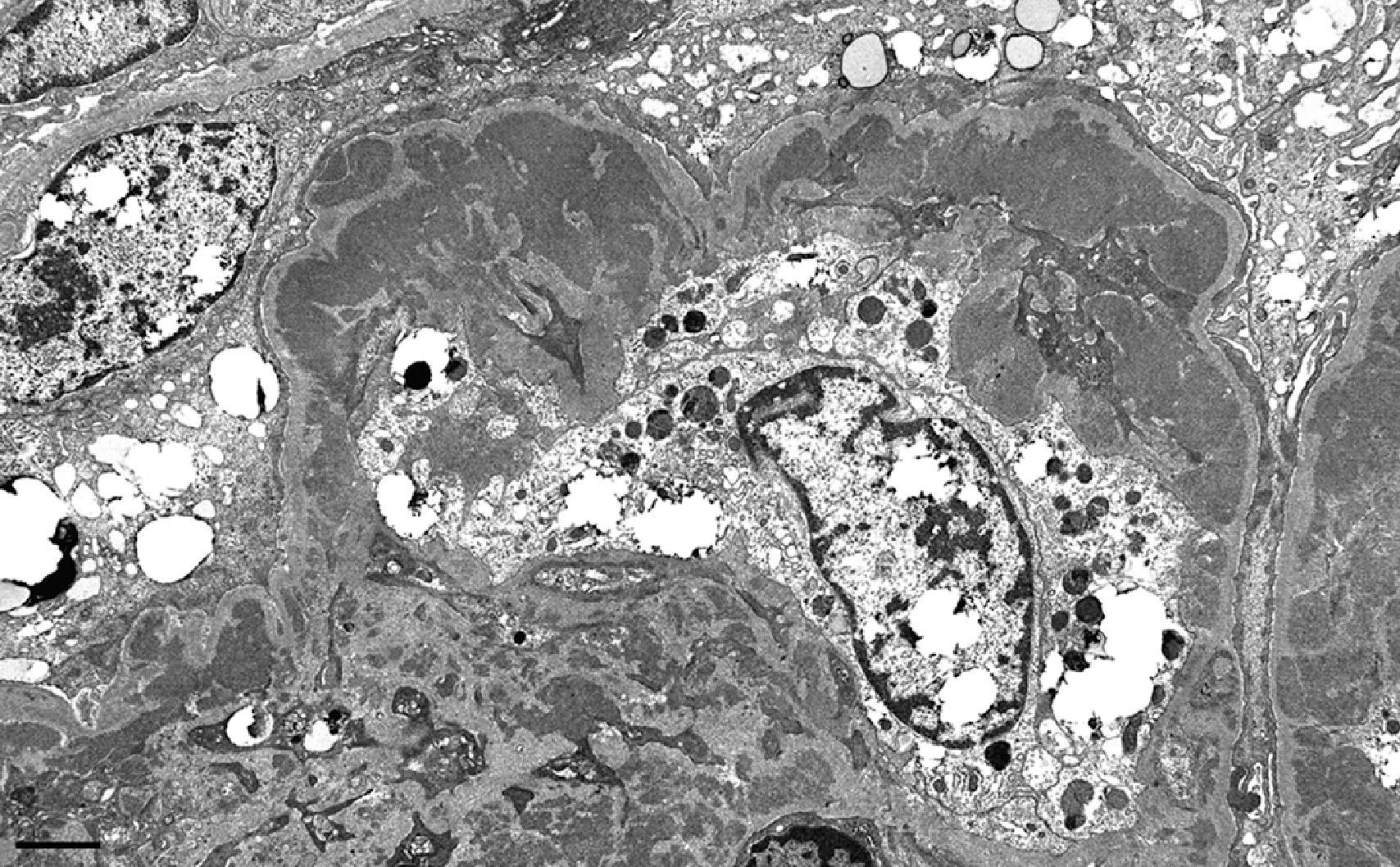
Unusual kidney biopsy findings in SLE patients include “lupus podocytopathy,” presenting as nephrotic syndrome with diffuse foot process effacement in the absence of peripheral capillary wall immune deposits. Mesangial immune deposits and variable mesangial hypercellularity (consistent with underlying class I or II LN) may accompany the podocyte alterations. Such cases resemble minimal change disease or focal segmental glomerulosclerosis in their histopathologic findings and response to glucocorticoids. An altered systemic cytokine milieu, rather than immune complex deposition, is thought to mediate direct podocyte injury. Lupus podocytopathy with collapsing features has been associated with APOL1 high-risk genotype in patients of African descent and carries a poor prognosis. Rare cases of LN have predominant tubulointerstitial nephritis with abundant tubulointerstitial immune deposits in the absence of significant glomerular lesions. Some cases with necrotizing and crescentic features and a paucity of peripheral capillary wall immune deposits are associated with circulating antineutrophil cytoplasmic antibody (ANCA), in addition to ANA. This “pauci-immune” variant is particularly common in those cases of LN class IV with diffuse but segmental lesions of necrosis and crescent formation. In any patient with SLE who develops thrombotic microangiopathy affecting the glomeruli and/or vessels, the possibility of a circulating lupus anticoagulant or an antiphospholipid antibody should be investigated. Other unusual vascular lesions include lupus vasculopathy (a noninflammatory narrowing of arterioles by a mixture of immune deposits and fibrin) and true inflammatory arteritis.
Kidney biopsy evaluation is not complete without an assessment of both histologic activity and chronicity as a guide to therapy. The use of modified National Institutes of Health (NIH) indices for activity and chronicity should be included in the kidney biopsy report. Active lesions include any combination of six features: glomerular endocapillary hypercellularity, neutrophil infiltration, fibrinoid necrosis, wire-loop deposits, cellular and/or fibrocellular crescents, and interstitial inflammation. Each component of activity is graded on a scale of 0 to 3+ based on the proportion of glomeruli or cortical area affected. Among features of activity, necrotizing lesions and cellular crescents carry the worst prognosis and are, therefore, accorded double weight. Thus, the total composite score for activity ranges from 0-24. Chronic changes include any combination of four features: global and/or segmental glomerular scarring (glomerulosclerosis), fibrous crescents, tubular atrophy, and interstitial fibrosis, each of which is graded on a scale of 0-3+ for a total composite score of 0-12. Because lesions of activity (A) and chronicity (C) can vary widely in a given biopsy, standard approaches to therapy weigh the extent and severity of active lesions (considered potentially responsive to immunosuppressive therapy) against the extent of chronic, irreversible disease.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here