Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The endemic mycoses are geographically restricted pathogens that exist as molds in specific environmental niches, where humans encounter them. In the United States, the major endemic mycoses are histoplasmosis, blastomycosis, and coccidioidomycosis. The extent of disease manifested by a given patient depends on both the size of the inoculum of the organism and the ability of the host to mount an effective immune response. The route of infection is almost always by inhalation into the alveoli of infectious conidia produced by the mold in the environment; therefore the major clinical manifestations are pulmonary. In addition, all of the endemic mycoses have the potential to disseminate hematogenously, and disease manifestations, especially in immunosuppressed patients, can reflect this widespread dissemination. In general, severe infection is initially treated with an intravenous amphotericin B formulation, which can later be changed to an oral azole agent. Mild to moderate infection is treated with an oral azole agent. With appropriate antifungal therapy, the prognosis is excellent for most such infections.
This chapter focuses on infection with the major endemic mycoses: histoplasmosis, blastomycosis, and coccidioidomycosis. Other systemic fungal infections—such as cryptococcosis, aspergillosis, and candidiasis, which primarily cause infection in immunosuppressed hosts—are not discussed here. The fungi that cause the endemic mycoses are all dimorphic; they exist as molds in the environment and as yeasts, or in the case of coccidioidomycosis, as spherules in tissues. Each organism occupies a different environmental niche and occurs in specific geographic areas ( Fig. 40.1 ). Infection occurs when a person is exposed to the conidia (sometimes called spores ) produced by the organism as it grows in the environment as a mold. These fungi cause infection in normal hosts as well as those who have defects in their immune response. A spectrum of disease manifestations can occur after inhalation of the conidia depending on the number of conidia inhaled and the immune status of the host. Hematogenous dissemination is common for all of these organisms, and it is possible for reactivation of a quiescent focus to occur years later.
Histoplasmosis is the most common endemic mycosis, likely infecting hundreds of thousands of persons yearly. Most persons in the highly endemic area are infected before adulthood . Histoplasma capsulatum thrives in highly nitrogenous soils in the Mississippi and Ohio River valleys , in multiple locations in Central and South America, and in focal areas of the eastern United States, Southeast Asia, and the Mediterranean basin. The organism grows to high concentrations in caves rich in bat guano. Activities that disperse the conidia include landscaping, demolition of old buildings, cleaning debris from attics or barns, and spelunking. Although most cases are sporadic, outbreaks of varying sizes frequently occur. The largest outbreak occurred during an urban demolition project in Indianapolis and caused infection in as many as a hundred thousand people.
Blastomycosis occurs most frequently in the south central and north central United States, the midwestern Canadian provinces, and areas bordering the St. Lawrence seaway, but Blastomyces dermatitidis is also found in the Middle East and Africa. The environmental niche for B. dermatitidis is thought to be soil and decaying wood. In many reports, middle-aged men account for most cases, and a well-described scenario is blastomycosis occurring in both a hunter and his dog.
There are two species of Coccidioides . Coccidioides immitis includes isolates from California, and Coccidioides posadasii includes isolates from all other areas. The endemic area for Coccidioides species includes the southwestern US desert regions known as the lower Sonoran life zone and areas of Central and South America that have the same type of ecosystem. Environmental cycles of rain and drought in the desert are important in the growth and dispersal of Coccidioides species. Dust storms and activities that involve the disruption of desert soil can lead to widespread dispersal of the conidia and increased risk for persons in the area. The exodus to the sun belt has contributed to an increase in the number of cases of coccidioidomycosis in older adults who are experiencing their first exposure to this organism.
Most patients infected with one of the endemic mycoses are asymptomatic or have such mild symptoms that they are thought to have a self-limited viral illness. Therefore the discussion of symptoms and signs that follows concentrates on fewer than 5% of persons exposed to these organisms. When symptoms do occur, the predominant manifestations are pulmonary, which is not surprising given that the portal of entry for these fungi is the lungs. For all of these fungi, dissemination during the early stages of infection is common; this occurs before the host establishes cellular immunity to the organism and is able to contain the infection. In most cases, dissemination is silent and not associated with clinical manifestations. However, patients who are immunosuppressed or exposed to a high inoculum of the organism can become acutely ill with disseminated infection. Patients also can be seen at a later time point with focal infection at a site to which the organism had spread hematogenously. Although the pulmonary manifestations are often similar among these three endemic mycoses, the clinical manifestations of disseminated infection are somewhat different for each organism.
A 66-year-old man with ankylosing spondylitis who had been on adalimumab for 10 months presented with fever and night sweats of 10 days’ duration and a dry cough and dyspnea that had lasted for 5 days. On examination, his temperature was 103°F and he appeared quite ill. Crackles were heard at both bases and hepatosplenomegaly was present. Computed tomography (CT) of the thorax showed diffuse reticulonodular lesions. His white blood cell count (WBC) was 3200/μL, hemoglobin 9.6 g/dL, platelets 86,000/μL. Liver enzymes were abnormal: AST 145 units, ALT 210 units, alkaline phosphatase 494 units, bilirubin 2.9 mg/dL. The laboratory called to say that they saw organisms in several neutrophils in the peripheral blood smear. Histoplasma antigen in urine was 19 ng/mL and in serum 11 ng/mL. Blood cultures yielded H. capsulatum 7 days later. Treatment with liposomal amphotericin B was begun, and the patient’s temperature rapidly returned to normal; within a week, he felt much better. After 2 weeks, therapy was changed to oral itraconazole, 200 mg twice daily for a total treatment course of 12 months. CT of the thorax showed complete resolution, pancytopenia resolved, and liver enzymes and bilirubin returned to normal.
COMMENT: Patients treated with tumor necrosis factor (TNF) antagonists, such as adalimumab, are at risk for developing widespread disseminated infection with H. capsulatum. Blood cultures are frequently positive, and the yeast form can be seen in neutrophils and monocytes in the peripheral blood smear. Involvement of liver, spleen, bone marrow, lungs, and adrenal glands is common. The diagnosis must be made quickly, allowing amphotericin B therapy to begin promptly if a fatal outcome is to be avoided.
Patients who have symptoms associated with acute pulmonary histoplasmosis usually manifest fever, nonproductive cough, anterior chest discomfort, myalgias, and fatigue. Patchy nodular infiltrates are noted on chest radiographs . Most patients are initially treated with antibiotics for community-acquired bacterial pneumonia. Only when the infection persists is the possibility of a fungal infection considered, and then appropriate diagnostic studies are undertaken.
Severe pneumonia develops in a minority of patients and manifests as high fever, dyspnea, nonproductive cough, and hypoxemia ( Fig. 40.2 ). Diffuse infiltrates are noted on the chest radiograph and may progress to acute respiratory distress syndrome (ARDS). Immunosuppressed patients are more likely to develop severe pulmonary infection.
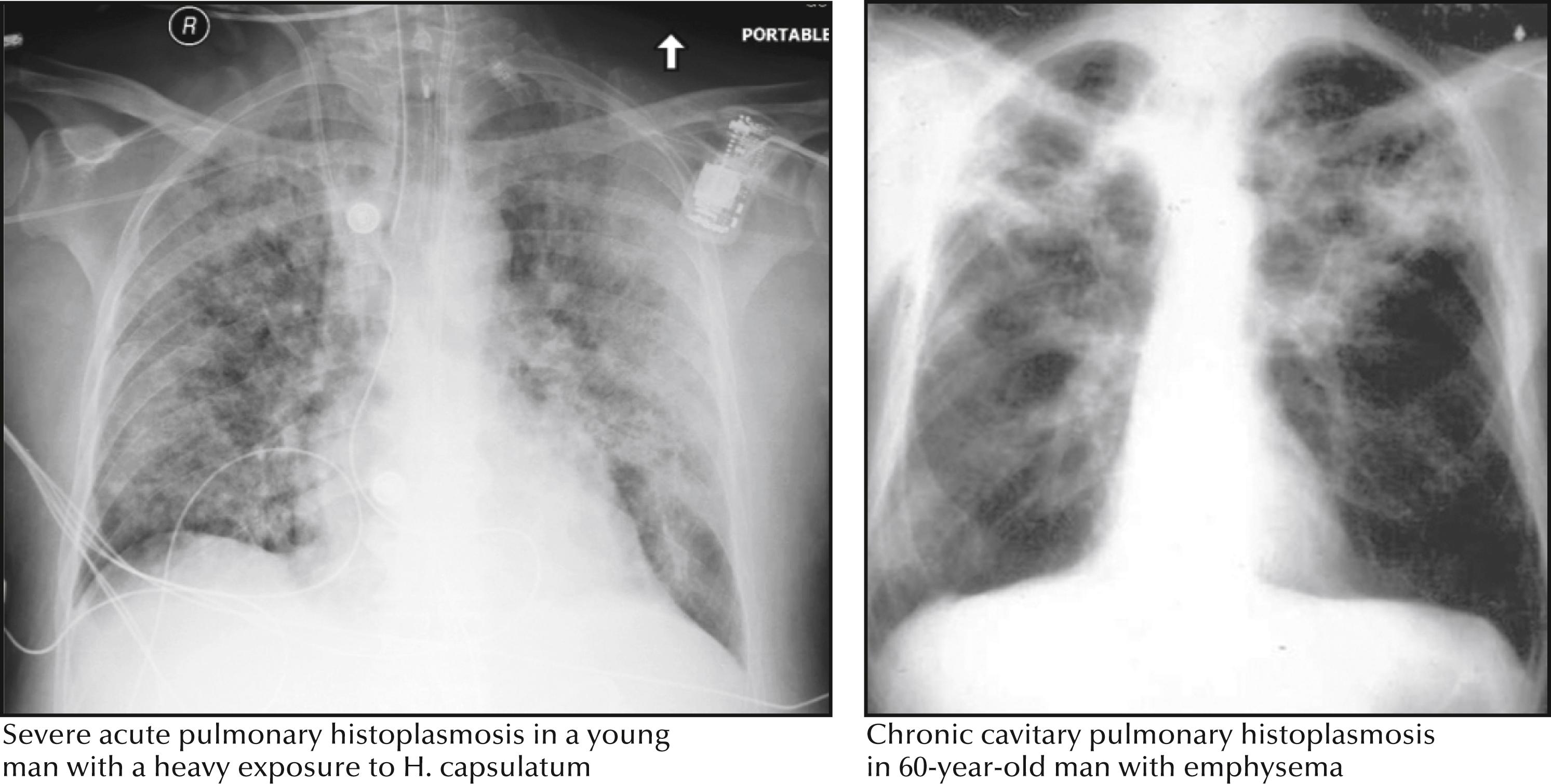
Chronic cavitary pulmonary histoplasmosis, which mimics tuberculosis, occurs mostly in older adults who have chronic obstructive pulmonary disease (COPD). Patients usually manifest fever, fatigue, anorexia, weight loss, cough productive of purulent sputum, and hemoptysis. Chest radiographs show upper lobe cavitary lesions, and fibrosis is seen in the lower lung fields (see Fig. 40.2 ).
The complications of pulmonary histoplasmosis include mediastinal granuloma and mediastinal fibrosis. Patients with mediastinal granuloma have persistent mediastinal and hilar lymphadenopathy; symptoms related to the enlarged nodes include dysphagia, chest pain, and nonproductive cough when the involved nodes impinge on mediastinal structures. Most patients have a gradual resolution of symptoms as the lymphadenopathy regresses. Mediastinal fibrosis is a rare and, in many patients, fatal complication in which excessive fibrosis occurs in response to mediastinal histoplasmosis; this can entrap the great vessels and bronchi. The symptoms are progressive and include dyspnea, cough, wheezing, and hemoptysis. Heart failure, pulmonary emboli, and superior vena cava syndrome can occur.
Acute disseminated histoplasmosis is seen mostly in patients who are immunosuppressed, including those who have acquired immunodeficiency syndrome (AIDS), have received a transplant, or have been treated with corticosteroids or TNF antagonists. Young infants are also at risk for disseminated histoplasmosis. Symptoms include chills, fever, fatigue, anorexia, and weight loss; sepsis syndrome with ARDS and disseminated intravascular coagulation can occur. Hepatosplenomegaly and skin and mucous membrane lesions are frequently present, and pancytopenia and elevated liver enzymes are common.
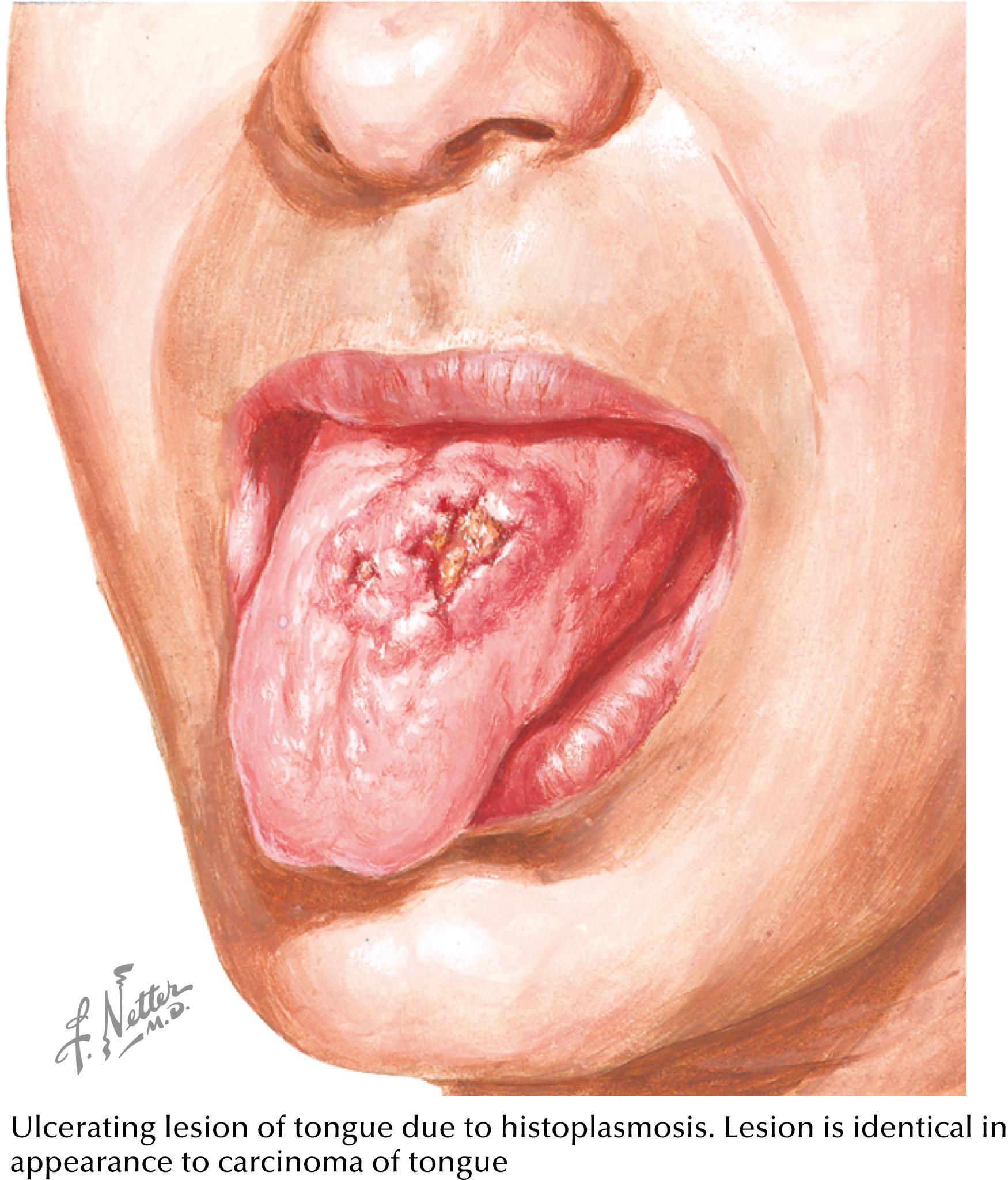
Chronic progressive disseminated histoplasmosis occurs mostly in middle-aged to elderly patients with no known immunosuppression. These patients often have fever of unknown origin with weeks to months of fever, night sweats, weight loss, and fatigue. Hepatosplenomegaly and mucosal ulcerations ( Fig. 40.3 ) and symptoms and signs of adrenal insufficiency are often noted.
A 36-year-old healthy woman from upper Michigan was seen with complaints of fevers, malaise, and left-sided chest pain of 2 weeks’ duration. She was given levofloxacin, 250 mg daily, but her symptoms progressed over the next week. A chest radiograph showed a left-lower-lobe pneumonia, and the levofloxacin was increased to 750 mg daily. One week later she was admitted to hospital with consolidation of most of the left lung and increased dyspnea. Further history revealed that in the preceding 2 months she had done most of the work of cleaning out a vacant cottage near the lakeshore that her family had purchased. Her temperature was 102°F, pulse 117 beats/min, oxygen saturation 93% on 3 L of oxygen. The WBC count was 21,600/μL. Treatment was changed to vancomycin and piperacillin-tazobactam, resulting in no improvement in her fever or dyspnea. On the fourth hospital day, she was intubated and bronchoscopy was performed. Bronchoalveolar lavage (BAL) fluid showed broad-based budding yeasts typical of B. dermatitidis. Culture later yielded B. dermatitidis . Treatment with liposomal amphotericin B, 5 mg/kg daily was begun; 2 weeks later, therapy was changed to oral itraconazole, 200 mg twice daily. She was treated for a total of 12 months and recovered completely.
COMMENT: This woman likely had exposure to a large number of conidia of B. dermatitidis in the course of cleaning out a vacant cabin in a highly endemic area for this fungus. Identification of the typical appearance of the yeast form of B. dermatitidis in the BAL fluid allowed treatment to begin before the culture revealed the organism.
The symptoms and signs of acute pulmonary blastomycosis are fever, cough, and myalgias; a localized pulmonary infiltrate is seen on the chest radiograph. The diagnosis is usually community-acquired bacterial pneumonia, and most patients are treated with antibiotics. Only when the infection persists is the possibility of a fungal infection considered, at which point appropriate diagnostic studies are undertaken. A minority of patients have severe pneumonia that progresses quickly to severe hypoxemia and ARDS ( Fig. 40.4 ).
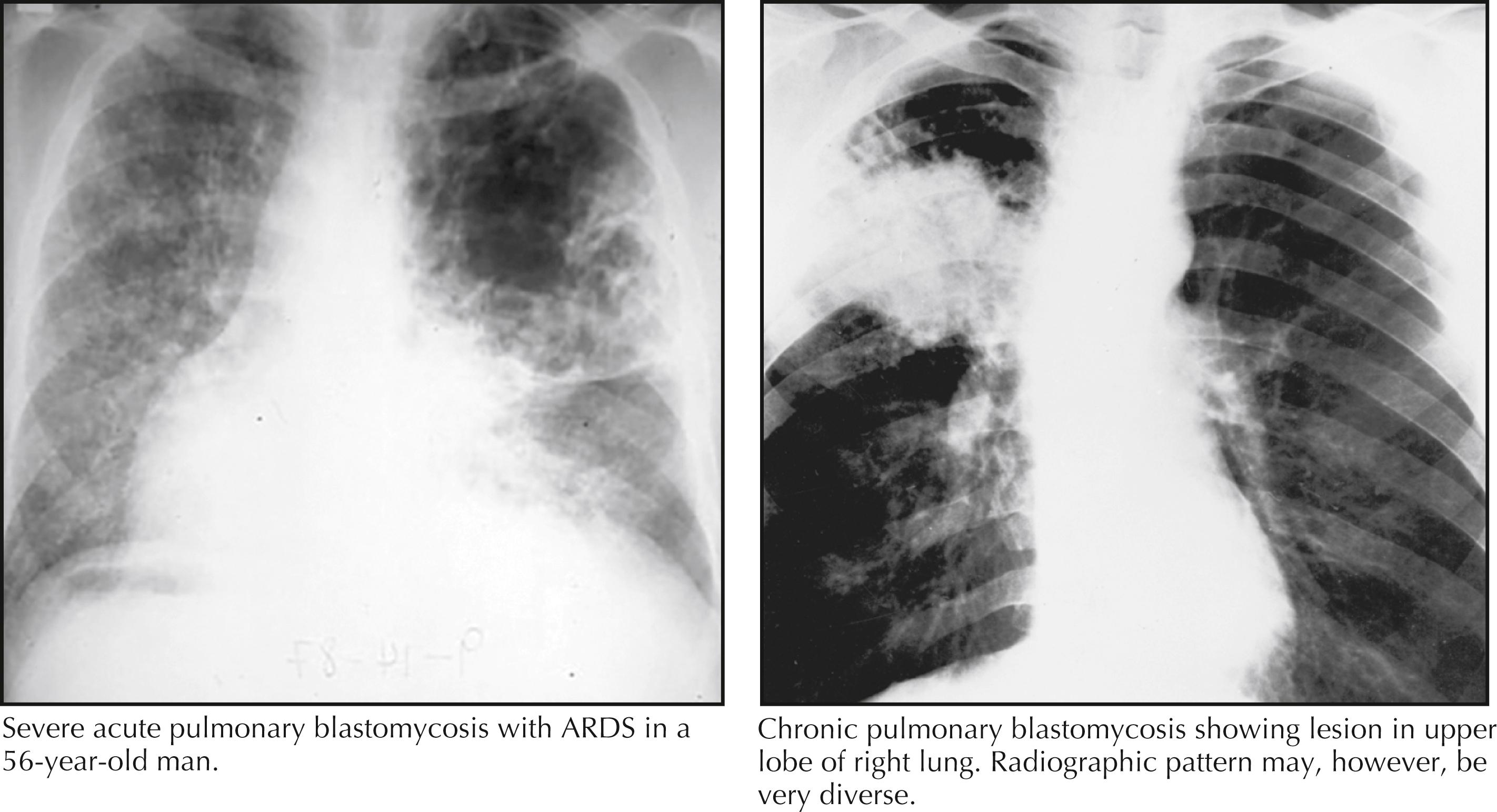
Chronic pulmonary blastomycosis can be mistaken for tuberculosis or lung cancer. The symptoms include fever, night sweats, weight loss, fatigue, dyspnea, cough, sputum production, and hemoptysis. The chest radiograph shows upper-lobe cavitary infiltrates, mass-like lesions, or multiple nodular lesions (see Fig. 40.4 ). Hilar and mediastinal lymphadenopathy occur less often than with histoplasmosis.
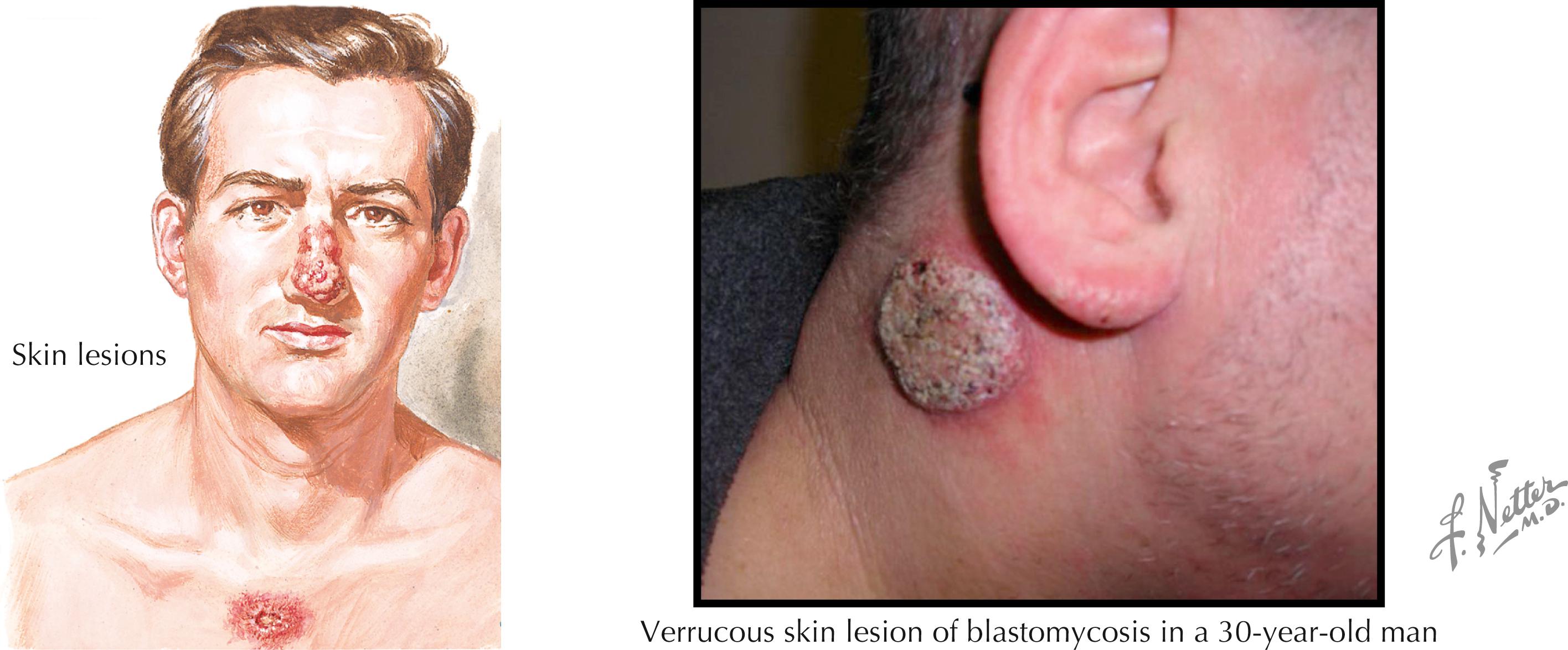
Cutaneous lesions are the most common manifestation of disseminated blastomycosis. The lesions are nonpainful well-circumscribed nodules or plaques that are verrucous and have punctate draining areas in their centers ( Fig. 40.5 ). The lesions can also manifest as painful ulcerations or pustular nodules. The skin lesions of blastomycosis can be mistaken for those caused by nontuberculous mycobacteria, bromide use, and pyoderma gangrenosum. Patients with skin lesions may or may not have pulmonary manifestations; in many, the pneumonia has resolved by the time the skin lesions appear. Osteoarticular blastomycosis may be contiguous to skin lesions or may occur at distant sites. The genitourinary tract is another frequent site of disseminated infection, and the prostate is the usual target organ. Prostatic nodules, with or without symptoms of prostatism, are found.
Blastomycosis is more severe in immunosuppressed patients, although it is seen much less commonly than histoplasmosis and coccidioidomycosis in patients with AIDS, transplant recipients, and patients receiving TNF antagonists. In these patients, the disease is usually disseminated to multiple organs; severe pulmonary manifestations also are more common.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here