Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Primary sclerosing cholangitis (PSC) is a chronic, cholestatic liver disease of unknown cause characterized by diffuse inflammation and fibrosis of intrahepatic and/or extrahepatic bile ducts and is strongly associated with inflammatory bowel disease (IBD). PSC is ultimately progressive, leading to obliteration of the biliary tree and subsequently to biliary cirrhosis. To date, the etiology of PSC remains unknown and effective medical therapy is not currently available. Patients with PSC have shortened life expectancy. Orthotopic liver transplantation (OLT) extends the life of patients with advanced-stage PSC. Development of colon cancer, gallbladder cancer, cholangiocarcinoma (CCA), and hepatocellular carcinoma are known complications of the disease.
PSC usually affects more young males than females. The mean age of diagnosis is the late 30s. In the United States, population-based studies have estimated an age-adjusted incidence for PSC to be 1.25 and 0.54 per 100,000 in men and women, respectively. Moreover, the calculated prevalence of PSC was 20.9 and 6.3 per 100,000 in men and women, respectively. Approximately 75% to 80% of northern European origin patients with PSC suffer from IBD, with chronic ulcerative colitis (CUC) being more common (approximately 90%) than Crohn disease.
PSC is associated with a lack of smoking. In one study the incidence of current smoking was 19% in patients with PSC compared with 38% of controls. In another study, 4.9% of PSC patients were reported to smoke compared with 26.1% of controls. The odds of having PSC in current smokers or former and current smokers compared with never-smokers were 0.13 and 0.41, respectively, regardless of the presence or absence of IBD. Studies have also reported that prior appendectomy may delay the onset of PSC but does not affect either the prevalence or severity of the latter.
The clinical presentation of PSC is heterogeneous and varies widely depending on the disease stage at the time of diagnosis. Asymptomatic individuals typically come to medical attention because of abnormal liver biochemistry detected during routine screening. Symptomatic patients present with symptoms/signs of cholestasis and complications of chronic cholestatic liver disease. The symptoms may include fatigue, pruritus, right upper quadrant pain, weight loss, and manifestations related to portal hypertension (i.e., ascites, gastrointestinal bleed from esophageal/gastric varices). Symptoms of bacterial cholangitis are less common, except if the patient has dominant biliary strictures and/or biliary stones. The physical examination of symptomatic patients may reveal jaundice, hepatomegaly, splenomegaly, skin excoriations, ascites, and peripheral edema.
A frequent clinical scenario is a patient with CUC who presents with a cholestatic pattern of liver enzymes. PSC can affect any age group, including children. Children may present with an overlap syndrome of PSC and autoimmune hepatitis (AIH), which can be as high as 35% according to a recent study.
The criteria used to diagnose PSC are based on the clinical presentation, biochemical abnormalities, histologic features, and the characteristic cholangiographic changes that affect the intrahepatic and extrahepatic bile ducts. In addition, secondary biliary cirrhosis needs to be excluded. The most frequent clinical presentation currently is an asymptomatic patient with persistently increased levels of alkaline phosphatase noted on routine serum biochemical testing.
Liver biopsy is recommended, but it is not always required to make the diagnosis. In a study of 79 patients with a PSC diagnosis established by cholangiography, liver biopsy performed following diagnosis did not affect the management in the vast majority of patients. The role of liver biopsy in PSC is to (1) exclude other causes of cholestatic liver disease; (2) diagnose or exclude small-duct PSC; and (3) define the PSC stage, which may have prognostic value. Small-duct PSC is a variant of PSC that accounts for approximately 5% of histologically proven cases. Small-duct PSC presents with a cholestatic pattern of liver enzymes and normal cholangiography, but liver biopsy reveals evidence of PSC. Small-duct PSC has a better long-term prognosis compared with classic PSC. However, a portion of patients with small-duct PSC progress to classic PSC over time.
In most patients the history, clinical presentation, serum biochemical profile, and cholangiography distinguish PSC from other causes of chronic cholestatic liver disease.
The pathologic findings in PSC depend on the duration and extent of the disease. Early on in the disease the liver may appear grossly normal, whereas in established PSC, biliary cirrhosis may be present with associated portal hypertension, ascites, and splenomegaly. The bifurcation of the hepatic duct is macroscopically involved in most cases. Enlarged lymph nodes are often present in the porta hepatis and along the proper and common hepatic arteries and the hepatic and common bile duct.
Liver biopsy is recommended for staging liver disease to determine the prognosis. PSC is staged histologically (1 to 4) according to the following system : stage 1 (portal stage), there is edema, inflammation, and ductal proliferation; stage 2 (periportal stage), periportal fibrosis and inflammation are noted; stage 3 (septal stage) is defined by septal fibrosis or bridging necrosis; stage 4 (cirrhotic stage) is characterized by biliary cirrhosis. Unfortunately, histologic changes can be markedly varied from segment to segment of the liver at any given point in time.
Cholangiography is required to diagnose a patient with PSC. Endoscopic retrograde cholangiopancreatography (ERCP) is the standard approach to evaluate the bile ducts. Magnetic resonance cholangiography (MRC) may suffice, and percutaneous transhepatic cholangiography (PTC) is sometimes necessary when ERCP is not possible. The classic cholangiographic findings of PSC include multifocal stricturing and beading throughout the biliary tree ( Fig. 117.1 ). Strictures are often diffusely distributed with intervening segments of dilated ducts (i.e., ectasia). The cholangiographic findings usually involve both the intrahepatic and extrahepatic bile ducts. Strictures can vary from 1 or 2 mm to several centimeters in length, and 30% to 40% of PSC patients may have mural irregularities, producing a shaggy appearance. These lesions may vary from a “fine brush border” to “frank nodularity.” Pseudodiverticula (i.e., tiny diverticulum-like outpouchings) of the extrahepatic bile ducts are nearly pathognomonic for PSC. In approximately 20% of PSC patients, only the intrahepatic and perihilar extrahepatic bile ducts are involved, and as many as 15% of PSC patients have involvement of the gallbladder and cystic duct. Moreover, approximately 5% of patients have small-duct PSC (i.e., normal cholangiogram but liver disease detectable on biochemical testing and histology).
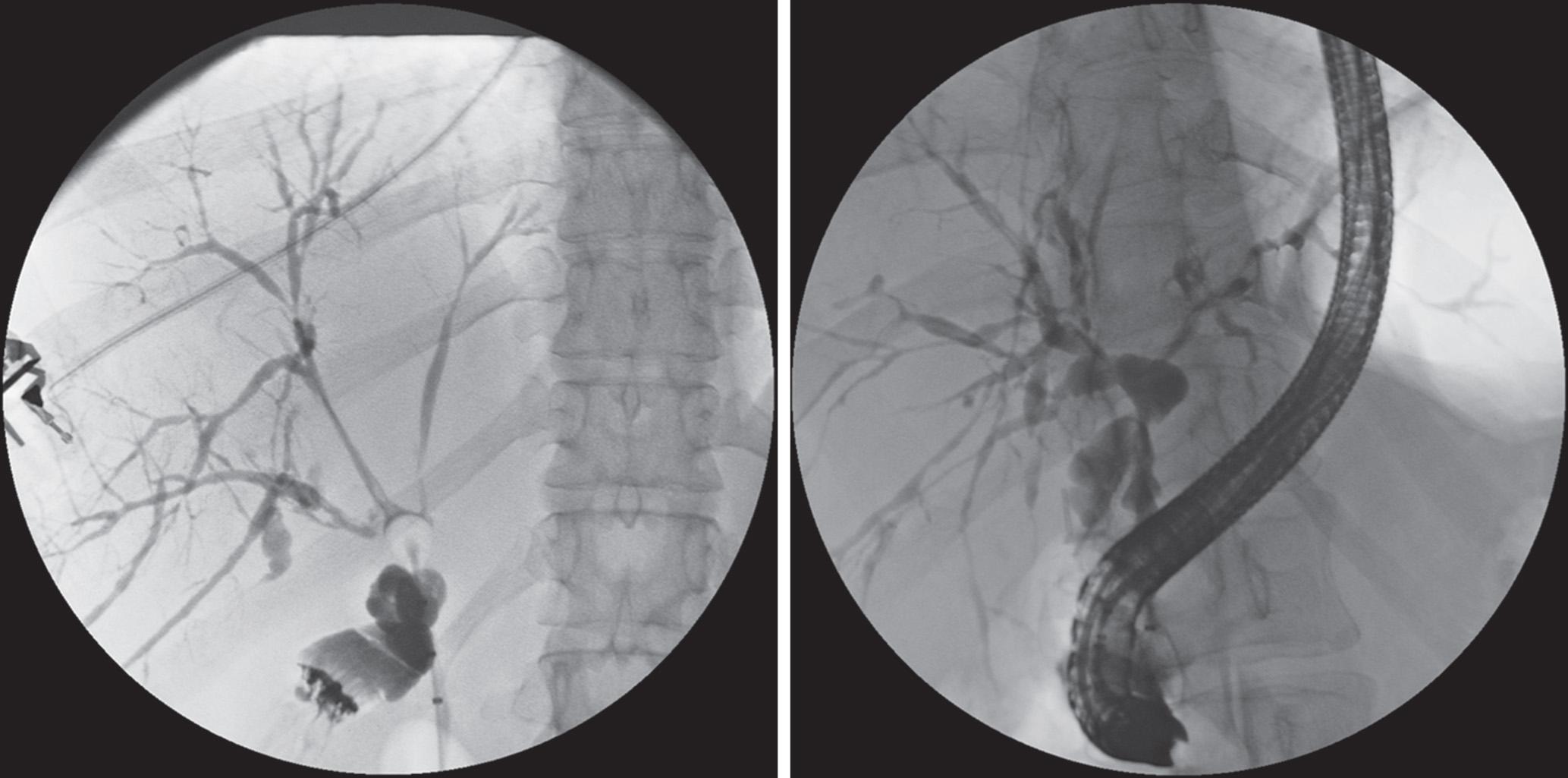
MRC is a noninvasive substitute for ERCP for the diagnosis of PSC. In a study of 73 patients with clinically suspected biliary disease, the sensitivity and specificity of MRC for diagnosing PSC were 82% and 98%, respectively. These authors reported that MRC had comparable diagnostic accuracy to ERCP, leading to reduced cost when used as the initial approach to diagnose PSC. In PSC patients, MRC could be used as a noninvasive imaging method for the detection of CCA. Indeed, some clinicians now prefer MRC over ERCP for the diagnosis of PSC.
PTC is used less frequently to image the bile ducts in patients with suspected PSC. PTC is an alternative approach to access the biliary tree when ERCP is not technically possible. Abdominal ultrasonography is valuable to evaluate the bile ducts for dilation and/or stones, and liver parenchyma for cirrhosis. Computed tomography (CT) can reveal morphologic features of liver cirrhosis. Atrophy of the left lateral segments and hypertrophy of the caudate lobe may differentiate cirrhosis associated with PSC from that seen in other causes of chronic liver disease. CT can also complement cholangiography in evaluating for malignancy, given its ability to detect peripheral, intrahepatic CCA and metastatic spread within the hepatic parenchyma or the abdomen. It is important to note that perihilar lymphadenopathy is common in PSC, and this finding alone cannot be taken as evidence of malignancy or metastasis.
IBD is seen in approximately 70% to 80% of patients with PSC and ulcerative colitis accounts for approximately 85% to 90% of those patients. Usually, the diagnosis of IBD is established approximately 8 to 10 years before the diagnosis of PSC, although cases of IBD occurring years after the diagnosis of PSC have also been reported. Conventional treatment of IBD does not alter the course of PSC, and severity of the former does not affect the disease seriousness of the latter. Proctocolectomy, the most aggressive treatment for CUC, has had no effect on PSC natural history. PSC patients with CUC have increased risk of colorectal dysplasia and neoplasia after OLT. The increased neoplastic potential is of concern in PSC patients following OLT particularly because of the life-long immunosuppression that is required. Thus, in PSC patients who undergo OLT, annual colonoscopy with surveillance biopsies is recommended.
PSC can coexist with AIH. These patients typically fulfill definitive criteria for both diseases and have elevated serum alkaline phosphatase and aminotransferases, increased IgG, and antinuclear and/or anti–smooth muscle antibodies. Liver biopsy shows moderate to severe interface hepatitis with or without biliary destruction. Aminotransferase levels are higher than what one would expect for classic PSC. Patients with overlap syndrome may show improvement of AIH with immunosuppressive therapy. Indeed, patients who present with AIH and do not respond entirely to immunosuppressant therapy should be suspected of having concurrent PSC.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here