Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Precise assessment of disease progression and adequate selection of initial therapeutic options should be performed through a multidisciplinary approach in the treatment of liver metastases.
Selected patients with colorectal liver metastases are candidates for curative hepatic resection with 5-year survival up to 58%.
Number, size, and bilobar distribution of liver metastases do not limit the indication of surgery when R0 resection is feasible.
Surgical resection should be deferred in patients with significant progression of metastatic disease during treatment with optimal preoperative chemotherapy until achieving disease control with second-line systemic chemotherapy or regional therapies.
Minimal requirement of standardized future liver remnant is dependent on the status of the underlying liver (>20% in normal liver, >30% in damaged liver, and >40% in cirrhosis).
Selected patients with advanced liver metastases benefit from reversed approach (resection of metastases prior to primary lesion) or staged hepatectomies.
Radiologic complete response to chemotherapy does not signify complete pathologic response. Therefore, surgical resection should include all the original sites of metastases.
Surgical resection of metastases from neuroendocrine tumor reduces symptoms and prolongs survival even after cytoreduction surgery. However, multidisciplinary approach is usually required in advanced disease.
Significance of surgical resection of metastases of noncolorectal and nonneuroendocrine origin is still unclear.
The liver is the most common site of hematogenous metastases from various malignancies, particularly those originating from the gastrointestinal tract. Although the patients having liver metastases are classified as stage 4 in TNM (tumor, node, metastasis) classifications, favorable long-term outcomes have been reported when curative resections are feasible especially in the patients with colorectal liver metastases (CLMs) , or neuroendocrine tumors (NETs). ,
With recent advancements in chemotherapy and growing use of portal vein embolization (PVE), resectability of CLM has dramatically increased, and long-term survival after resection of CLM has significantly improved. The practical keys in initial clinical management of patients with CLM are (1) precise assessment of extension of disease and (2) adequate selection of therapeutic options. Surgical resection is potentially the most curative therapeutic strategy for liver metastases. However, to select the patients who would truly have prognostic advantages from surgery, a multidisciplinary approach by surgeons, medical oncologist, radiologist, and pathologist is essential ( Figure 13-1 ).
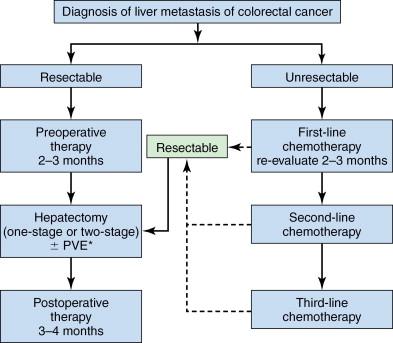
Colorectal cancer is the second leading cause of cancer-related death in the United States. The 20%–25% of patients are found to have synchronous CLM, , and 35%–55% of patients develop CLM during the course of the disease. However, the 5-year survival after curative resection of CLM has been reported to be up to 58%, whereas median survival of CLM without any treatment is approximately 6 months. Therefore, adequate assessment and preoperative management are important to select the patient with resectable or potentially resectable CLM.
In pretreatment assessment of oncological status of CLM, liver dynamic computed tomography (CT) is a standard imaging modality with an adequate sensitivity and specificity. Magnetic resonance imaging (MRI) and positron emission tomography (PET) can be added in selected cases to improve the diagnostic accuracy, especially for diagnosis of indeterminate lesions, detection of local recurrence, or screening of extrahepatic disease. However, the choice of imaging modalities should be based on the local expertise and the balance between the costs and benefits.
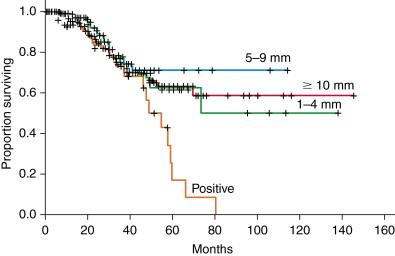
Previously, four or more tumors, size >5 cm, bilateral disease, and narrow surgical margin <1 cm were reported to be negative prognostic factors, and accordingly, they have been considered as contraindications for liver resection. In the era of effective systemic therapy and PVE, however, the oncologic criteria for liver resection is shifting toward complete resection of all viable disease because favorable outcomes would be expected in a selected patient amenable to curative resection even with extrahepatic diseases. Also, a multicenter study of 557 patients has reported that width of surgical margin does not affect the overall survival if negative margin can be obtained ( Figure 13-2 ). Therefore, large number, size, and bilobar distribution of tumor are no longer absolute contraindications for hepatic resection as long as R0 resections are feasible ( Table 13-1 ).
| Conventional Criteria | Current Consensus | |
|---|---|---|
| ONCOLOGIC CRITERIA | ||
| Number | ≤3 | No limitation if R0 resection is feasible |
| Size | ≤5 cm | No limitation if R0 resection is feasible |
| Distribution | Unilateral distribution | No limitation if R0 resection is feasible |
| Extrahepatic disease | Contraindication | If curatively resectable or long-term control can be expected |
| TECHNICAL CRITERIA | ||
| Surgical margin | ≥1 cm | Microscopic R0 resection |
| Future liver remnant (FLR) | Anticipated ability to preserve adequate FLR volume with intact blood flow and biliary drainage | Anticipated ability to preserve adequate FLR volume (>20% in normal liver, >30% in extensively treated liver) a with intact blood flow and biliary drainage |
Technical resectability is determined by the balance between the feasibility of curative resection (R0 resection) and preservation of adequate inflow–outflow pathways for the liver remnant. Technical feasibility of liver resection basically depends on the distribution of tumors and vascular anatomy. Because the metabolic performance of the liver is highly dependent on both intact inflows (i.e., arterial and portal flows) and adequate outflows (i.e., venous flow and biliary drainage), patency of these four vessels is essential to expect a full performance of the future liver remnant (FLR).
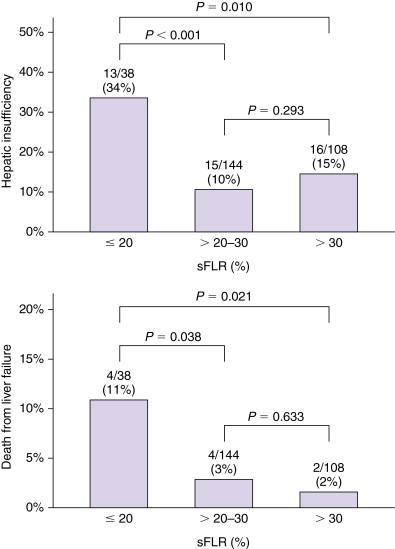
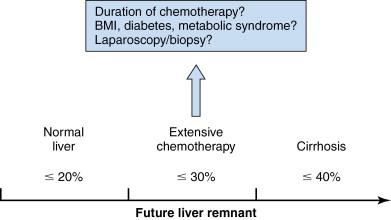
Currently, functional reserve of the liver is estimated by both static and dynamic measurements. The most reliable static variable is the FLR volume. Because absolute volume of FLR against standardized liver volume (SLV) (i.e., sFLR: standardized FLR) has strong correlation with the rates of postoperative morbidity and mortality ( Figure 13-3 ), , minimal requirements of sFLR has currently been set at >20% in normal liver, >30% in damaged liver after extensive treatment, and >40% in cirrhotic liver ( Figure 13-4 ). These cutoff values offer a good practical decision making in patients requiring major hepatectomy. Dynamic measurements such as degree of hypertrophy, indocyanine green clearance test, or hepatic scintigraphy have also been reported to be a sensitive variable to evaluate functional reserve of the liver. Because FLR volume itself is not correlated with functional reserve, dynamic measurements should be integrated to estimate the total functional reserve of FLR in individual patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here