Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The origins and development of current vascular surgical trauma care are largely based on experiences from wartime casualties and practice. Care for this segment of the population has always presented challenges for physicians to improve medical and surgical practices. Advances in limb salvage were achieved in the mobile surgical hospitals and vascular specialty centers of the Korean and Vietnam Wars. Arterial repairs and vein bypass grafting performed far forward in these wars resulted in a decrease in amputation rates from 50% with arterial ligation to 13%. In the Vietnam Vascular Registry, data was collected and analyzed to develop best practices for vascular trauma management. As such, lessons learned about the management of vascular trauma in combat and war zones were disseminated to the civilian sector. In this day-and-age, civilian trauma algorithms are deeply rooted in these lessons learned from our military heritage.
The application of endovascular techniques has revolutionized the modern management of vascular trauma. A 9-year analysis of 23,105 patients in the National Trauma Data Bank demonstrated a marked increase in the use of these techniques. During this 9-year period, the prevalence of using endovascular techniques increased from 0.3% in 2002 to 9.0% in 2010. This was particularly true among blunt trauma patients (from 0.4% in 2002 to 13.2% in 2010; P < .001), and injuries of the internal iliac artery (from 8.0% in 2002 to 40.3% in 2010; P < .001), thoracic aorta (from 0.5% in 2002 to 21.9% in 2010; P < .001), and common/external iliac arteries (from 0.4% in 2002 to 20.4% in 2010; P < .001).
The pivotal step in performing vascular surgery is to have control of the vessel being operated on. In open surgery, the operator needs to have proximal and distal control before any intervention is performed. In endovascular surgery, the operator needs to have a wire across the lesion before any intervention is performed. These basic tenets are the same in the management of traumatic vascular injuries but are much more challenging to achieve. The vascular trauma patient is unique due to the severe physiologic derangements secondary to massive hemorrhage and concomitant injuries. These hurdles can be compounded by limited resources, which are sometimes encountered in the battlefield or in small community hospitals.
Mean 1-year cost per patient of trauma care in our population was $75,210. On average, 58% of cost was accounted for by the index hospitalization. Total 1-year treatment cost of adult major trauma in the United States was conservatively estimated to be US$27 billion annually in 2005. In 2013, the CDC estimated the total lifetime medical and work loss costs of injuries and violence at $671 billion. In both civilian and combat settings, the incidence of vascular injury is approximately 0.2% to 7% of all injured patients. The social impact of trauma goes beyond the cost of health care for trauma victims. It extends to the lost lifetime productivity, lost wages, and the need for social support such as unemployment wages and workers compensation. Trauma is the leading cause of death in the 1- to 44-year-old age group in the United States, as a consequence of unintentional injury, assault, homicide, and suicide. Iatrogenic vascular injury has also been increasing in prevalence as the number of invasive procedures increases. In fact, it has been described as high as 33% of all vascular trauma at a Level I trauma center.
Hippocrates was credited with saying, “War is the only proper school for a surgeon.” Vascular trauma management is no exception. As we already mentioned in the onset of this chapter, many of the trauma management paradigms used in the civilian society are extension of the experiences encountered by surgeons in combat during war. Similarities exist between civilian and military vascular trauma that allows us to extrapolate on our military experience and utilize the acquired knowledge to guide trauma management in the civilian setting. Nonetheless, it is important to point out that there are many differences between patterns of military versus civilian vascular trauma.
Among civilian victims, gunshot wounds are responsible for the majority of peripheral vascular injury, stab wounds for a smaller percentage, and the remainder is due to blunt trauma. As such, most civilian gunshot wounds are generally due to low-velocity projectiles. This is in contradistinction to military trauma where arterial injury usually occurs in the setting of massive extremity involvement, where orthopedic fractures, large soft tissue defects, nerve injury, and vein injury are common. This pattern is seen in one-third of patients presenting with vascular injury to combat trauma-receiving centers; it can also be seen in civilian close-range shotgun blasts. The majority of military traumatic injuries are due to explosive devices (74%) with high-velocity gunshot wounds responsible for most of the remainder (17%); blunt injury is infrequent.
Vascular trauma, whether in the civilian or military setting, most commonly causes the patient's demise due to uncontrolled hemorrhage. If the patient survives the initial physiologic insult, it can still lead to death if the lethal triad of hypothermia, coagulopathy, and acidosis sets in and takes control of the victim. Care of vascular trauma patients should first aim at hemorrhage control and managing the trauma and hemorrhagic homeostatic derangement consequences. If repair can be done in a safe and expedited fashion without further compromising the patient's homeostasis, then it should be performed. Otherwise, damage control resuscitation approach for reversal of the lethal triad and stabilizing the patient might be the best chance at survival. The principle of live to fight another day is based on immediate control rather than immediate repair. The basic tenet of damage control surgery is to stop ongoing contamination and hemorrhage, transfer the patient as rapidly as possible to an intensive resuscitative care unit, and employ all medical resources at hand to halt the progression of the lethal triad.
In order to address concerns that damage control resuscitation (DCR) might have a deleterious effect on vascular reconstructions, Fox and colleagues performed a case-control study of battlefield casualties who underwent vascular repair and compared patients treated by a rigorous DCR protocol versus conventional crystalloid and nonmatched erythrocyte resuscitation. There was no worse outcome among patients when the coagulopathy was treated. The author's admonition was to “fix the injury, fix it well, and trust that your patient doesn't need to be coagulopathic for the graft to work.” Damage control options for vascular trauma include tourniquet use, temporary vascular shunting (TVS), ligation, or balloon tamponade. None of these technique has proven superior to the others in temporizing vascular injury. Taken as a whole, however, damage control for major trauma is associated with improved physiology, increased limb salvage, and decreased mortality.
Initial management of vascular trauma should follow the guidelines of advanced Trauma Life Support (ATLS) with attention to the airway, ventilation, external hemorrhage control, and restoring circulating blood volume. Once the initial survey of the trauma patient is complete, the secondary and tertiary assessment of the trauma patient with a detailed history and physical exam followed by the necessary diagnostic imaging.
Trauma victims usually have multisystem injuries. Before embarking on a complex and potentially lengthy vascular repair, the provider must ensure there are no missed and more life threatening injuries which need to be addressed first. Unrecognized severe intraabdominal or intracranial injuries can lead to disastrous outcomes if not addressed early.
Hard signs, such as active arterial hemorrhage, expanding hematoma, loss of distal pulse, signs of ischemia, and a bruit or thrill are highly suggestive of arterial injury. Profound signs of acute ischemia include the classic 6 “Ps”: paresthesia, paralysis, pulselessness, pain, pallor, and poikilothermia. With such findings, no diagnostic imaging is usually required before surgical exploration of extremity vessels. Soft signs raise the suspicion of arterial injury and include moderate-sized hematoma, history of significant blood loss or hypotension at the prehospital scene, diminished distal pulses, and ipsilateral neurologic deficits. Soft signs warrant additional diagnostic imaging ( Table 48.1 ).
| Hard Signs a | Soft Signs a |
|---|---|
|
|
a Hard signs require surgical exploration and control while soft signs provide time for further diagnostic workup.
b Paresthesia, paralysis, pulselessness, pain, pallor, and poikilothermia.
Diagnostic evaluation of the trauma patient starts with the primary survey on presentation. An unstable patient would need to go directly to the OR for either operative repair or damage control surgery. Once the primary and secondary surveys have been completed, further investigation would assist in localizing the injury if not obvious. Ankle-brachial index (ABI) is a diagnostic exam that is instrumental in the management of lower-extremity trauma patient and is feasible to perform at bedside. An ABI of <0.9 may suggest arterial injury and need for further diagnostic imaging. A good H&P and examination of the uninjured contralateral limb is paramount to appropriately interpret the ABI exam. Older patients with peripheral arterial disease may present with diminished ABIs, confounding the exam. In general, young healthy patients with lower extremity injuries in proximity to major vascular structures but with an ipsilateral ABI of greater than 0.9 can be observed.
In the stable trauma patient without moderate to severe soft tissue damage, an ultrasonography evaluation might provide valuable information to the presence or absence of blood flow, intimal flap, vessel transection, and vessel thrombosis. Albeit operator-dependent, ultrasound imaging provides high quality vascular images. This allows assessments, which are reproducible low cost, and few if any side effects. This modality is particularly helpful in extremity trauma. Wani and colleagues showed that Doppler had a sensitivity of 94% and specificity of 82.5% in diagnosis of vascular injury.
CAT scans have become a main stay diagnostic modality in many if not all trauma centers and emergency departments. Many trauma victims will undergo CAT scan evaluation of various body parts, depending on the nature of the injury. CT angiography is so sensitive and specific to detecting and localizing vascular injuries that it has become the gold standard. In their meta-analysis of 11 studies, Jens and colleagues estimated the sensitivity and specificity of CTA were 96.2% (95% CI 93.5% to 97.8%) and 99.2% (95% CI 96.8% to 99.8%), respectively.
In patients where extremity vascular injury is suspected by mechanism or physical findings, angiography has been the gold standard diagnostic test. However, new research is suggesting that multislice (32 or 64) CT angiography may have sufficient sensitivity to detect these injuries. A prospective study of 21 patients with potential extremity vascular injuries, primarily due to gunshot wounds, and abnormal ankle brachial indices underwent CT angiography followed by either conventional angiography or operative exploration. CTA had 100% sensitivity for clinically relevant findings. Fifty percent of patients had concomitant orthopedic injuries as well. Vascular surgeons at Walter Reed Army Medical Center evaluated the use of CTA to detect vascular injuries and found that it performed well even in the presence of metallic fragments or external fixators.
MRI and MRA long acquisition time has been the limiting factor in utilizing these modalities in the setting of acute trauma. Other shortcomings include the high cost, noncompatibility in trauma patients with potential for metallic fragments, whether in civilian or military settings from gunshot wounds, improvised explosive devices, and so on. There is also the potential for nephrogenic systemic fibrosis in patients with advanced renal insufficiency.
Historically speaking, angiography has always been the gold standard for diagnosing vascular injury. In this day and age, high resolution, multidetector CT angiography had largely replaced angiography for diagnostic purposes, as it provides similar quality imaging while bypassing the risk of the invasive nature of angiography. Endovascular techniques are largely reserved nowadays for intervention. Nonetheless, it remains as another diagnostic modality in the armamentarium of the vascular surgeon or interventionalist.
As mentioned earlier in the chapter, once operative repair is deemed necessary, basic principles for management of vascular surgery apply irrespective of types and location of injury. Before exploring the injured vessel, proximal and distal control must be obtained. Vessel control can be obtained by various methods, such as vascular tapes or loops, vascular clamps, manual direct pressure with a finger or sponge stick, or balloon occlusive devices. Debridement of the injured segment of vessel is the initial step in vascular repair. Damaged intima, if unattended to, increases the risk of postrepair thrombosis significantly. Resection and debridement of the vessel might need to be extended beyond the site of obvious injury to include the total zone of damaged artery. External examination of the artery alone might not permit proper assessment of the extent of injury. If a simple laceration of large vessels is encountered, lateral suture repair can suffice as long as the vessel is large enough and the laceration is less than 50% of the circumference. Larger lacerations (over 50% of the vessel diameter) or smaller vessels may require a patch angioplasty to prevent stenosis. Autogenous vein is the preferred patch material. Also, segmental resection with an end-to-end anastomosis can be performed for more complex injuries spanning a distance of less than or equal to 1 cm. The vessel ends should be spatulated to avoid anastomotic stricture. The goal of the repair is to produce a tension-free anastomotic repair. Generally, 3 cm of artery need to be mobilized for every 1 cm resected. For injuries greater than 1 cm in length, an interposition graft is usually required. Ideally, the contralateral saphenous vein, if available, should serve as the conduit of choice. The ipsilateral saphenous vein should not be sacrificed if there is any concern for concomitant ipsilateral venous injury, as it is an important collateral venous return pathway. If the autogenous vein is unavailable, prosthetic graft is an alternative, and polytetraflouroethylene (PTFE) is most commonly used. In multiply injured extremities, finding adequate conduit may be challenging. PTFE provides a rapidly accessible option and can be used if the autologous vein is not available or if the patient will not tolerate a long vein harvest; this latter scenario may also argue for TVS. One major concern with prosthetic graft is its use in infected surgical beds. Vertrees and colleagues demonstrated that in patients with limited autologous vein, prosthetic graft can be used temporarily in contaminated wounds in multiple anatomic locations with acceptable short-term patency and an opportunity for later revision.
Cervical trauma poses a diagnostic and therapeutic dilemma, as the neck houses numerous vital structures in a very limited space. Anatomically, this spans the area from the base of the skull to the thoracic outlet. The aerodigestive tract, brachial plexus, and nerves, as well as the carotid sheath, subclavian, and vertebral vessels, are all at risk.
During the 1990s, work from trauma groups in Memphis and Denver uncovered a much larger incidence of this type of injury that had been previously suspected. One of the initial reports evaluated 20,349 trauma admissions at the Presley Regional Trauma Center; 67 patients (0.33%) were found to have 87 blunt cerebrovascular injury (BCVI). In a pilot study of all patients undergoing post-injury thoracic aortograms performed at the University of Colorado, 3.5% of patients were found to have a documented BCVI, only half of whom manifested symptoms. Disconcerted by these findings, the trauma surgeons in Denver undertook a prospective study in 249 patients, 40 with symptoms consistent with BCVI and 209 with high-risk features ( Table 48.2 ). This report documented BCVI in 28 (70%) of the symptomatic patients and 57 (27%) of the asymptomatic patients. Injuries included 65 isolated carotid artery injuries, 10 isolated vertebral artery injuries, and 10 combined carotid and vertebral artery injuries. In another report from the same year, their overall incidence was reported at 0.38% of trauma admissions, but was 1.07% among admissions during the time they performed aggressive screening. Contemporary estimates of BCVI incidence is 0.4% to 1.0% of blunt trauma admissions. The mechanism of the majority of these injuries is motor vehicle crash, reported in 45% to 82% of series, followed by pedestrian trauma (12%), motorcycle crash (11%), falls (9% to 10%), and assaults (5% to 6%). Nowadays BCVI is estimated to occur in 3% to 10% of blunt trauma victims, with a stroke rate up to 25%. Guidelines for BCVI screening are based on clinical and radiographic findings, as noted in Table 48.2 .
| Hard Signs of BCVI | Signs Suggestive of Occult BCVI |
|---|---|
| Hemorrhage from the mouth, ears, nose, or wounds | Severe cervical hyperextension and rotation or hyperflexion if associated with displaced midface or complex mandibular fracture or closed head injury consistent with diffuse axonal injury |
| Expanding cervical hematoma | Near hanging resulting in anoxic brain injury |
| Cervical bruit in a patient <50 years old | Seat belt abrasion or other soft tissue injury of the anterior neck resulting in significant swelling or altered mental status |
| Evidence of cerebral infarction on computed tomography scan | Basilar skull fracture |
| Unexplained or incongruous neurologic deficit, transient ischemic attack, amaurosis fugax, or Horner syndrome | Cervical vertebral body fracture |
The grading system proposed by the Denver group in 1999 is still in use today to guide the management of BCVI ( Table 48.3 ). This system correlates well with mortality and stroke risk, and has been used as the basis for treatment recommendations. In a recent report, the overall stroke risk for patients with BCVI was 14% and the in-hospital mortality was 10%. Another report cited a 26% in-hospital mortality. Among those who survived to discharge, only 31% were discharged home, whereas 43% were transferred to another inpatient facility such as rehabilitation or skilled nursing. By injury grade, mortality was 0 % to 33% for grade I to III lesions, 11% to 60% for grade IV, and 100% for grade V; stroke risk was 3% for grade I, 11% for grade II, 33% for grade III, 44% for grade IV, and 100% for grade V.
| Injury Grade | Description |
|---|---|
| I | Luminal irregularity or dissection with <25% luminal narrowing |
| II | Dissection or intramural hematoma with ≥25% luminal narrowing, intramural thrombus, or raised intimal flap |
| III | Pseudoaneurysm |
| IV | Occlusion |
| V | Transection with free extravasation |
There are no level I evidence to guide management recommendations. EAST (Eastern Association for the Surgery of Trauma) statement recommends diagnostic evaluation should be pursued for the screening and diagnosing of DCVI based on level II evidence in patients presenting with any neurologic abnormality that is unexplained by an already diagnosed injury; blunt trauma patients presenting with epistaxis from a suspected arterial source after trauma should be evaluated for BCVI. EAST also recommends considering diagnostic evaluation based on level III evidence in asymptomatic patients with significant blunt head trauma are at significantly increased risk for BCVI, and screening should be considered. Risk factors are as follows:
Glasgow Coma Scale score ≤8
Petrous bone fracture
Diffuse axonal injury
Cervical spine fractures particularly those with
Fracture of C1 to C3 and
Fracture through the foramen transversarium
Cervical spine fracture with subluxation or rotational component
Lefort II or III facial fractures
Based on level II evidence, diagnostic four-vessel cerebral angiography (FVCA) remains the gold standard for the diagnosis of BCVI. Nonetheless, multislice (eight or greater), multidetector CTA has a similar rate of detection for BCVI when compared with historic control rates of diagnosis with FVCA and may be considered as a screening modality in place of FVCA.
Once diagnosed, management of BCVI is based on the grade of injury sustained. Based on level II evidence, in the absence of contraindications, grade I and II injuries are treated with antithrombotic agents. Based on level III evidence, either antiplatelet or anticoagulation therapy can be used with seemingly similar outcomes. If anticoagulation is selected for treatment, the infusion should be started without a bolus and continued till an oral agent is started and therapeutic levels are achieved. Therapy for 3 to 6 months is recommended. In grade III injuries, pseudoaneurysms rarely resolve with observation or heparinization, and invasive therapy (open or endovascular approach) should be considered. If carotid stents are placed without subsequent antiplatelet therapy, a high rate of thrombosis was noted in this population. If the patient is presenting with early neurologic deficit and an anatomically accessible carotid lesion, operative (open or endovascular) repair should be considered to restore flow. Grades IV and V are highly morbid and lethal injuries. Some authors feel that outcomes are generally independent of treatment efforts, although others have suggested decreased stroke risk with heparin therapy for grade IV lesions. Interestingly, one center reported treating occlusion of the internal carotid artery in asymptomatic patients after penetrating trauma with embolization to prevent an occluded artery “reopening” and providing a substrate for distal embolization after vasospasm dissipates. Grade V transections require obtaining proximal and distal control, which can be extremely challenging depending on the location of the injury. Outcomes are miserable; one series reported 100% mortality from this injury.
Penetrating cervical injuries are inherently differently than BCVI, due to differences in pathophysiology, diagnosis, treatment, and outcomes. The mechanism of injury is different between military and civilian population. In a series of 63 combat-trauma-related penetrating cervical injuries, 33 (53%) had positive findings. A total of 25 patients were explored in-theater, and eight had findings detected after evacuation to the United States and underwent repair in a delayed fashion. Blast fragments were responsible for the majority of these wounds (79%), with high-velocity gunshot wounds being responsible for the remainder. This incidence did not change much over the years. Brennan and colleagues reported 98% of these neck injuries were due to high-velocity projectiles. In civilian trauma, the most common mechanism is gunshot wounds. In a series of 38 civilian gunshot wounds to the internal carotid artery (ICA), 89% of patients had symptoms including neck hematoma (84%), active hemorrhage (32%), and neurologic deficits (26%). The current mortality rate in civilians with penetrating neck injuries ranges from 3% to 6%.
Traditionally, the cervical area was divided into three zones progressing from caudal to cephalad. This is based on the classification proposed by Monson and colleagues in 1969.
Zone I extends from the thoracic outlet to the cricoid cartilage; Zone II extends from cricoid cartilage to the angle of the mandible; and Zone III extends from angle of the mandible to the base of the skull. Knowledge of vascular structures that commonly traverse each zone is paramount as initial triage and management of the cervical trauma is dependent on the zone and type of injury incurred ( Table 48.4 ).
| Zone I: Thoracic Outlet to Cricoid Cartilage | Zone II: Cricoid Cartilage to Angle of Mandible | Zone III: Angle of Mandible to Base of Skull |
|---|---|---|
|
|
|
In a hemodynamically stable patient, diagnostic evaluation would be considered. Sclafani and colleagues found that the presence of nearby fragments decreased the sensitivity of this modality in military trauma situations and recommended checking a plain film first if MDCT-A is planned. The gold standard remains FVCA. In civilian trauma, the current practice trend is to rely on MD-CTA evaluation and resort to angiography if intervention in planned or if the CTA images are suboptimal. Duplex examination is often limited by the wound, dressing, or injuries to the cervical spine.
Hemodynamic instability would mandate immediate exploration in the operating room regardless of the zone of injury without further imaging beyond cervical plain films. In this day and age, endovascular techniques would provide the advantage of obtaining further diagnostic imaging intraoperatively, with the ability of vascular control and possibly intervention. This is particularly important for vascular injury in Zones I and III where anatomical restraints makes it difficult to obtain vascular control and intervention.
Proximal control is obtained via a median sternotomy incision with extension of the incision on to the neck as necessary. The aortic arch, innominate, and left common carotid artery can be controlled through this incision. If injury is suspected to the left proximal subclavian artery, a second or third left intercostal space anterolateral thoracotomy would be the preferred approach for control and intervention.
A cervical incision along the anterior border of the sternocleidomastoid muscle is required for Zone II injury (a generous version of a carotid endarterectomy incision). If the bleeding is then noted to be coming from a more proximal source, then the cervical incision can be extended into a median sternotomy. If bilateral cervical vascular injuries are present, either a transverse Kocher incision or bilateral cervical incisions will gain exposure to both carotid sheathes. If the arterial injury is known to involve only the left subclavian artery, a supraclavicular incision can be utilized for exposure of the mid to distal left subclavian artery. An infraclavicular incision can be utilized for exposure of the distal right and left subclavian arteries and the proximal axillary arteries.
For Zone III arterial injury, subluxation or resection of the angle of the mandible and resection of the posterior belly of the digastric muscle may be needed. In this zone, control of the distal internal carotid artery is particular difficult due to the anatomic constraints. Other methods to gain distal control are balloon occlusive devices with access through the common carotid artery or site of injury. Due to the difficulty in exposure and control of Zone III injuries, endovascular approach is the preferred option when possible.
There are several damage control options available in the cervical region. Rapid insertion of a balloon catheter such as a Foley into the wound tract, inflation, and outward traction may provide tamponade and temporize bleeding. In Zone III, exposure is generally difficult and the ICA may be difficult to compress. One option is to perform an arteriotomy on the common carotid after controlling it with vessel loops, insert a number 3 or 4 Fogarty embolectomy catheter, and advance and inflate in a serial fashion until bleeding from the distal ICA abates. This may also be done under angiographic guidance if the patient is in the fluoroscopy suite. An additional advantage of intramural balloon occlusion is that a determination of patient tolerance to ligation can be determined; if the patient manifests a focal neurologic deficit with occlusion, some form of repair should be attempted if at all possible. Finally, for resistant bleeding high in Zone III, packing of the carotid canal with bone wax may allow tamponade for life-saving purposes. After hemostasis is achieved with a damage control strategy, the options include formal high cervical exploration for repair or ligation; arteriographic embolization or/and stenting; or 48 hour balloon occlusion. The Fogarty balloon is filled with contrast and the patient is monitored for signs of cerebral ischemia. If there is no sign of ischemia, the balloon remains in place until 48 to 72 hours postplacement, at which time it is deflated and removed. The patient should be imaged with a crossover angiogram to rule out a pseudoaneurysm above the occlusion. If the patient manifests signs of ischemia, a bypass from the cervical ICA to the petrous ICA is necessary and will require the assistance of a neurosurgeon.
The types of injuries that can be present with penetrating trauma are very similar in type and recommended treatment to those for BCVI, although major wall disruption is much more likely. For intramural hematomas, intimal defects, or dissection with preserved distal flow, treatment similar to grades I and II BCVI is appropriate—namely heparinization. Stenting may be appropriate to tack down the intima for major dissections. Patients should undergo repeat imaging in 1 to 2 weeks. Pseudoaneurysm with contrast extravasation should be either observed (for small aneurysms) or stented with or without coil embolization. According to Feliciano, heparin is risky in this situation given the possibility of rupture. Antiplatelet therapy should be used if a stent is placed, however. For occlusions, anticoagulation is recommended to prevent distal propagation or embolization. As previously mentioned, Sclafani and colleagues recommend embolization of the thrombosed ICA. Finally, carotid-jugular arteriovenous fistulae may be asymptomatic if small, but if sufficiently large may produce high-output heart failure or coma secondary to elevated intracranial pressure. Treatment is with operative repair for lesions in Zone II and embolization in Zone III.
There has historically been controversy regarding the surgical management of the patient with a cervical vascular wound and a neurologic deficit, with concern that revascularization would convert an area of ischemia into hemorrhage. This issue was addressed in an article in 1978 by Liekweg and Greenfield. Among noncomatose patients, 85% of patients with neurologic deficits who are revascularized have positive outcomes versus 50% of those ligated ( P < .05). However, among comatose patients, favorable outcome with repair was seen in 27% versus 25% of patients undergoing ligation ( P = NS). Current practice is to revascularize all patients with penetrating injury regardless of neurologic deficits except for coma. Patients with GCS less than 8 not related to hypovolemia, hypothermia, or intoxication are likely to have an adverse outcome regardless of management. If the presentation is delayed more than 3 to 4 hours postinjury, a CT scan should be obtained; if there an ischemic stroke is present, revascularization is contraindicated, as this may convert to a hemorrhagic infarct.
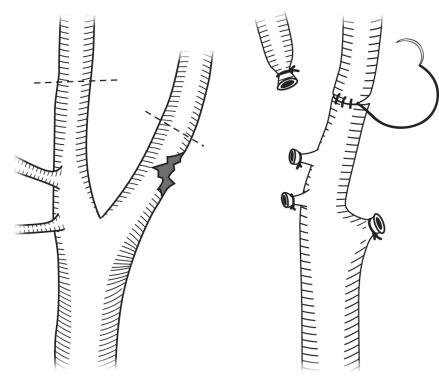
Surgical options include ligation, primary repair, bypass graft with autogenous vein or prosthetic graft, patch graft, or external carotid to internal ( Figs. 48.1 and 48.2 ) for injuries near the carotid bulb. As previously mentioned, Liekweg and colleagues demonstrated improved outcomes for revascularization over ligation for noncomatose patients. For comatose patients, they recommend repair only if there is evidence of prograde flow. If bypass grafting is performed, PTFE is appropriate for common carotid injuries; autogenous vein should be used for ICA repairs, given the improved patency rates. As always, prosthetic grafts should be avoided if possible in contaminated wounds. If surgical repair is performed, a careful search for injury to other structures, especially the aerodigestive tract. If such an injury is encountered, in addition to repairing it appropriately, the vascular suture line should be isolated by means of interposition of vascularized tissue. The sternocleidomastoid muscle can be detached from its sternal head and rotated to cover the repair. Nonoperative therapy options include angioplasty to tack down intimal flaps and coil embolization and stents for pseudoaneurysms. In patients where observation is selected as the treatment plan, repeat imaging is necessary to ensure the resolution of the lesion. In the series by Fox, vertebral artery occlusions were safely observed.

Trauma victims sustaining thoracic vascular trauma are generally divided into three categories. The first group dies immediately at the scene of trauma due to rapid exsanguination. The second group becomes unstable en route to medical care, and the majority of this group (>96%) die secondary to multisystem trauma. The third group represents those who survive the initial trauma and remain relatively stable till their arrival to a medical care; their chance of survival goes up substantially to approximately 70% to 95%, with deaths in this group often being secondary to neurologic injury.
On presentation at the trauma bay, there are several findings, which should raise the clinician's index of suspicion for a thoracic vascular injury. These include hypotension, upper extremity hypertension, intermittent paralysis or focal neurologic findings, inequality of blood pressure or pulses between extremities, external evidence of chest trauma, an expanding hematoma at the thoracic outlet, an intrascapular murmur, a palpable fracture of the sternum or the thoracic spine, or a left-sided flail chest. If the patient is suspected of having a hemothorax based on physical exam and chest x-ray, we recommend placing a 36 to 40 French chest tube in, as a large bore chest tube might serve better in draining a hemothorax without clotting which can happen with a smaller (e.g., 20 French) chest tube. Once chest tubes are placed as indicated, output greater than 1500 mL initially or ongoing hemorrhage at or over 200 to 250 mL/h are indications for thoracotomy.
Initially, care for these patients should be no different than any other trauma patients following the guidelines of Advanced Trauma Life Support (ATLS) protocol. The one lesson to keep in mind in these patients is that fluid resuscitation should be minimized for several reasons. Permissive hypotension will help stabilize early soft platelet plugs. Any patients presenting with military antishock trousers (MAST) in place and the suspicion of a proximal vascular injury should have them removed immediately, as this is effectively a “distal cross-clamp” and can lead to instability of clots and worsened bleeding. If there is blunt injury, aggressive prevention of hypotension helps avoid propagation of the injury. Finally, concomitant pulmonary contusions or other injuries are commonly associated with thoracic vascular injury, and minimizing fluid resuscitation can avoid iatrogenic worsening of respiratory status.
Traditional open repair, while effective in the long-term, is associated with periprocedural mortality between 10% and 20%, along with a 5% risk of stroke and up to a 5% risk of spinal cord ischemia. The advent of endovascular stent-graft repair has allowed single centers to cut periprocedural mortality in half and decrease the risk of major neurologic events to less than 2%.
Historically, aortic injury (predominantly located at the isthmus) is the second most common cause of death in blunt trauma. Despite the advances in the management of these patient, these injuries remain the second most lethal condition in blunt trauma patients, after head trauma. Parmley and colleagues first described thoracic blunt aortic injury in his series in 1958. He advocated for urgent surgical intervention in these patients due to the high mortality rate in his series, a 61% mortality rate at 7 days. Operative mortality has historically ranged from 15% to 28%, and paraplegia has complicated nearly 9% of cases prior to the advent of alternative treatment modalities. In Parmley's initial report, 57% of cases of BAI were secondary to automobile crashes and 16% were due to airplane crashes. This remains true today; more than 80% of BAI is due to automobile accidents.
On plain film chest radiography, findings associated with blunt aortic injury vary dramatically. Most commonly, there is a widened mediastinum, seen in 85% of cases. Other findings include an indistinct aortic knob (24%), a left pleural effusion (19%), an apical cap (19%); uncommon findings include fracture of the first or second rib, tracheal deviation, a depressed left bronchus, and nasogastric tube deviation. A completely negative film is seen, however, in 7% of patients. Historically, aortography was the most common diagnostic modality, with transesophageal echocardiography being a distant second. However, both of these modalities have been supplanted by MDCTA in the updated report by the American Association for the Surgery of Trauma multicenter trial (AAST), known as the AAST II study. Aortography decreased from 87% to 8% of cases, echocardiography from 12% to 1%, with CT angiography increasing from 35% to 93% of cases. MDCTA now stands as the modern gold-standard diagnostic modality for BAI.
Table 48.5 reflects the currently accepted grading system of blunt thoracic aortic injury, which was adopted by the SVS in their statement regarding clinical practice guidelines for thoracic BAI management.
| Injury Grade | Description |
|---|---|
| I | Intimal tear |
| II | Intramural hematoma |
| III | Pseudoaneurysm |
| IV | Rupture |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here