Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Modern sunscreens were first developed during the 1940s with the primary aim of preventing sunburn. Today, sunscreens are also used to prevent other aspects of photodamage, including carcinogenesis and photoaging
The active ingredients in sunscreens include: (1) soluble organic filters, i.e. “chemical” compounds, that absorb UV radiation; and (2) insoluble metal oxide filters, i.e. “physical” agents, that both absorb and scatter UV radiation
The ability of sunscreens to prevent actinic keratoses and cutaneous squamous cell carcinoma has been demonstrated in human studies, and there is growing evidence that they can also prevent photoaging and cutaneous melanoma
Side effects from sunscreens include irritation (which is common) and allergy (which is rare)
Photoprotection and sun avoidance may lead to a decrease in vitamin D levels in some individuals; optimum vitamin D levels can be achieved with a balanced diet and oral supplementation
Evidence exists that even ancient civilizations sought protection from the damaging effects of the sun. The concept of excessive sun exposure as unhealthy or at least cosmetically unacceptable eventually spread to Northern Europe, especially among the aristocracy, and continued into the nineteenth century, until the period of the Industrial Revolution. In 1890, Palm noted that children in urban slums of England had rickets whereas those in similar housing conditions in sunny India did not, and linked the lack of sun to the development of rickets . Physicians then began to treat such children with artificial UV radiation. At the same time, Finsen began to treat a number of other diseases, including cutaneous tuberculosis and, interestingly, lupus erythematosus, with artificial light; he was awarded the Nobel Prize for this work in 1903. At the turn of the twentieth century, the image of tanned skin as aesthetically pleasing began to emerge. The French fashion designer Coco Chanel is often credited with developing the concept of tanned skin as beautiful that still persists today. Indeed for decades consumers applied various oils to the skin and used metal reflectors to enhance tanning. However, the twentieth century also saw the recognition of sunburn as an effect of UVB, the discovery of UVB-specific DNA mutations, and establishment of UVB as a complete carcinogen.
During the 1990s, dermatologists and other healthcare professionals made serious efforts to educate consumers regarding the damaging effects of excessive sun exposure as well as exposure to artificial tanning devices. Despite these efforts, recent studies show that while understanding of the adverse effects of exposure has increased, there has been very little positive change in behavior. This is especially true for adolescents, who continue to suffer sunburns and who continue to seek tans through natural and artificial exposure. Over 20 million Americans use tanning salons every year and sunscreen usage has actually been declining in some studies .
Sunscreens attenuate UV wavelengths which would otherwise interact with molecules in the skin (chromophores) to generate photochemical reactions, leading to photobiologic change. These preparations are usually combinations of active filters that absorb or scatter incident radiation, and they are dispersed in a variety of topical vehicles. The active ingredients are not meant to be absorbed in any large quantity into the living epidermis, dermis, or general circulation.
In 1928, the first modern sunscreen, a product containing benzyl salicylate and benzyl cinnamate, was introduced in the US. However, widespread use of sunscreens did not occur until World War II, when the US military provided preparations containing red petrolatum to prevent sunburn in troops stationed in sunny areas. After the war, there was an increased interest in UV-absorbing chemicals, and para-aminobenzoic acid (PABA), patented in 1943, was the first of these to gain widespread usage. It was not until the mid-1970s that sunscreens gained a degree of consumer acceptance.
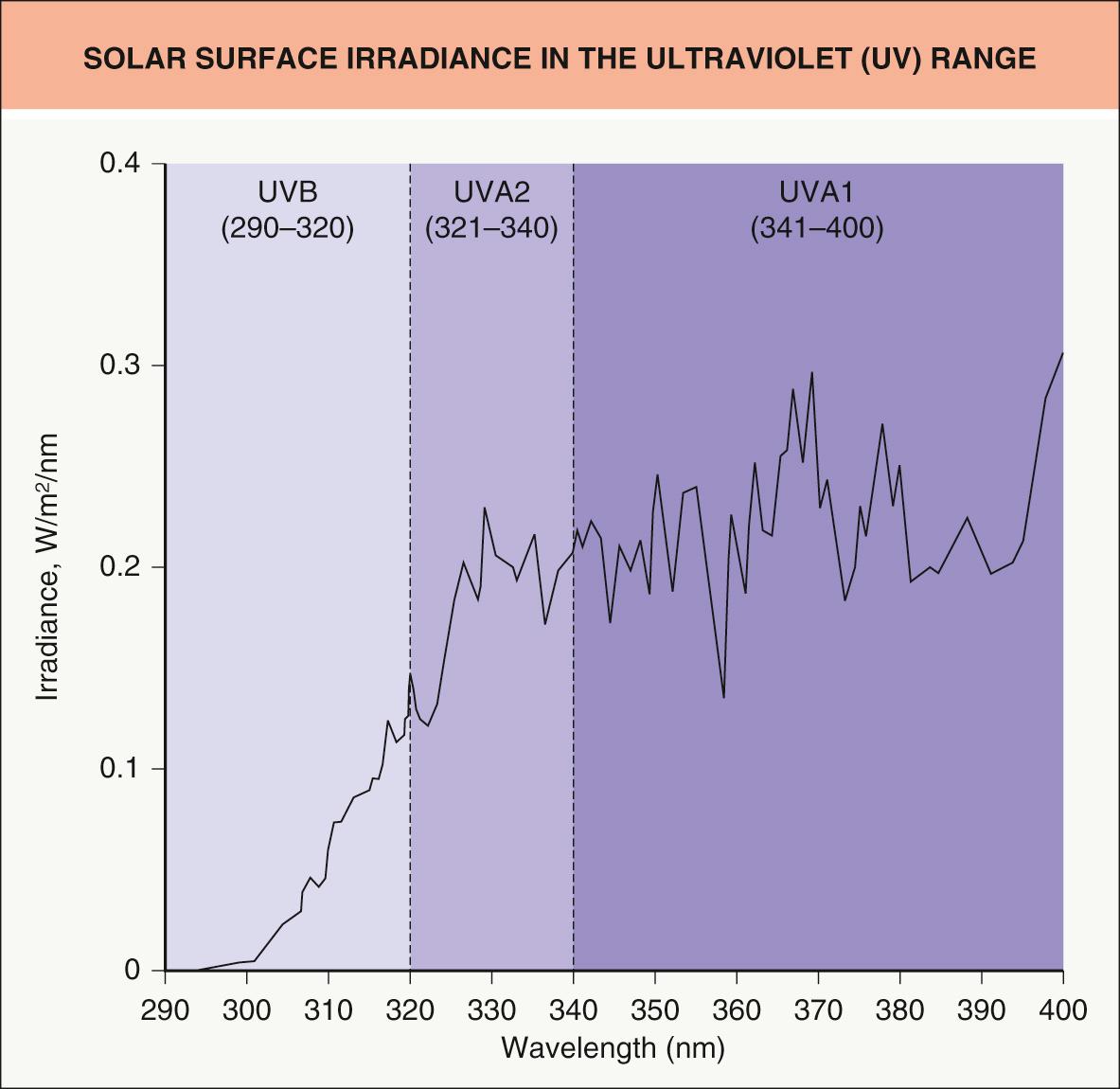
While solar radiation reaching the surface of the earth and human skin includes ultraviolet, visible, and infrared rays (see Fig. 134.1 ), the initial sunscreens were formulated primarily to prevent sunburn and therefore contained primarily UVB-absorbing agents ( Fig. 132.1 ). As scientific evidence grew documenting the deleterious effects of longer-wavelength UVA radiation and as manufacturers strove to develop higher levels of protection against burning, agents with absorption in the longer wavelengths (e.g. benzophenones) were combined with UVB absorbers. The inorganic sunscreens (e.g. titanium dioxide) were used from the early days of sunscreen manufacturing, but, because of large particle size, were not cosmetically acceptable in high concentrations. Newer technology allowed for micronization of such particles, with an increase in transparency and therefore greater cosmetic acceptability. More recently, chemical agents with greater UVA absorption have been introduced (see below) and are gaining in popularity.
Today, a wide array of sunscreen-containing preparations, including foundations, moisturizers and lipsticks, are commercially available. Another improvement in sunscreen products has been the development of agents with increased substantivity. Such agents, usually marketed as “sports” products, have bases which persist on the stratum corneum for longer than do the usual bases, allowing for vigorous outdoor activity, sweating, and even swimming with retained photoprotection.
Since sunscreens alter the structure or function of skin, they are considered over-the-counter drugs in the US and are regulated by the Food and Drug Administration (FDA). Regulations include the maximum concentration of active ingredients, safety labeling, and the methodology for determining the sun protection factor (SPF).
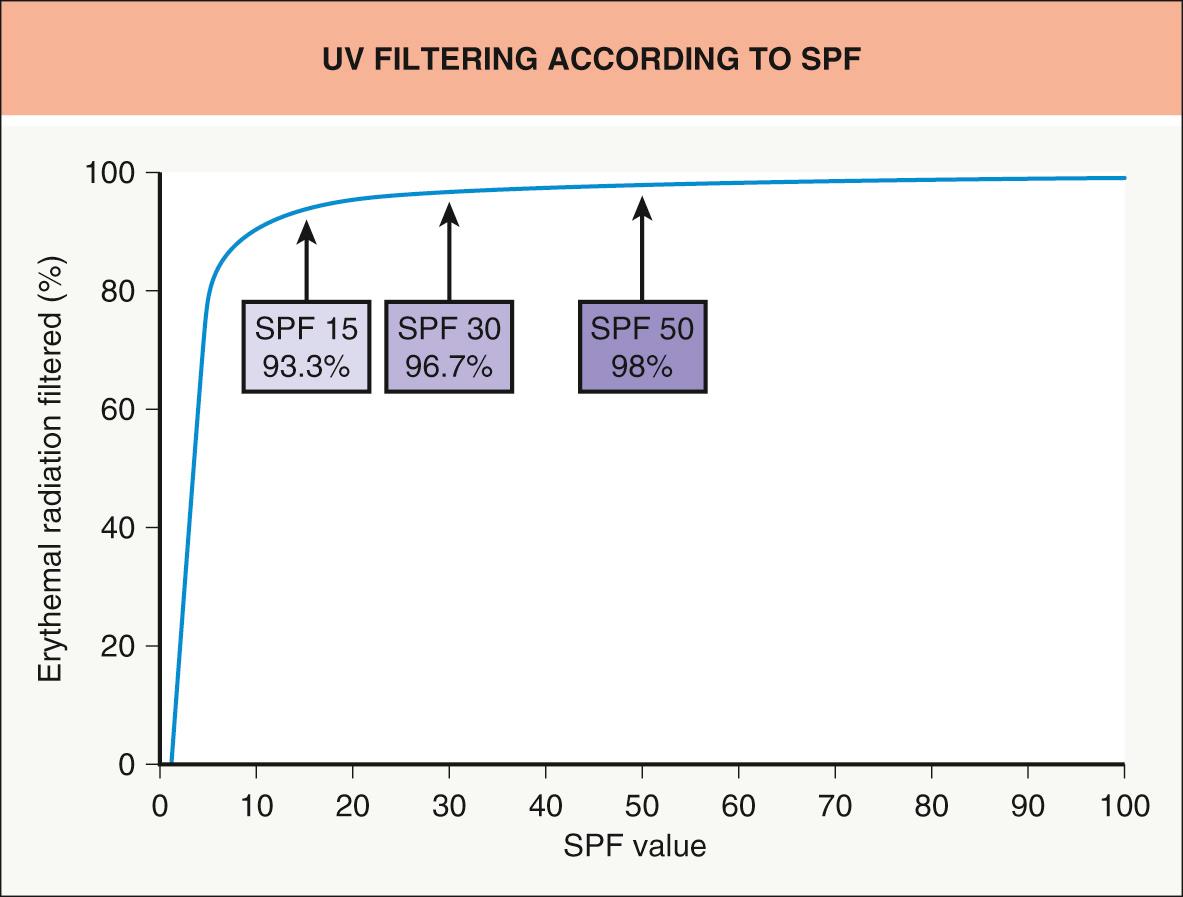
Sunscreen efficacy is assessed based upon its sun protection factor (SPF), which is determined by cutaneous phototesting utilizing artificial sunlight ( Table 132.1 ). If properly tested and applied, a product with an SPF of 15 would allow 15 times as much time in the sun with the same resultant level of erythema as without the product in a given individual (by attenuating 93.3% of the UV reaching the skin). Similarly, an SPF 50 product would allow 50 times as much exposure with the same result (via 98% UV blockage). Since the SPF is calculated as a ratio, the theoretical percentage of UV filtering basically levels off at SPF 30 and above ( Fig. 132.2 ).
| DETERMINATION OF THE SUN PROTECTION FACTOR (SPF) |
|
Since scientists became aware of the damage that can be caused by photons longer than 315–320 nm, many attempts have been made to develop a standard method of testing for protecting against long-wave or UVA radiation. Because there is no easily reproducible cutaneous biologic endpoint that is relatively specific for UVA radiation (as erythema is for UVB), photobiologists have suggested a number of assays, including both in vivo and in vitro methodologies.
The most commonly utilized human-skin-based assays include immediate pigment darkening (IPD) and persistent pigment darkening (PPD), each with endpoints that measure a different aspect of the skin's tanning response to UVA radiation. The former, as discussed in Chapter 86 , is dependent upon radiation-induced darkening of pre-existing melanin, while the latter is a more complex response which involves de novo synthesis and transfer of melanin. Both endpoints can be produced with relatively small amounts of UVA radiation, making them reasonable for human phototesting. In Japan, the assay for PPD is favored .
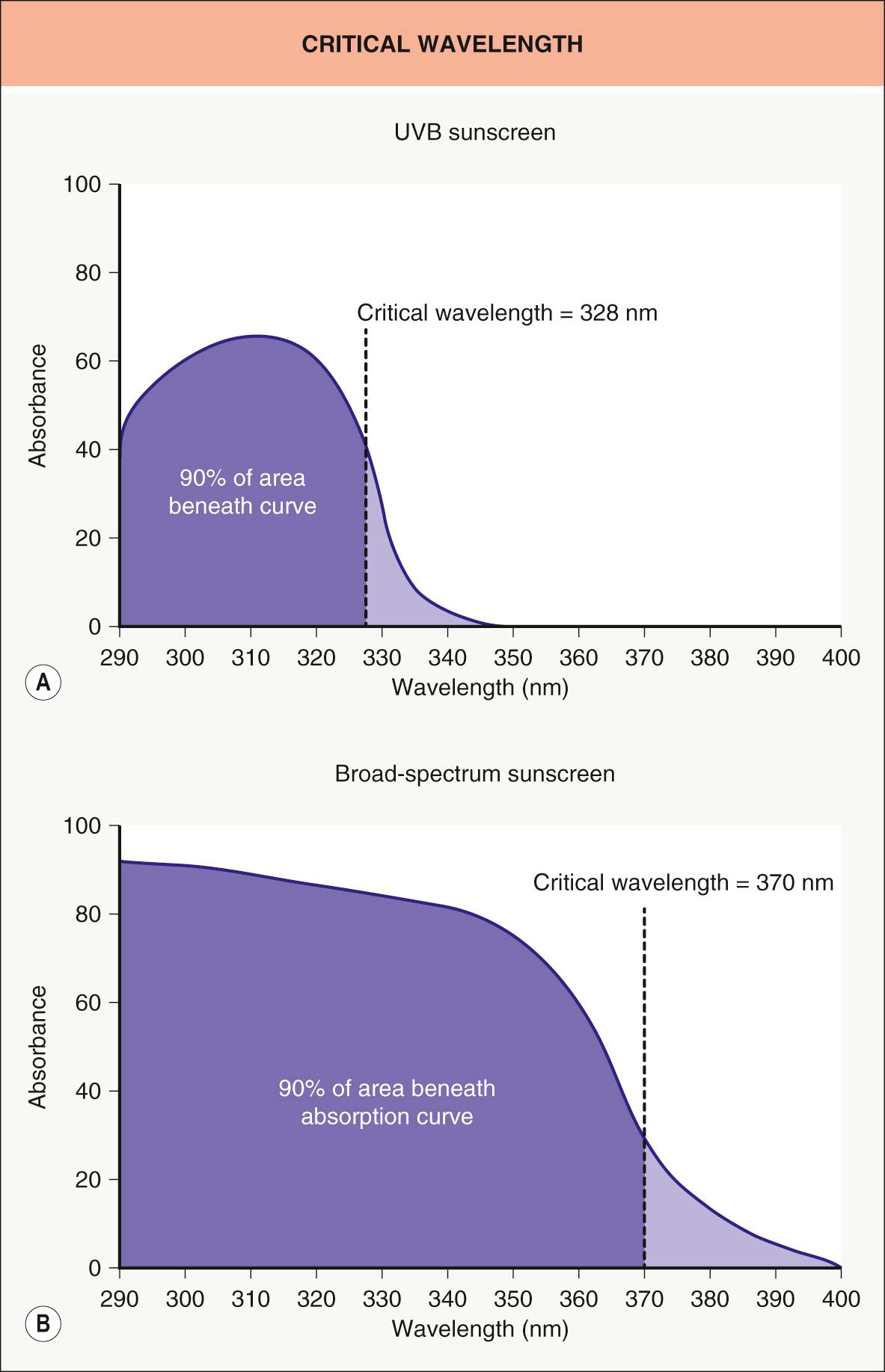
The most commonly accepted in vitro method for assessing UVA protection is the critical wavelength (CW) determination, which is utilized in the European Union, Australia, and the US. This is a relatively simple assay in which the sunscreen is dissolved in solvent and the absorption spectrum is determined by a spectrophotometer. The CW for the product represents the wavelength at which the cumulative absorption of solar-simulated radiation above 290 nm is 90% ( Fig. 132.3 ).
In 1999, the FDA published sunscreen regulations that included: (1) limiting the maximum allowable SPF value on the label; and (2) changing the terminology for rating substantivity. As a result, manufacturers had to limit their labeling and marketing of efficacy claims regarding protection from sunburn. Methods for testing and labeling UVA protection were not addressed.
Based upon additional input from industry, dermatologists and photobiologists, the FDA published its Final Rule for “Labeling and Effectiveness Testing: Sunscreen Drug Products for Over-the-Counter Human Use” in 2011 ( Table 132.2 ). The major label on each product consists of the SPF value and, if efficacy in the UVA range is deemed sufficient, the term “Broad Spectrum”. UVA testing is limited to an in vitro CW determination (see Fig. 132.3 ), with a CW ≥370 nm meriting the broad spectrum designation. Substantivity ratings are limited to “Water Resistant (40)” and “Water Resistant (80)”, with the number reflecting minutes. The maximum SPF is capped at 50+, although the FDA will consider the use of higher SPF threshold values in the future if there are new data to support the increased clinical efficacy of such formulations.
| FDA LABELING OF SUNSCREENS – ULTRAVIOLET B (UVB) AND ULTRAVIOLET A (UVA) PROTECTION (2011) |
| Ultraviolet B (UVB) protection |
|
| Ultraviolet A (UVA) protection |
|
| Substantivity |
|
| Additional labeling |
|
The manufacture and regulation of sunscreens varies throughout the world ( Table 132.3 ). While the methodology for measuring SPF (which predominantly represents UVB protection) is nearly universal, there are multiple methods for determining UVA protection. In many countries, in vivo measurement of the UVA protection factor (UVA-PF), based on the biologic phenomenon of persistent pigment darkening (PPD), has been replaced by in vitro methodologies such as the CW (see Fig. 132.3 ). While specific regulations will undoubtedly continue to evolve, one issue in the US has been the limited number of active sunscreen ingredients approved by the FDA.
| COMPARISON OF TESTING AND LABELING OF SUNSCREENS GLOBALLY | |||||
|---|---|---|---|---|---|
| US | Canada | European Union | Australia, New Zealand | Japan | |
| Regulatory category | Drugs | Drug | Cosmetics | Drugs | Cosmetics |
| No. of allowed ingredients | 16 | 20 | 28 | 29 | 32 |
| Ultraviolet B | |||||
| Efficacy testing | SPF | SPF | SPF | SPF | SPF |
| Label descriptors | None | Medium to very high | Low to high | Low to very high | None |
| Highest SPF allowed | 50+ | 50+ | 50+ | 50+ | 50+ |
| Ultraviolet A | |||||
| Efficacy testing | CW ≥370 nm |
|
|
|
|
| Label descriptors | Broad spectrum | Broad spectrum | UVA seal | Broad spectrum | PA+ (2–4), PA++ (4–8), or PA+++ (>8) |
When applied properly, sunscreens form a film or coating on the surface of the stratum corneum that attenuates or “filters out” radiation that would otherwise reach the living epidermis and dermis. The active agents in sunscreen products do this by either absorbing or scattering photons before they penetrate the skin surface. Nowadays, many sunscreen products contain combinations of agents that work by one or both mechanisms. Active sunscreen ingredients have conventionally been divided into so-called organic “chemical” agents which are water- or oil-soluble and function through photon absorption versus inorganic metal oxide “physical” agents which are insoluble and scatter light energy. However, there are limitations and exceptions to this seemingly clear-cut distinction.
When organic sunscreen agents absorb radiation, they are initially raised to a higher-energy, excited molecular state. Upon reverting to their ground state, this stored energy is usually dissipated in the form of heat, which is barely perceptible and non-damaging to the skin. However, some sunscreen agents, once excited, can undergo photochemical reactions with other sunscreen components (e.g. ecamsule), resulting in loss of their photoprotective efficacy. As a result, such agents (particularly avobenzone) lack photostability, i.e. they are deemed to be photolabile. In order to preserve efficacy, sunscreen agents such as avobenzone are combined with photostabilizers (e.g. salicylates, octocrylene).
In the past, opaque topical sun protection products consisting of insoluble metal oxide filters were considered “sunblockers”, but since 1999 the FDA has not considered the concept of achieving complete blockage via topical photoprotection to be valid. Whereas absorption of solar radiation is directly related to chemical structure , optical physical scattering (and reflection) by inorganic agents is based primarily on particle size . Because sunscreens formulated with large-particle inorganic agents are cosmetically unacceptable due to their opaque appearance, particles are now micronized and the resultant products are significantly less visible. However, there is evidence that within the UV spectrum these inorganic agents such as particulate titanium dioxide, especially when micronized, also exhibit significant direct photon absorption that may be more important than their photon scattering effects .
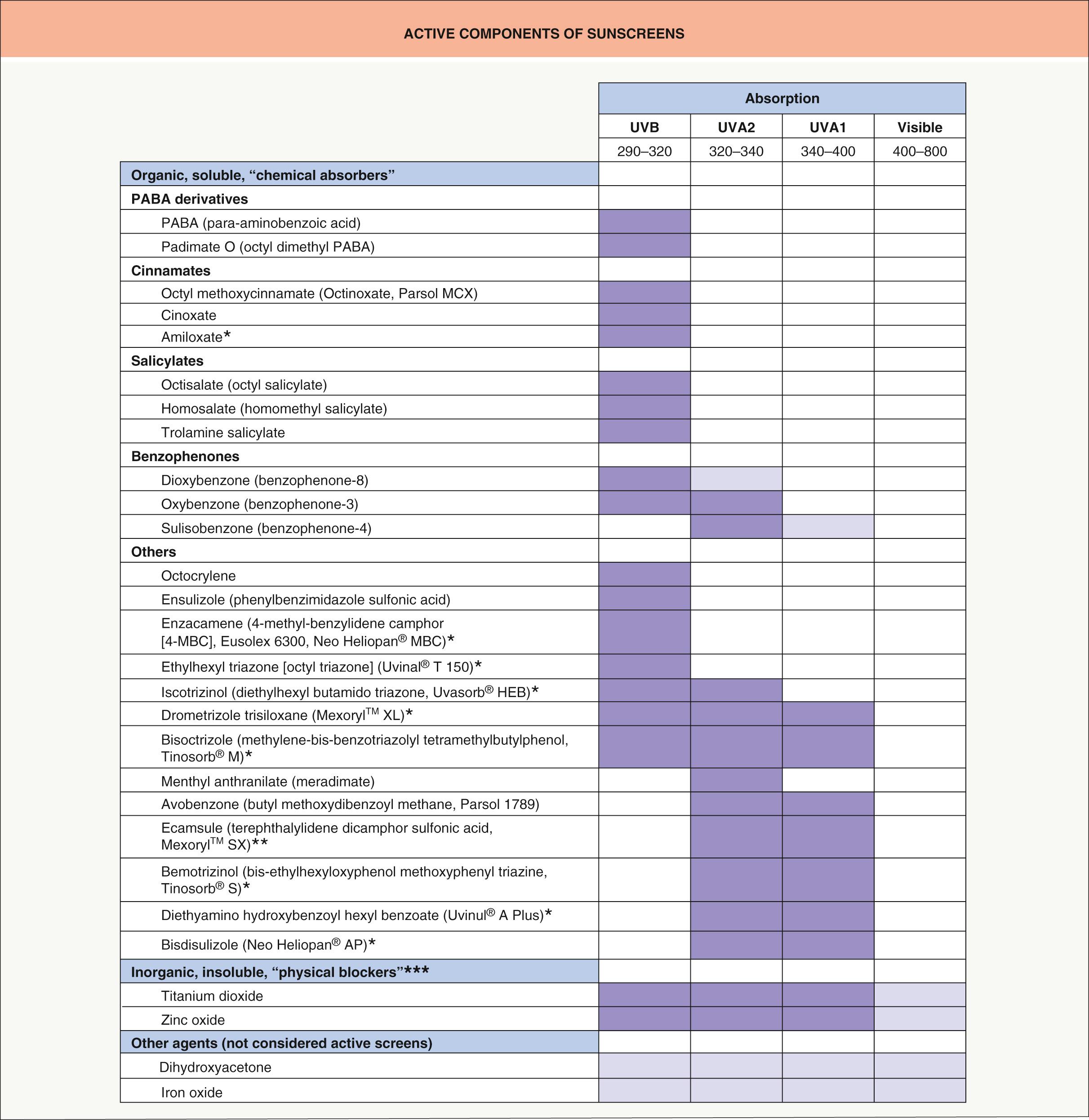
Currently, most sunscreens contain a combination of two or more agents in order to achieve broad-spectrum protection. Fig. 132.4 lists, according to type of activity and spectrum of protection, the active agents currently used in sunscreens .
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here