Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Submucosal tumors (SMTs) of the esophagus and gastroesophageal junction (GEJ) are a group of rare, benign lesions. This chapter will review the various types of esophageal and GEJ SMTs. We intend to focus on advancements in the imaging modalities, medical treatment, and surgical options for these tumors, including descriptions of evolving surgical techniques.
Our understanding of the prevalence of esophageal SMTs comes largely from autopsy studies from the mid-20th century, which identified an overall prevalence of about 0.5% for benign esophageal tumors and cysts. Only 18% of all esophageal tumors were benign; the remaining 82% were malignant. Benign esophageal and GEJ SMTs are found in people of all ages from infancy to elderly, with the majority presenting in the fourth through sixth decades, and with male predominance.
The development of symptoms related to esophageal SMTs is largely correlated to the size of the tumor. Presenting symptoms may include dysphagia and regurgitation due to intraluminal obstruction, pain, pulmonary symptoms due to extramural mass effect on the airways, or bleeding. Physical examination is usually unrevealing unless the tumor is quite large; therefore, discovery of these tumors is often incidental. About 50% of esophageal SMTs are asymptomatic, and identified on endoscopy or imaging obtained for other reasons. A chest x-ray may show a mediastinal prominence in the case of a large tumor. Computed tomography (CT) of the chest can provide a clearer depiction of a small lesion, but findings may be subtle, suggesting esophageal wall thickening only. A contrast esophagram usually shows a smooth-walled indentation on the column of contrast. The differential diagnosis for SMTs is broad, including benign solid tumor, cyst, vascular anomaly, or cancer. As advancements in imaging technology have developed, confidence in determining the benign nature of these lesions without tissue for diagnosis has grown. However, in some cases, multiple imaging modalities may still not definitively rule out malignancy. Standard endoscopic forceps biopsy or endoscopic ultrasound (EUS)-guided fine-needle aspiration (FNA) may be inadequate or indeterminate due to the submucosal location of the tumor, and whether biopsy is advisable. For large or symptomatic SMTs, surgical resection is generally recommended. However, surveillance versus surgical resection may be considered for small, asymptomatic tumors.
Due to the overall rare incidence of SMTs, evidence-based recommendations are mostly limited to findings from a few small series. The exception to this is leiomyoma, which is relatively frequent. Observation has been generally recommended for asymptomatic lesions less than 3 cm in diameter. However, concerns for patient compliance, the cost of repeat surveillance, the potential for delayed diagnosis of malignancy, and patient anxiety may lead physicians to recommend resection of small esophageal SMTs.
Treatment options for esophageal SMTs have evolved in recent years. Targeted molecular therapy has greatly improved outcomes for gastrointestinal stromal tumors (GIST), and recommendations for optimizing medical therapy are evolving. Observation and surveillance of small and asymptomatic esophageal SMTs may be possible now with better diagnostic techniques in cases of well defined, small, asymptomatic, benign esophageal lesions with low potential for malignancy. Historically, surgical resection was recommended for esophageal or GEJ SMTs, for both treatment and definitive diagnosis. Open laparotomy or thoracotomy for surgical enucleation or esophagectomy was recommended. Minimally invasive techniques including laparoscopy, video-assisted thoracoscopic surgery (VATS), and robotic surgical techniques have gained acceptance with improved outcomes in postoperative pain, pulmonary complications, and length of stay. More recently, purely endoscopic techniques for resection of select esophageal SMTs have been shown to be feasible and safe when performed by experienced endoscopists.
Surgical resection is the mainstay of treatment for leiomyoma. Indications for surgical resection include symptomatic lesions, inability to rule out malignancy or distinguish from GIST, atypical imaging findings, overlying mucosal erosion or dysplastic changes, regional lymphadenopathy, large tumors (size recommendation is variable), and tumor growth during surveillance.
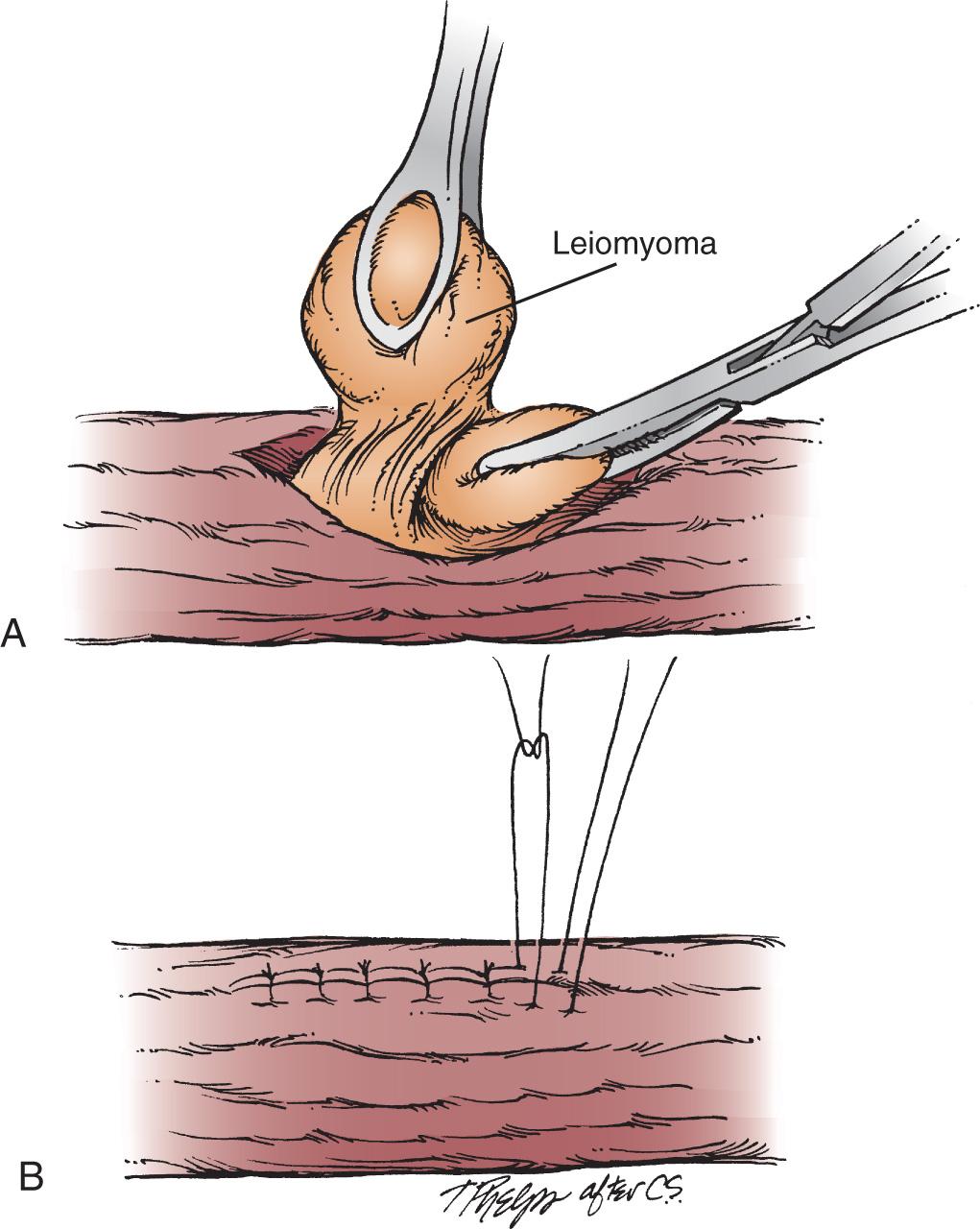
Traditionally SMTs, particularly leiomyomas, were resected by either enucleation via right or left thoracotomy, or laparotomy for GEJ tumors. With open access to the esophagus or GEJ, the surgeon can visualize and palpate the tumor to precisely localize it. The overlying pleura is incised, and the longitudinal and circular muscle split. The tumor is dissected from the surrounding muscle, submucosa, and enucleated from the underlying mucosa. Any mucosal injuries (incidence reported at 7% in open cases) are closed primarily, and the overlying muscle layers and pleura are generally closed ( Fig. 46.1 ). The safety and efficacy of the open approach has been well documented. Mortality of thoracotomy with enucleation is less than 1.3%, and around 90% of patients were reported to be symptom free after 5 years. Esophagectomy may be indicated for tumors greater than 8 cm or diffuse leiomyomatosis. Esophagectomy historically has been required for about 5% to 10% of leiomyoma patients.
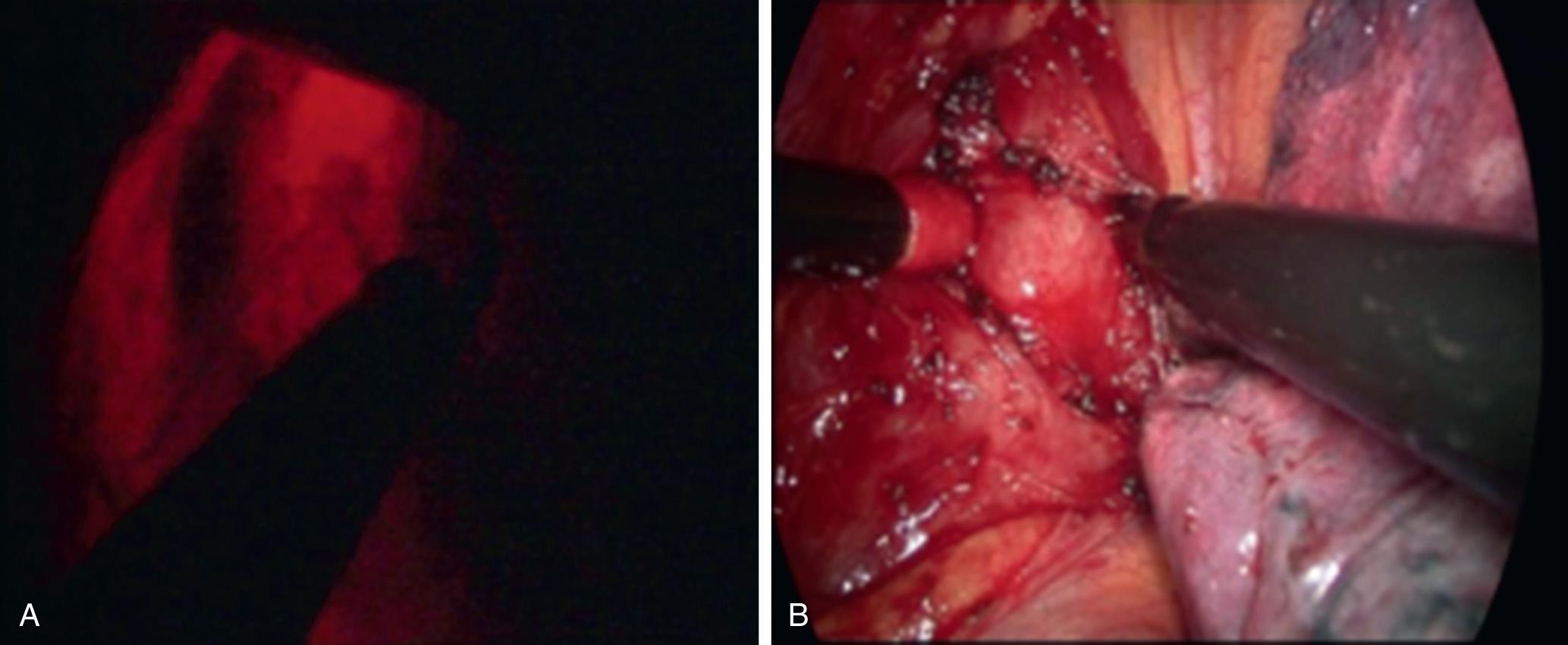
Laparoscopic and video-assisted thoracoscopic resection is now widely accepted and found to be safe in case series. Robotic minimally invasive surgery is also a safe and feasible option. The length of stay for minimally invasive enucleation is about half that expected for open thoracic surgery. Laparoscopic enucleation guided by intraoperative frozen section has been reported for tumors that displayed features concerning for GIST. However, frozen section has been shown to be unreliable for differentiating GIST from leiomyoma, as immunohistochemistry is required to confirm the diagnosis. The thoracoscopic approach may be required for more proximal lesions, and the esophagus is usually accessed through the right chest. Compared to thoracotomy, VATS reduces pulmonary complications, postoperative pain, and length of stay. Intraoperative endoscopy is beneficial for the identification of small tumors and testing mucosal integrity after enucleation ( Fig. 46.2 ). A few cases of robotic enucleation for esophageal leiomyoma have been reported. Surgeons who have used this technique suggest that the benefits include improved dexterity, precision, and visibility, allowing minimized esophageal mobilization and avoidance of mucosal injury. Up-front cost, the requirement of a bedside surgeon, and docking time are concerns that must be individually weighed against the potential benefits of this approach.
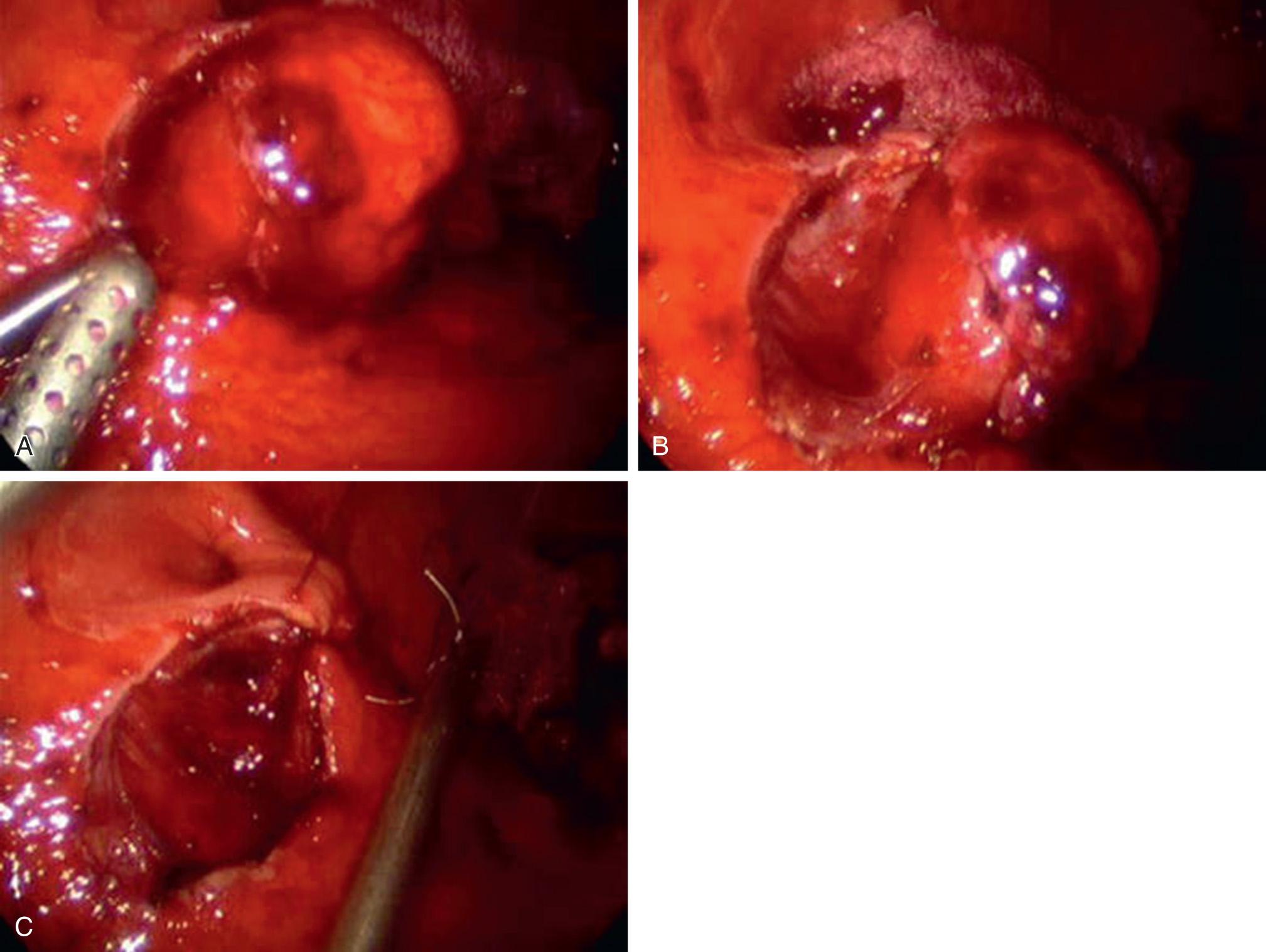
Minimally invasive tumor localization can be improved with intraoperative endoscopy for specific localization of small lesions, and intraluminal balloons or bougies to help optimize the position of the lesion for surgical resection. Laparoscopic transgastric or intragastric surgery has been reported for difficult-to-reach leiomyomas that are posteriorly located at the GEJ ( Fig. 46.3 ). With this technique, trocars are placed through the abdominal wall and then directly into the lumen of the stomach. Capnogastrum is achieved with or without the aid of a coda balloon to occlude the gastric outlet. This approach may help to avoid excess resection and minimize the disruption of the angle of His at the GEJ, and has been successful for tumors as large as 7 cm. Enucleation may be possible with combined endoscopic and laparoscopic instruments, but full-thickness resection is performed if enucleation with clear margins is not possible. The specimen can be removed via a specimen bag through a trocar site or transorally with the endoscope via a grasper or snare. The gastrotomy incisions are primarily closed with suture, but large defects from resection may be closed with an endoscopic stapler. If resection of a portion of the cardia is required and will leave a significant deformity of the GEJ, fundoplication should be considered to prevent postoperative reflux. The development of subsequent worsened or new onset gastroesophageal reflux disease (GERD) after resection of GEJ lesions or enucleation of midesophageal lesions has been reported.
Endoscopic resection has been used more frequently in the last several years. Originally endoscopic resection techniques were used for the curative resection of well-differentiated squamous cell carcinoma of the esophagus. Later, the technique was used for Barrett esophagus with high-grade dysplasia or intramucosal adenocarcinoma. The endoscopic submucosal dissection (ESD) technique is commonly used for tunneling in peroral endoscopic myotomy (POEM), a natural orifice transluminal endoscopic surgery (NOTES) procedure for the treatment of achalasia. Endoscopic resection has grown in popularity and indications have expanded as more surgeons and gastroenterologists gain experience with ESD and resection techniques. The particular approach depends upon the characteristics of the tumor and the experience of the endoscopist. A disadvantage of endoscopic resection is the learning curve, though it is reported to be relatively short for experienced endoscopists.
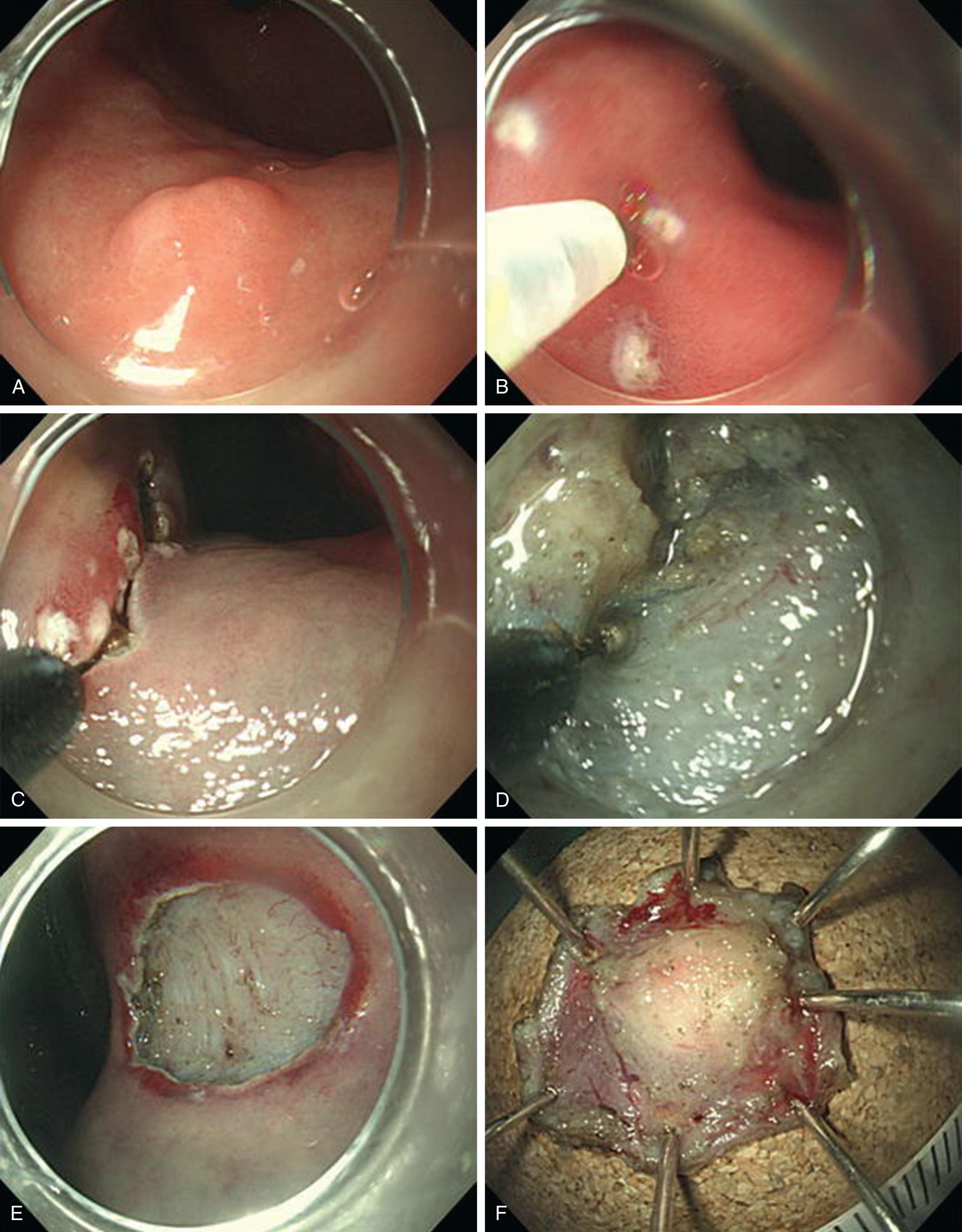
Endoscopic mucosal resection (EMR; Fig. 46.4 ) may be used for small, superficial tumors (<2 cm, superficial to submucosa). EMR separates the full thickness of mucosa from the underlying muscle layers. With this technique, a lifting solution is injected into the submucosal layer beneath the tumor to create a pseudopolyp, which can then be banded and snared to complete the resection.
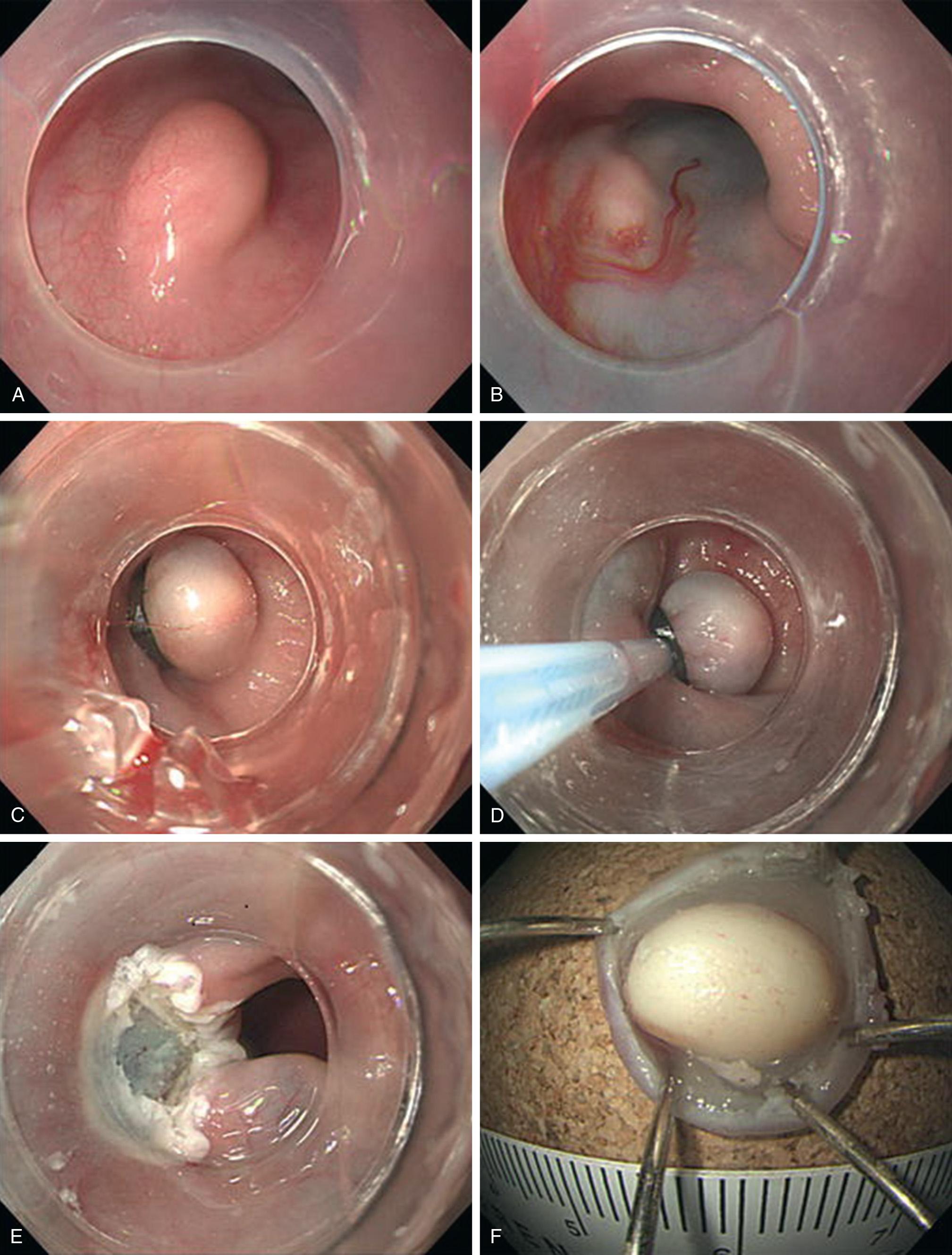
Endoscopy is performed with a high-definition single- or double-channel endoscope. A 23- or 25-gauge endoscopic needle is used to infiltrate the submucosa deep to the lesion with dilute indigo carmine or methylene blue solution, with or without epinephrine, to lift the lesion and expand and highlight the plane between it and the adjacent layers. To aid visualization in the limited working space of the esophagus, the injection is begun distal to the lesion and then sequential injections are placed proximally. The lesion should lift on the cushion of infiltrated submucosa in order to allow for en bloc resection. Failure to lift indicates a misplaced tip of the needle, most commonly too deep into the muscle, which can be adjusted by slowly withdrawing while gently injecting. If adjusting the needle fails to create an adequate lift, this suggests infiltration or scarring of the tumor into the deeper muscle layers. A simple cautery snare can be used for a well-lifted small lesion, but usually other techniques may be required to avoid piecemeal resection for an adherent tumor.
Cap-assisted EMR can be used with a single-channel endoscope with the cap in place. The targeted lesion then can be fully suctioned into the cap prior to snaring it. For more precise resection, a variceal band can be applied to ligate and position the lesion as a pseudopolyp so that the snare can be placed under direct vision. As an alternative to the cap-assisted technique, an endoscopic grasper can be used to lift the lesion for snaring, but this requires a dual-channel endoscope. The resection bed is then checked for bleeding or perforation. The specimen can be sucked into the cap and retrieved with the scope, or with grasping forceps or a retrieval net.
ESD ( Fig. 46.5 ), though currently performed most frequently in Asia, endoscopists worldwide have begun to accept and use this technique. ESD is indicated for larger, deeper tumors. ESD variations include endoscopic submucosal tunneling or, in select cases, even full-thickness resection. ESD is indicated for tumors larger than 2 cm or involving layers deep into the mucosa, which cannot be captured en bloc by the limited dimensions of the EMR cap. Feasible size ranges from 2 to 8 cm depending on author, though greater than 5 cm usually requires piecemeal resection or at least dividing the lesion to allow extraction. ESD involves a customized incision through the normal mucosa and dissection beneath and around the lesion through the submucosal plane. The dissection is performed with one of several varieties of endoscopic electrosurgical knives.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here