Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The essential role of the spleen in the defense against bacterial organisms is well documented. King and Schumacker first described the susceptibility of splenectomized infants to infection in 1952. The immunologic role of the spleen led pediatric surgeons to initiate a nonoperative approach to splenic injuries in children, which has evolved into the preferred method for treating children and also adults. In the modern era, splenic surgery is primarily centered in the management of hematologic disorders. The most significant change in management over the past 25 years has been the introduction of laparoscopic splenectomy in adults by Delaitre and Maignien and subsequently in children by Tulman and Holcomb.
The splenic primordium develops as a mesenchymal bulge in the dorsal mesogastrium between the stomach and the pancreas, initially observed at the 8- to 10-mm embryo stage. A true epithelium is noted at the 10- to 12-mm stage as the splenic sinusoids begin to communicate with capillaries. The spleen produces white and red cells by the fourth month of fetal life, although this function ceases later in gestation. The anatomic arrangement of the spleen is consistent with the various functions of the spleen. The splenic artery branches into segmental vessels, which further branch into trabecular arteries. After further bifurcations, small arteries enter the white pulp, which is composed of lymphocytes and macrophages arranged as a germinal center around the central artery. The central artery delivers particulate material into the white pulp, an arrangement that facilitates antibody formation in response to antigens. The red pulp consists of the endothelial cords of Billroth, which receive the blood after it passes through the white pulp. The red pulp destroys old and defective cells. The spleen also removes Howell–Jolly bodies (nuclear remnants), Heinz bodies (denatured hemoglobin), and Pappenheimer bodies (iron granules). These particles are noted on peripheral smear after splenectomy. The immune response occurs in the white pulp as antigens come in contact with macrophages and helper T-cells. T-cells initiate cytokine synthesis, and activated T-cells circulate to modulate the response. A humoral response occurs as macrophages and helper T-cells come in contact with antigens.
Splenic function also involves removal of particulate matter as well as production of nonspecific opsonins, which further activate the complement system. In addition, the spleen serves as a biologic filter. If little antibody is available for opsonization of bacteria, the spleen assumes a greater role. This may be a factor in the age-related differences in postsplenectomy infections in young children who lack an adequate antibody response. The spleen also serves as a reservoir for platelets and factor VIII.
Asplenia is often noted with complex congenital heart disease as well as bilateral “right-sidedness” such as bilateral three-lobed lungs, a right-sided stomach, and a central liver (Ivemark syndrome). Intestinal malrotation also has been observed with asplenia. These infants are at risk for overwhelming infection and should receive antibiotics for prophylaxis.
Polysplenia usually consists of a cluster of very small splenic masses and is often found with biliary atresia. Other associated conditions include a preduodenal portal vein, situs inversus, malrotation, and cardiac defects. In the instance of heterotaxy, the splenic mass(es) are always on one side of the body and along the greater curvature of the stomach. These children have adequate splenic immune function.
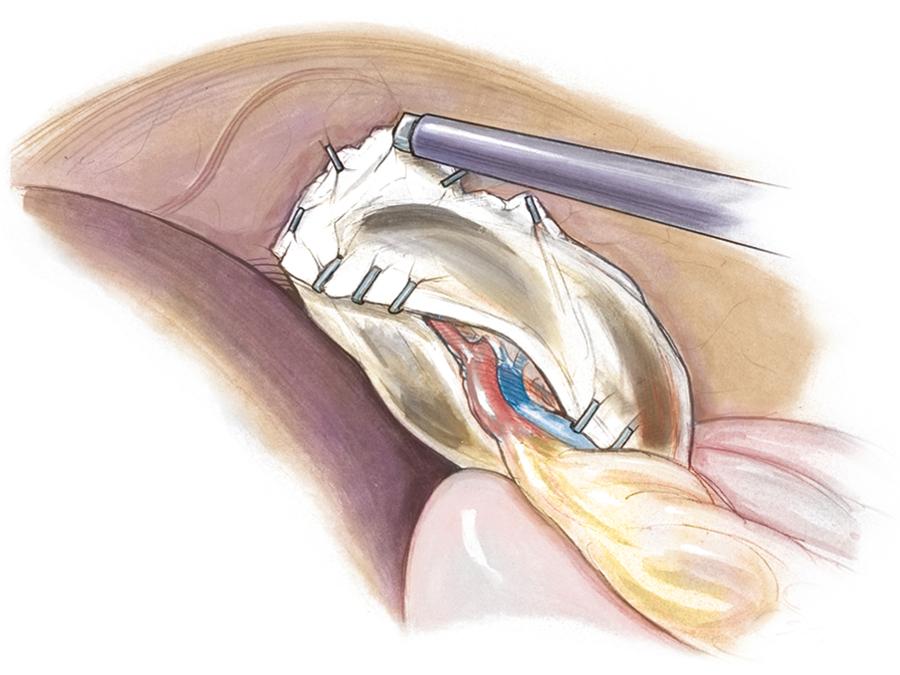
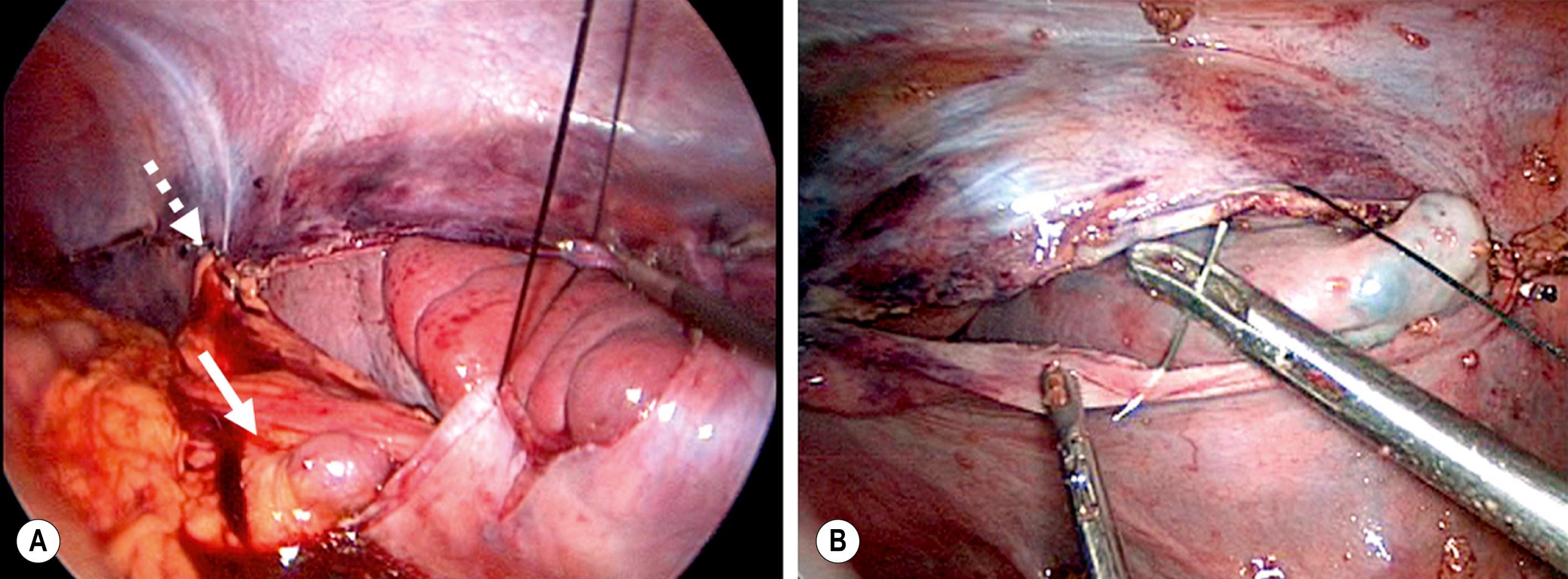
This condition is characterized by a lack of ligamentous attachments to the diaphragm, colon, and retroperitoneum, resulting in a mobile spleen. This is likely due to failure of development of the splenic ligaments from the dorsal mesentery. Children can present with an abdominal mass and episodic pain, but they can also present acutely with torsion and infarction. Recently, two cases of portal/mesenteric varices were reported in adolescent patients, both of which resolved with splenectomy. Pancreatitis has also been noted as a presenting sign. Splenopexy is the preferred method of treatment and can be performed with placement of the spleen into a mesh basket, suture splenopexy, colonic displacement with gastropexy, positioning in an omental basket, or placement in an extraperitoneal pocket. The laparoscopic approach is the preferred technique, and the use of an absorbable or nonabsorbable mesh with fixation in the left upper quadrant is seen in Figure 47.1 . Placement of the spleen in an extraperitoneal pocket is seen in Figure 47.2 . Partial splenectomy and splenopexy also have been described. Torsion with infarction requires splenectomy. Cases of chronic torsion have also been reported with massive splenomegaly that may necessitate splenectomy.
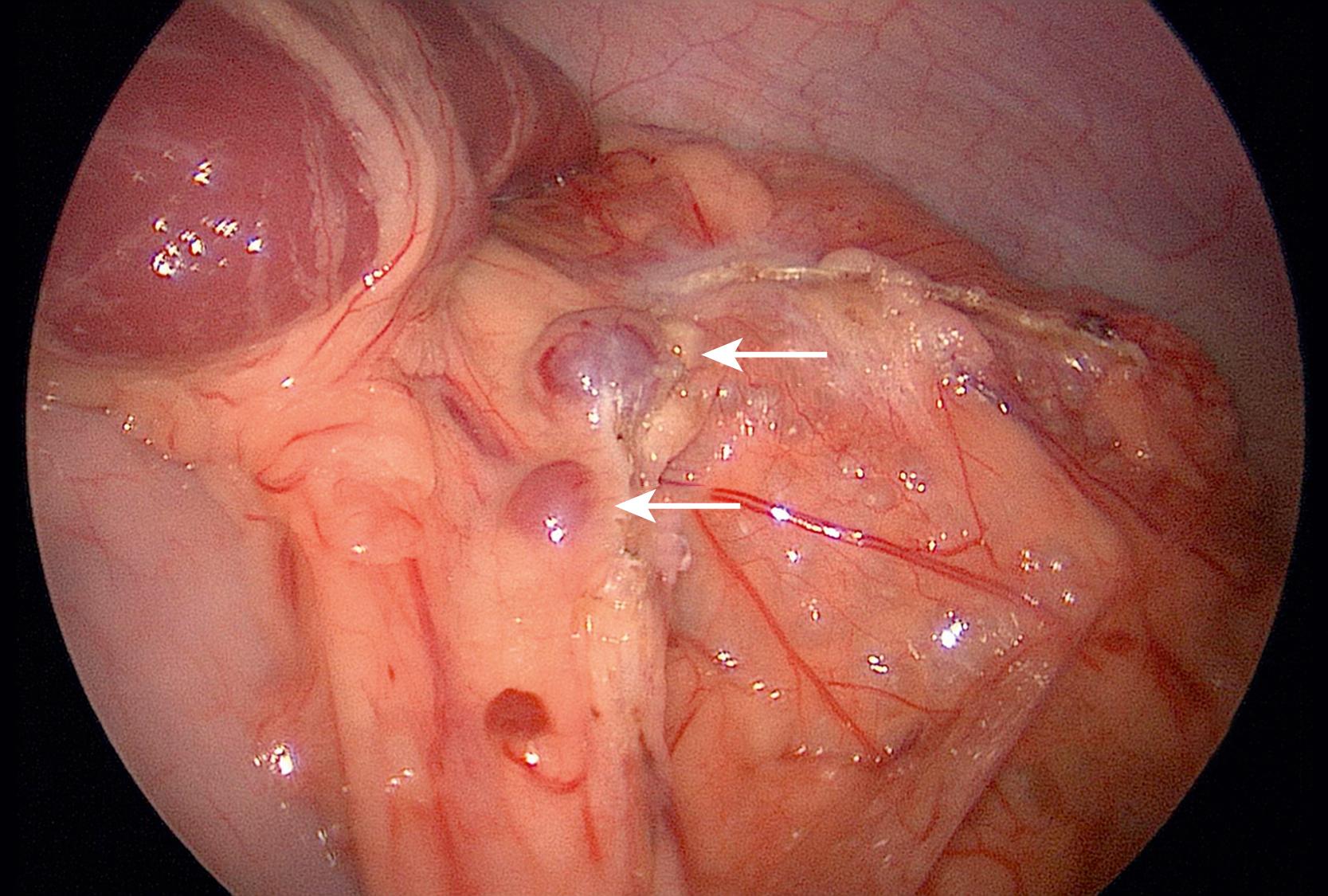
Accessory spleens have been noted in 15–30% of children, with a large series noting a 19% rate. Accessory spleens likely originate from mesenchymal remnants that fail to fuse with the main splenic mass. Most (75%) are located near the splenic hilum ( Fig. 47.3 ). Other locations that should be evaluated at the time of splenectomy include the lesser sac along the splenic vessels, omentum, and retroperitoneum. When found, most (86%) of accessory spleens are single, 11% have two, and 3% have three or more. A missed accessory spleen at the time of planned total splenectomy can lead to recurrence of the primary disease process, which in cases of immune thrombocytopenic purpura (ITP) is early and with hereditary spherocytosis (HS) is later.
This condition in which the left gonad and the spleen are attached is a result of early fusion between the two structures prior to descent of the testis. The remnant can be a continuous band or can be discontinuous with splenic tissue attached to the gonad. A splenic remnant also has been noted in the left scrotum as an accessory splenic remnant type of abnormality.
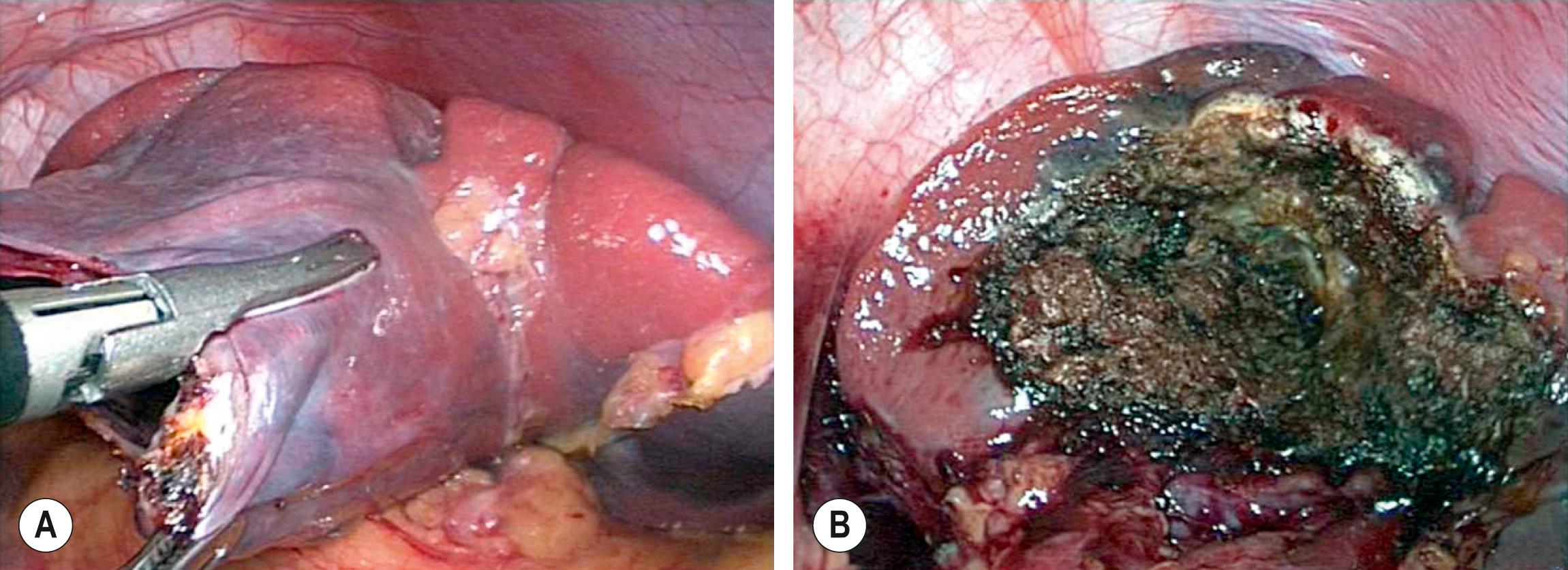
Cysts of the spleen are most frequently primary splenic cysts containing an epithelial lining ( Fig. 47.4 ). They are also referred to as epithelial or epidermoid cysts. Post-traumatic pseudocysts are occasionally seen. Inclusion of surface mesothelium into the splenic parenchyma is the most likely etiology of epithelial cysts. Patients may present with symptoms related to their size with gastric compression or pain, an abdominal mass, rupture, or infection with an abscess. Simple cysts less than 5 cm can be observed, but cysts that are enlarging, symptomatic, or larger than 5 cm usually require treatment. Most symptomatic cysts are larger than 8 cm. Percutaneous aspiration and sclerosis utilizing alcohol or other agents have been reported with variable success. A recent study showed sclerosis with alcohol caused disappearance in 20% of patients. In this study, in patients with symptoms, 67% had symptom resolution with cysts larger than 5 cm.
![Fig. 47.4, (A) A large epithelial splenic cyst (arrow) is seen on the CT scan. (B) At laparoscopy, the large cyst (seen in [A] ) is seen to occupy most of the spleen. Fig. 47.4, (A) A large epithelial splenic cyst (arrow) is seen on the CT scan. (B) At laparoscopy, the large cyst (seen in [A] ) is seen to occupy most of the spleen.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SplenicConditions/3_3s20B978032354940000047X.jpg)
Marsupialization is commonly performed but has been associated with a high recurrence rate if an adequate amount of cyst wall is not removed ( Fig. 47.5 ). In addition, a high recurrence rate with laparoscopic partial excision has also been observed. However, others have had good success with this technique. Some authors recommend partial splenectomy associated with cyst resection. Our group has reported good results with partial splenectomy, emphasizing resection of a margin of normal spleen so that the cut surfaces cannot oppose, which might lead to recurrence. Other techniques involve lining the cyst with Surgicel (Ethicon, Inc., Somerville, NJ) and omentopexy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here