Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Spinal dysraphism is a spectrum of disorders resulting from incomplete formation of the neural tube or abnormal development of structures immediately associated with the neural tube and nearby somites.
Several forms of dysraphism, including myelomeningocele, have been linked to maternal nutritional deficiencies. Folate supplementation for expecting mothers and women of childbearing age has resulted in a dramatic decline in the incidence of open neural tube defects worldwide.
Disorders of primary neurulation result from incomplete fusion of the neural folds and include open neural tube defects (eg, myelomeningocele), spinal lipomas, and dermal sinus tracts. Elements of the caudal spinal cord and sacrum develop during a process called secondary neurulation. Disorders at this stage result in terminal myelocystocele, caudal agenesis, and fatty filum, among other conditions. The split cord malformations are thought to result from aberrant gastrulation.
Myelomeningocele is traditionally treated postnatally via cyst decompression, isolation, and closure of the dura, followed by skin closure. In a select population of fetuses with myelomeningocele, intrauterine repair during gestation weeks 19 to 25 has been shown to reduce the need for hydrocephalus shunting, in addition to a measured improvement in cognitive and motor function.
Occult spinal dysraphism refers to a heterogeneous group of skin-covered anomalies including lipomyelomeningocele, dermal sinus tract, split cord malformation, and fatty filum terminale. Patients often present with tethered cord syndrome, which may include a variety of skin stigmata as well as neurologic, urologic, and orthopedic deficits.
Ultrasonography, CT, and MRI in particular can help define and delineate the type and extent of dysraphism present and assist with preoperative counseling and surgical planning.
The primary goal of surgical management for spinal dysraphism is to prevent future neurologic deterioration or halt existing neurologic decline.
The most common early complication following surgical correction of spinal dysraphism is CSF leak. Spinal cord retethering is the most important late complication, usually presenting during times of axial skeletal growth and requiring surgical detethering.
Most patients with severe forms of spinal dysraphism require lifelong care by a multidisciplinary team including neurosurgeons, urologists, and pediatricians.
Spinal dysraphism represents a spectrum of congenital anomalies resulting from incomplete or aberrant neural tube fusion. If the lesion is confined to the bony posterior arches at one or more levels, it is termed spina bifida. Simple spina bifida of the lower lumbar spine is a common radiologic finding, especially in children, and by itself carries no significance; in contrast, bony spina bifida may accompany any of several complex anomalies involving the spinal cord, nerve roots, dura, and even the pelvic visceral structures. In these cases, spinal dysraphism constitutes a major source of disability among children and adults.
Spinal axis formation begins during the second and third gestational weeks, during a process called primary neurulation, which includes formation of three distinct germ cell layers and the notochord. The neural tube enfolds and fuses first at the occipital region on gestational day 21. Then in a zipper-like fashion the fusion extends rostrally and caudally until complete closure is achieved at the cranial neuropore (day 24) and caudal neuropore (day 26), respectively. Embryologic dysjunction describes the process in which the neural tube separates from the overlying ectoderm. When dysjunction occurs too early (premature dysjunction), mesenchymal cells destined for lipomatous differentiation may migrate into the underlying neural structures, giving rise to a spectrum of conditions including intradural lipoma and lipomyelomeningocele. If dysjunction fails (nondysjunction), an open communication develops between the ectoderm and neuroectoderm. Widespread nondysjunction results in the familiar myelomeningocele, whereas less overt manifestations of nondysjunction include dermal sinus tracts and myelocystocele. Secondary neurulation commences following the closure of the caudal neuropore and includes canalization and retrogressive differentiation. Both canalization and retrogressive differentiation below the upper lumbar spine follow a less defined course than primary neurulation. Thus errors during this stage result in a more heterogeneous group of anomalies, including tethered spinal cord (TSC), terminal myelocystocele, anterior sacral meningocele, and caudal regression syndrome.
Beyond the embryologic categorization of neural tube defects (NTD), spinal dysraphism is also commonly subdivided into two broad categories based on the outward appearance on physical exam: (1) spina bifida cystica , which includes myelomeningocele, is observed as herniation of neural elements through the skin as well as the bony defect and is obvious at birth; and (2) spina bifida occulta , describes as a lesion wherein the underlying neural defect is masked by intact overlying skin. The external signs can be subtle, and symptoms may not develop until late childhood or even adulthood, as the result of spinal cord tethering. Included in the latter group are diastematomyelia, lipomyelomeningocele, tethered filum terminale, anterior sacral meningocele, myelocystocele, and the caudal regression syndromes. Early recognition of these entities is important, because neurologic function may be preserved only by early (prophylactic) and appropriate surgical intervention.
Myelomeningocele is the most common significant birth defect involving the spine, affecting nearly 1 out of every 4000 infants. The etiology is unknown, but evidence exists for both environmental and genetic influences. A role for genetic risk factors is supported by numerous studies documenting familial aggregation of this condition. A strong relationship between folate deficiency and neural tube defects has ushered in public health efforts to supplement dietary folate in women of childbearing age worldwide. This has resulted in a dramatic decline in the incidence of myelomeningocele, though to a lesser degree in the developing world where nutritional deficiencies remain a burden. Since the early 1980s the prevalence of spina bifida in industrialized countries has been further decreasing because of the steadily increasing proportion of affected fetuses that are detected prenatally and electively terminated. As a result of multiple clinical trials demonstrating the protective effect of folate supplementation, the Centers for Disease Control and Prevention (CDC) currently recommends a daily dose of folic acid of 0.4 mg for all women of childbearing age who are capable of becoming pregnant. Still, fortification of the food supply may be a more effective strategy to prevent neural tube defects, rather than individual supplementation. Newer therapies aimed at regeneration of the dorsal arch structures, including nanotechnologies and stem cell therapy, provide further promise though they remain experimental.
As outlined earlier, myelomeningocele is an open neural tube defect that results from faulty disjunction during primary neurulation. The timing of failure between days 26 to 30 correlates with the closure of the caudal neuropore, thereby explaining the predominance of this defect in the lumbosacral spine. Ultimately, the neuroectoderm incompletely separates from the ectoderm, and neural tissue (including nerves, meninges, and cerebrospinal fluid) herniates through the bony spinous defect. The spinal abnormality is part of a more widespread complex of central nervous system abnormalities, which often also include hydrocephalus, gyral anomalies, and the Chiari II malformation of the hindbrain.
Developments in the prenatal diagnosis of fetal anomalies have made antenatal recognition of myelomeningocele commonplace. Families at risk are routinely offered amniocentesis for amniotic alpha-fetoprotein and acetylcholinesterase, which are important in separating open lesions from skin-covered masses, such as myelocystocele. Amniocentesis along with ultrasound screening has a combined accuracy of more than 90%. Prenatal magnetic resonance imaging (MRI), using ultrafast T2-weighted sequences, may also be used to characterize the Chiari II malformation and other associated anomalies. Furthermore, fetal MRI may augment ultrasound by detecting spinal cord abnormalities underlying bony abnormalities. Studies indicate that such prenatal imaging studies can help to determine prognosis. Specifically, lesion level determined by prenatal imaging studies appears to predict neurologic deficit and ambulatory potential, but not the degree of fetal ventriculomegaly or the extent of hindbrain deformity. Families can be professionally counseled regarding the expected prognosis and decisions about abortion or the option of fetal closure.
The majority of fetuses with spina bifida that are not electively terminated receive no specific treatment until after birth. In the United States, these babies are generally delivered by cesarean section, though the benefit of this approach over vaginal delivery has not been clearly demonstrated. The parents should be told the infant's prognosis based on the functional spinal level, and it should be emphasized that closure of the defect is a lifesaving measure but will not alter the preexisting neurologic deficits.
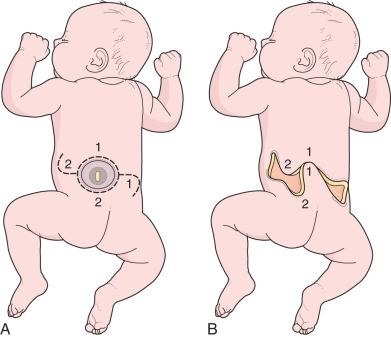
The initial step in managing the newborn with myelomeningocele is a careful physical examination by a pediatrician and neurosurgeon ( Fig. 7.1 ). A thorough evaluation should reveal associated anomalies, including cardiac and renal defects that might contraindicate surgical closure of the spine defect. Data suggest that if broad-spectrum antibiotics are administered, closure of the myelomeningocele can be safely delayed for up to a week to allow time for discussion with the parents. In most instances, however, the closure is performed within 48 to 72 hours of birth. The myelomeningocele is inspected; the red, granular neural placode is surrounded by a pearly “zona epitheliosa” that must be entirely excised to prevent the appearance of a dermoid inclusion cyst. Most myelomeningoceles are slightly oval with the long axis oriented vertically. If the lesion is oriented more horizontally, a horizontal skin closure may be preferable. Large defects that span a relatively large horizontal surface area may not be amenable to the traditional skin closure described later. In these cases, involvement of a plastic surgeon is advised, particularly when complex flap reconstruction is needed to achieve a complete, robust closure. While awaiting surgical closure, the infant is nursed in the prone position with a sterile, saline-soaked gauze dressing loosely applied over the sac or neural placode. Care is taken to ensure fecal contents from the nearby anus are not able to migrate superiorly into the defect. This can be achieved by placement of a thin barrier adhered horizontally below the caudal margin of the defect and above the anus.
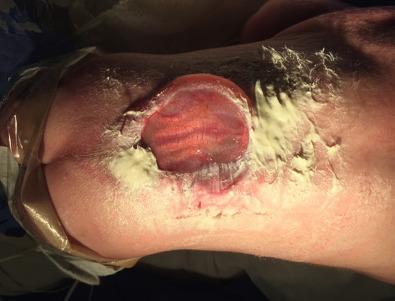
Approximately 85% of myelomeningocele patients either present with hydrocephalus or develop it during the newborn period. A large head or bulging fontanelle suggests active hydrocephalus and indicates the need for a head ultrasound or computed tomography (CT) scan. Stridor, apnea, or bradycardia in the absence of overt intracranial hypertension suggests a symptomatic Chiari II malformation, which carries a poor prognosis. Neurologic examination is difficult in a newborn infant, and distinguishing voluntary leg motion from reflex movement may be difficult. It is generally assumed that any leg movement in response to a painful stimulus to that limb is reflexive. Contractures and foot deformity denote paralysis at that segmental level. Virtually all affected neonates have abnormal bladder function, but this is difficult to assess in the newborn. A patulous, insensate anus confirms sacral denervation.
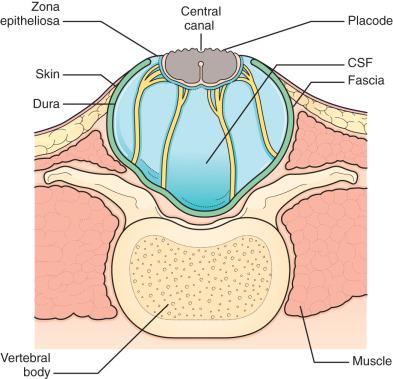
Typically, the back is closed first, and a CSF diverting procedure is deferred unless necessary. In cases with overt hydrocephalus, the back closure and the shunt can be performed simultaneously—if elevated intracranial pressure is felt to compromise the integrity of the spinal closure, resulting in a CSF leak. However, liberal employment of CSF diversion during—or prior to—back closure may equate to unnecessary treatment in a patient who would otherwise not develop persistent hydrocephalus. The goal of back closure is to seal the spinal cord with multiple tissue layers to inhibit the entrance of bacteria from the skin and to prevent CSF leakage while preserving neurologic function and minimizing the risk of future spinal cord tethering. Accomplishing this goal requires a thorough understanding of the three-dimensional anatomy of the tissue layers involved ( Fig. 7.2 ).
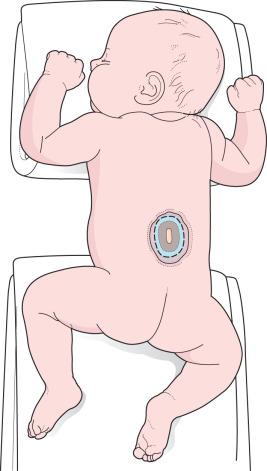
The patient is intubated in the lateral position or supine on a donut gel roll and then transferred to the prone position, with rolls under the chest and hips to allow the abdomen to hang freely ( Fig. 7.3 ). The surgeon gently attempts to approximate the base of the sac or defect vertically or horizontally, to determine which direction of closure results in the least amount of skin tension. The defect and surrounding skin are prepped and draped in the standard fashion, but special care must be taken not to permit contact of iodine solution with the central neural placode. If lateral, relaxing incisions are anticipated for a complex flap closure, the prepped area should be widened accordingly.
An elliptical incision is made, oriented along the chosen axis, just inside the junction of the normal skin and the thin, pearly zona epitheliosa. Full-thickness skin forming the base of the sac is viable and should not be excised. The incision is carried through the subcutaneous tissue until the glistening layer of everted dura or fascia is encountered. The base of the sac is mobilized medially until it is seen to enter the fascial defect. Any adhesions between the pial sac as it enters the fascial defect are released with a combination of blunt and sharp dissection to minimize the risk of tethering. The sac is entered by radially incising the cuff of skin surrounding the neural placode. The zona epitheliosa and skin is sharply excised circumferentially around the placode with scissors and discarded. It is important to excise all of the zona epitheliosa to prevent later formation of an epidermoid or dermoid cyst. At this point, the neural placode is lying freely above the everted dura ( Fig. 7.4 ).
Next, the neural placode is “reconstructed” so that it fits neatly within the dural canal and a pial surface is in contact with the dural closure. This is also thought to minimize the risk of future spinal cord tethering. Interrupted 6-0 sutures approximate the pia-arachnoid-neural junction of one side of the placode with the other, folding the placode into a tube. The central canal is closed along its entire length.
Attention is then directed to the dura, which is everted and loosely attached peripherally to the underlying fascia. Using a combination of low-voltage monopolar, and sharp and blunt dissection, the dura is undermined bluntly and reflected medially on each side until enough has been mobilized to enable a closure (see Fig. 7.4 ). The dura is closed in a watertight fashion using a running suture of 4-0 neurilon. If possible, the fascia may be closed as a separate layer by incising it laterally in a semicircle on both sides, elevating it from the underlying muscle, and reflecting it medially. Like the dura, the fascia is closed with a continuous stitch of 4-0 suture material ( Fig. 7.5 ). The fascia is poor at the caudal end of a lumbar myelomeningocele as well as with most sacral lesions; thus closure at this level may be incomplete. On rare occasions a bovine pericardial graft may be used to complete the caudal closure, if insufficient dura precludes a watertight closure.
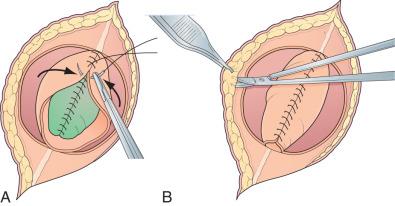
The skin is mobilized by blunt dissection with dissecting scissors or a finger. It may be necessary to free up the skin ventrally all the way to the abdomen ( Fig. 7.5 ). In most instances, primary skin closure in the midsagittal (vertical) plane is easiest, but occasionally horizontal closure results in less tension. A two-layer closure using vertical interrupted mattress skin sutures is preferred.
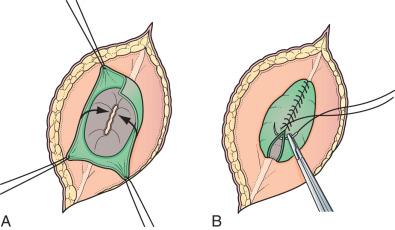
Very large lesions require special techniques. Wide circular defects can be closed using a simple rotation flap ( Fig. 7.6A-B ). Various types of “Z-plasties” and relaxation incisions have been described and may be necessary in very large or difficult lesions. Particularly large and challenging defects may necessitate a myocutaneous flap, which offers a stronger, multilayer closure with a rich vascular supply. Alternative techniques such as allogeneic skin grafts and tissue expansion are rarely needed.
Postoperatively, the patient remains in the prone position with hyperextended hips, if possible, to minimize secondary tension on the wound. The closed defect is loosely covered with a moist, antibiotic-impregnated dressing that allows for multiple daily wound inspections. Hydrocephalus develops in the majority of these patients and may present during the period of wound healing. Although definitive CSF diversion preferably ensues after the back has healed, early intervention may be necessary, particularly if increasing intrathecal pressure places the wound at risk for CSF leak and wound breakdown. Timing of hydrocephalus treatment is based on daily head circumference measurements, fontanel assessment, wound inspections, interval head ultrasounds, and signs and symptoms of elevated intracranial pressure (eg, apneic or bradycardic spells).
Care of the child with a myelomeningocele is lifelong; it only begins with the surgical closure. A deterioration in neurologic function may indicate a progressive process such as shunt malfunction, hydromyelia, tethered cord, or symptomatic Chiari II malformation. Given the multisystem influence of myelomeningocele, multidisciplinary collaboration between specialists in urology, orthopedic surgery, and pediatrics is imperative to optimize functionality and quality of life. Among those who undergo early back closure, 92% will survive to 1 year. From prospective outcome cohort data, it is known that the survival rate until age 17 is 78%, but it drops to 46% by the fourth decade of life. Death is the result of problems associated with the Chiari II malformation, restrictive lung disease secondary to chest deformity, shunt malfunction, or urinary sepsis. A sensory level higher than T11 is associated with increased risk of mortality, likely due to increased risk of urosepsis. Approximately 75% of children with myelomeningocele are ambulatory, although most require braces and crutches. Approximately 75% of surviving infants will have normal intelligence (defined as IQ > 80), although only 60% of those requiring shunts for hydrocephalus will have normal intelligence. Normal intelligence drops to 70% in surviving adults.
The role of endoscopic third ventriculostomy (ETV) in the management of myelomeningocele-related hydrocephalus has gained modest traction. Kulkarni and colleagues reported successful CSF diversion in >60% of patients treated via ETV, thereby avoiding the long-term morbidity associated with shunting. Meanwhile, Dr. Benjamin Warf has addressed hydrocephalus in myelomeningocele patients via the combined ETV with choroid plexus cauterization (ETV/CPC) procedure. In a cohort of 115 patients, 76% successfully avoided a shunt at a median follow-up of nearly 20 months. While promising, future studies with long-term cognitive and quality-of-life outcomes measures are likely needed before widespread adoption of ETV/CPC in this population.
Because myelomeningocele is routinely detected before 20 weeks of gestation, it is one of only a few conditions that is amenable to surgical therapy prior to delivery. Intrauterine myelomeningocele repair has gained popularity as an alternative to postnatal repair within a select patient population. The rationale for intrauterine repair emerges from evidence that neurologic deterioration occurs during gestation. Normal lower extremity movement can be seen on sonograms of affected fetuses before 17 to 20 weeks of gestation, but most late-gestation fetuses and newborns with myelomeningocele have some degree of deformity and paralysis. Such deterioration could be the result of exposure of neural tissue to amniotic fluid and meconium or direct trauma as the exposed neural placode impacts against the uterine wall. Protection of intrathecal contents after in utero closure may reduce or eliminate neural tissue damage. Animal studies (in which a model for spina bifida is created by laminectomy and exposure of the fetal spinal cord to the amniotic fluid) have demonstrated improved leg function if the lesion is closed before birth. Additionally, there is evidence that the Chiari II malformation, which occurs in the majority of individuals with spina bifida, is acquired and may be prevented by in utero closure.
The first successful in utero repair of spina bifida was performed in 1997 by hysterotomy at Vanderbilt University Children's Hospital and at the Children's Hospital of Philadelphia. Three years earlier, in utero closure had been attempted via an endoscopic technique, though it was deemed unsatisfactory and promptly abandoned. Fetuses treated in utero are delivered by cesarean section because the forces of labor are likely to produce a uterine dehiscence. The early experience at both institutions suggested that relative to babies treated postnatally, those treated in utero had a decreased incidence of hindbrain herniation and possibly a decreased need for shunting. The combined experience at the Children's Hospital of Philadelphia and Vanderbilt demonstrated a reduction in hydrocephalus requiring shunting for patients treated in utero relative to those treated postnataly. It is hypothesized that fetal closure of the spinal lesion reduces the need for shunting by eliminating the leakage of spinal fluid, which puts back pressure on the hindbrain. This allows reduction of the hindbrain hernia and relieves the obstruction of the outflow from the fourth ventricle.
In utero spina bifida closure appears to be generally well tolerated by the expectant mothers. Approximately 5% of fetuses have died from complications associated with uncontrollable labor and premature birth. Continued modifications to and improvement of the surgical technique surrounding uterine manipulation and closure may reduce perinatal morbidity. An analysis of lower extremity function in children treated prenatally revealed no significant difference from a set of historical control subjects who were treated with conventional postnatal repair. However, many of the children evaluated in this series had lower limb paralysis at the time of the surgery, which may have diluted any possible benefit. In contrast, a series from the Children's Hospital of Philadelphia suggested potentially improved leg function in patients with prenatally confirmed intact leg movement on ultrasound prior to fetal surgery. Problems with delayed development of dermoid inclusion cysts and tethered cord may adversely affect outcome in the long term. Prenatal surgery does not appear to reduce the need for intermittent catheterization during childhood, though a slight improvement in urodynamic studies has been observed relative to postnatal repair. The incidence of the Chiari II malformation and the need for shunting may decrease, but there are currently no long-term data.
Prior to the Management of Myelomeningocele Study (MOMS) trial, outcomes for spina bifida babies treated in utero were assessed relative to outcomes in conventionally treated, historical control subjects. Such comparisons are, however, prone to substantial biases because fetuses that undergo in utero closures represent a highly selected subset of cases. In addition, the medical management of spina bifida is continuously improving, making comparisons with historical control subjects particularly problematic.
A consortium of three institutions (Children's Hospital of Philadelphia, Vanderbilt, and University of California San Francisco) performed an unblinded, randomized, controlled trial of in utero treatment of spina bifida ([MOMS] to obtain definitive answers regarding the benefits of fetal myelomeningocele closure ). Pregnant women who receive a prenatal diagnosis of spina bifida between 16 and 25 weeks of gestation were randomized to either in utero repair at 19 to 25 weeks' gestation or cesarean delivery after demonstration of lung maturity. The primary study end points were the need for a shunt procedure at 12 months and fetal/infant death. Secondary end points included neurologic function, cognitive outcome, and maternal morbidity. The intent to treat analysis demonstrated a significant risk reduction with regard to the primary end point, and the study was closed early due to efficacy of prenatal surgery. The prenatal surgery group benefited from decreased shunt requirement (40% versus 82%) and a higher proportion of normal hindbrain anatomy, and it was more likely to ambulate independently at 30 months compared to the postnatal group. There were no maternal deaths, and adverse neonatal outcomes were similar between groups; however, prenatal surgery was associated with more pregnancy complications, increased frequency of preterm delivery, and a higher rate of respiratory distress syndrome in the neonate. To date, this is the only randomized study that demonstrates clear benefits of in utero treatment of spina bifida. These benefits were realized at experienced centers with strict inclusion criteria and must be carefully weighted against the higher rates of prematurity and maternal morbidity. Additionally, a subsequent, subanalysis of MOMS participants found that patients with larger ventricles at initial screening carried an increased requirement for shunting when undergoing fetal surgery, which approached the rate observed among postnatal repairs. Specifically, among fetuses with a lateral horn ventricle size >15 mm, 79% treated prenatally required a shunt. Meanwhile, only 20% of those with a ventricle size <10 mm required a shunt, and 45% with ventricles of 10 to 15 mm required a shunt. Thus patients and families should be counseled with this consideration in mind before fetal surgery is pursued.
In an attempt to reduce maternal morbidity and uterine manipulation, some have proposed an in utero endoscopic technique utilizing a biopatch overlay with simple skin closure. Results from a small (n = 10), single arm trial demonstrated feasibility but no clear advantage over the more traditional technique utilized in the MOMS trial. Similar hydrocephalus and neuromotor outcomes were demonstrated with endoscopic repair. However, aborted procedures, premature rupture of membranes, and more premature delivery may limit the widespread adoption of endoscopic repair until a safer, more refined technique is developed.
Long-term outcomes for in utero repair are limited but thus far encouraging. Ten-year neurofunctional outcomes data in 42 children found that nearly 80% were community ambulators, and the majority scored within the average range on select behavioral and cognitive metrics. Patients undergoing fetal myelomeningocele repair, however, demonstrated significantly more deficits in executive function than their community peers. Overall, early neurodevelopmental outcomes predicted 10-year outcomes, and the need for shunting following prenatal repair was associated with adaptive behavior deficits. Although more extensive follow-up is needed, early data suggest fetal myelomeningocele repair improves long-term functional status.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here