Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Common nonmelanoma skin cancers (NMSCs) include basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). The common forms of skin cancer arise, respectively, from the upper layers (epidermis) of the skin:
Basal cell: arises from the basal layer of the epidermis
Squamous cell: arises from the superficial layer of the epidermis
Melanoma: arises from the basal pigmented cells of the epidermis
Surgeons are often called upon to evaluate cutaneous lesions and malignancy. This chapter reviews cutaneous nonmelanoma malignancies and provides suggestions for evaluation, planning, and treatment.
NMSCs are the most common human malignancies, occurring more frequently than all other solid tumors combined. There are over 80 different NMSCs. The most common of these tumors are BCC and SCC, although several other types exist and will be discussed briefly. The frequency of these cancers is such that almost everyone living in the United States to 80 years of age will have one; as the life span increases, these malignancies become more common as the effects of sun exposure and other causative factors become more important. These are as follows:
Reduction of protective factors in the atmosphere
Reduction with aging in previously very effective local skin immune mechanisms responsible for tumor surveillance
Increased sun exposure and the general reduction of immunity with progressive aging
These factors and the constantly increasing life span keep the incidence of these common malignancies increasing rapidly. Progress in the past 40 years has produced vast improvements in the classification, diagnosis, and treatment of NMSCs. Accurately identifying lesions early in their course allows classification and directed differential treatment for improved outcomes. Not infrequently, patients ignore their initial lesions, permitting development of large tumors. Disfigurement may result as not only skin tissue structures are involved but also muscle, nerves, cartilage, and bone.
Recent figures indicate that the annual cost of treating basal and squamous cell cutaneous cancer in the United States will exceed over $8.1 billion dollars. Although most of these skin lesions have a rather benign behavior as far as cancers are concerned, certain more aggressive subtypes must be identified and treated differently with wider excisions, margin evaluation, possible sentinel lymph node biopsies, and continued periodic observation for regional and distant metastatic spread. Specifically, these subtypes need complete negative peripheral and deep margin confirmation before reconstruction and, subsequently, consideration for frequent postoperative observation. Less commonly, some of these patients will need radiation and medical oncologic treatment, in addition to surgery. Although cutaneous SCC is curable with local excision for the most part, tumor recurrence, metastasis, morbidity, and even disease-specific death does occur. It is estimated that 5600 to 12,600 US patients with cutaneous SCC will develop nodal metastasis, and 4000 to 8800 will die from the disease. Although few surgeons would recognize it initially, these numbers of disease-specific death are similar to deaths from melanoma, leukemia, non-Hodgkin’s lymphoma, and renal and bladder cancers.
Risk factors for BCC and SCC include a number of carcinogens. The most agreed on is sunlight. Cumulative exposure to sun, especially tropical sun without protection, in fair-skinned individuals who burn rather than tan, is the most generally discussed and accepted risk. Tumors are more common on sun-exposed surfaces such as the face, scalp, and neck. Sun lamp or ultraviolet light treatment for psoriasis is a risk factor for squamous cell skin cancers. Microtrauma to the skin contributes to the etiology of basal cell skin cancers. Radiation exposure, such as historically commonly delivered for acne in teenagers, also predisposes to skin cancer and thyroid cancer.
Because sun exposure has been implicated in the predisposition to these lesions, there has been considerable attention to programs aimed toward preventing excess exposure, especially sunburns. Two guides to this prevention are “Safe Sun Guidelines” and “Skin Protection from Ultraviolet Light Exposure” (available from the Skin Cancer Foundation). The exact effectiveness of prophylaxis is not known but is under extensive study. Sun-protective clothing is also offered.
Genetics and hereditary conditions such as basal cell nevus syndrome predispose to the development of hundreds of multiple cutaneous basal cell skin cancers. Northern Europeans, such as those of Irish and English heritage, also seem more prone to the development of skin cancer. Mutations in the RIS and DOS oncogenes have been implicated as well as the sonic hedgehog-signaling pathway for basal cell skin cancer. The PTCH (patched) gene on chromosome 9Q has been suggested in the genesis of these lesions.
A very significant factor in the development of NMSC is associated with the advances in solid organ transplantation and the medical immunosuppression management for the maintenance of those organs. This progress has rendered longer life spans for these patients but is associated with a high risk of NMSC development as well as poor disease prognosis. We have also seen an increase of immunomodulating drugs for many medical conditions such as IBD, rheumatoid arthritis, and several others.
Fifty percent to 70% of OTR patients are affected by NMSC, and posttransplant BCC risk linearly increases, whereas SCC incidence rises exponentially ( Fig. 1 ). This is reflected in the switch from a usual 4:1 ratio of BCC to SCC. SCC is the predominant OTR NMSC observed (although BCC is still highly prevalent). Merkel cell carcinoma occurs in OTRs at 5 to 10 times greater rates compared with the general population. Functional immunosuppression via HIV, lymphoma, and leukemia is a risk factor for NMSC and portends poor disease prognosis.
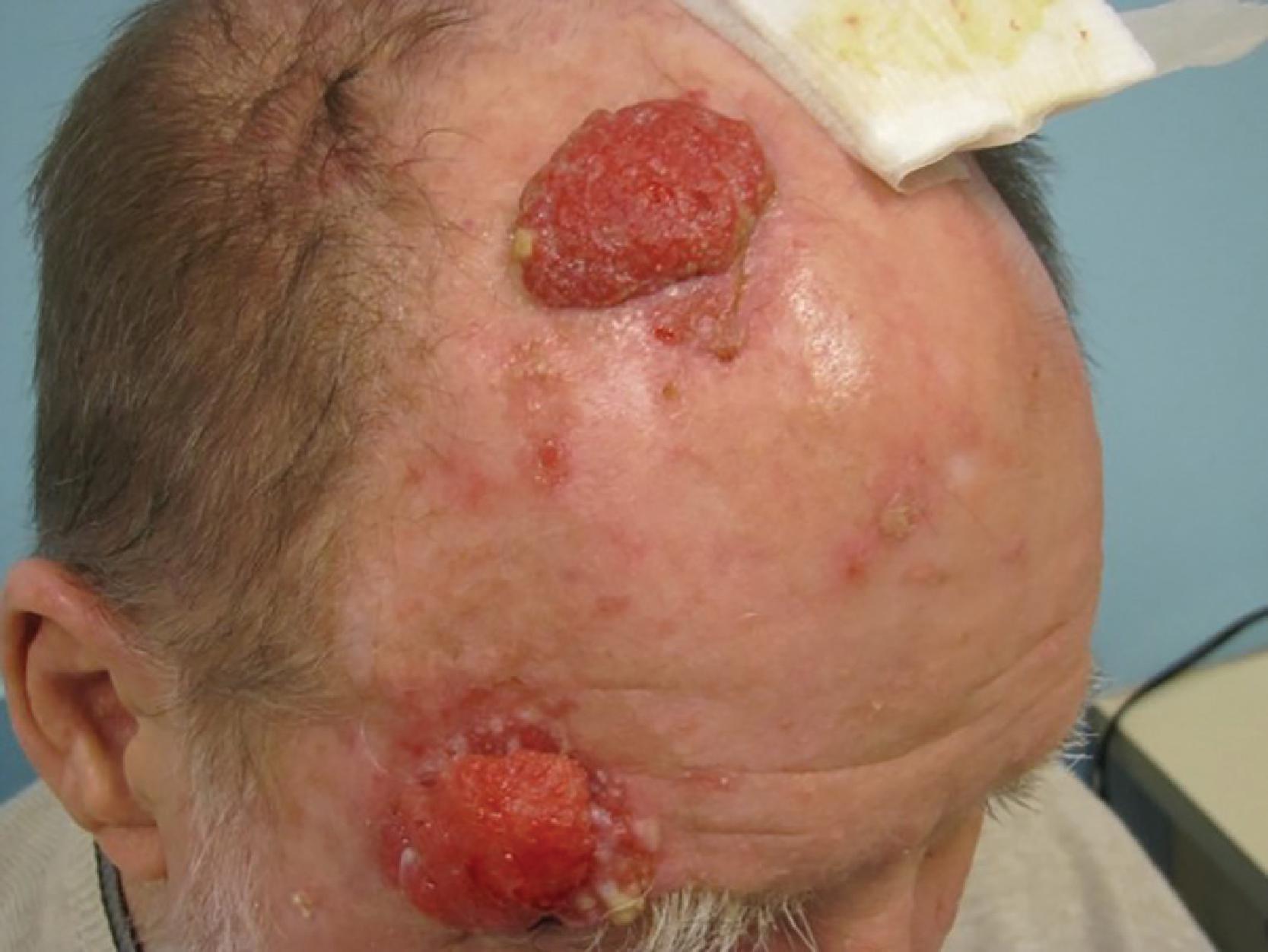
History and physical examination remain the mainstay of initial diagnostic workup in cases of suspected cutaneous malignancy, noting identification of relevant risk factors, especially as they relate to prior carcinogen exposure, genetic cancer syndromes, or immunosuppression. A generalized skin and physical examination must include several key elements: (1) comprehensive skin evaluation of the surrounding area; (2) for lesions in the head and neck region, suspicion should be raised for those in the hair-bearing scalp or behind the ears. A complete regional examination of the skin and lymph nodes, and a complete, careful skin examination is performed because of the frequency of additional lesions. Attention should be directed particularly to sun-exposed skin sites. Patients with BCC and SCC are at increased risk for developing melanoma as well as additional NMSCs.
The clinical appearance of lesions such as squamous cell and basal cell are characteristic. They include color change, nodularity, and ulceration. Scaling and telangiectasia are frequent. Generally, the skin is reddened and thickened. Flaking or keratoses may be present, and both types of lesions frequently demonstrate surrounding premalignant lesions, such as keratoses. Frequently, squamous cells arise in areas of actinic keratoses, known precursor lesions .
The physical examination and evaluation of BCCs and SCCs first includes an analysis of tumor location and size. The tumor should be characterized by its apparent appearance as a particular skin cancer type. This evaluation should map the borders of the tumor, the entire local and regional area, and examine local lymph nodes. The lesion is classified as primary, recurrent, or one with less common features. The general health of the patient may dictate treatment considerations. Certain locations have been known to predispose to recurrence (such as fusion planes, which are areas where the primitive parts of the face come together in the fetus) in head and neck BCC, the areas of the nasal labial fold, and the groove between the nose and the cheeks. Certain areas such as the ear, mucosal surfaces, genitalia, and lip, tend to produce squamous cell skin cancers that possess a greater capacity for metastasis ( Table 1 ). The “mask area” of the face has been known to have deeper skin cancers that are prone to incomplete resection and therefore have a higher risk for recurrence or persistence. Those lesions greater than 2 cm in diameter or more than 2 mm in thickness present more risk of persistence and recurrence than do smaller lesions.
|
In general, all lesions that have higher risk should have surgical complete margin assessment, preferably at the time of resection. Enlarged nodes must be carefully documented and warrant further diagnostic workup. Contrast-enhanced CT or MRI can be used for evaluation of clinically suspicious lymphadenopathy. Soft tissue disease including nerve invasion is best discerned by the use of MRI. Bone or cartilage invasion is best evaluated with CT. Ultrasound-guided fine-needle aspiration biopsy can be used to evaluate suspicious lymphadenopathy.
BCC is the most common malignancy observed in adults. There are at least 5.5 million new BCC cases diagnosed each year in the United States alone; the risk of developing a BCC is more than 20% over the course of an entire lifetime, and its incidence more than 4 times that of SCC. The predominant risk factor for the development of BCC is ultraviolet light exposure, primarily UVB. The strength of the causal relationship is not as clear as in the case of SCC. UV light induces inflammation of the skin that clinically results in visible erythema. On the molecular level, prostaglandin synthesis is markedly increased through the induction of cyclo-oxygenase-2 (COX-2). COX-2 is induced by inflammation. COX-2 inhibitors markedly decrease the incidence of cutaneous SCC and BCC. Other important risk factors include individuals with fairer skin, freckling, or blonde or red hair coloring. Genetic syndromes such as basal cell nevus syndrome and xeroderma pigmentosum are less common but strong risk factors. Finally, a history of immunosuppression, especially in the context of solid organ transplantation, poses a substantial risk for the development of BCC.
BCC is a mostly indolent entity from the perspective of metastatic or mortality risk. Untreated BCC is most concerning for local tissue destruction as opposed to systemic involvement or death. As a result, the goal of treatment is complete elimination of the primary lesion, sparing of as much healthy tissue as is reasonably achievable. Surgery is typically the preferred treatment and will be discussed in detail later. Less commonly, in circumstances such as very large tumors, cosmetically challenging areas, patients who cannot tolerate surgery, or for palliative treatment of BCC, radiotherapy can be employed with an expected success rate of 90% or more. The problem arises with identification of recurrence in the irradiated bed. There is no pathologic margin status to review and no specimen for microstaging. Medical therapies with topical 5-fluorouracil (5FU), topical imiquimod cream, and intralesional injection of interferon-α-2b have been studied and show some success. More recently, the novel hedgehog pathway inhibitor, vismodegib, has become an option for treatment of challenging or metastatic disease.
The majority (80%) of BCCs will manifest as lesions of the sun-exposed areas of the head and neck. Although it is tempting to treat the subtypes of BCC as superfluous minutiae, knowledge of the different pathologic entities that exist under the umbrella of BCC is essential to prognostication and appropriate treatment.
The most common subtype of BCC is the nodular type, comprising about 50% of all BCC tumors. This entity often presents with a shiny, pearly papule or nodule with a smooth surface and surrounding telangiectasias. Over time, lesions enlarge, ulcerate, and develop a more prominent rolled edge. The most likely location is the face, but other hair-bearing skin areas can also develop nodular BCC; non–hair bearing areas rarely develop these lesions.
Superficial BCC is the second most common, accounting for approximately 20% of all BCC tumors. Masses of this type are most often well-circumscribed, erythematous, and either truly flat macules/patches versus thin papules/plaques. An atrophic quality with associated hypopigmentation is common.
Nonaggressive subtypes of basal cell skin cancers include the keratotic, infundibulocystic, and nodular types and superficial types. Primary lesions in this group may generally be managed with 5-mm resection margins. It is very unusual for BCCs to exhibit lymph node or systemic metastasis. Lesions that do exhibit these aggressive traits have frequently been neglected, previously radiated, or multiply recurrent lesions.
Aggressive subtypes possessing more risk have more aggressive growth. These include the micronodular, infiltrative, sclerosing, morpheaform, and desmoplastic types and “basosquamous” carcinoma.
Some basal cells are pigmented and are easily confused with melanoma by appearance. The difference is often only established pathologically. Micronodular, infiltrative, and sclerosing tumors are notorious for a subclinical component that spreads laterally, leading to a high recurrence rate compared with other BCC subtypes. The final two subtypes worthy of a brief discussion are the basosquamous and morpheaform BCCs. The basosquamous family of tumors exhibit the worst features of BCC and SCC, most notably with tendencies for aggressive local growth, recurrence despite treatment, and metastasis in nearly 10% of cases. Morpheaform lesions have an indurated quality that is usually light pink to white in color with ill-defined borders and areas of regression. Aggressive local tissue destruction is the rule as opposed to the exception.
Basal cell nevus or Gorlin syndrome or nevoid BCC syndrome is an inherited disorder that presents with multiple cutaneous basal cells, multiple odontogenic keratocysts of the jaws, musculoskeletal and neurologic abnormalities, and pitting of the palmar and plantar surfaces of the skin. The disease is autosomal dominant. The lesions behave as basal cell skin cancers and are usually managed with surgical excision.
Usually, patients will have hundreds of basal cell skin cancers over the course of their lifetime, and many eventually succumb to the disease as sheer numbers overwhelm treatment. Topical chemotherapy agents have been used in an attempt to eradicate early superficial nonfacial basal cells, but surgical excisions are the treatment of choice.
SCC of the skin is the second most common malignancy, with more than 3.3 million new cases diagnosed per year in the United States alone. Similar to BCC, cumulative UV radiation exposure is considered the strongest risk factor for the development of SCC. In addition to the mentioned risk factors for BCC, which also augment risk for SCC, tobacco smoking, human papillomavirus infection, and chronic/nonhealing wounds are other notable risk factors for SCC. There are many histologic features of these tumors that can affect prognosis ( Table 2 ). Originally, squamous cell skin cancers were divided into several grades; however most pathologists presently feel that two general groups of behavior, can be identified for squamous cell cancers: (1) well-differentiated lesions and (2) moderately differentiated, poorly differentiated, and undifferentiated lesions.
|
Although the prognosis within the latter groups becomes dramatically worse in the poor and undifferentiated categories (which, like melanomas, are capable of in-transit, lymphatic, and systemic metastasis) the tendency is to treat these high-risk lesions with wider and deeper resection margins. Sentinel lymph node biopsies in the latter group may be considered if the lesion is greater than 2 cm area or greater than 2 mm thickness ( Fig. 2 ). Histologic confirmation of negative peripheral and deep margins before defect closure or flap reconstruction must be routinely accomplished.
SCC carries a risk of metastasis (about 5%) in patients with less differentiated lesions and in certain anatomic areas, such as the lip and genital region. The frequency is also increased for immuno- incompetent individuals, who tend to have less differentiated lesions. The mainstay of treatment for most lesions is surgical excision.
Radiotherapy has a role in the treatment of BCC for patients who are poor surgical candidates, those receiving palliative treatment, or those in whom the sheer volume or anatomy of the surgical resection would challenge appearance or function. Of course, radiation is used in the adjuvant setting for patients with aggressive disease and or metastasis to regional nodal beds. There is a subset of SCC lesions that manifest with perineural invasion (as evidenced by the development of neuropathic symptoms or on pathology). These lesions are at higher risk when named rather than unnamed nerves are involved, and contrast MRI will assist in defining the extent of involvement.
Any discussion of SCC requires mentioning important precancerous lesions and in situ malignancies that are both direct precursors to the frankly malignant lesions of SCC. Actinic keratoses are exceedingly common and represent disordered keratinocyte growth that is limited to the epidermis. The growth occurs as a direct result of photodamage, which explains the predilection for the head, neck, upper trunk, and extremities. Although the morphology of these lesions can be quite variable, they classically present as a rough erythematous papule with prominent white to yellow scaling. Associated tenderness portends a higher probability of associated squamous carcinoma.
Keratoacanthomas comprise a group of cutaneous neoplasms that share many features of SCC but are unique enough to merit individual discussion at a genetic level. Keratoacanthoma lesions may double in size quite rapidly. The more rapidly growing lesions should be removed for diagnosis as soon as feasible because most of these lesions are malignant. Keratoacanthomas most often manifest as a solitary lesion that grows rapidly over several weeks into a sharply circumscribed nodule with a cratered, keratotic core. The lesions are characteristically sensitive or painful when touched, and sensory nerve symptoms such as numbness, pain, burning, and paresthesia are concerning and should raise suspicion of SCC. Motor nerve involvement is much less frequent and occurs in extensive or deeply penetrating lesions that exhibit paralysis, weakness, or double vision, placing treatment of the tumor in a high-risk category. A contrast MRI is the best modality to document major nerve invasion.
Most lesions occur on the head and neck or in sun-exposed areas of the extremities. Occasionally, the lesion will spontaneously resolve over several months, leaving behind an inconspicuous atrophic scar. Because 95% of these lesions will continue to grow and not involute, creating a much larger lesion which is difficult to treat, it is our recommendation to excise them as growth is frequently alarmingly rapid (doubling every few weeks), and complete removal is always more challenging when larger. They should be treated identically to SCC as even pathologic differentiation from the benign variant is difficult. Recently, syndromes of elderly individuals with multiple keratoacanthoma squamous cells have occurred (frequently on the legs in severely sun-damaged skin), coming back frequently, even in resected squamous cell cancer scars, challenging treatment. Some have postulated a viral etiology. Injections of 5FU have been utilized with success in some patients, especially when multiple lesions appear almost simultaneously in elderly patients or if patients are not good candidates for operative resection.
Subtypes of squamous cell variants, such as the adenoid cystic type or adenosquamous carcinoma, are tumors whose more aggressive behavior characteristics increase the risk of incomplete resection, recurrence, and metastasis.
Desmoplastic cutaneous squamous cell carcinoma has a higher risk of recurrence as a result of incomplete resection because of subtle deeper extensions along fascial planes with failure to achieve clear margins. Some have suggested that invasion of a tumor into the fat or invasion deep enough to penetrate lymphatics constitutes more potential for recurrence and metastasis, as is known with melanoma.
Squamous cell carcinoma in situ represents full-thickness atypia of the upper skin layer (epidermis). Because the lesions are noninvasive, they are treated by simple complete excision because metastasis is not possible. As a pathologic entity, SCC in situ involves the abutment of neoplastic cells onto the epidermis but not into the dermis. Clinically, these lesions resemble eczema or psoriasis and have a characteristic pink to red color with scaling and sharply demarcated borders. Patients who have had development of SCC from SCC in situ are thought to have a roughly twofold to threefold higher chance of metastasis, especially in cases of mucosal, lip, or perineal SCC in situ. Topical chemotherapy agents such as 5FU, imiquimod, or cryotherapy can be utilized for SCC in situ; however, excision with margin confirmation is preferred for recurrence, persistence, or previous treatment failures. It is difficult to predict the cure rate of these treatments because they are not predicated on complete biopsies, and follow-up is generally incomplete and lacking biopsy proof of eradication. Immune modulators can also be offered for extensive disease or for patients who are not candidates for surgical treatment, and success has been described with vismodegib.
Certain precursor lesions, such as actinic keratoses, are precancerous. Actinic keratoses are red, crusty lesions that weep and sometimes develop cutaneous horn proliferation or thick layers of flaky skin. They have an incidence of in-situ and invasive squamous cell skin cancer development of at least 10% (0.1%/year); therefore, treatment of these lesions with liquid nitrogen, a topical chemotherapy, or excision seems reasonable. However, recurrence should be a marker for progression to SCC and should be treated accordingly. Actinic keratoses may regress and reappear. SCC may be preceded by SCC in situ. Two syndromes are Bowen’s disease and Paget’s disease, which are types of SCC in situ that affect the breast and perineal areas and may progress to invasive SCC.
Bowen’s disease is an erythematous, plaquelike lesion that represents a low-grade, slow-growing intraepithelial variant of SCC. Essentially, the growth of this tumor is intraepidermal, and its history and progression are similar to other intradermal carcinomas such as erythroplasia of Queyrat or Paget’s squamous cell disease. It is a less common malignancy that appears as a circumscribed, red or pink patch that can weep and present a scaly appearance. Many lesions occur on the trunk, lower extremities, and genitalia, but the lesions can involve the head and neck region. The progression of the lesion is slow and limited and extends normally over years. Invasive SCC can arise in these lesions, and if so, metastasis is possible. Excision to clear the margins with an initial 5-mm margin is recommended, and Mohs excisions with margin evaluation are favored. Larger lesions may require flap coverage or skin grafting. In the head and neck, cosmetically sensitive flap reconstructive techniques are preferred when primary closure is not possible.
The gold standard in diagnosis of cutaneous lesions is full-thickness skin biopsy to at least the reticular dermis (infiltrative histology, deeper penetration, higher-risk lesions). Pathologic information that results from this biopsy depth includes lesion type, differentiation, histology, histopathologic subtype (for SCC and BCC), ulceration, perineural involvement, vascular involvement, degree of differentiation (SCC), tumor depth (SCC), and tumor thickness (SCC). A shave biopsy of a squamous cell skin cancer will frequently be read as SCC in situ and not confirm any invasive component.
Recall that SCCs with poorly or undifferentiated histologic characteristics and those greater than 2 cm in area or more than 2 mm thick (tumor can invade lymph vessels in the dermis) are at higher risk for infiltrative spread and lymph node involvement. Based on the histologic confirmation of aggressive characteristics, a sentinel lymph node examination or more aggressive resection may be considered. Clinically enlarged nodes should be evaluated with an ultrasound-guided fine-needle aspiration biopsy. The use of the ultrasound guidance allows for a more accurate biopsy, focusing on the most suspicious area of the node based on vascularity and echo texture. This process will help avoid the problem of false-negative biopsies.
Appropriate management of NMSC depends on formulating an appropriate risk estimate. A small, slowly growing SCC found in an immunocompetent patient is an entirely different entity than a large, rapidly growing, painful SCC located in the amputation stump of an immunocompromised patient. The workup and treatment for these lesions would be different and is influenced by the risk stratification of a lesion. This includes advanced imaging or diagnostic procedures, different surgical margins, need for lymph node biopsy, consideration for referral to medical or radiation oncology, and follow-up (see Table 1 ).
Area L: trunk, extremities; Area M: cheeks, forehead, neck, scalp; Area H: “mask areas” of face (central face, eyelids, eyebrows, periorbital, nose, lips, chin, mandible, preauricular and postauricular skin/sulci, ear, temple), genitalia, hands, and feet.
Once a diagnosis is made, (either clinical diagnosis with a high degree of certainty or pathologic diagnosis), a surgical plan may be formulated. All surgical plans have a few commonalities, including expected margins, plan for margin evaluation and amount of resection desired, and type of wound closure or reconstruction.
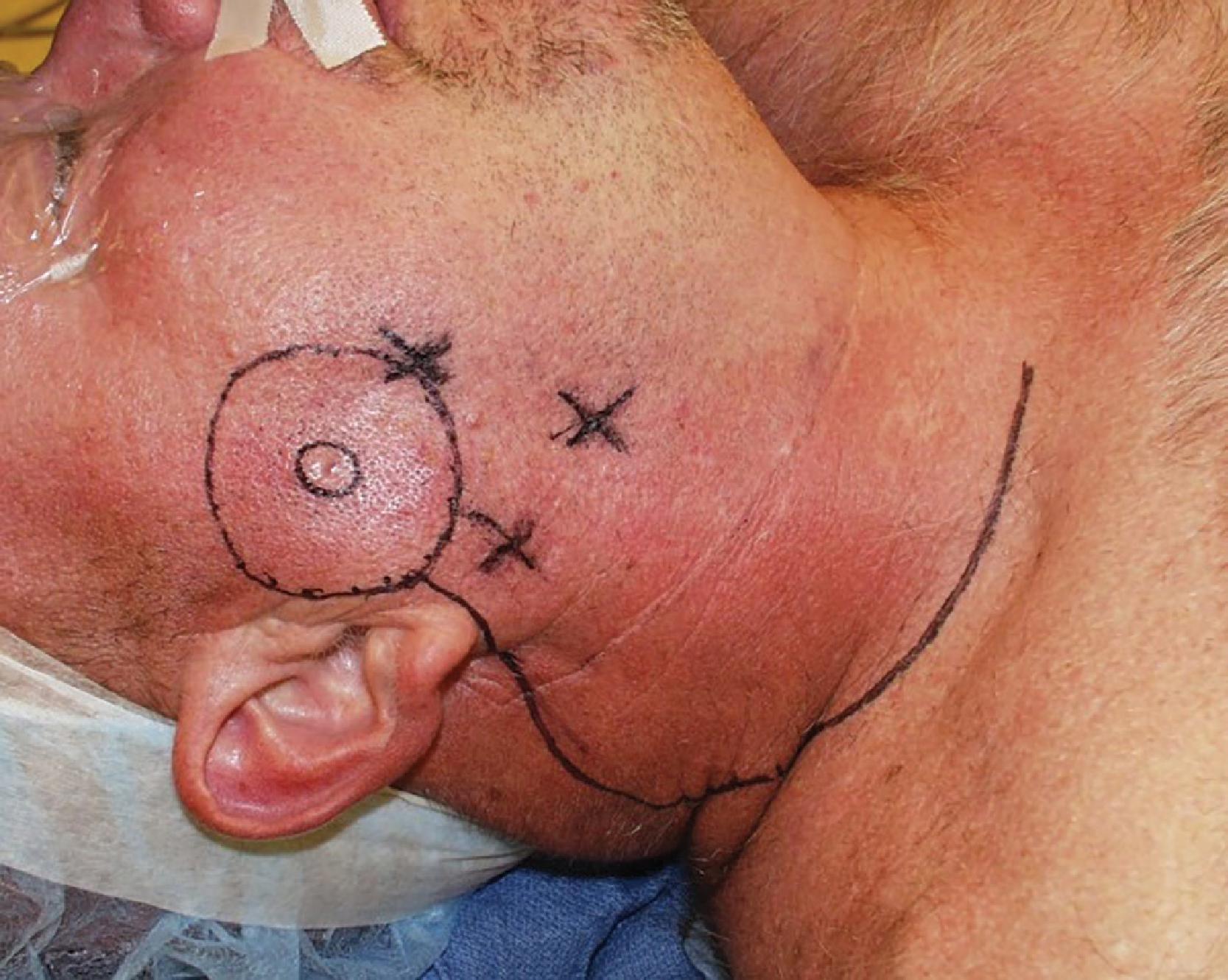
A systematic approach to marking begins with first tracing the extent of the lesion itself. In straightforward cases, there will be stark differentiation between the abnormal tissue and the normal tissue with an obvious border. Commonly, confounding factors such as marginal erythema, telangiectasias, or dyspigmentation may add confusion in determining which areas of skin constitute the lesion.
Next, an uninvolved segment of tissue is added: the surgical margin. There are published and widely circulated national guidelines for resection margins for both BCC and SCC that are confirmed by high-level clinical data from expert national group consensus. Our practice is to follow recommendations from the National Comprehensive Cancer Network (NCCN) in the United States listed in Table 3 , which are intelligent, comprehensive, and pragmatic.
| Tumor Type and Risk | Recommended Margin |
|---|---|
| Local, low-risk SCC | 4–6 mm |
| Local, high-risk SCC | 10–15 mm |
| Local, low-risk BCC | 4 mm |
| Local, high-risk BCC | 3 mm followed by 3 mm if first margin positive; consider Mohs surgery |
| Dermatofibrosarcoma protuberans | 3–5 cm and down to investing fascia; consider Mohs surgery |
| Merkel cell carcinoma | 1–2 cm; include sentinel node biopsy |
Marginal evaluation is the most critical aspect of tumor extirpation. An operation that removes some tumor but not all introduces the morbidity of surgery without the benefit of cure. With this in mind, surgeons must be well equipped to fully understand that both all peripheral and deep margins are evaluated. Guidelines exist for peripheral margin distance, as listed earlier, but deep margins are often determined by the surgeon’s decision making or, in the case of Mohs micrographic surgery, real-time pathologic evaluation.
Techniques for rapid or intraoperative evaluation of margins include frozen-section assessment and Mohs micrographic surgery. Frozen-section assessment is done by laboratory-based pathologists and considers representative “breadloaf” or interval margin samples. The Mohs technique produces an immediate determination of the full length of the cutaneous margin as well as the deep margin. In either case, the surgeon may request permanent sectioning when unclear. In the case of equivocal margin status, the wound should be temporized with allograft or dressings until final permanent margins are available.
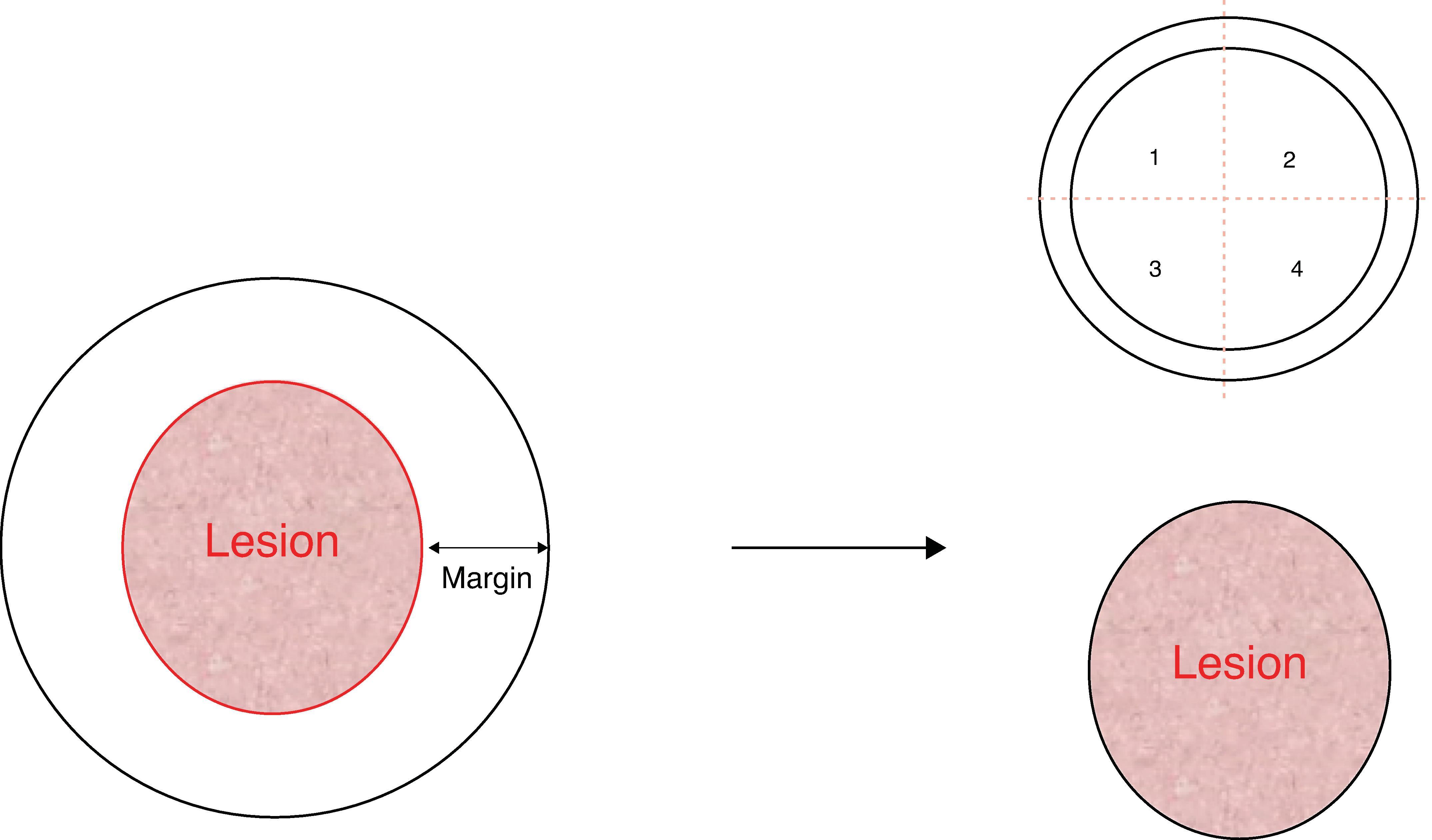
An alternative approach to rapid margin evaluation is to excise a thin rim of tissue from all quadrants at the estimated periphery of the lesion. Each skin section from each quadrant is then labeled and sent individually for analysis guaranteeing identification, and each thin defect is closed primarily ( Fig. 3 ).
The situation of a patient being referred for reconstruction following resection by an outside physician is worth a brief discussion. One course of action is re-resection of a thin margin around the initial resection bed, regardless of the outside pathology report, which serves to even and square the edge, trim a contaminated or devascularized edge, and provide an additional clean margin for further pathologic confirmation. It has been our experience that the rate of residual tumor identified on such secondary marginal examinations may reach 10%.
If an enlarged node is identified or the lesion is high risk, lymph node sampling is suggested. Sentinel node biopsy or fine-needle aspiration biopsy with ultrasound guidance would be the first options for nodal interrogation. If these modalities are not available, directed, open biopsy may be considered. The author prefers at least consideration of sentinel node evaluation in poorly differentiated or undifferentiated high-risk squamous cell skin cancer; however, the benefit of routine lymph node evaluation in less differentiated squamous cell cancers has not been proven. In the event of nodal disease, surgical resection or consultation with medical and radiation oncology should proceed. Many institutions have multidisciplinary conferences in which these discussions occur weekly.
Surgical excision is most often the preferred therapy for cutaneous nonmelanoma malignancies. There are a small number of patients who are not candidates for surgery. The most common reason for this is substantial medical morbidity that limits the safe administration of anesthesia. Other patients are resolutely against surgical excision for a variety of reasons. Finally, certain lesions may not be well treated with surgery, such as diffuse lesions of Bowen’s disease or those in anatomically sensitive areas that are functional and difficult to reconstruct.
Also, it must be understood that small, noninvasive in situ and precancerous lesions may be treated very effectively with nonsurgical modalities.
Imiquimod is a topical immunomodulator that now provides two different formulations (1% and 3%) that are approved by the US Food and Drug Administration (FDA) for precancerous actinic keratosis, actinic cheilitis, and small truncal superficial BCC lesions; dosing frequency and duration depends on the indication and formulation. Imiquimod functions by inducing local antitumoral cytokines that produce a striking inflammatory breakdown of areas involving superficial BCC and precancerous actinic keratoses. Erosions can be quite extensive depending on the treated area; erosions are predisposed to infection and may require antibiotic treatment during imiquimod therapy. It is important to note that many practitioners now use imiquimod for off-label, non–FDA-approved uses, such as low-risk BCC (other than superficial) histology or anatomic sites other than the trunk (such as the face).
Liquid nitrogen is very effective for precancerous actinic keratoses and low-risk BCC. Actinic keratoses can be treated with a few seconds of exposure, while BCC requires successive rounds of tissue freezing.
5FU is topical chemotherapy, a derivative of the deoxyribonucleic acid uracil, that intercalates into DNA; rapidly dividing cells (such as NMSC and precancerous actinic keratosis) take up more 5FU, inducing necrosis. Similar to imiquimod, it can cause extensive erosions, and superinfection is common. 5FU has generally fallen out of favor because it is mutagenic on normal tissue and predisposes treated patients to cancer, which makes it a less appealing option compared to nonmutagenic imiquimod.
Electrodessication and curettage effectively treats truncal or extremity low-risk BCC, whereby the tumor is aggressively debulked with a curette, and then a broad-tipped curette tip is used under the electrodessication setting to ablate the bed of the tumor. This is repeated two additional times. Advantages are that it is quick, simple, has a good cure rate, and has favorable healing (a small circular white scar). Disadvantages include wound care required sometimes for weeks, which may be inconvenient if a patient does not have anybody assisting them at home.
Radiation therapy in cases of primary straightforward NMSC of the basal or squamous type can often produce good cure rates (90%) and excellent cosmetic results. Multiple treatments are required over a period of weeks, and there is no confirmation of negative margins, nor is there complete examination of the histology of the lesion to confirm complete histologic characteristics. The authors have found that many elderly patients with lesions of the lower extremities do not do well in terms of cure or residual symptoms following radiation treatment. Young patients in whom there is risk of further radiation-induced malignancy are generally treated with surgery. The use of radiation as a primary treatment modality will often preclude its further use as an adjuvant modality in the same area, thus limiting the possibility of using radiation for the management of recurrence or new disease.
Radiation therapy has also been utilized as adjunctive treatment in patients in whom the margins are histologically but not clinically positive or for perineural involvement or where the danger of intralymphatic spread into adjacent skin is high. Patients who have extracapsular extension, referred to as soft tissue disease, of tumor are also candidates for postoperative radiation. Patients with more than two positive nodes will be considered for adjuvant radiation to the nodal basin.
Treatment of metastatic disease often employs medical oncologic or radiotherapy. Some encouraging results have been obtained with immune modulators, such as vismodegib. Patients with disease remaining following radiation may be candidates for vismodegib, a hedgehog pathway inhibitor. This drug has shown some efficacy in the management of metastatic and inoperable BCC.
Although several chemotherapeutic agents have been described in case studies or small patient cohorts, only one randomized controlled trial (RCT) has been conducted, demonstrating a 34% overall response rate for a cisplatin-based regimen. EGFR inhibitors evaluated for use in cutaneous SCC by RCTs include cetuximab, panitumumab, and gefitinib; response rates ranged from 15% to 31%. Inhibitors of the immune checkpoint programmed death-1 (PD-1) have yielded promising outcomes in advanced cutaneous SCC. The PD-1 inhibitor cemiplimab recently received FDA approval for use in advanced cutaneous SCC. Despite these advances, the preferred regimen for systemic treatment of cutaneous SCC remains unclear, particularly in immunocompromised populations.
All patients with BCC and SCC, especially those with high-risk lesions, should have a complete skin examination periodically with special attention to the area of origin, where recurrence, either locally or regionally, is likely. Although most recurrences of lesions develop within a 2-year period, rarely a tumor will recur after 5 to 10 years.
Nonaggressive skin cancer types will recur in about 50% of lesions with positive histologic margins. One-third to one-half of patients having a basal or squamous cell skin cancer will develop a second primary lesion within the next 5 to 10 years. Follow-up schedules should be tailored to the particular severity and extent of disease and to the risk of recurrence.
There are dozens of cutaneous malignancies that do not arise from keratinocytes, all of which are substantially less common than BCC and SCC. There is a handful worthy of a brief mention, but we encourage interested readers to pursue further reading in a cutaneous oncology text.
Merkle cell carcinoma is a rare neuroendocrine skin cancer with about 1600 cases diagnosed in the United States annually. It is characterized by rapid growth and very aggressive behavior. A firm, intradermal, red nodule rapidly progresses to ulceration. Recently, some lesions have been traced to a polyomavirus infection, and the disease is more frequent in patients receiving immunosuppression. The disease progresses rapidly by intravascular and lymphatic spread. Therefore, initial positron emission tomography (PET) imaging should evaluate the possibility of regional and distant spread. Prognosis is often predicted by positive regional lymph nodes, which occur in 25% to 30% of N0 patients (no clinical evidence of nodal disease). The disease often tends to be undertreated by limited surgical resections; thus, local failure is common (up to one-third of patients). Local control may be improved by larger resection margins that often generate the need for skin grafting. The current NCCN guidelines recommend excision with a 1- to 2-cm margin down to fascia or periosteum (level III evidence). The creation of surgical wide margins must be balanced with the potential morbidity of this surgery. For instance, would it be appropriate to resect upper and lower eyelids for a Merkel cell carcinoma of the lateral cheek? The rate of local recurrence, even in the face of wide local excision, ranges from 25% to 40%. Sentinel node biopsies are routinely recommended.
Periodic follow-up with local, regional, and systemic monitoring is recommended. Although most recurrences occur in the first 2 to 3 years, survival is best predicted by the presence of nodal and distant disease. The behavior of Merkle cell cancer is usually more aggressive than significant invasive melanomas, and in fact, the mortality exceeds that of melanoma. The histologic diagnosis is confirmed by performing immunohistochemical staining (IHC). Because of the frequency of local failure in treatment when surgical margins are compromised, some have recommended postoperative radiation therapy for the primary tumor, draining lymphatics, and regional lymph node basins. The added value of this treatment when surgical margins are generous, negative sentinel lymph node biopsy is obtained, and PET scans are negative has not been established. Adjuvant chemotherapy for regional disease has also been recommended (positive regional node disease) in addition to surgery, but its value is unproven. The diagnosis of Merkle cell cancer portends a high risk of local, regional, and distant recurrence. The 5-year disease-specific survival ranges from 60% to 87% for local disease, 39% to 62% for nodal disease, and 20% for distant metastatic disease. The median time to recurrence is 7 to 9 months, with 80% to 90% of recurrences occurring within the first 2 years. The draining nodal basin is the most common site for recurrence. Because of the high recurrence rates, patients should be evaluated every 3 to 4 months for the first 2 years following treatment.
Dermatofibrosarcoma protuberans (DFSP) is a rare intermediate-grade soft tissue sarcoma that usually features indolent irregular, trabecular, and infiltrative growth with projections a far distance from the primary tumor. The final diagnosis is often delayed because of the innocuous appearance of the initial tumor. This will often lead to inadequate resection and recurrence. Approximately 85% to 90% of DFSPs are low grade; the remainder contain a high-grade sarcomatous component (which is usually a fibrosarcoma, designated DFSP-FS) and are considered to be intermediate-grade sarcomas.
The age range of affected individuals is usually between 20 and 50 years of age. Most lesions are located on the trunk or proximal extremities, with the head and neck area affected less commonly. Some patients provide a causal history of trauma; however, the relationship has not been proven. The lesion is slowly growing as a rule (and for practical purposes does not metastasize); however, it has a strong tendency for local fascial extension along tissue planes in small, thin fingers, that defy clinical and pathologic perception. Standard surgical excisions without Mohs pathology have yielded up to a 60% recurrence rate, generating the recommendation that 3- to 4-cm clinical margins or better Mohs surgery be utilized.
The DFSP presents clinically as a painless, firm, red to brown or purple plaque. It can demonstrate soft areas or protrusions. This indolent presentation is often enough to prevent early diagnosis because of its benign nature. The diagnosis can prove difficult, and differentiating it from other fibrotic skin processes can require special stains. Negative biopsies in one area should not be reassuring but should prompt more biopsies and/or complete excision of a suspicious lesion. Immunostaining with CD-34 factor, XIIIA metallothionines, tenascin, and stromelysin-3 are helpful in making the diagnosis. The lesion is frequently misdiagnosed both clinically and pathologically and underestimated surgically.
Treatment for DFSP is surgical excision with wide margins, and up to 3 to 4 cm have been recommended. Currently, Mohs micrographic surgery is the surgical method of choice, which has reduced recurrence rates to about 2%. Regional lymph node biopsies are not indicated because the incidence of metastatic spread is so low, probably 1%. Although adjuvant radiation has been recommended following standard surgical treatment, the value of supplemental radiation in patients who have widely negative surgical margins has not been established.
Lymphomas may present as cutaneous lesions and include mycosis fungoides and Sezary syndrome, which are cancers originating from T4-helper cells. These are rarer tumors that appear as a reddish patch, plaque, or lump. They are sometimes oozing or have a “psoriatic” appearance. Itching is a prominent clinical symptom. Because of its benign appearance, diagnosis is commonly delayed because it overlaps in appearance with many other cutaneous dermatologic conditions. Some patients progress to frank systemic lymphoma over time, and 5-year survival rates of the skin lesion itself vary from 50% to 100% and often relate to the development of another internal serious blood neoplasm.
Therapy varies from medication such as Rituxan to excision, chemotherapy agents, or topical radiation, such as skin electron-beam therapy. In some cases, surgery provides control of local symptoms and apparent cure for limited lesions.
This rare and aggressive vascular tumor of the skin generally occurs as a rapidly enlarging nodule or plaque, sometimes simulating a bruise. In-transit metastasis occurs quickly, and initial presentation with satellite nodules is not uncommon. There is a predilection for occurrence in the head and neck area and in the elderly. In one series, the mean age was 70 years and seemed to favor male patients. Aggressive treatment is indicated with 3- to 5-cm margins, frequently requiring flap or skin graft reconstruction. Sentinel lymph node biopsy is recommended. Local and systemic recurrence is the routine because of the tendency for in-transit metastasis, and the prognosis is quite limited, with 20% survival. These statistics and the proven aggressive behavior of this tumor have generated the recommendation that radiotherapy and chemotherapy accompany the planned surgery. Despite these additions, the prognosis remains largely unaffected by additions to the surgical treatment proposed.
Adnexal tumors occur from skin appendages such as eccrine, apocrine, and sebaceous glands as well as hair follicles. Some occur from fibrous or neural tissues. Sebaceous carcinomas usually present as a slightly reddened nodule whose diagnosis is dependent on biopsy. The prognosis varies with the aggressiveness of the lesion, and wide excision with 2-cm margins to the fascia are recommended. Although some practitioners perform sentinel lymph node evaluations in more aggressive lesions, the value of these interventions remains to be proven.
NMSCs vary in severity from the relatively common and benign varieties to very aggressive and frequently lethal lesions. Treatment favors surgical resection, predicated on accurate diagnosis and classification, with the aggressiveness of treatment based on good pathologic definition of the lesion type and proper margins.
The authors wish to express their appreciation for the contributions of the panels who created the NCCN guidelines, which form an excellent and carefully considered basis for the recommendations for diagnosis and treatment of these lesions that are summarized in this chapter. The authors recommend these guidelines in the strongest terms for those wishing further information and clarification as they serve as a yearly, up-to-date summary of current recommendations for diagnosis, evaluation, and treatment.
Melanoma is a malignant neoplasm that arises from melanocytes, most often as a cutaneous lesion, but primary tumors also arise in other tissues where neural elements penetrate. Although melanoma accounts for approximately 5% of all cutaneous malignancies, it is the deadliest form of skin cancer, responsible for greater than 90% of deaths. It is the third most common malignancy responsible for death, and its rate of increase is much higher than for other common cancers except breast cancer. Melanoma is the ninth most common malignancy.
Ninety percent of cases present with local or regional disease, for which the primary treatment is surgical and potentially curative. Current surgical approaches have reduced disease extent and morbidity compared with historical techniques but have equivalent or superior effectiveness.
Melanoma is a rare disease, with an incidence of 22.1 per 100,000. Melanoma misdiagnosis accounts for more pathology and dermatology malpractice claims than any other cancer except breast cancer.
Prognostic factors relate to thickness, location, ulceration, and the presence of lymphatic and nodal disease. It may spread unpredictably to any organ. Melanoma also has a proclivity for arising in middle-aged adults compared with other cutaneous malignancies, which tend to appear in later life.
Given the substantial risk for morbidity and mortality, a thorough understanding of diagnosis, initial management, and subsequent follow-up care is important for surgeons who encounter melanoma in practice. Although there is an increasing trend toward nonsurgical treatment of melanoma in this era of advancing immunotherapy and targeted molecular therapy, we believe that identification by inspection and surgical tumor extirpation remains the backbone of comprehensive initial melanoma care. Thankfully, for the great majority of affected individuals, this is all that is necessary.
There has been an alarming increase in the incidence of melanoma since the 1950s, especially in young Caucasian women. At that time, the incidence was approximately 1 per 100,000 in the adult population in the United States. Compare this to recent estimates reporting the incidence to be closer to 40 per 100,000 for the same population. This makes melanoma one of the fastest-growing neoplasms. Experts argue whether this is the result of heredity, environment (sun), or overdiagnosis.
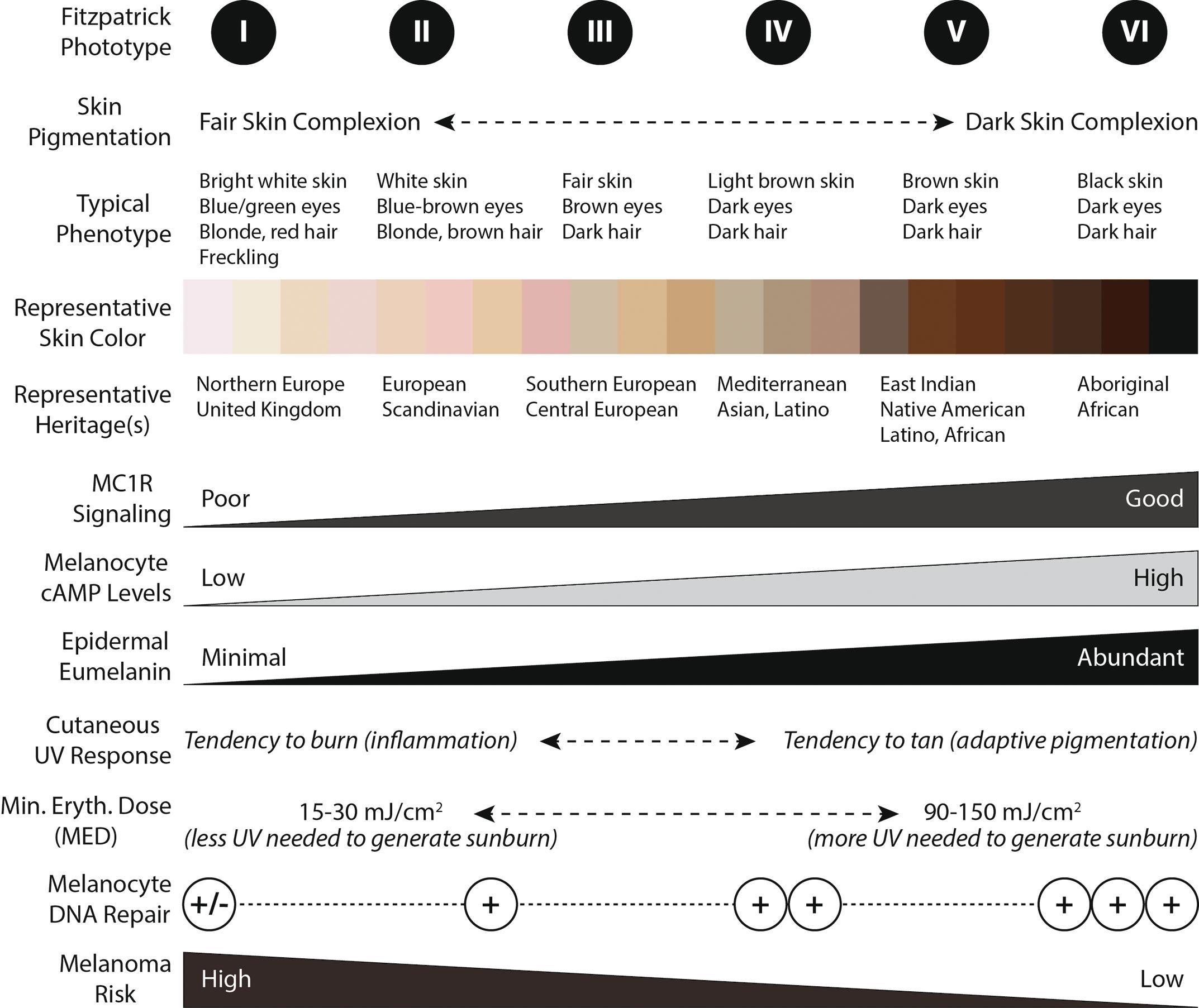
Substantial time and resources have been invested in identifying risk factors for the development of melanoma. From a patient-level perspective, relevant risk factors are lightly pigmented skin, red hair color, and an inability to tan/tendency to sunburn easily (Fitzpatrick types I and II; Fig. 1 ). Environmental factors include cumulative dose of ultraviolet (UV) radiation (either from chronic sun exposure or intense intermittent sun exposure), use of UV tanning beds, and immunosuppression. Important precursor lesions are dysplastic nevi. Finally, recent research has highlighted the important role that genetics play; numerous single-gene mutations have been implicated along with the classically associated DNA repair conditions such as xeroderma pigmentosum. Oncogenesis is thought to be based on gain-of-function mutations in oncogenes and loss-of-function mutations in suppressor genes. Two-thirds of the risk generation is felt to come from UV light exposure (natural or man made), and reduction of UV exposure, particularly sunburns, results in decreased frequency. The sun-drenched head and neck contains 9% of the body surface but accounts for 30% of melanoma cases. Whereas keratinocytes subjected to UV damage undergo programmed cell death, melanoma cells survive this insult and may change their characteristics toward malignant behavior.
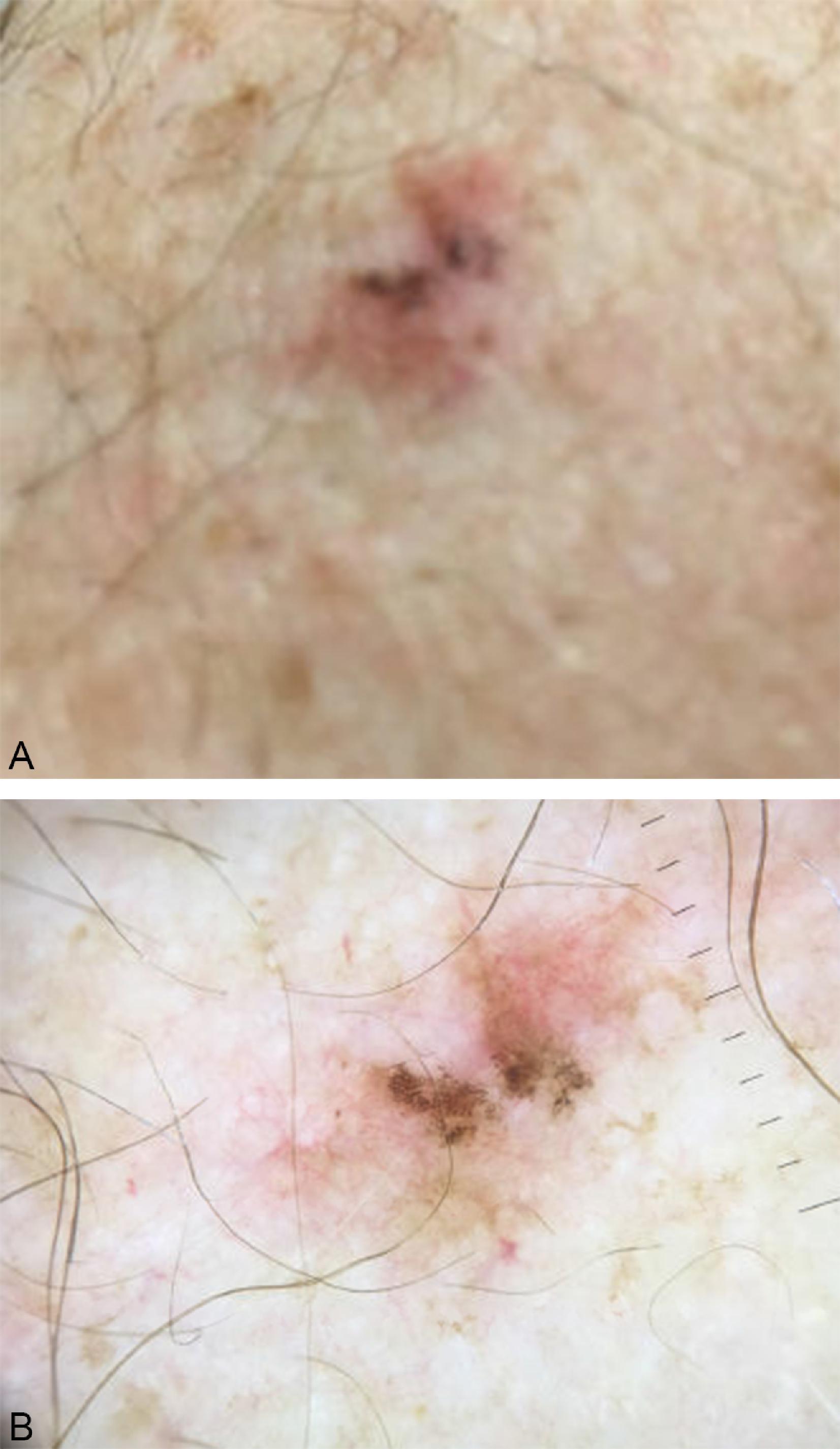
Important precursor lesions exist in melanoma in 30% of cases, some of which impart different risks for the subsequent development of malignancy. Currently the bulk of melanoma is felt to arise de novo. Acquired melanocytic nevi are considered the most closely related pathologic entities to melanoma; development of >100 acquired melanocytic nevi imparts a tenfold increase in risk for developing melanoma. Atypical melanocytic nevi (AMN) have both sporadic and dominant forms, as the development of >5 atypical melanocytic nevi carries a fivefold increase in risk for melanoma ( Fig. 2 ). Finally, solar lentigines, when found in groups, are responsible for a fourfold increase in melanoma risk. Other risk groups include FAMMM (familial atypical multiple mole syndrome), AMS (atypical mole syndrome), DNS (dysplastic nevus syndrome), congenital nevi, and cellular blue nevi; all increase the frequency of malignancy. Elder in 1985 extended the theory of nevus-melanoma transition, postulating dysplastic nevi as a precursor to sporadic melanoma. Dysplastic nevi exist in 2% to 8% of the population.
The plain fact, accounting for the challenge and difficulty of the diagnosis both clinically and pathologically, is that there are no ubiquitously agreed-upon criteria by which to make the diagnosis. Seventy percent of melanomas detected are now less than 1 mm, and many of these smaller lesions are in a diagnostic gray zone. Pathologists are increasingly evaluating lesions in a diagnostic “overlap” zone in which penalties exist for underdiagnosis but not overdiagnosis. Recently, in order to prove that a part of the increased frequency of melanoma is generated by overdiagnosis, a group of pathologists were asked to re-read their own slides from 20 years ago, and there was a significant increase (60%) in diagnoses of melanoma versus benign/dysplastic nevi. The conclusion is that a portion of the increased frequency of melanoma is physician generated by threat of litigation for “underdiagnosis.” This fact would also account for some of the increased survival.
There are two forms of melanin produced by melanocytes: the black/brown pigment eumelanin, and the red/yellow pigment pheomelanin. The ratio of these compounds produces skin color, rather than the absolute number of melanocytes. Eumelanin is a better UV shield, whereas pheomelanin is less protective and has a higher DNA damage in response to sunlight.
Melanoma is an umbrella term that encompasses a collection of heterogenous neoplastic lesions arising from melanocytes. Understanding the major differences among these entities is paramount to providing appropriate surgical care. The heterogeneity of forms makes clinical and histologic diagnosis difficult. Melanomas of the skin start from the basal layer of pigment cells. They first grow horizontally or radially within the epidermis. In this stage, they are at first melanoma in situ (see Fig. 2 ) and are not invasive. This stage may be long or short. They then may change their surface antigens and cytogenetic profiles to develop a vertical growth phase. Steps in the process may be rapid or slow. As melanoma invades the mid-dermis, it reaches the area where blood vessels and lymphatics are present, and metastasis is then possible, either into the adjacent skin (in transit metastasis) or regionally and distally through lymph or blood.
This is the most common subtype of melanoma, accounting for approximately 75% of all cases ( Fig. 3 ). Although the risk of any individual nevus progressing to melanoma is very low, nevi that progress to malignant transformation manifest as superficial spreading lesions most often. They have first a radial and then a vertical growth phase; 30% begin in a preexisting nevus, but the majority arise de novo. The majority of superficial spreading melanomas are observed on the trunk and extremities. Classically, these lesions develop with the widely known ABCDE characteristics: asymmetric shape, irregular borders, uneven/multiple colors, diameter in excess of 6 mm (though 50% of diagnosed lesions are now smaller than that threshold), and unpredictable evolution in time. Because smaller lesions are frequent, this criterion should now be modified. A very common history provided by the patient will be a period of slowly progressive expansion in a radial direction followed by comparatively rapid vertical growth, usually causing a papule or nodule. These changes relate to growth limited to the epidermis followed by invasion more deeply into the dermis. Partial regression with corresponding loss of pigment, interval pale and atrophic areas, are also common.
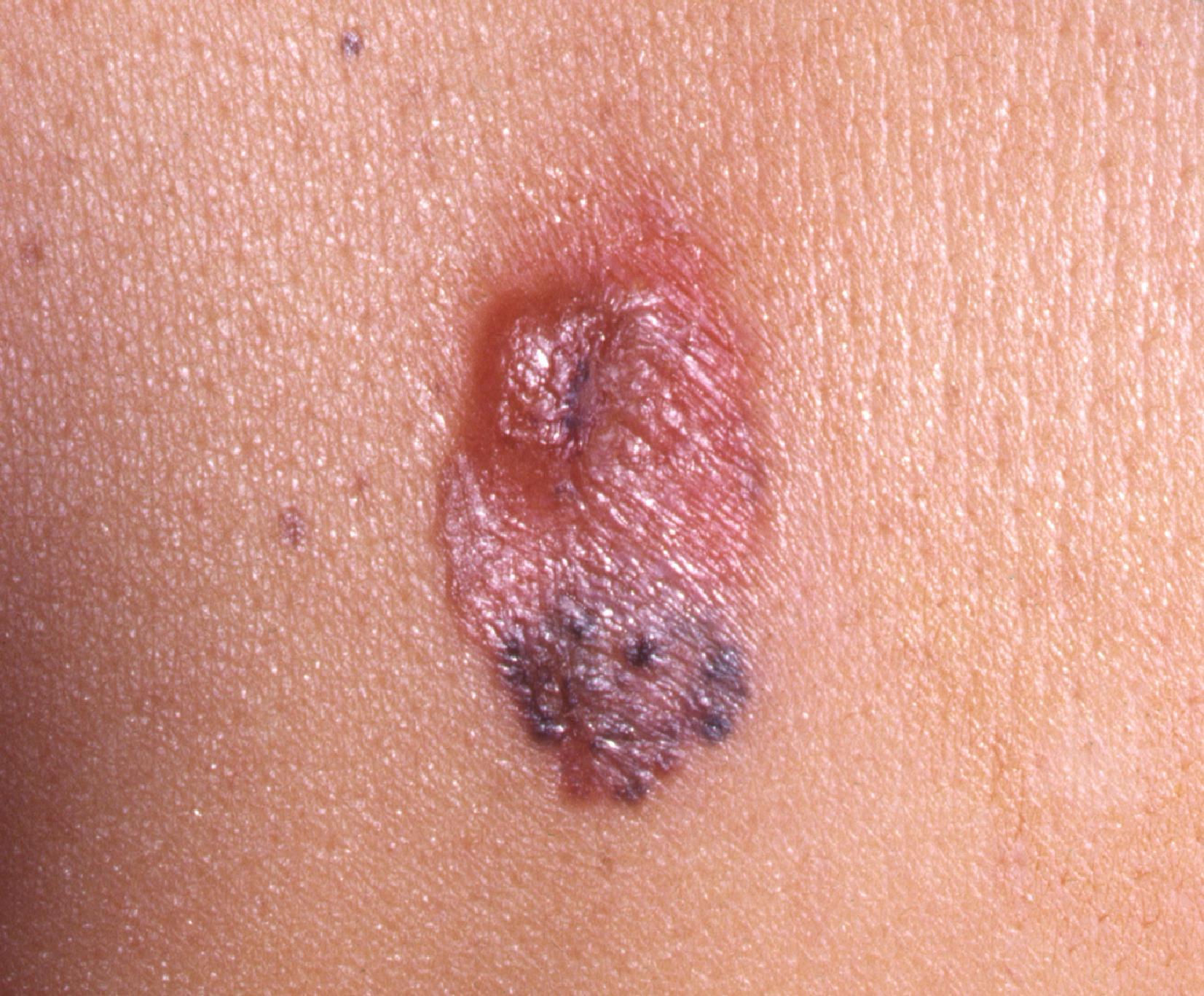
The second most common subtype of melanoma is the nodular melanoma, identified in 15% to 30% of all melanoma cases ( Fig. 4 ). The physical manifestation of nodular melanomas can be variable, making clinical diagnosis challenging. The ABCDE criteria are less applicable because nodular tumors may be symmetric or asymmetric; pink, red, blue, black, and nonpigmented lesions are all well described; the tumor often appears as an isolated nodule or papule without an ensuing increase in size, and the timeline for growth is variable. Much of this clinical variability arises from the pathologic behavior of the tumor, namely that it lacks a superficial-radial phase and instead tends to grow almost exclusively in a vertical fashion. Much like an iceberg, the majority of the neoplasm often exists beneath the visible surface of the skin. A high index of suspicion is required to pursue further workup and accurately identify this tumor. Nodular melanoma is often diagnosed at a later stage than other subtypes of melanoma.
Lentigo maligna melanoma is the third most common form of melanoma (10%–15%) and the subtype most strongly linked to chronic UV irradiation ( Fig. 5 ). As a result, these lesions are almost exclusively identified in older adults in areas of chronic sun exposure, such as the distal extremities and the head and neck region. The clinical examination classically features a variably pigmented patch in the setting of notable photodamage. Radial growth is often slowly progressive, leading to highly irregular borders and substantial pigment variations. Lentigo maligna melanomas are notorious for exerting a field effect in which surrounding dermis has suffered the cumulative mutagenic effects of chronic UV irradiation. This allows the primary tumor to infest its vulnerable surroundings with foci of malignant cells, termed skip lesions. Incisional biopsy specimens may fail to identify malignant cells surrounding the clinically obvious melanoma, placing patients at higher risk for recurrence, reoperation, or advanced disease. Negative margins at the time of definitive surgical excision may also be difficult to interpret as a result of the skip lesions/regression frequent in lentigo maligna melanoma.
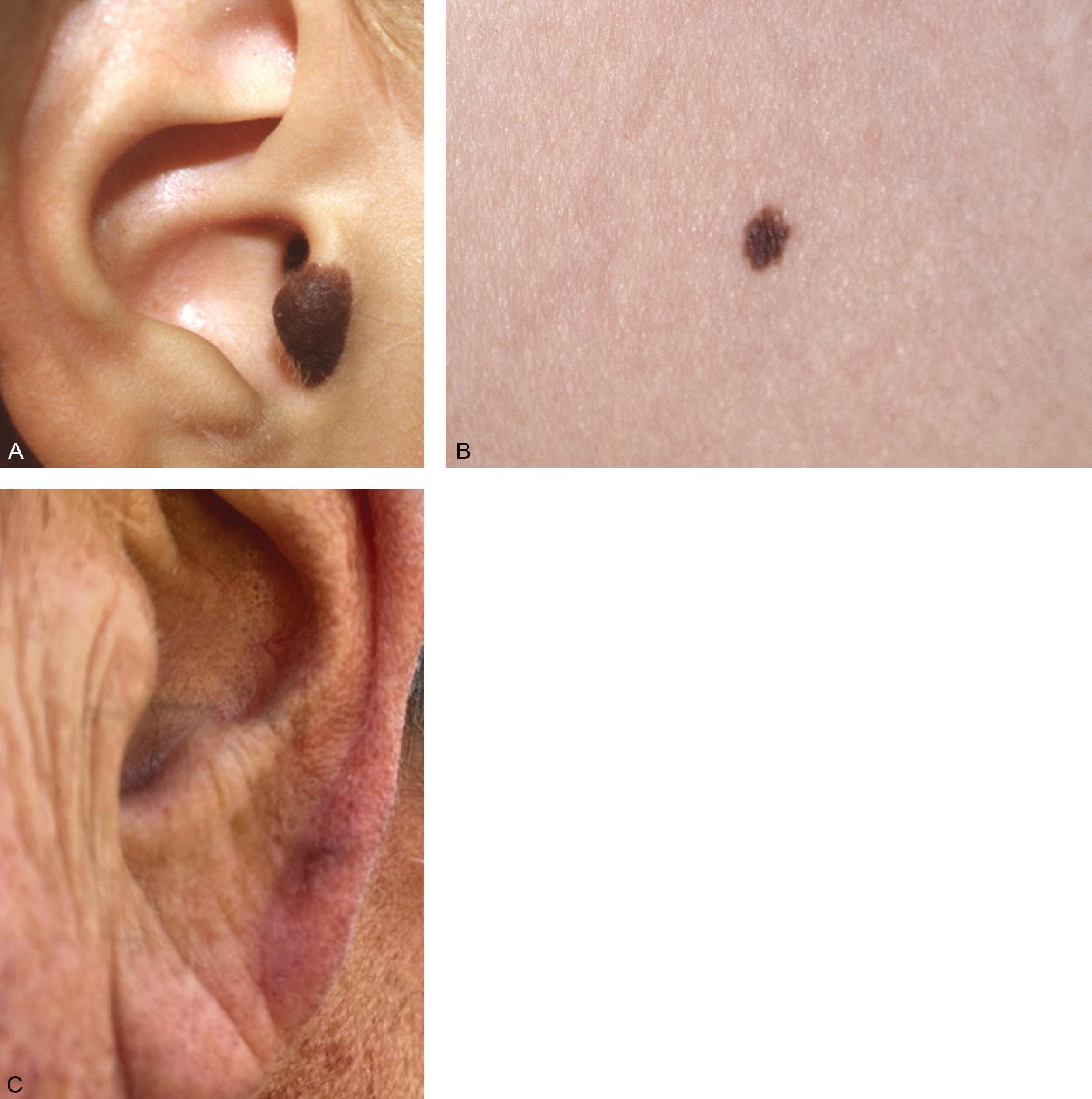
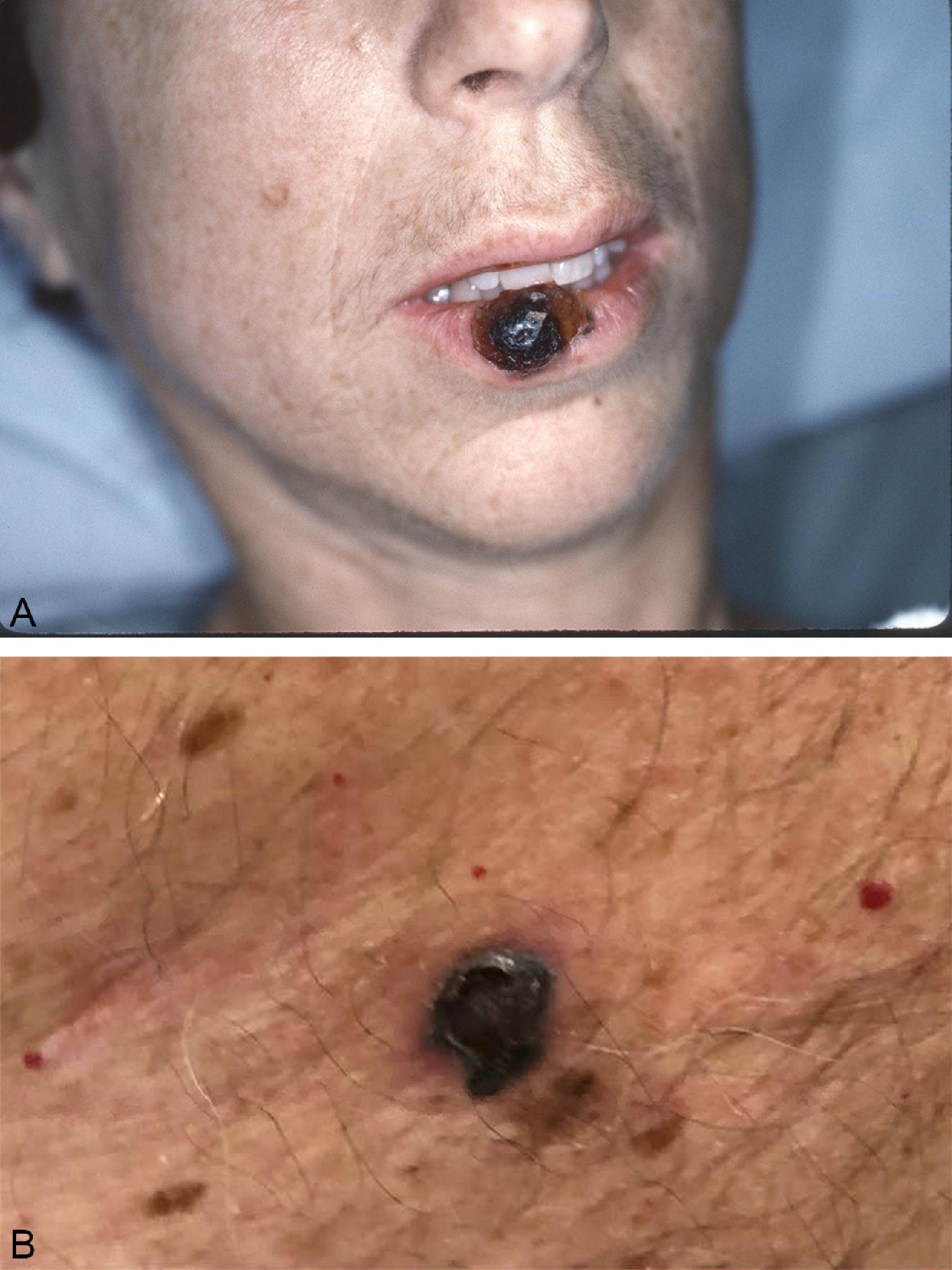
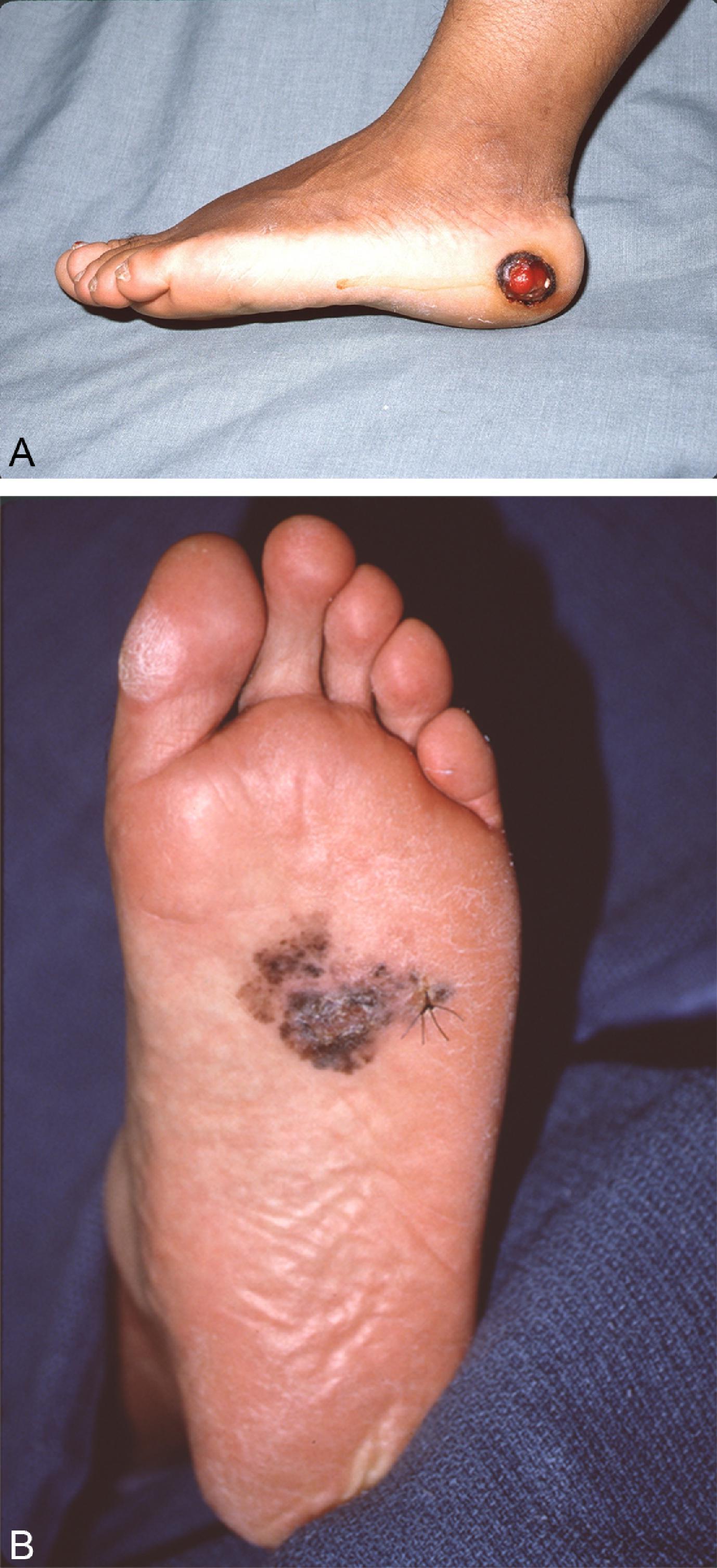
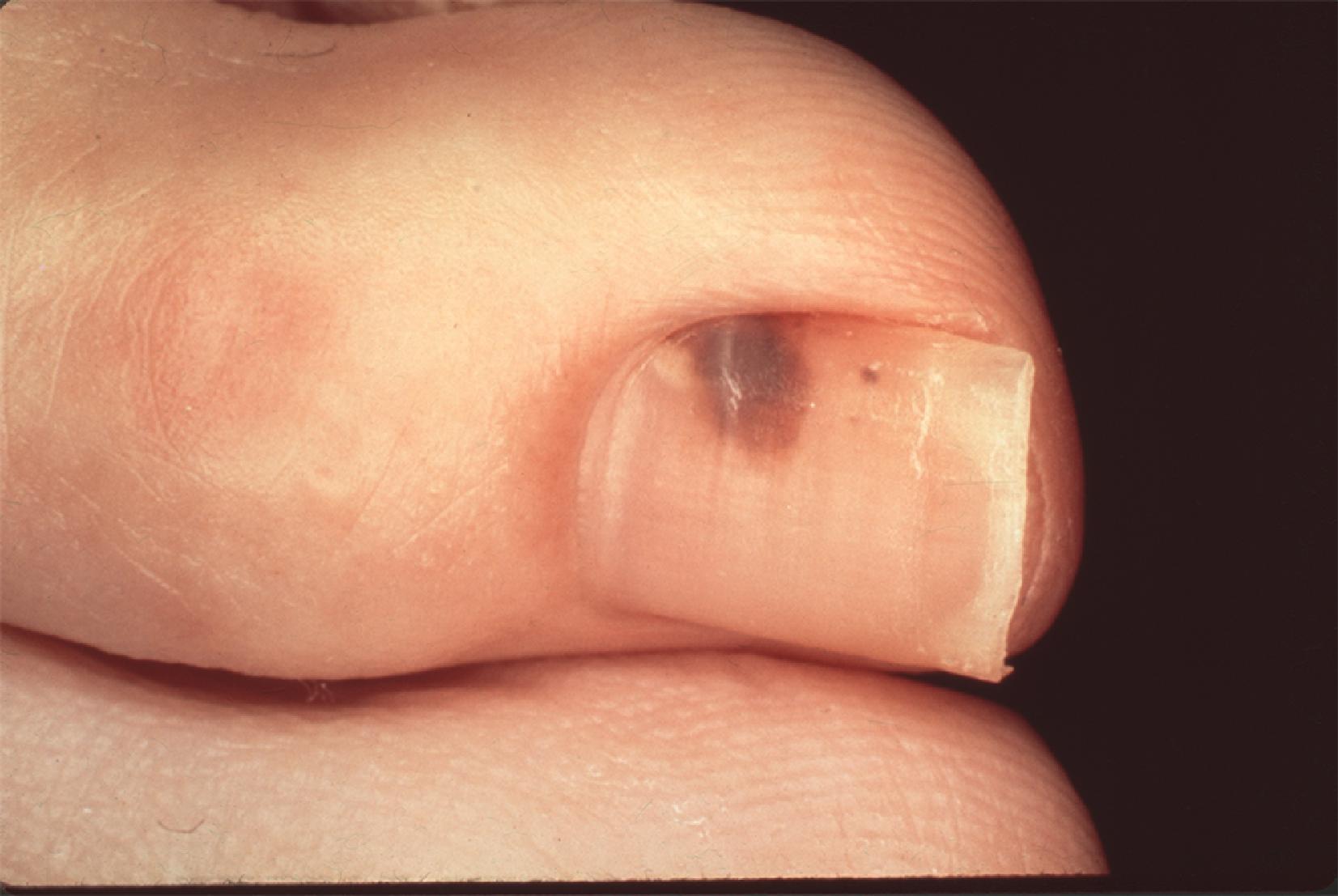
Acral lentiginous melanoma is an uncommon manifestation of melanoma, accounting for less than 5% of cases ( Fig. 6 ). However, these lesions tend to make up a higher proportion of all melanoma cases in dark-skinned populations. Arising in the skin of the palms and soles or intimately within the nail structure, a high degree of suspicion is required for accurate and timely diagnosis. The usual presentation is similar to other melanoma subtypes; usual features include an asymmetric, brown to black macule with variable coloring and irregular borders. With regard to lesions of the nail bed itself, melanoma can be very difficult to distinguish from posttraumatic changes or benign nevi ( Fig. 7 ). A guideline to help identify candidates for biopsy includes subungual lesions in fair-skinned patients, darkly pigmented lesions, irregularly pigmented lesions, those wider than 3 mm, or lesions that extend beyond the nail plate either into the hyponychium, paronychium, or eponychium (Hutchinson sign).
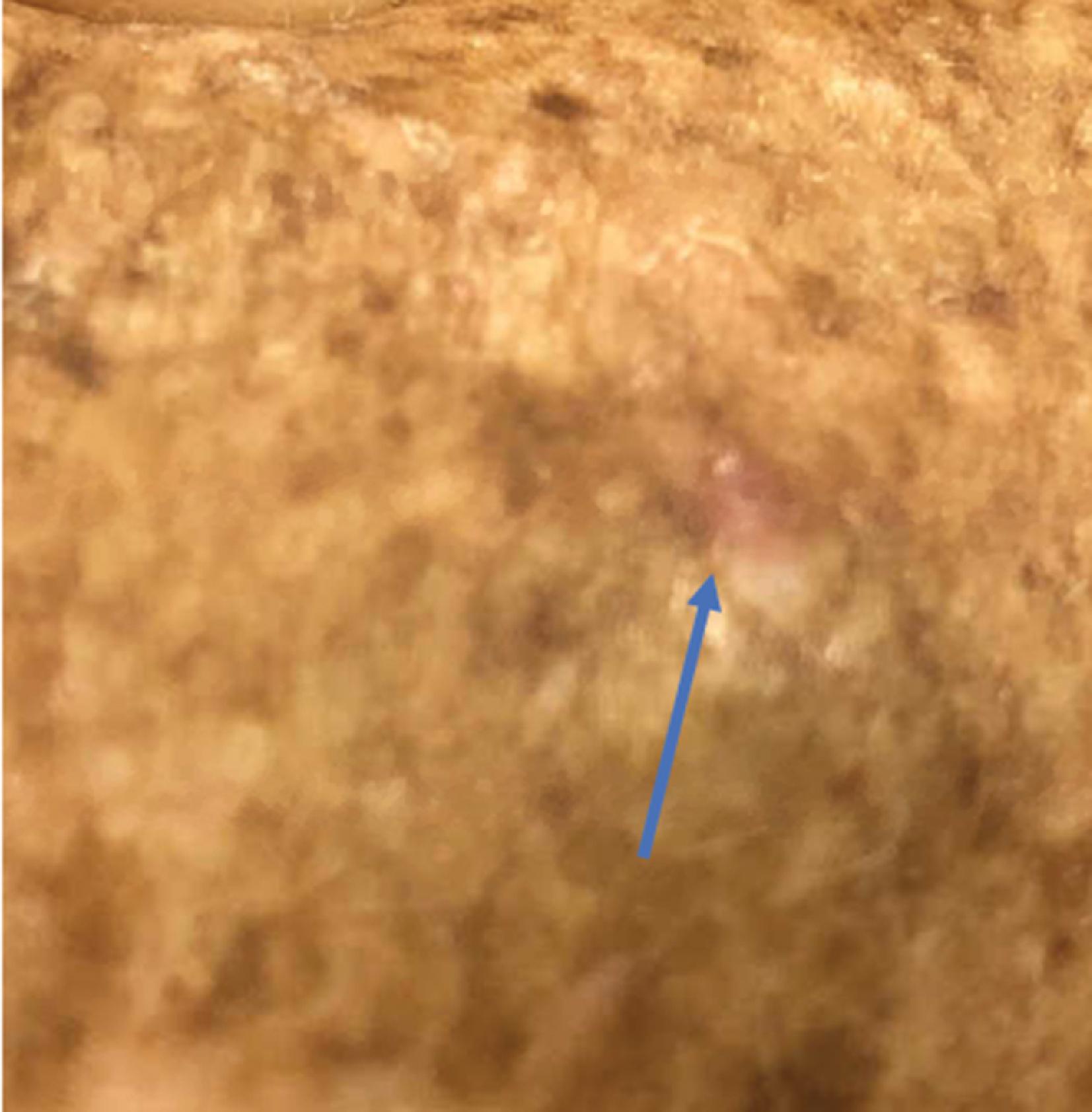
The majority of melanomas are pigmented in some manner, facilitating their detection. However, melanoma without any evidence of pigmentation is also possible ( Fig. 8 ). These lesions appear similar to a squamous cell carcinoma or a wart, and they may be red or occasionally pale. There should thus be a low threshold to biopsy unidentified lesions. Repeat observation of lesions in this category will reduce diagnostic errors, and clinicians should not hesitate to perform a full-thickness skin biopsy recognizing the atypical appearance of 10% of the subset of all melanomas. Odd lesion colors or the lack of pigment have no impact on prognosis.
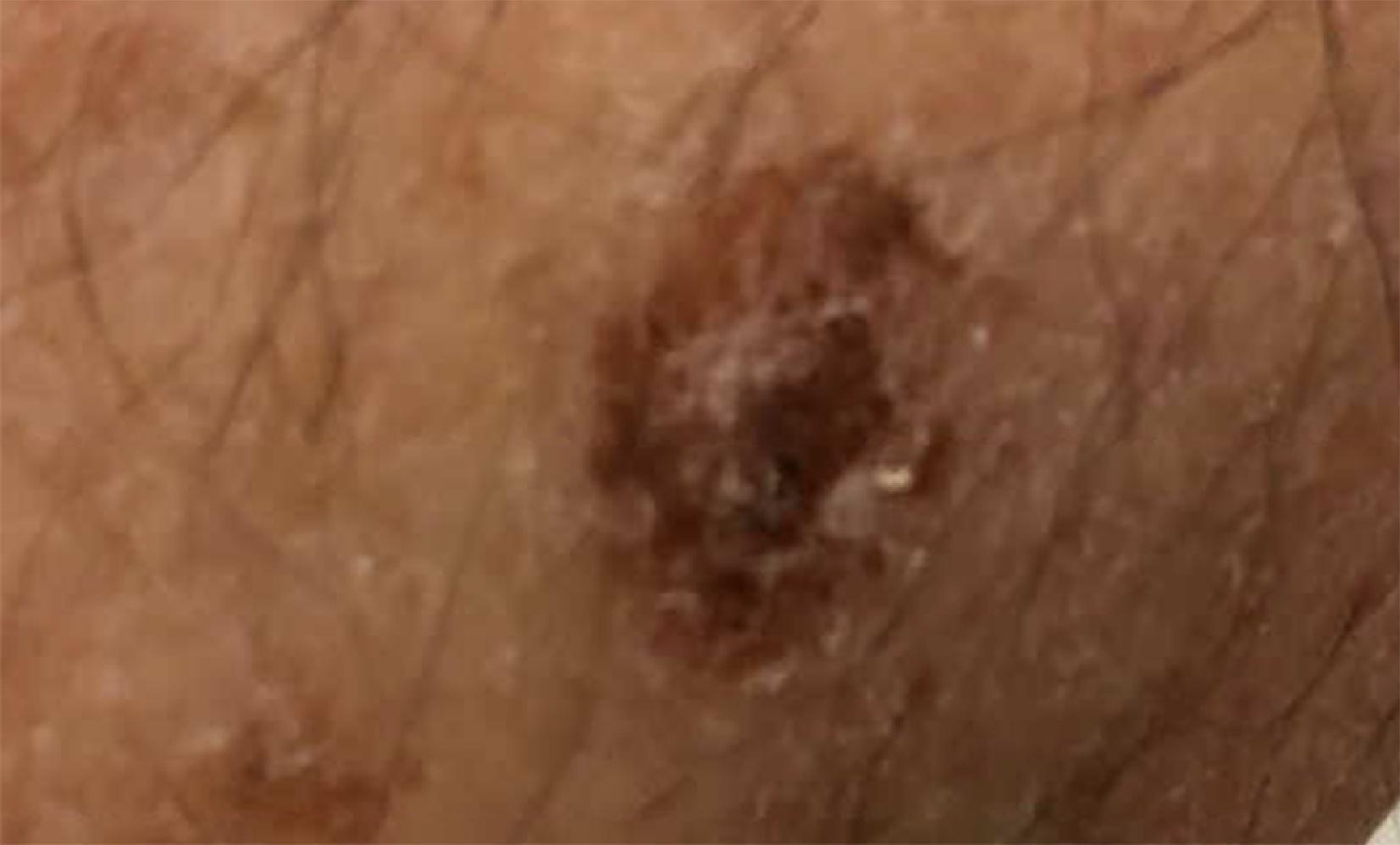
Desmoplastic melanoma is a particularly notable variant as this lesion poses substantial risk for destructively aggressive local growth and recurrence as a result of inadequate resection ( Fig. 9 ). Because it has many of the appearances of a keratosis, the lesion is easily missed. The most frequent clinical manifestation of desmoplastic melanoma is a flesh colored plaque in sun-bathed skin, though red, pink, brown, or black lesions are possible. Exact identification of desmoplastic type is exclusively a histologic diagnosis. Identification and classification require full-thickness tissue sampling as the superficial layers of the lesion are classically nondiagnostic or vaguely resemble fibrosis, cutaneous lesions, or even scars. Some feel that, because of the depth of the lesion, traditional depth-based staging may overestimate the seriousness of the tumor; this melanoma variant seems less likely to metastasize to nodes or beyond, and it is usually discovered at a later stage. (Warning: Fully 10% of melanomas are not easily identifiable by their presenting color or clinical appearance characteristics. Adequate biopsy remains the single confirmation of pathologic type. Remember that pathologic identification may also be challenging.)
Clear cell sarcoma is a very rare tumor that recently has been identified as a molecular relative of melanoma. Most clear cell sarcomas arise from tendon sheaths and fascial aponeuroses, especially in the lower extremities; the trunk is the second most common site for tumor growth. Clinically, the lesion is almost always noted to be a tender flesh- or gray-colored mass that ranges in size from one to several centimeters. A hallmark is intimacy with underlying tendon or aponeurotic structures. Metastasis is common, and prognosis is poor.
The family of mucosal melanomas comprises a diverse group of tumors characterized by their anatomic locations and their unique genetic signatures. These tumors develop in the mucosa of the mouth, nasopharynx, larynx, and anogenital regions along the junction of epithelial morphologies. Clinical examination usually discloses a pigmented, sessile, or slightly raised mass, though as many as 40% may be amelanotic. As with several other variants, a low threshold to perform biopsies characterizes a prudent approach.
The staging of melanoma is based on the American Joint Committee on Cancer (AJCC) staging system, which was most recently updated to the eighth edition and considers tumor size, node status, and metastases. We have provided the citation for the most updated AJCC system and encourage readers to review this. Specifically, T status is driven by tumor size and the presence or absence of ulceration. The N status characterizes both clinically apparent and clinically occult nodal metastases and non-nodal local spread. Finally, M status is determined by the anatomic site of distant spread and the serum lactate dehydrogenase level. Melanoma staging is vital for providing accurate prognosis.
Prognosis determination proceeds directly from staging. Four tumor features have been specifically repeatedly associated most closely with prognostic outcomes:
Primary tumor thickness
Presence of ulceration in the primary lesion
Mitotic rate of tumor cells
Presence of microscopic metastasis on analysis of resected lymph nodes
Survival varies from >95% at 10 years for promptly treated stage 1 tumors to <20% for metastatic stage 4 disease. These figures may be changing, however, in the era of advanced immunotherapies and targeted molecular therapies.
A detailed history and physical examination are both very important in the initial encounter for a patient presenting with suspected melanoma. Specific historical elements that should be collected include risk factors for melanoma (see earlier) and family history of cutaneous malignancies. Questions pertaining to the lesion should include time course (including possible precursor lesions present at birth or in development thereafter), circumstances surrounding lesion development (e.g., in a preexisting nevus, following trauma), associated dermatologic symptoms such as itching or burning, and the presence of constitutional symptoms, ulceration, or bleeding.
A total body skin examination is a must, especially if there are numerous melanocytic nevi or clinically atypical appearing nevi. It is very frequent that additional melanomas or melanoma precursor lesions will be discovered. Some dermatology experts utilize dermoscopy (examination with a magnified light device) in screening for atypical nevi.
Particular attention should be paid to the examination of local/regional lymph nodes. Clinically palpable nodes must be further evaluated, as one-half of the clinically palpable nodes are positive, and one-half are negative for melanoma involvement, emphasizing the importance of ultrasound or sentinel lymph node biopsy (SLNB).
Routine laboratory and imaging studies were formerly a cornerstone of preoperative workup in cases of suspected melanoma. Recent meta-analyses have determined that the diagnostic utility of these measures is minimal. Up-to-date recommendations from several national societies, both in the United States and elsewhere, do not include these evaluations for asymptomatic patients. For patients with concerning pathology or signs or symptoms of metastatic disease on presentation, targeted workup (e.g., head imaging for a patient with headaches and suspected central nervous metastasis) should be pursued and may be performed in coordination with a medical oncologist or other specialist.
Excisional biopsy is the cornerstone of diagnosis in cases of suspected melanoma. The recommended technique is excisional biopsy with 1- to 2-mm margins, followed by straight-line closure with minimal undermining. If needed, closure with allograft skin or negative-pressure therapy devices are lesser options while awaiting final pathology. All samples should be appropriately oriented and sent for pathologic review. Ideally, a pathologist with specific expertise in the review of melanocytic neoplasms is identified for consultation.
Excisional biopsy is considered ideal because it allows for an accurate assessment of tumor architecture, including Breslow depth, mitotic rate, invasive characteristics, and identification of the deepest areas for classification. This method further reduces the error of a sampling bias that may occur with melanoma arising in a previously benign nevus or in cases of Lentigo maligna melanoma. An extremely important principle of biopsy technique is orientation of the incision. The surgeon’s mindset should be that all suspicious lesions are malignant until proven otherwise, implying that wider resection may eventually be needed. The subsequent resection, therefore, must necessarily remove the scar of the biopsy incision plus the appropriate margins, which is best accomplished when the biopsy excision has been properly planned with the possibility of later excision in mind. On extremities especially, careful planning of the initial biopsy incision is crucial for facilitating tissue salvage in a radical future operation. Therefore, virtually all extremity incisions should be oriented longitudinally and within the relaxed skin tension lines (RSTLs) in a proximal-to-distal fashion, and not transversely (medial to lateral), as depicted in Figure 10 . Some favor making the long ellipse of the skin excision parallel to the direction proximally of presumed lymphatic drainage to have a chance of capturing more lymphatics in the long axis of the excision.
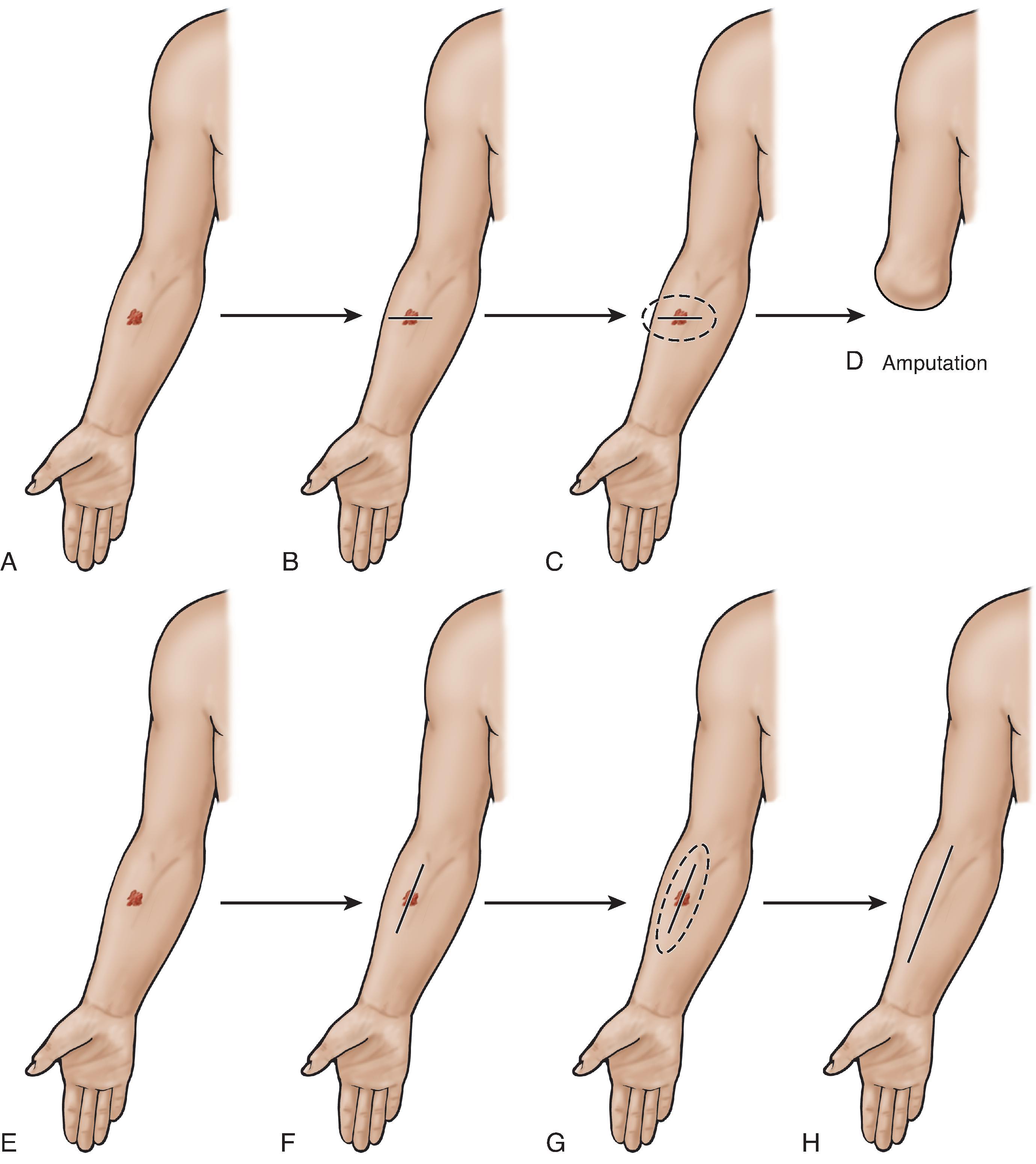
If excisional biopsy is not a feasible option, full-thickness skin punch biopsy is the next best modality. Shave biopsy is discouraged because it does not permit conclusive review of tumor depth, which all too frequently cannot be accurately assessed once scar tissue from the shave biopsy is present. The biopsy process itself almost always obscures future attempts at depth determination. Saucerization is a partial-thickness skin sampling and is also discouraged.
Biopsy reports must include several crucial pieces of information: pathologic diagnosis (ultimately, the pathologist must confirm the suspected clinical diagnosis or provide an alternative diagnosis); tumor thickness (otherwise known as Breslow depth ) must also be measured accurately to the nearest 0.1 mm; presence or absence of ulceration; the positive or negative status of both the deep and peripheral margins; and the mitotic rate of the lesion. Additional information includes any histologic subtyping presence or absence of vascular, lymph, or nerve invasion character of the inflammatory infiltrate and the presence or absence of microscopic satellite lesions. Immunohistochemical staining techniques also provide further characterization of tumors, but as yet are not of consequence to surgical planning. The Clark level may also be mentioned, but please note that the Clark level is clinically inconsequential in modern practice. Instead, the Breslow depth is used.
Surgical excision is the principal treatment for cutaneous melanoma. There has been a trend away from radical excisions with very wide margins and extensive lymphadenectomy to conservative excisions with targeted lymphatic tissue removal. Results from important randomized, controlled trials have shed light on critical questions pertaining to surgical care, but many important components of surgical resection are still practiced based on traditional surgical experience.
The purpose of surgical excision is to provide complete excision of the lesion and its immediate surrounding area to provide durable, recurrence-free survival in the local area. Removal of tissue adequate to ensure elimination of the primary tumor, surrounding nests of malignant cells (microsatellites), and even closely situated second primary tumors is the major goal. Currently, there is a strong desire to avoid debilitating or disfiguring wide resections, large wounds that may necessitate advanced reconstructive techniques, or wounds of such size or location as to predispose to an unnecessarily high risk of surgical site infection, or cause disability. A number of high-quality, multicenter, randomized, controlled trials have evaluated the optimal margins for surgical excision and concluded that exceptionally wide margins offer no benefit in either survival or in local recurrence. Tumor thickness is the predominant factor that dictates the appropriate margin.
The recommended guidelines for surgical margins are shown in Table 1 . It is crucial to point out that margins represent the distance from the scars of prior excisions and/or biopsies of the lesion or from the edges of any residual tissue that appears to be involved. In other words, if a punch biopsy is performed and there is residual pigmented tissue as well as a thin halo of erythematous tissue around the lesion that appears suspicious, the proper margins should be measured from the edge of the suspicious erythema, not just the pigmented lesion itself.
* For tumors in areas in which 2-cm margins would introduce substantial functional or aesthetic morbidity, 1-cm margins can be considered.
† Individual microscopic tumor characteristics, satellites, and tumor location should be considered, as “wider margins” have a degree of variability.
Despite robust evidence supporting cutaneous margin dimensions when removing melanoma, there is relatively little published on the optimal deep margin. Thus, by tradition, the deep margin is typically through one extra tissue layer of clear uninvolved tissue (frequently to or less commonly including the muscle fascia or deep fascia). Although some surgeons resect fascia, many only resect this layer if the tumor appears to be closely invading the tissue layer above the fascia.
We do not recommend frozen/rapid section analysis of melanoma samples. Although skill can be obtained in the analysis of melanoma samples with stains made for nonpigmented basal cells or squamous cells, there is an error rate with rapid techniques both for margins and depth assessment of tumors. The techniques associated with frozen-section processing have been shown to introduce errors of up to 0.4 mm in the measurement of tumor depth. Surgeons should base their final judgment recommendations exclusively on final pathology measurements in fully stained specimens.
The role of Mohs micrographic surgery is unproven for invasive melanoma but seems valuable in challenging (repetitively positive) margin cases to provide temporary margin assessment. It is useful in those (less frequent) cases of melanoma in situ where the microscopic extent of the lesion exceeds the visible dimensions of the lesion. This assessment, using tissue stains developed for basal cell and squamous cell, is 75% accurate on the immediate Mohs stains for those who practice. Provisional analysis must be confirmed with standard pathologic processing before any reconstruction is accomplished. Less commonly, melanoma in situ is still present on permanent stains after clear temporary margins with Mohs surgery, but it has not been recognized. Society-level recommendations state that Mohs micrographic surgery can be considered for melanoma in situ or lentigo maligna melanoma for cases in which conventional margins may be disfiguring or disabling (e.g., the face) in order to limit extensive excisions. Definitive outcomes in these cases are not currently available.
The exact role of immunostains for margin assessment has not been confirmed. They may highlight extensive disease whose relevance for a particular patient may justify thought before extensive deforming resection is accomplished to clear an immunostain margin.
Our experience has shown that Mohs micrographic surgical excisions of melanoma in situ have a 25% rate of positive permanent margins on follow-up with regular hematoxylin and eosin (H&E) stains, so permanent sections should always be employed for confirmation of rapid margins by Mohs surgery or frozen sections. Reconstruction must be deferred awaiting permanent negative margins.
Historically, patients with all but thin melanoma were treated with early, elective complete lymphadenectomy because experience demonstrated that cutaneous melanoma had a proclivity for nodal and distant metastasis. The passage of time demonstrated that this approach introduced substantial morbidity without measurable reduction in regional or distant metastasis or improved survival for thin tumors. Two techniques have accurately predicted which patients are at risk for nodal and systemic disease: (1) specimen risk analysis outlined earlier with histologic examination and (2) analysis of the sentinel lymph node biopsy or ultrasound biopsy of the nodal basin. The evolution from routine widespread complete lymphadenectomy to targeted sentinel lymph node biopsy permit both approaches to have differing and sequential roles in the surgical management of regional lymphatic areas. Nodes that have a length-to-depth ratio of less than 2, a hypoechoic center, absent hilar vessels, or focal nodularity with increased vascularity are suspicious for tumor involvement.
Physical examination of regional nodes alone is unable to confirm 50% of those with clinically positive nodes, and similarly 20% of patients with clinically negative nodes have or will have microscopic involvement. Metastasis to regional nodes or into the bloodstream occurs when the melanoma reaches the mid-dermis, where blood vessels and lymphatics are present.
Most patients with regional lymph node metastasis on sentinel lymph node biopsy can select nodal observation, ultrasound observation, or completion nodal dissection, as approximately three-fourths of patients with positive sentinel nodes have one to two positive nodes.
Staging of regional lymph nodes may be accomplished with SLNB or with ultrasound-guided sampling for regional lymphatic mapping-sampling, fine-needle biopsy, or excisional biopsy; one or several lymph nodes may be identified for lymphatic mapping.
The sentinel lymph node biopsy is much more commonly indicated than complete lymphadenectomy; the technique has a low false-negative rate, is very important for determining prognosis, and is a basis for surgical or adjuvant therapy or for clinical trials. When nonsentinel nodes in the basin are positive (11%), the prognosis for systemic spread is increased.
At a theoretical level, the sentinel node concept depends on proof that a given area of skin will reliably drain first into one single or perhaps several closely associated nodes within the larger lymph node basin. Sampling the sentinel nodes can thus provide an accurate pathologic examination of the relevant nodes in the basin, assuming that lymphatic fluid from the affected skin patch does indeed pass through the sentinel nodes before encountering the other nodes in the basin. Initially the accuracy of the technique was 75%, but with time and practice the accuracy now approaches 94%. Studies have shown variability in lymphatic drainage over time, and some sites drain to several node basins simultaneously. Some studies have demonstrated that the positive node may not be the node with the highest count, and node excision should define all nodes within 80% to 90% of the highest node sampled. The false-negative rate is 11%, and currently the procedure has a negative predictive value of 97%.
Survival is poorer in patients with thicker primary tumors, ulceration of the primary tumor, or involvement of nonsentinel nodes. SLNB is performed in thick melanomas more for purposes of regional disease control, adjuvant therapy, or investigational drug treatment.
Complications from sentinel lymph node biopsy are much less common than from complete lymphadenectomy; most recent high-quality studies show the rate of surgical site occurrences (e.g., seroma, wound infection, dehiscence) for sentinel lymph node biopsy to be 4% to 10% and for complete lymphadenectomy to be 25% to 40%. Complete lymphadenectomy also carries a substantially higher risk of extremity lymphedema, whereas lymphedema is rare after removal of one or two nodes (less than 1%) and probably reflects prior deficient lymphatics on a congenital or infectious basis. Extremity complications include lymphedema (sentinel node vs. completion lymphadenectomy) (0.7% vs. 12%), wound infection (1% vs. 7%), hematoma and seroma (2% vs. 6%), and sensory nerve injury (0.2% vs. 1.8%). Shoulder dysfunction occurs after axillary dissection, and arm delayed wound healing (16%). Lymphedema (32% overall in complete node removals) is more frequent with inguinal dissections, which have 8% and 51% complication rates for SNLB versus complete inguinal dissections.
The indications for sentinel lymph node biopsy have grown in recent years. Their rationale is to determine prognosis, to select patients who may benefit from lymphadenectomy or additional adjuvant therapy, or to select patients who may benefit from clinical trials. Current recommendations are to perform sentinel lymph node biopsy in all patients with clinically uninvolved nodes and melanoma ≥1 mm in thickness or those with melanoma <0.8 to 1 mm in thickness but with high-risk features (e.g., ulceration, >2 mitoses/mm 2 , histologic evidence of lymphovascular invasion). The status of the sentinel node has been shown to have a profound effect on prognosis. The findings on sentinel lymph node biopsy may significantly change a patient’s stage for a given T status. Patients with a positive sentinel lymph node biopsy are equally likely to have metastases elsewhere in the nodal basin or at distant sites. Moreover, the role of completion lymphadenectomy after positive sentinel lymph node biopsy has been called into more question considering the prevalence of systemic disease when multiple positive lymph nodes are found.
To perform the sentinel lymph node biopsy, a radioactive tracer, technetium-99 sulfur colloid is injected preoperatively into the dermis at the location of the suspicious skin lesion or directly into the scar from the prior excisional biopsy. It is of vital importance to ensure that the injection is done into the dermis because the subcutaneous tissue has a much less predictable lymphatic drainage pattern than the dermis. Radioactivity is then sensed with a scintillation counter as early as 15 minutes after injection of the radiotracer solution and for as many as 6 hours afterward. Simultaneously, a blue dye (isosulfan blue) may also be injected intradermally into the skin lesion in surgery. The dye will also transit via the lymphatics and help visually mark the involved lymphatic vessels and nodes with blue staining. Scintigraphy counts further direct the surgeon to the appropriate lymph nodes in the basin. An appropriately placed skin incision is made, and the combination of blue dye marking and radioactivity counts obtained via intraoperative probe identify the relevant sentinel node or nodes for excision. We inject the lesion with the radiotracer and dye and then perform the sentinel lymph node biopsy before the wide local excision of the lesion or biopsy scar. Some feel that more radical biopsy excisions of the original lesion may change the location of the draining node to a different and inappropriate node for consideration. Node harvest should continue until the count falls below 10% to 20% of that in the highest sentinel node.
In intermediate-thickness melanomas (1.2–3.5 mm), sentinel lymph node biopsy followed by regional lymph node dissection had a potential survival advantage (MSLT I) of up to 23% over those having delayed lymph node dissections when late lymphatic metastases (85% immediate survival vs 62% in delayed node dissection survival). With thicker melanomas, the survival statistics were 71% survival with early node dissections versus 64% observation. Thus, this study indicates there is the possibility of decreased nodal recurrence, decreased distant metastasis, and death with early complete node dissection.
More recent studies (MSLT II) have not shown a significant survival difference at 3 years, but they do show better local control of the appearance of new tumor deposits. This included a 69% decrease in local recurrence, 5% better survival at 3 years, and better disease control (92% vs. 77%). Long-term studies are awaited to highlight and further confirm survival differences.
In thick melanomas (>3.5 mm), these figures in MSLT 1 are 64% survival with immediate node dissection versus 48% in delayed-node dissection survival, with 17% of patients in the delayed group having lymph nodes appear with an average presentation time of 19 months. Thick melanoma sentinel node biopsies had an average 9% relapse rate over time following the original lymph node sampling, where the initial positive rate was 33% and with follow-up reached 42% for delayed presentation of nodes in the sentinel basin.
Low-volume involvement of a lymph node is infrequently accompanied by additional lymph node involvement in the node basin. The number, size, and character of lymph node tumor deposits yields a further classification of sentinel node involvement which predicts safe observation alone for minimal disease. Seventy percent of positive sentinel lymph nodes have single node involvement, and many have peripheral (less threatening location) as compared with central deposits, which indicate early lesions. Thus, many are observing these patients with serial ultrasound examinations of the remainder of the lymph node basin, doing delayed lymph node biopsies or delayed node dissection when indicated for late appearing and nonsentinel nodes. When the largest area in the involved lymph node is less than 0.1 mm, there is a 9% chance of further involvement; when the area is 0.1 to 1.0 mm, there is a 16% chance, and when it is greater than 1.0 mm, there is a 25% chance. One large study showed a sentinel lymph node biopsy group had a 4% late recurrence in the original lymph node basin. After thorough review of the original samples, 80% of those with late positive nodes could be identified in retrospect in the original slides. Immunostains increase detection accuracy of positive nodes as they are able to detect 1 in 100,000 cells versus 1 in 10,000 cells with H&E. Complex staging systems are offered for types and character of lymph node involvement, and observation and immunotherapy are now offered more widely versus completion lymph node dissection. In several studies, long-term disease-free 10-year survival occurred in a retrospective subset of 20% to 40% of intermediate-thickness melanoma patients after regional lymph node dissection.
For patients with clinically suspicious nodes on physical examination, biopsy is still indicated. Only 50% of these are positive. Most often, ultrasound-guided fine-needle or core biopsy is appropriate, convenient, and 90% accurate. Once the pathologic diagnosis of nodal metastasis is made, complete lymph node dissection (preferred) versus frequent observation are considered to accurately characterize the patient’s N status and manage regional disease. In summary, sentinel lymph node biopsy is not routinely performed for lesions less than 1 mm thick in the absence of aggressive histologic characteristics, but it is considered “therapeutic” for lesions 1 to 4 mm thick and “prognostic” for lesions greater than 4 mm thick. Early positive sentinel lymph node evaluations in intermediate-thickness melanoma occur with a 16% to 20% frequency, with 4% demonstrating late positivity after SLNB. In patients who did not undergo SLNB, late follow-up demonstrates appearance of positive nodes in 17% in intermediate-thickness melanoma, usually at around 20 months. In those who had SLNB and regional lymph node dissection, there was often a small but distinct survival advantage of 10% to 20% in both local control and survival; however, recent short-term studies have indicated that early survival statistics are identical. Lymph node metastases were found in 33% of patients with thick melanomas (>3.5 mm), and a further 9% had an additional late appearance of node metastases over time.
In 2% to 5% of patients, no clear sentinel node can be found; if no sentinel node can be identified, planned completion lymphadenectomy demonstrates 1% to 2% positive nonsentinel nodes. Curiously, late node dissections when interval follow-up demonstrates late appearance of further positive nodes have fewer complications (5% vs. 25% as compared with early lymphadenectomy).
Deep lymph node dissections (e.g., pelvis) may occasionally be individually considered based on the original pathology, despite their increased difficulty and morbidity.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here