Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Caused by three genera of imperfect fungi— Epidermophyton, Trichophyton, and Microsporum —that cause superficial infections involving keratinized tissues such as the cornified layer of epidermis, the hair, and the nails
Dermatophytosis involving different anatomic sites named with site-specific terms (scalp-tinea capitis, beard-area-tinea barbae, face-tinea faciei, trunk-tinea corporis, intertriginous areas-tinea cruris, feet and hands-tinea pedis et manus, and nails-tinea unguium)
Typical lesions present as sharply demarcated annular patches and plaques with an arcuate border
Tinea capitis and barbae present as folliculitis; tinea unguium is characterized by yellow-gray discoloration of nails
Parakeratosis with neutrophils, mild/minimal spongiosis ( Figure 2.1A )
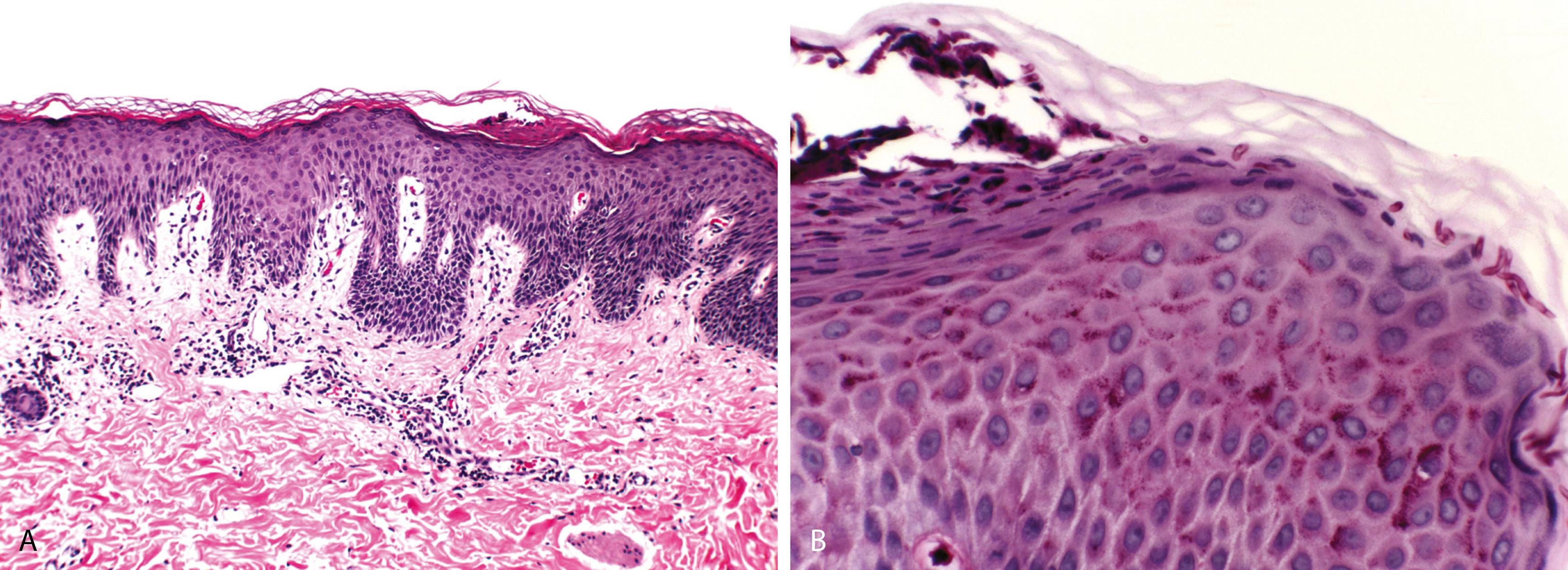
Mild/minimal, superficial perivascular lymphocytic infiltrate
Fungi present as filamentous hyphae, spores, or yeast forms in the cornified layer and in the hair shafts in cases of tinea capitis and tinea barbae (endothrix infection—dermatophytes within the hair shaft, ectothrix infection—dermatophytes confined to the surface of the hair shaft)
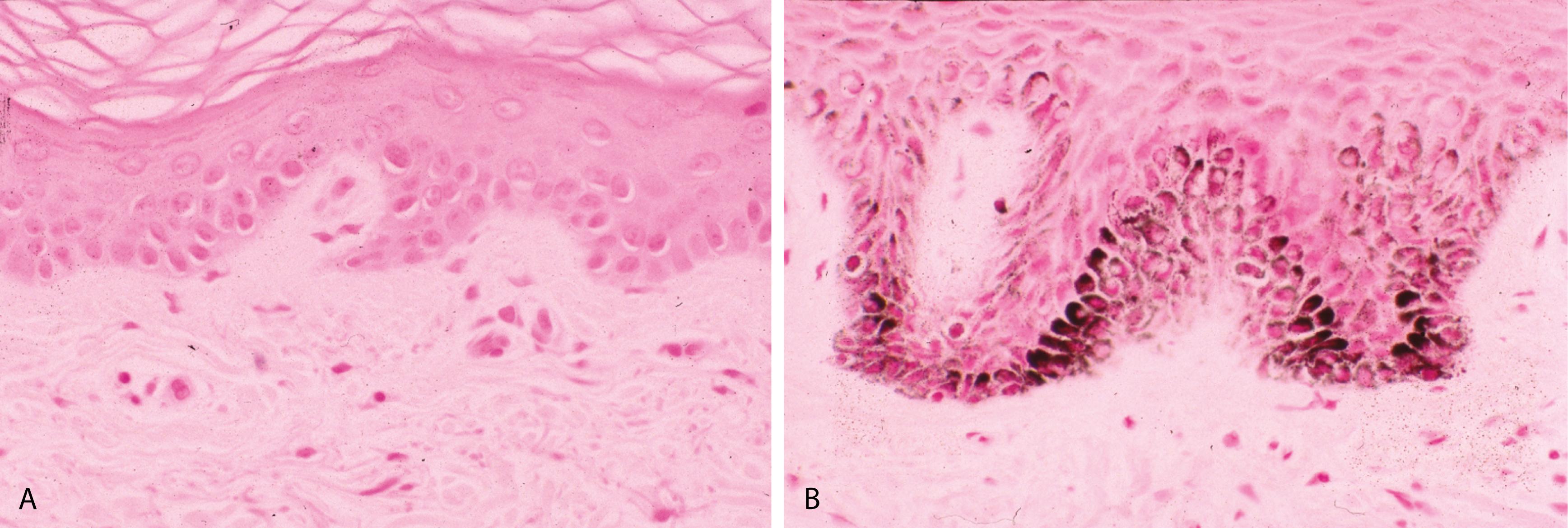
Periodic acid–Schiff (PAS) reaction stains fungi deep red to pink, and Gomori methenamine silver (GMS) stains fungi black ( Figure 2.1B )
Microbiologic cultures useful in speciation of the fungal organisms
Polymerase chain reaction (PCR) techniques particularly helpful for detecting dermatophyte infections of the nail (onychomycosis)
Vitiligo
Decreased density/complete of absence of basal melanocytes
Urticaria
Presence of dermal edema, telangiectases, and eosinophils
Candidiasis
Seen in patients with impaired host response, especially patients with hematologic malignancies and diabetes
Can present as cutaneous, mucocutaneous, or systemic infection
Presence of spongiotic or subcorneal pustules in which pseudohyphae can be demonstrated with PAS/GMS stain
Pityriasis (Tinea) Versicolor Caused by Genus Malassezia
Affects upper trunk with brownish discoloration that may become hypopigmented
Presence of round spores and thick, short hyphae (“spaghetti and meatballs”) recognizable as faintly basophilic, refractive structures in routine hematoxylin and eosin (H&E)–stained sections diagnostic
Identification of fungal organisms on routine H&E-stained sections aided by lowering the microscope condenser, which enhances the refractile nature of the fungi
Fungi in the cornified layer are sandwiched between a lower zone of parakeratosis and an upper zone of orthokeratosis (“sandwich sign”)
The presence of neutrophils in a slightly parakeratotic cornified layer and mild superficial perivascular dermatitis should always prompt a PAS stain in search of fungal elements
Deep folliculitis with intense acute and granulomatous inflammation due to dermatophyte infection is known as “Majocchi granuloma”
Acquired, possibly autoimmune disease with a strong familial association
Characterized by patches of pigment loss (“hypopigmentation”) in skin
Localized disease may show linear, segmental pattern; generalized disease involves face, upper trunk, dorsa of hands, periorificial areas, and genitalia; scalp and eyelashes not typically affected
Stable patches of vitiligo are sharply demarcated and may be surrounded by a zone of hyperpigmentation; in active lesions, areas of total depigmentation may be surrounded by a zone of partial depigmentation and have a slight rim of erythema at the border
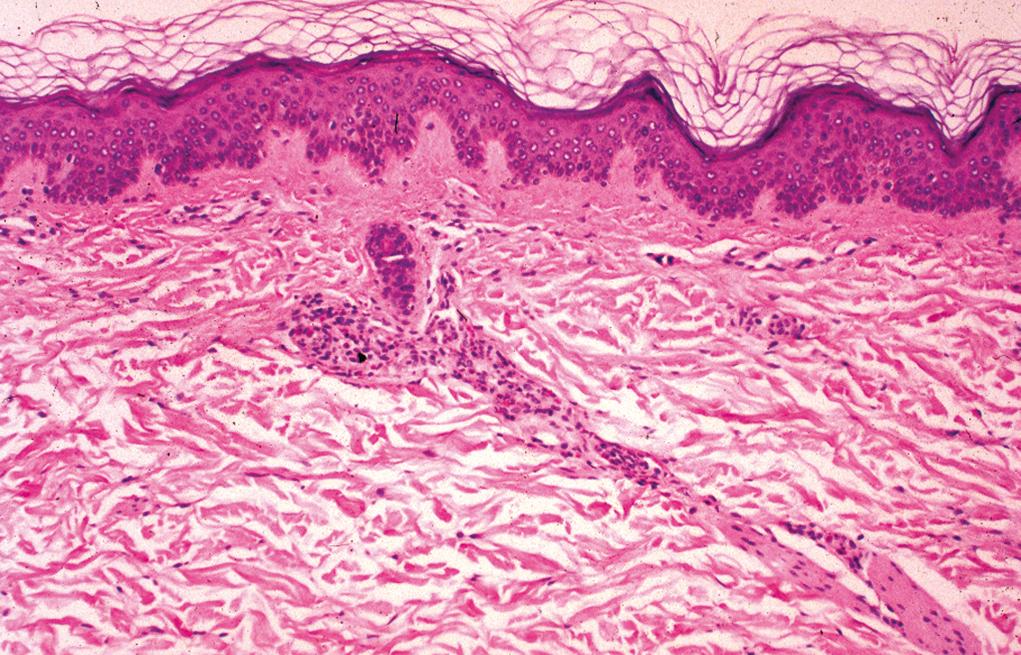
Sparse/mild superficial perivascular inflammation with scattered melanophages
Complete loss of melanin from the basal layer and total absence of melanocytes only seen in well-established lesions and in the depigmented center of expanding lesions
Advancing border-enlarged melanocytes with increased dendritic processes and a mild superficial perivascular inflammation
Normal-appearing skin (adjacent to the vitiliginous patches)—patchy interface change and a mild superficial perivascular and lichenoid lymphocytic infiltrate
Active lesions—lymphocyte aggregates (“Pautrier-like” microabscesses) in the basal layer
Silver stains or the dopa reaction (Fontana-Masson) highlight loss of basal layer melanin pigment ( Figures 2.2A and B )
Immunohistochemical stains for SOX10 or Melan-A are useful in confirming decreased density/absence of basal melanocytes
Noncontributory
On routine H&E-stained sections, other diseases manifesting with minimal histologic alterations (normal-appearing skin), such as tinea versicolor, urticaria, and urticaria pigmentosa (macular variant), should be considered.
Mycosis fungoides-epidermotropism and Pautrier microabscesses in all epidermal layers composed of lymphocytes with hyperchromatic and convoluted nuclei diagnostic
Biopsy of uninvolved/normal skin in addition to that of lesional skin (for comparative purposes) is essential for a diagnosis of early/evolving lesions.
Vitiligo can be seen in association with other autoimmune disorders such as thyroid disorders, pernicious anemia, and alopecia areata.
Attili VR, Attili SK. Lichenoid inflammation in vitiligo: a clinical and histopathologic review of 210 cases. Int J Dermatol . 2008;47:663–669.
Le Poole IC, Das PK. Microscopic changes in vitiligo. Clin Dermatol . 1997;15:863–873.
Silverberg JI, Silverberg NB. Clinical features of vitiligo associated with comorbid autoimmune disease: a prospective survey. J Am Acad Dermatol . 2013;69:824–826.
Typically, presents with pruritic, raised, erythematous, and edematous areas ( wheals or hives )
Acute urticaria lasts <6 weeks, more common in the pediatric age group; chronic urticaria lasts >6 weeks, more common in adults
An underlying predisposing condition can be identified in only up to 25% of patients; certain foods, drugs, contact allergens, and physical stimuli such as pressure, cold temperature, and occult infections may be factors
Urticarial vasculitis is a syndrome consisting of recurrent urticaria, arthralgia, and abdominal pain; individual cutaneous lesions persist for >24 hours
In angioedema, dermal edema extends into subcutaneous fat and presents with large wheals
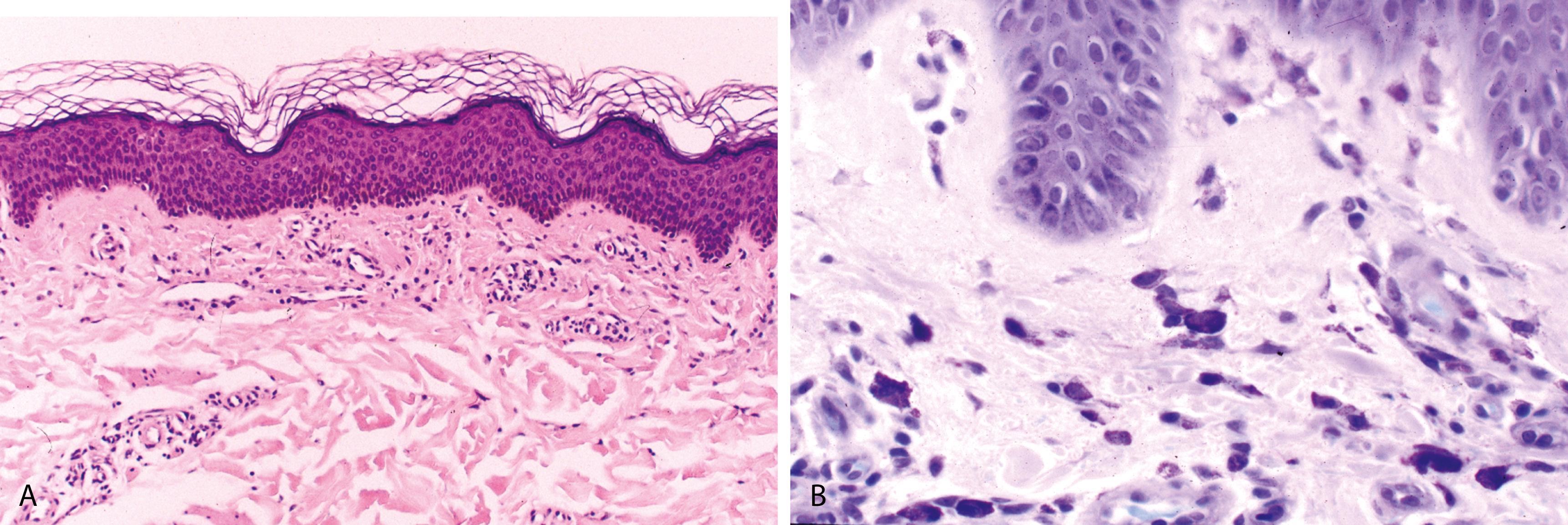
Interstitial edema, telangiectases, and a sparse/mild superficial perivascular mixed inflammatory cell infiltrate composed of lymphocytes, eosinophils, and neutrophils ( Figure 2.3 )
Urticarial vasculitis—features of early leukocytoclastic vasculitis characterized by mild perivascular infiltrate of neutrophils, neutrophilic nuclear dust, and extravasated red blood cells; minimal or absent fibrin deposits in the vessel walls
Noncontributory
Hypocomplementemia, seen in 32% of patients with urticarial vasculitis; measurements of CH50 and C1q binding assays are helpful
Patients with hereditary angioedema have a low serum level of C1 esterase inhibitor during the attack
Direct immunofluorescence (DIF) studies: vascular deposits of immunoglobulins, complement, or fibrin are seen in one third of patients with urticarial vasculitis
Macular Variant of Urticaria Pigmentosa (Telangiectasia Macularis Eruptiva Perstans)
Generally occurs as an extensive eruption of brownish-red macules
Dilated blood vessels in the upper dermis and a mild superficial perivascular mononuclear cell infiltrate composed mostly of mast cells are characteristic( Figure 2.4A )
Eosinophils are generally absent; dermal edema is not prominent
Giemsa, toluidine blue, Leder, or immunohistochemical stain for mast cell tryptase and CD117 can help demonstrate the increased number of mast cells ( Figure 2.4B )
Leukocytoclastic Vasculitis Due to Other Causes
Fibrinoid necrosis of vessels, leukocytoclasia, and an abundance of extravasated erythrocytes
Fibrinoid necrosis and thrombosis are not typically seen in urticarial vasculitis
In hereditary angioedema, a form of dominantly inherited angioedema, recurrent attacks of edema involve skin and oral, laryngeal, and gastrointestinal mucosa; death due to laryngeal edema can occur if not treated
Urticarial vasculitis may be associated with infectious mononucleosis, infectious hepatitis, and autoimmune diseases such as systemic lupus erythematosus (SLE)
Barzilai A, Sagi L, Baum S, Trau H, et al. The histopathology of urticaria revisited-clinical pathological study. Am J Dermatopathol . 2017;39:753–759.
Cugno M, Castelli R, Cicardi M. Angioedema due to acquired C1-inhibitor deficiency: a bridging condition between autoimmunity and lymphoproliferation. Autoimmun Rev . 2008;8:156–159.
Wisnieski JJ. Urticarial vasculitis. Curr Opin Rheumatol . 2000;12:24–31.
Disorder of unknown etiology involving skin, mucous membranes, hair, and nails
Typically presents as pruritic, polygonal, violaceous, flat-topped papules with a fine scale
Predilection for flexor surfaces of extremities, lower back, and glans penis
Surface of lesions may show a network of white lines ( Wickham striae )
Oral lesions (lacy, reticular network of papules involving buccal mucosa or the tongue) may be the sole manifestation
Association with hepatitis C
Compact hyperkeratosis, irregular epidermal hyperplasia (“saw-toothed”) with wedge-shaped hypergranulosis that corresponds to the openings of follicles and acrosyringia ( Figure 2.5A )
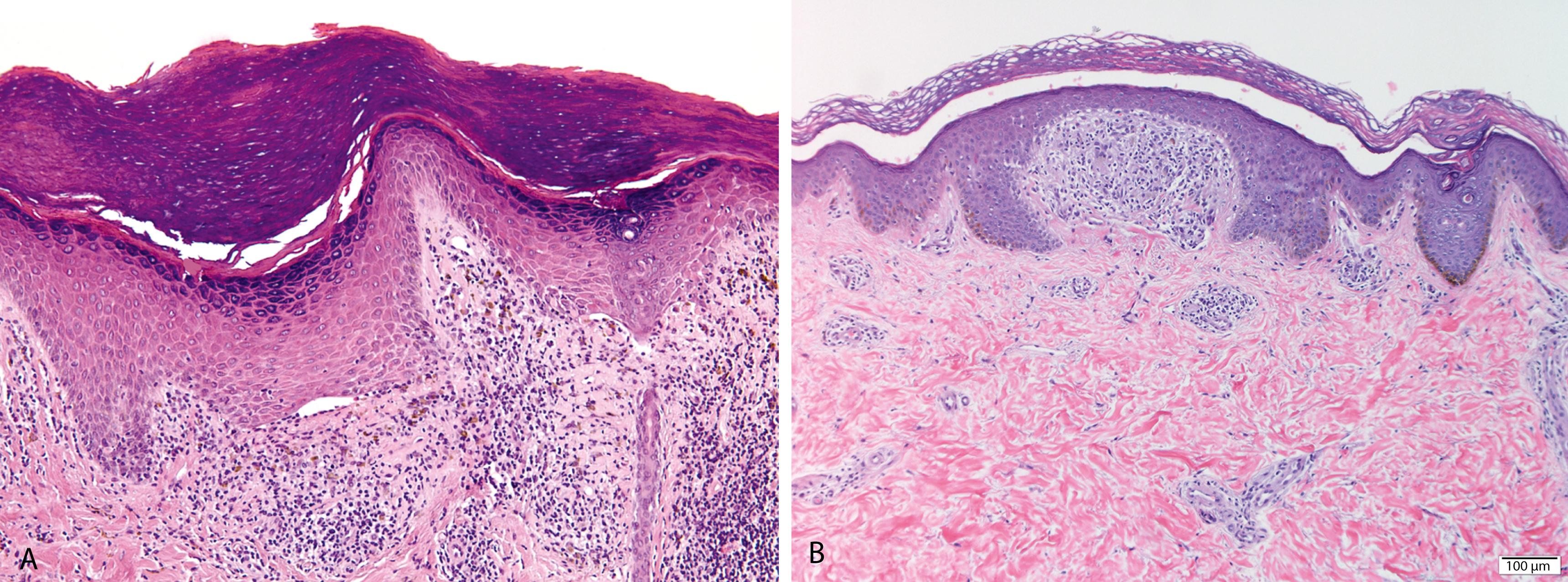
Bandlike, predominantly lymphocytic infiltrate in the superficial dermis that obscures the dermoepidermal junction
Eosinophilic colloid bodies (“Civatte bodies”), representing damage to the basal cell layer, are present at the dermoepidermal junction
Small clefts known as Max-Joseph/Caspary-Joseph spaces may be seen at the dermo-epidermal junction
Hypertrophic lichen planus—typically characterized by irregular acanthosis and interface change with an underlying inflammatory infiltrate confined only to the tips of the rete ridges
Oral lesions—typically show parakeratosis, less acanthosis, and are frequently ulcerated
Lichen planus of hair follicles (lichen planopilaris)—active lesions typically show a dense lymphocytic infiltrate surrounding the infundibulum of the follicle ; in later stages, there is perifollicular fibrosis with loss of follicles with advanced stages resulting in scarring alopecia
PAS stain confirms interface change
Noncontributory
Lichenoid Drug Eruption
Focal parakeratosis and necrotic keratinocytes particularly at and above the dermoepidermal junction
Presence of eosinophils in the inflammatory cell infiltrate
Lichen Planus–Like Keratosis (Benign Lichenoid Keratosis)
Solitary lesion
Absence of epidermal hyperplasia with hypergranulosis and presence of parakeratosis in addition to lichenoid pattern of inflammation
Adjacent areas may show changes of solar lentigo
Lichenoid Graft-Versus-Host Disease (GVHD)
Generally the inflammatory cell infiltrate is sparse
Foci of parakeratosis and thinning of epidermis may be present
Lichen Striatus
More common in children than adults and presents as a unilateral eruption along Blaschko lines on extremities, trunk, or neck
Lichenoid inflammatory cell infiltrate similar to lichen planus; however, the infiltrate extends deep in the reticular dermis around hair follicles and sweat glands
Epidermal spongiosis and an admixture of histiocytes in the inflammatory cell infiltrate
Lichen Nitidus
Asymptomatic dermatosis of childhood, characterized by round, flat-topped papules that measure only a few millimeters
Histologically, the inflammatory cell infiltrate is bandlike but the lesion is small and discrete; infiltrate is confined to widened dermal papillae and enclosed by elongated rete on either side (“ball and claw”) ( Figure 2.5B )
Frequent histiocytes in the infiltrate, epidermal atrophy, and focal parakeratosis are helpful in differentiating lichen nitidus from lichen planus
Parakeratosis and eosinophils in the inflammatory infiltrate are not features of cutaneous lichen planus, and indicative of other causes of lichenoid dermatitis such as lichenoid drug eruption
Shai A, Halevy S. Lichen planus and lichen planus-like eruptions: pathogenesis and associated diseases. Int J Dermatol . 1992;31:379–384.
Shiohara T. The lichenoid tissue reaction: an immunological perspective. Am J Dermatopathol . 1998;10:252–256.
Tziotzios C, Lee JYW, Brier T, et al. Lichen planus and lichenoid dermatoses: clinical overview and molecular basis. J Am Acad Dermatol . 2018;79(5):789–804.
Acute cytotoxic cell-mediated hypersensitivity reaction to infections, most commonly herpes simplex virus infection, and drugs, in particular sulfonamides
The eruption is multiform and consists of macules, papules, vesicles, and occasionally large flaccid bullae; often associated with fever
Herpesvirus-associated erythema multiforme (EM) involves the extremities and presents with typical target-like lesions, whereas drug-associated EM shows truncal involvement and a purpuric type of macular eruption; mucosal involvement (Stevens-Johnson syndrome) is characteristic
The most severe form, toxic epidermal necrolysis (a widespread blotchy erythema) is characterized by large flaccid bullae with “sloughing” of the skin and is most often caused by drugs, including sulfonamides, β-lactam antibiotics, and nonsteroidal anti-inflammatory drugs; associated with a high mortality rate
Cornified layer is unaltered, attesting to the acute nature of the disease
Confluent interface change/vacuolar alteration of the basal cell layer and a sparse superficial perivascular lymphocytic infiltrate that may focally obscure the dermoepidermal junction ( Figure 2.6A )
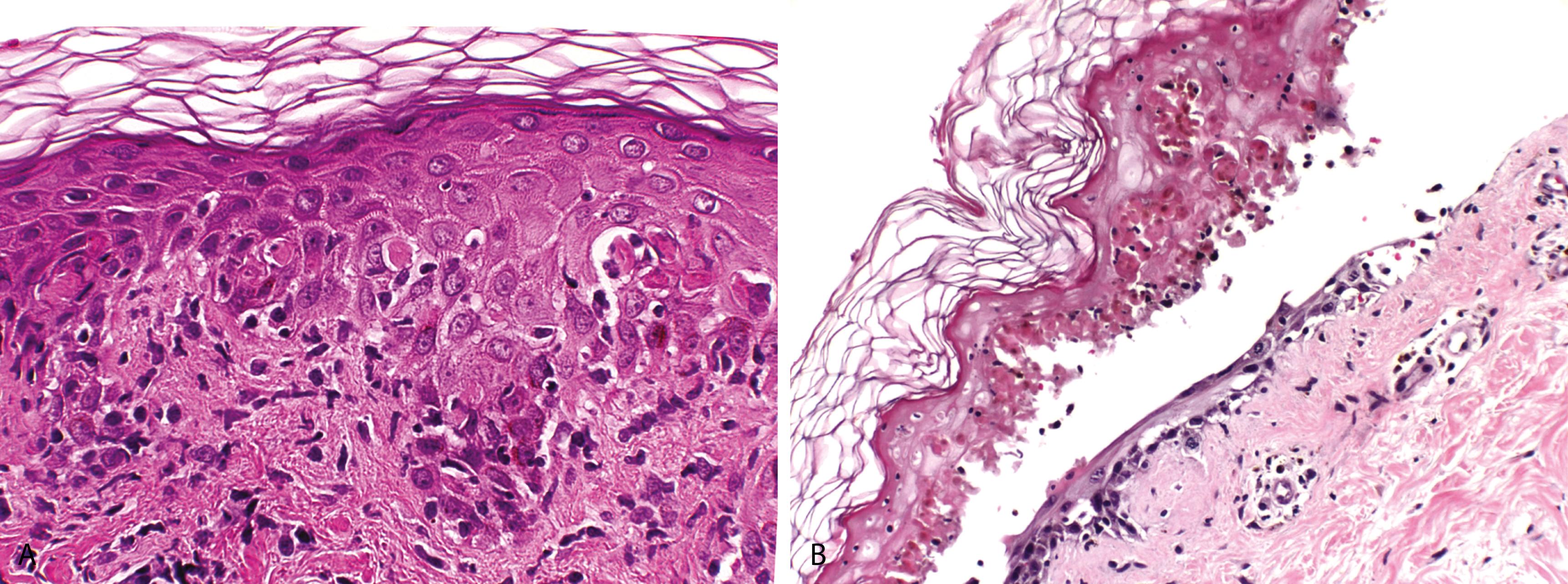
The hallmark of EM is the presence of necrotic keratinocytes, initially as single cells and later as small clusters; the necrosis is more widespread in drug-induced EM
In toxic epidermal necrolysis, there is full-thickness epidermal necrosis resulting in subepidermal bullae ( Figure 2.6B )
In late lesions, the papillary dermis may contain melanophages (a sign of damage to the basal cell layer)
Noncontributory
DIF studies of lesional skin <48 hours old will show immunoglobulin M (IgM) and C3 in the walls of superficial dermal vessels
Herpes simplex virus DNA has been detected within lesions of EM using PCR and in situ hybridization (ISH)
Staphylococcal Scalded-Skin Syndrome
Clinical appearance may be similar to toxic epidermal necrolysis
Microscopically, staphylococcal scalded-skin syndrome shows a split in the granular layer diagnostic (in contrast to toxic epidermal necrolysis where there is separation at the dermoepidermal junction)
Acute GVHD
May be histopathologically indistinguishable from early EM
Fixed Drug Eruption
Interface dermatitis with necrotic keratinocytes similar to EM
Deeper extension of inflammatory cell infiltrate containing eosinophils
Prominent pigment incontinence
Erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis are best regarded as a spectrum of the same disease process.
Ackerman AB, Ragaz A. Erythema multiforme. Am J Dermatopathol . 1985;7:133–139.
Roujeau JC. Stevens-Johnson syndrome and toxic epidermal necrolysis are severity variants of the same disease which differs from erythema multiforme. J Dermatol . 1997;24:726–729.
Samim F, Auluck A, Zed C, Williams PM. Erythema multiforme: a review of epidemiology, pathogenesis, clinical features, and treatment. Dent Clin North Am . 2013;57(4):583–596.
Occurs most frequently in bone marrow transplant recipients and less commonly in recipients of blood products and solid organ transplants
Incidence is 30% to 40% in related donor-recipient versus 60% to 80% when unrelated; risk increases with age
Acute phase
Presents with the triad of skin lesions, hepatic dysfunction, and diarrhea; develops within 3 months after transplant
Skin lesions are characterized by extensive erythematous macules, purpuric to violaceous papules and plaques, and, in severe cases, toxic epidermal necrolysis–like eruption; oral lesions may be present
Chronic phase
Occurs >3 months to a year after transplantation
In the early lichenoid stage, the eruption is similar to lichen planus
Late sclerotic stage is characterized by skin induration and tightening
Acute phase—4 grades
Grade I: vacuolar alteration of the basal cell layer, which may be focal or diffuse
Grade II: intraepidermal necrotic keratinocytes occasionally surrounded by lymphocytes (satellite necrosis) ( Figure 2.7 )
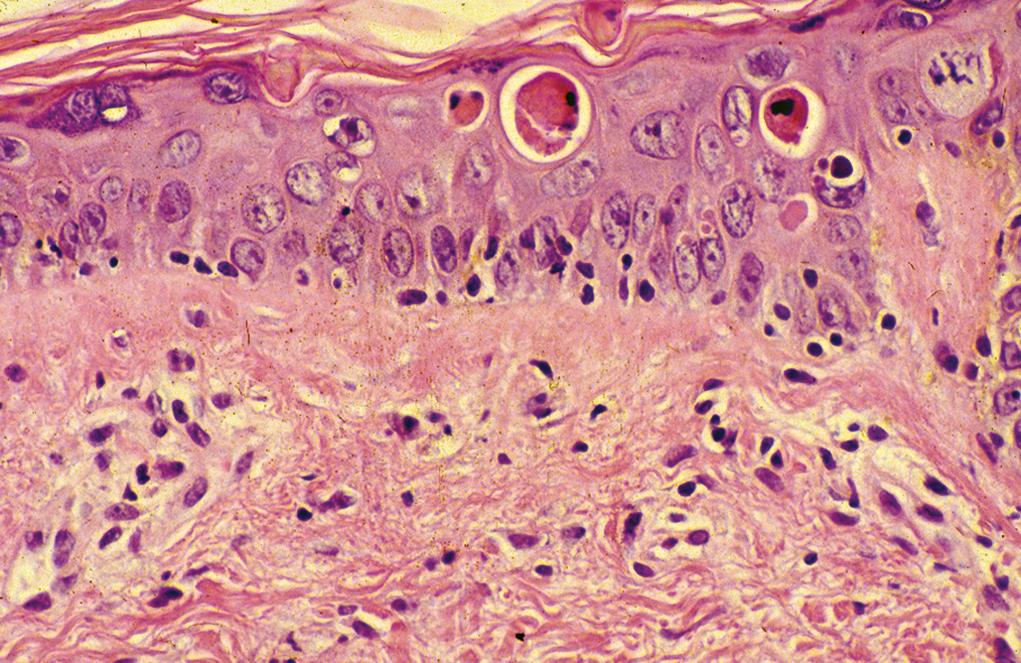
Grade III: widespread necrosis of keratinocytes with separation at the dermoepidermal junction
Grade IV: full-thickness necrosis and loss of epidermis
Sparse superficial perivascular lymphocytic infiltrate is usually present in acute GVHD
Follicular papules when evident clinically exhibit histopathologic changes in follicular epithelium similar to those evident in the epidermis
Chronic phase
Early lichenoid phase-histopathologic features of lichen planus; satellite necrosis may still be seen in GVHD
Late sclerotic phase-histopathologic changes similar to scleroderma with dermal sclerosis extending into the subcutis plus epidermal atrophy
Noncontributory
Noncontributory
Erythema Multiforme
May be histopathologically indistinguishable from acute GVHD
Lichen Planus
May be histopathologically indistinguishable from lichenoid phase of GVHD
Scleroderma
May be histopathologically indistinguishable from sclerotic phase of GVHD
Epidermal atrophy, if present, helps in differentiating sclerotic phase of GVHD from scleroderma
Histopathologic findings are phase dependent
Acute phase characterized by an interface (vacuolar type) dermatitis caused by the attack of donor immunocompetent T lymphocytes against histocompatibility antigens exposed on recipient cells
Chronic phase—initially characterized by an interface (lichenoid type) dermatitis and later as a sclerosing dermatitis and caused by immunocompetent lymphocytes that differentiate in the recipient
Target cells in GVHD are the stem cells in the regenerating compartment—that is, the basal keratinocytes in skin and the epithelial cells at the base of the crypts in the gastrointestinal tract
Aractingi S, Chosidow O. Cutaneous graft-versus-host disease. Arch Dermatol . 1998;134:602–612.
Hillen U, Hausermann P, Massi D, et al. Consensus on performing skin biopsies, laboratory workup, evaluation of tissue samples and reporting of the results in patients with suspected cutaneous graft-versus-host disease. J Eur Acad Dermatol Venereol . 2015;29:948–954.
Zhou Y, Barnett MJ, Rivers JK. Clinical significance of skin biopsies in the diagnosis and management of graft-vs-host disease in early post-allogeneic bone marrow transplantation. Arch Dermatol . 2000;136:717–721.
Chronic multisystem autoimmune disease that affects the connective tissue and vasculature of various organs
Three classic cutaneous forms: discoid, subacute, and systemic lupus
Discoid lupus erythematosus (DLE)—clinically, mildly scaling, erythematous, edematous, sharply demarcated plaques measuring up to 15 cm, involving the scalp, face, upper trunk, and upper extremities (photosensitive distribution); follicular plugging may be seen
Older lesions—atrophic with variable pigmentation
Subacute lupus erythematosus—presents as a symmetric photodistributed eruption of psoriasiform/annular plaques
Tumid lupus—indurated plaques and nodules without overlying erythema or atrophy
Verrucous lesions—seen in 2% of patients with chronic cutaneous lupus erythematosus
Panniculitis—seen in select patients with chronic cutaneous or systemic forms of lupus erythematosus
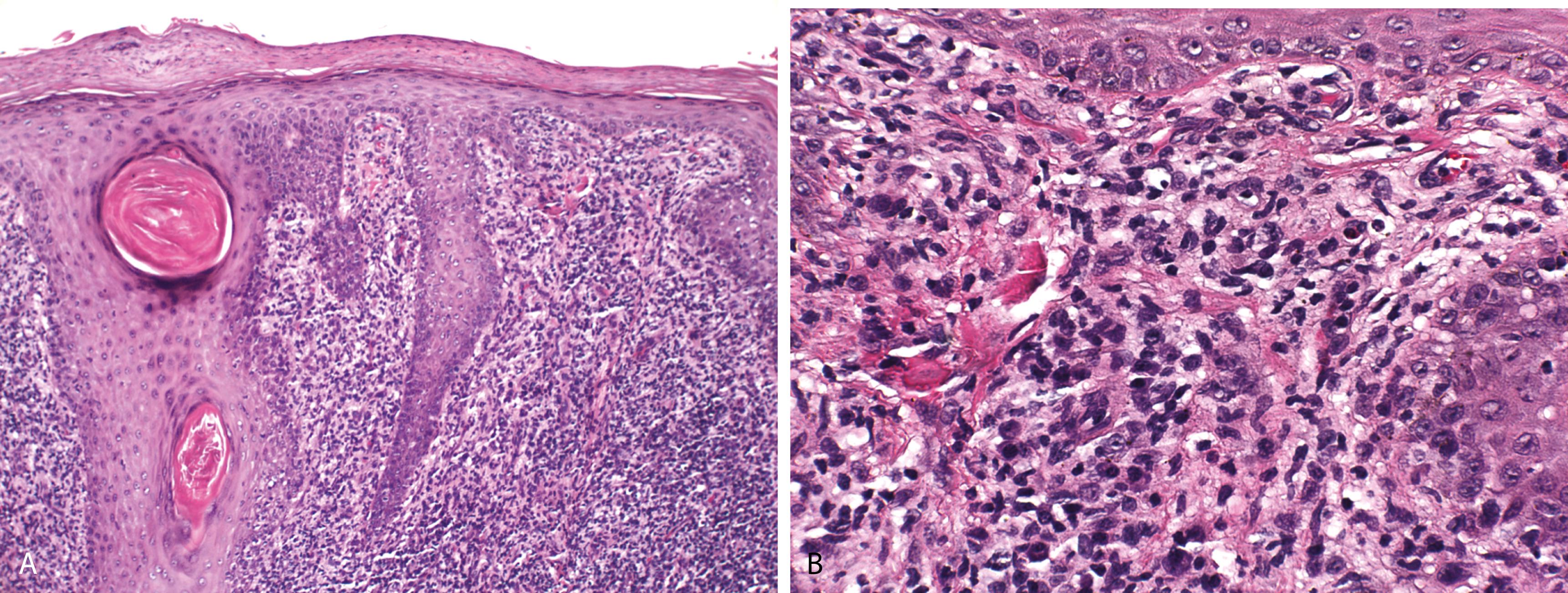
DLE—epidermal atrophy, hyperkeratosis with follicular plugging and confluent interface change that involves follicular epithelia and marked thickening of the basement membrane ( Figure 2.8A )
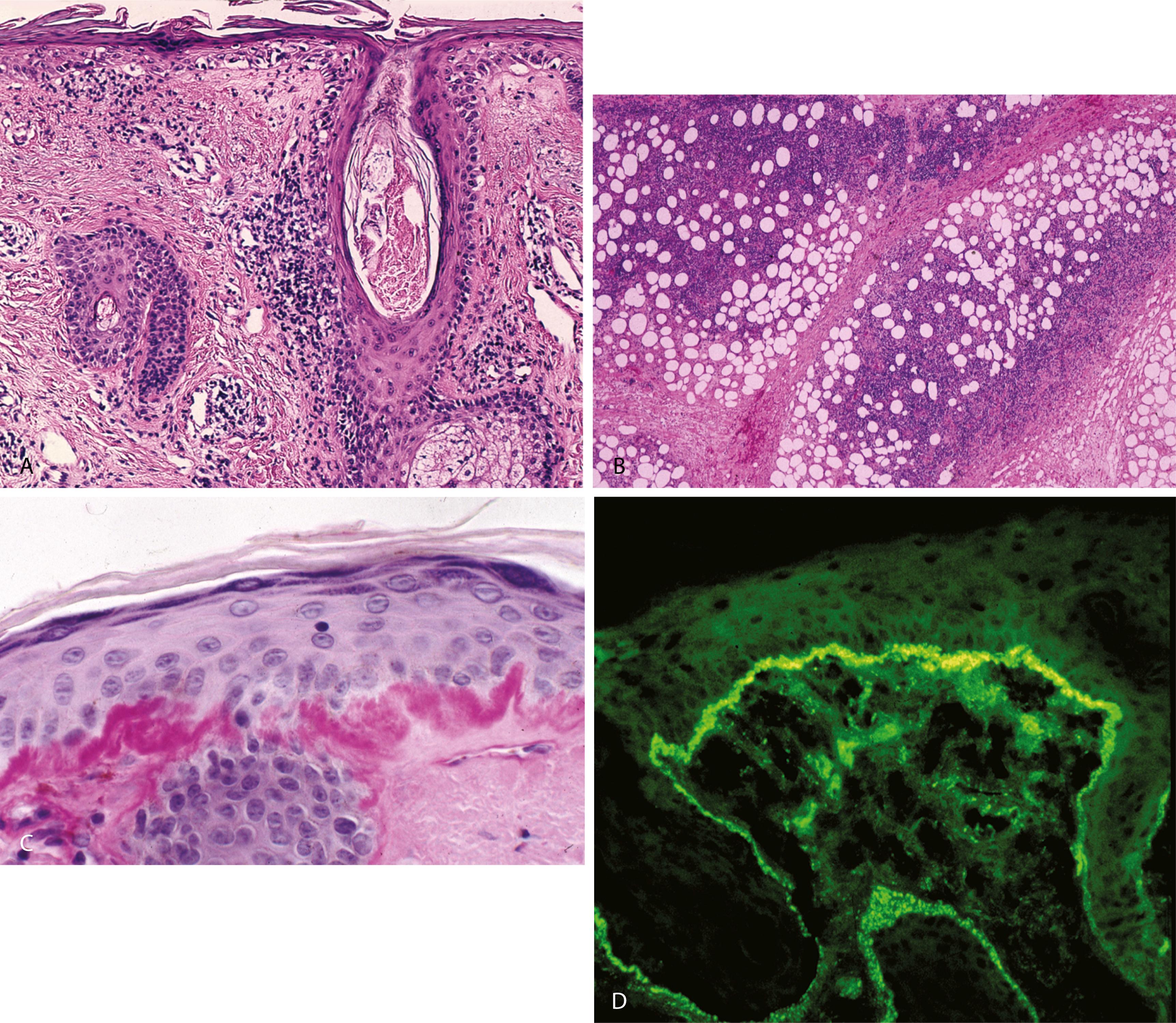
A moderate perivascular and periadnexal lymphocytic infiltrate and a mild increase in dermal mucin
Subacute cutaneous lupus erythematosus (SCLE) and neonatal lupus erythematosus-prominent interface change with numerous individually necrotic dyskeratotic keratinocytes, less prominent hyperkeratosis and inflammatory cell infiltrate than DLE
SLE—may show interface change with involvement of follicular epithelia, periadnexal and perivascular lymphocytic infiltrate, and increased dermal mucin similar to discoid lupus but more often the histologic changes are subtle
Tumid lupus erythematosus—superficial and deep perivascular and periadnexal lymphocytic infiltrate and increased dermal mucin but no epidermal changes
Lupus panniculitis/lupus profundus—lobular lymphocytic panniculitis with hyaline fat necrosis, superficial and deep perivascular and periadnexal lymphocytic infiltrate, interstitial mucin, with or without epidermal changes ( Figure 2.8B ), reactive lymphoid follicles not uncommon in the subcutis
Verrucous lupus—verrucous epidermal hyperplasia, interface change involving follicular epithelia, superficial and deep perivascular and periadnexal lymphocytic infiltrate and increased dermal mucin
PAS stain—helpful in demonstrating interface change and thickened basement membrane ( Figure 2.8C )
Colloidal iron stain—highlights interstitial mucin deposits
DIF—continuous granular deposition of immunoglobulin G (IgG), IgM, and C3 in a band along the dermoepidermal junction ( Figure 2.8D )
Dermatomyositis
Interface change, pauci-inflammatory, minimal increase in dermal mucin, and lupus band test is generally negative
Lichen Planus
Irregular epidermal hyperplasia with hypergranulosis, and lichenoid lymphoid cell infiltrate with pigment incontinence
Polymorphous Light Eruption
Superficial and deep perivascular lymphocytic infiltrates of lupus (especially tumid form) must be differentiated from polymorphous light eruption, which usually shows marked edema of papillary dermis
Subcutaneous Panniculitis-Like T-Cell Lymphoma
Can mimic lupus profundus
Rimming of the subcutaneous fat cells by neoplastic T lymphocytes and presence of lymphocyte nuclei within the cytoplasm of histiocytes (emperipolesis) are characteristic features
Interface dermatitis with involvement of follicular epithelia diagnostic for all forms of lupus erythematosus
Increase in interstitial mucin is minimal in SCLE and DLE
Site of biopsy crucial for DIF-exposed normal skin for SLE and lesion >3 months duration for DLE
Well-defined lesions of DLE occur in 15% of patients with subacute lupus erythematosus
Arps DP, Patel RM. Lupus profundus (panniculitis): a potential mimic of subcutaneous panniculitis-like T-cell lymphoma. Arch Pathol Lab Med . 2013;137:1211–1215.
Biazar C, Sigges J, Patsinakidis N, Ruland V, et al. Cutaneous lupus erythematosus: first multicenter database analysis of 1002 patients from the European Society of Cutaneous Lupus Erythematosus (EUSCLE). Autoimmun Rev . 2013;12:444–454.
Jerdan MS, Hood AF, Moore GW, et al. Histopathologic comparison of the subsets of lupus erythematosus. Arch Dermatol . 1990;126:52–55.
Connective tissue disease characterized by inflammatory myositis involving the proximal muscles and cutaneous lesions consisting of a heliotrope rash (violaceous, slightly edematous periorbital patches involving the eyelids), Gottron papules (discrete red-purple papules over bony prominences of knuckles, knees, and elbows), and photodistributed erythematous-edematous lesions over the upper chest and back
The disease has two peaks, one in childhood and one between the ages of 45 and 65 years
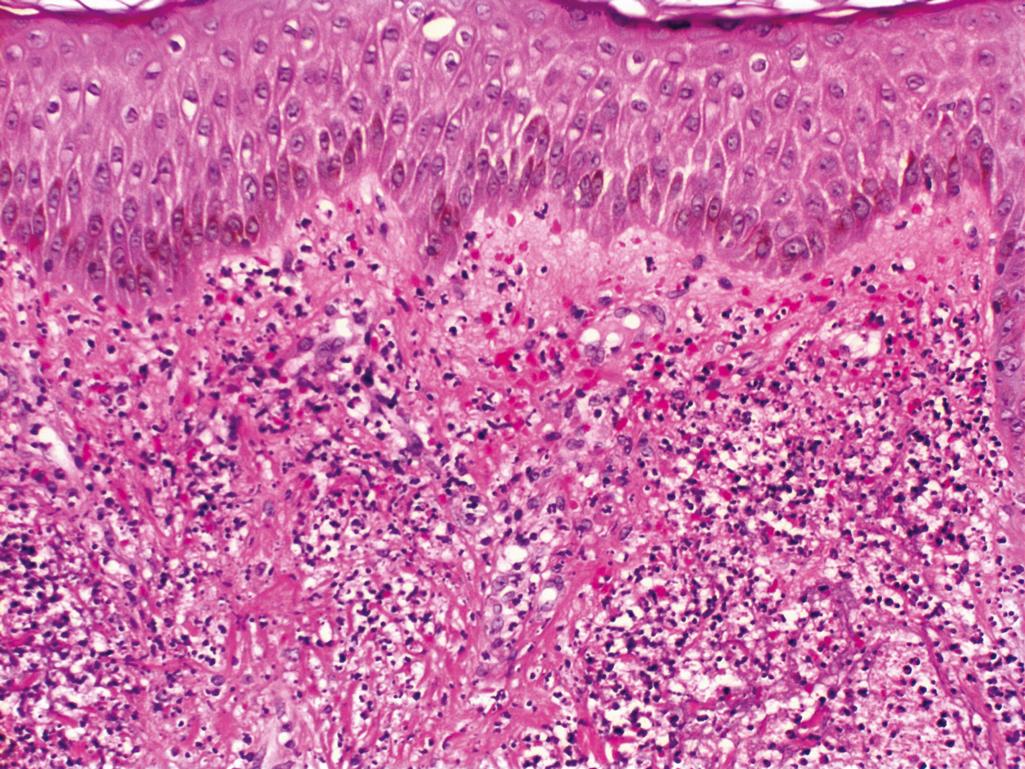
Histopathologic changes of the erythematous-edematous lesions of the skin include epidermal atrophy, vacuolar interface change with scattered individually necrotic keratinocytes, a sparse superficial perivascular lymphocytic infiltrate ( Figure 2.9 )
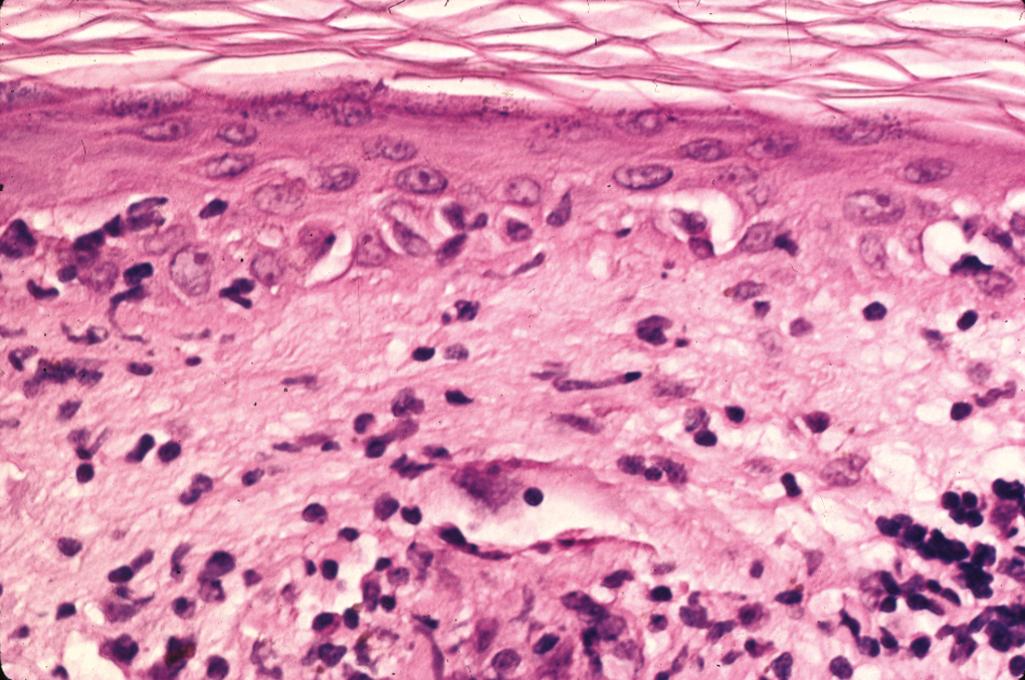
Mild increase in interstitial mucin
Subepidermal fibrin deposits can be seen
Gottron papules—epidermal hyperplasia in addition to mild interface change
Panniculitis and calcification of the subcutaneous tissue may be seen at a later stage
PAS stain confirms interface change and a thickened basement membrane zone
DIF—may show complement (C5b-C9) around blood vessels
Subacute Cutaneous Lupus Erythematosus
Histopathologic features identical to those of dermatomyositis, clinical correlation including evaluation for myositis critical
Positive lupus band test helpful (especially in early stages of dermatomyositis when the muscular weakness is not apparent)
Pauci-inflammatory interface dermatitis with a minimal increase in dermal mucin
Histopathologic reaction pattern that of a subtle interface dermatitis and is therefore not diagnostic
Clinicopathologic correlation key to making the histopathologic diagnosis
Callen JP. Dermatomyositis. Lancet . 2000;355:53–57.
Mendese G, Mahalingam M. Histopathology of Gottron’s papules-utility in diagnosing dermatomyositis. J Cutan Pathol . 2007;34:793–798.
Sontheimer RD. Cutaneous features of classic dermatomyositis and amyopathic dermatomyositis. Curr Opin Rheumatol . 1999;11:475–482.
Spongiotic dermatitis refers to reaction pattern characterized histologically by the presence of intercellular edema (spongiosis) in the epidermis
Group includes allergic contact dermatitis, photoallergic dermatitis, nummular dermatitis, atopic dermatitis, dyshidrotic dermatitis, and Id reaction
Allergic Contact Dermatitis
Most commonly caused by poison ivy, nickel, and rubber compounds, presents with pruritic, edematous, erythematous papules and occasional vesicles usually within 1-3 days of exposure
Photoallergic Dermatitis
Due to topical application (photocontact) or ingestion of an allergen
Presents with pruritic and erythematous papulovesicular lesions on sun-exposed skin; usually on face, arms, and neck
Nummular Dermatitis
Disease of unknown etiology characterized by coin-shaped, pruritic, erythematous, scaly, crusted plaques on the exterior aspects of extremities
Atopic Dermatitis
Inherited chronic, pruritic, scaly eruption affecting face and extensor aspects of extremities in children
Dyshidrotic Dermatitis
Characterized by numerous pruritic vesicles along sides of fingers and toes and palms and soles
Autoeczematization or Id Reaction
Sudden localized or generalized eruption of pinhead-sized vesicles developing in association with a defined local dermatitis or with infection
Most common cause is a remote dermatophyte infection
Three distinct reaction patterns— acute , subacute , and chronic
Acute spongiotic dermatitis
Spongiotic vesicles with minimal acanthosis ( Figure 2.10A )
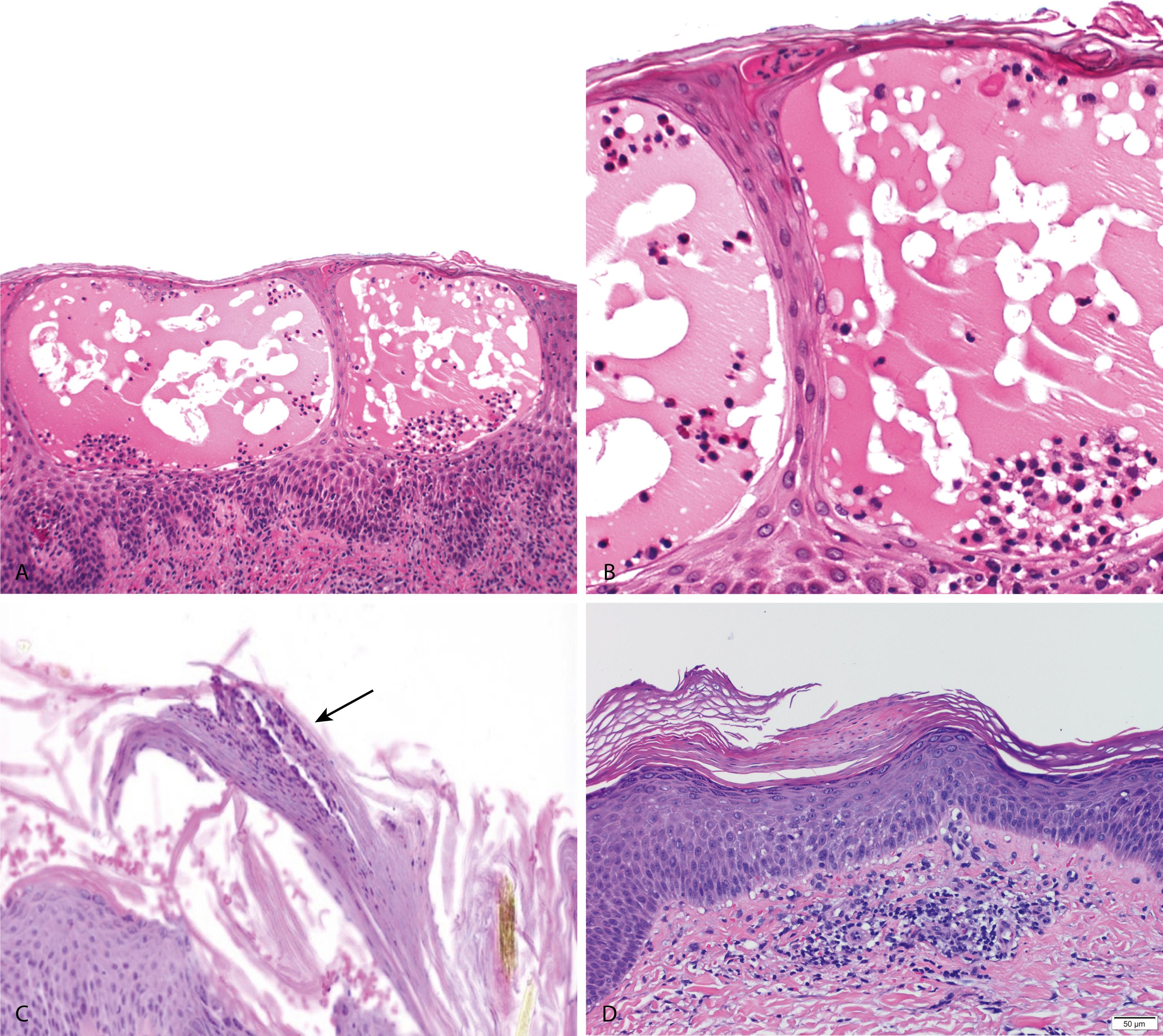
In allergic contact dermatitis, eosinophils may be present in the vesicles ( Figure 2.10B )
Mild papillary dermal edema and a superficial perivascular lymphohistiocytic inflammation are present
Subacute spongiotic dermatitis
Parakeratosis with plasma, mild to moderate spongiosis, epidermal hyperplasia, and superficial perivascular lymphohistiocytic infiltrate
Chronic Spongiotic Dermatitis
Hyperkeratosis, acanthosis with hypergranulosis, minimal spongiosis, and papillary dermal fibrosis
PAS stain useful in excluding dermatophytosis with spongiosis
Noncontributory
Seborrheic Dermatitis
Histopathology varies with age of the lesion, follicle-centered, “shoulder” parakeratosis ( Figure 2.10C ) and a mild superficial perivascular lymphoid cell infiltrate with admixed neutrophils
Pityriasis Rosea
“Moundlike” parakeratosis ( Figure 2.10D ), mild spongiosis ( only in the “herald patch” ), mild superficial perivascular lymphoid cell infiltrate, and extravasated red cells
Identical changes may be seen in superficial form of erythema annulare centrifugum
Spongiotic Drug Eruptions and Insect Bite Reactions
Deeper extension of the inflammatory infiltrate with admixed eosinophils
Psoriasis
Chronic spongiotic dermatitis (lichen simplex chronicus) may resemble psoriasis but lacks confluent parakeratosis with neutrophils and thinning of suprapapillary plates
Eczema is a nonspecific term used clinically to describe erythematous vesicular lesions with scaly crust that show spongiotic dermatitis on histology
Three distinct reaction patterns—acute, subacute, and chronic
Histopathology typically varies with age of the lesion
Several entities can exhibit the same reaction pattern
Ackerman AB, Chongchitnant N, Sanchez J, et al. Histologic Diagnosis of Inflammatory Skin Diseases . Baltimore: Williams & Wilkins; 1997.
Ackerman AB, Ragaz A. A plea to expunge the word “eczema” from the lexicon of dermatology and dermatopathology. Am J Dermatopathol . 1982;4:315.
Weedon D. Skin Pathology . 2nd ed. New York: Churchill Livingstone; 2002:112.
An X-linked–dominant dermatosis that affects mostly females
Characteristic cutaneous manifestations seen at birth include crops of vesicles and bullae on extremities arranged in a linear or whorled pattern
Lesions heal with hyperkeratosis and verrucous epidermal hyperplasia; the verrucous lesions heal with streaks and whorls of hyperpigmentation that are later replaced by faint hypochromic patches
Vesicular stage—characterized by marked epidermal spongiosis with eosinophils, individually dyskeratotic keratinocytes, and whorls of squamous cells with central keratinization ( Figure 2.11 )
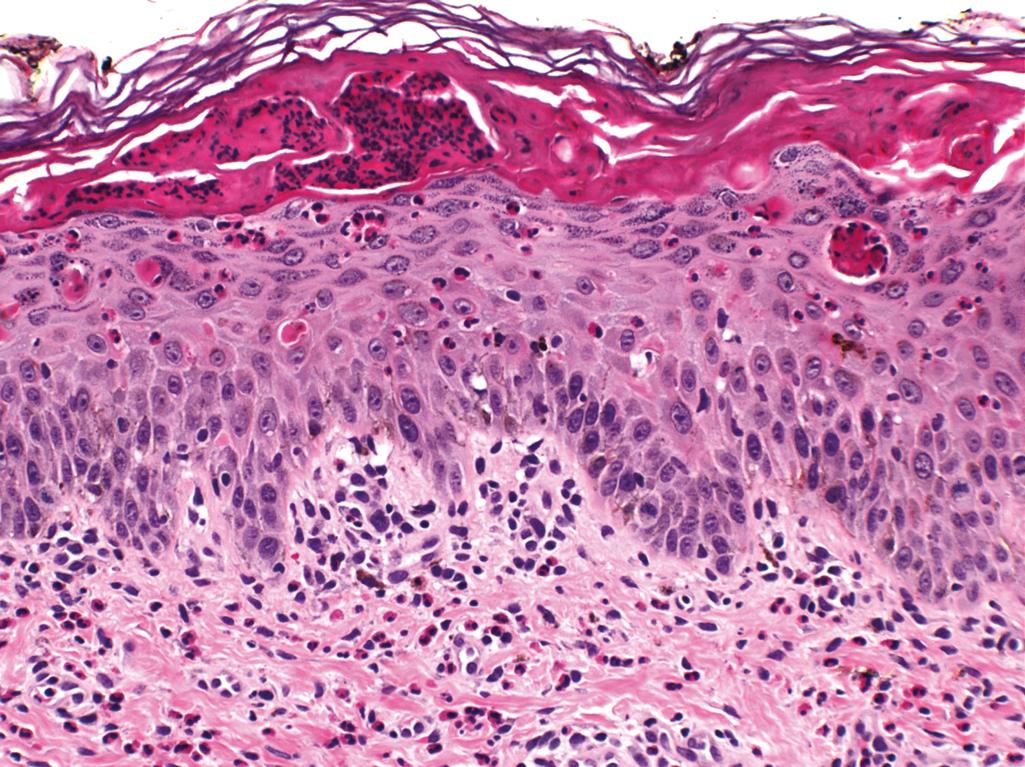
Verrucous stage—characterized by hyperkeratosis, verrucous epidermal hyperplasia, individually dyskeratotic keratinocytes, and occasional eosinophils
Hyperpigmented stage—characterized by numerous melanophages in the superficial dermis
Hypopigmented stage—characterized by epidermal atrophy, decreased melanin in the basal cell layer, and absence of adnexae
Noncontributory
Noncontributory
Eosinophilic Spongiosis
Seen in a spectrum of unrelated entities that include allergic contact dermatitis, precursor lesions of pemphigus and bullous pemphigoid; clinical history essential in differentiation
Epidermal Nevus
Verrucous stage of incontinentia pigmenti may resemble epidermal nevus
Postinflammatory Pigmentary Alterations
Hyper- and hypopigmented stages of incontinentia pigmenti resemble postinflammatory pigmentary changes
Histopathology typically varies with age of the lesion with four distinct histopathologic reaction patterns
Several entities can exhibit the same reaction pattern, clinicopathologic correlation essential in making a conclusive diagnosis
Eosinophilic chemotactic activity has been shown in the blister fluid of patients with incontinentia pigmenti
In up to 80% of patients, systemic findings with involvement of the central nervous system and eye may be seen; teeth abnormalities may be present
Extent of systemic involvement determines the clinical course
The genetic defect in the familial form has been traced to a mutation in the IKBKG- gene (called NEMO) localized to Xq28 region
Ackerman AB, Chongchitnant N, Sanchez J, et al. Histologic Diagnosis of Inflammatory Skin Diseases . Baltimore: Williams & Wilkins; 1997.
Berlin AL, Paller AS, Chan LS. Incontinentia pigmenti: a review and update on the molecular basis of pathophysiology. J Am Acad Dermatol . 2002;47:169–187.
Ehrenreich M, Tarlow MM, Godlewska-Janusz E, Schwartz RA. Incontinentia pigmenti (Bloch-Sulzberger syndrome): a systemic disorder. Cutis . 2007;79:355–362.
Chronic dermatosis of unknown etiology affecting up to 2% of the population
Males and females affected equally
Variably sized, well-demarcated plaques covered by thick, silvery white scale that have a predilection for areas with trauma, including scalp, lumbosacral skin, and extensor surfaces of elbows and knees
Several manifestations that include localized or generalized pustular psoriasis, eruptive or guttate psoriasis, and erythrodermic psoriasis
Involvement of nails, oral mucosa, and tongue can occur
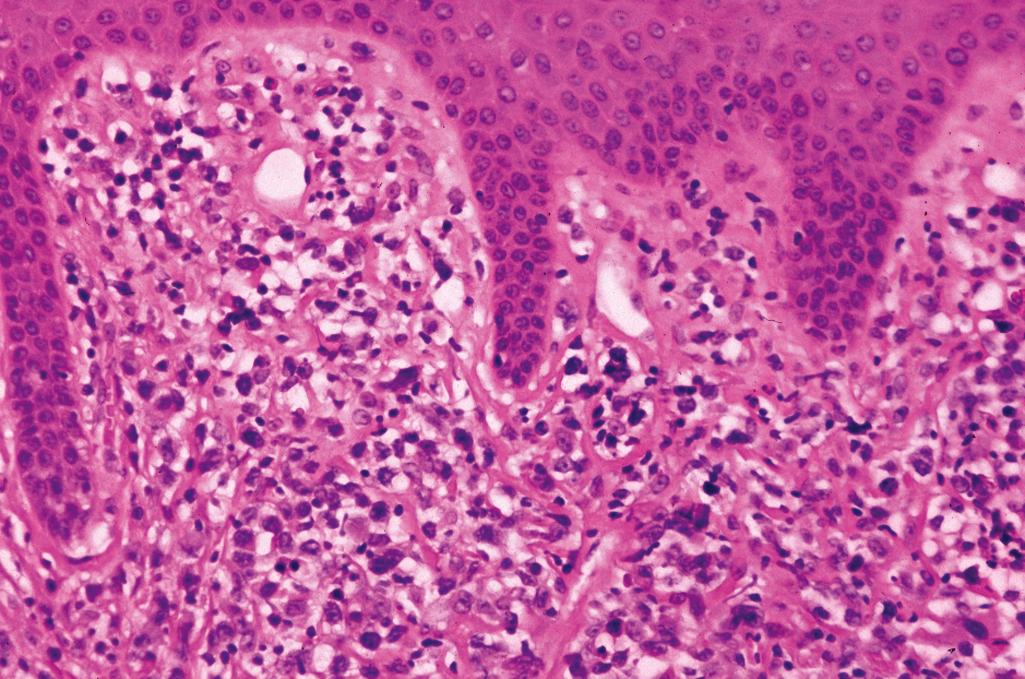
Confluent parakeratosis with neutrophils and neutrophilic microabscesses (intracorneal-Munro microabscesses and intraspinous-spongiform pustule of Kogoj)
Hypogranulosis corresponding to zones of parakeratosis
Regular (psoriasiform) epidermal hyperplasia with elongation of rete ridges and thinning of suprapapillary plates ( Figure 2.12A )
![Figure 2.12, A, Psoriasis. Confluent parakeratosis with collections of neutrophils, diminished granular layer, regular (psoriasiform) epidermal hyperplasia with thinning of suprapapillary plates, dilated blood vessels in the papillary dermis, and mild superficial perivascular inflammation are seen. B, Pustular psoriasis. Prominent Munro microabscess. C, AIDS-associated psoriasis. Scale crust with neutrophils, psoriasiform epidermal hyperplasia, absence of thinning of the suprapapillary plate [a], and numerous plasma cells [b]. Figure 2.12, A, Psoriasis. Confluent parakeratosis with collections of neutrophils, diminished granular layer, regular (psoriasiform) epidermal hyperplasia with thinning of suprapapillary plates, dilated blood vessels in the papillary dermis, and mild superficial perivascular inflammation are seen. B, Pustular psoriasis. Prominent Munro microabscess. C, AIDS-associated psoriasis. Scale crust with neutrophils, psoriasiform epidermal hyperplasia, absence of thinning of the suprapapillary plate [a], and numerous plasma cells [b].](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SkinandAdnexalStructures/11_3s20B9780323661652000028.jpg)
Dilated tortuous blood vessels in the dermal papillae
Mild superficial perivascular lymphocytic infiltrate
Pustular psoriasis—Exhibits the above but has in addition prominent Munro microabscesses and spongiform pustules of Kogoj ( Figure 2.12B )
Guttate psoriasis—psoriasiform epidermal hyperplasia not as pronounced
Erythrodermic psoriasis-histopathologic changes may be nonspecific
AIDS-associated psoriasiform dermatitis
Distinguishing features from psoriasis include individually necrotic/dyskeratotic keratinocytes, absence of thinning of the suprapapillary plates, and presence of plasma cells ( Figure 2.12C-a,b )
PAS stain helpful in excluding dermatophytic infections (which also exhibit neutrophils in the stratum corneum)
Noncontributory
Subacute/Chronic Spongiotic Dermatitis Such As Allergic Contact or Nummular Dermatitis
Presence of spongiosis and eosinophils helpful in differentiation
Dermatophyte Infection
Presence of superficial fungal spores and hyphal forms diagnostic (typically evident in PAS stained sections)
Bacterial Impetigo
Subcorneal pustule diagnostic of an early lesion (rarely biopsied)
Gram stain may demonstrate the bacteria
Pityriasis Rubra Pilaris
Alternating ortho and para-keratotic scale, psoriasiform epidermal hyperplasia, follicular plugging with “shoulder” parakeratosis diagnostic
Contrasting features with psoriasis include absence of neutrophils in the stratum corneum and retention of the granular cell layer
“Dry” horn typical-scale crust with neutrophils but no inspissated serum
Intraepidermal pustule not specific for psoriasis—can be seen in Reiter syndrome, AIDS-associated psoriasiform dermatitis, acrodermatitis enteropathica, and glucagonoma syndrome
Basal keratinocyte atypia not uncommon and related to etiopathogenesis
Psoriatic arthritis is seen in about 15% of patients with psoriasis and characteristically involves terminal interphalangeal joints
Severe expression of psoriasis is seen in patients with AIDS who may present with extensive erythroderma, inverse psoriasis, and palmoplantar psoriasis
De Mozzi P, Johnston GA, Alexandroff AB. Psoriasis. An evidence-based update. Report of the 9th evidence-based update meeting, 12 May 2011, Loughborough, UK. Br J Dermatol . 2012;166:252–260.
Nestle FO. Psoriasis. Curr Dir Autoimmun . 2008;10:65–75.
Ragaz A, Ackerman AB. Evolution, maturation, and regression of lesions of psoriasis. Am J Dermatopathol . 1979;1:199.
Chronic follicular-based erythematous papular eruption of unknown etiology that progresses to form orange-red scaly plaques containing islands of normal-appearing skin
Generalized erythroderma may occur with progression
Palmoplantar keratoderma and scales on the face and scalp not uncommon
Alternating orthokeratosis and parakeratosis in horizontal and vertical directions (“checkerboard pattern”) ( Figure 2.13A and B )
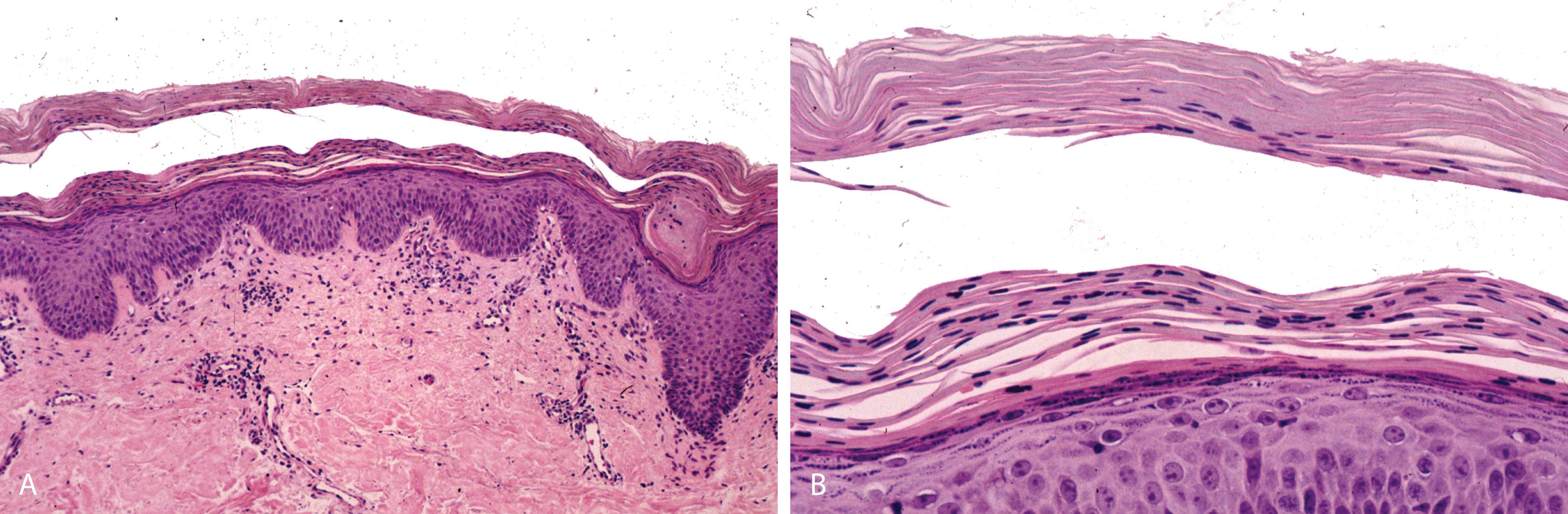
Psoriasiform epidermal hyperplasia with short rete and thick suprapapillary plates
Follicular papules demonstrate “shoulder” parakeratosis and dilated follicular infundibula with follicular plugging
Acantholysis restricted to adnexal epithelium
Noncontributory
Noncontributory
Psoriasis
Confluent parakeratosis with neutrophils and neutrophilic microabscesses, psoriasiform epidermal hyperplasia with hypogranulosis ( granular cell layer is retained in pityriasis rubra pilaris ) and basal keratinocyte atypia, thinning of suprapapillary plates ( suprapapillary plates are thick in pityriasis rubra pilaris ), and papillary dermal telangiectases are diagnostic features
“Checkerboard” cornified layer distinctive
Acantholysis not uncommon but restricted to adnexal epithelium when present
“Shoulder” parakeratosis not specific to pityriasis rubra pilaris and can also be seen in seborrheic dermatitis
Barr RJ, Young EM Jr. Psoriasiform and related papulosquamous disorders. J Cutan Pathol . 1985;12:412–425.
Mobini N, Toussaint S, Kamino H. Noninfectious erythematous, papular, and squamous diseases. In: Elder DE, Elenitsas R, Johnson BL Jr, et al., eds. Lever’s Histopathology of Skin . 10th ed. Philadelphia; Lippincott Williams & Wilkins; 2008:169.
Piamphongsant T, Akaraphant R. Pityriasis rubra pilaris: a new proposed classification. Clin Exp Dermatol . 1994;19:134–138.
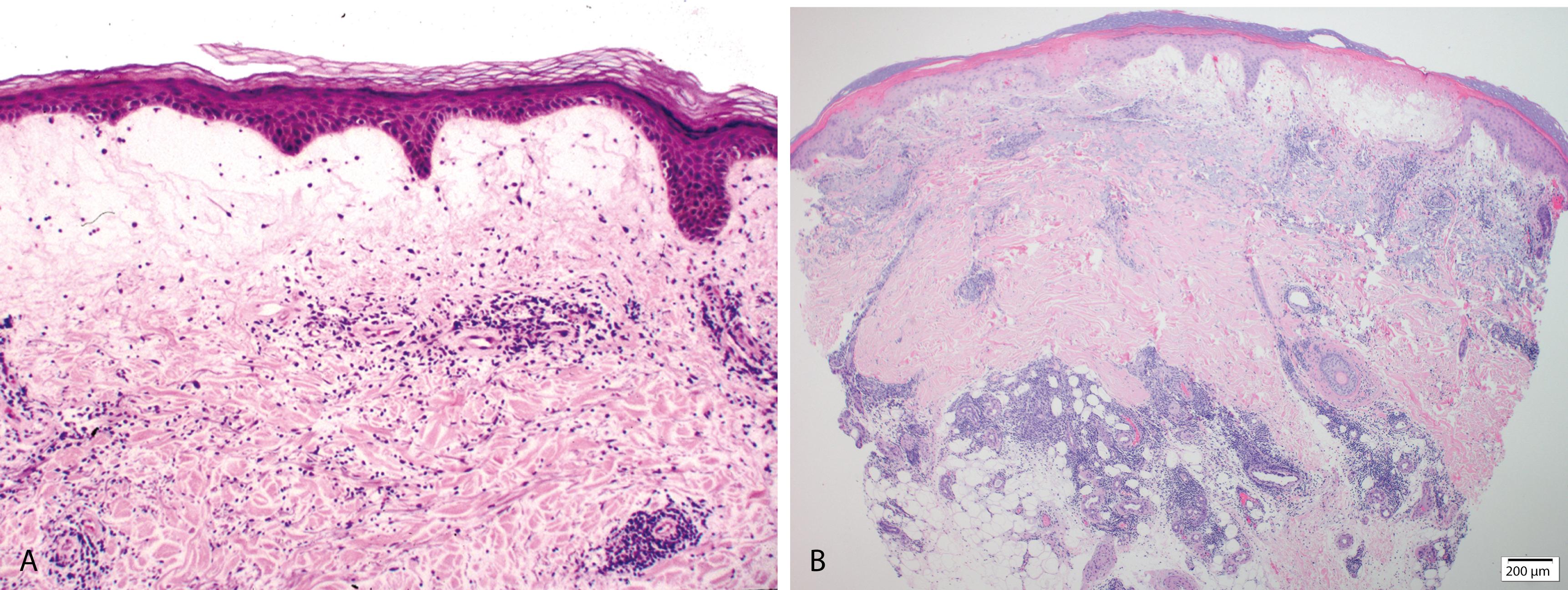
Recurrent pruritic papules and plaques that typically occur in young women mostly during summer, induced by ultraviolet radiation (UVR)
Eruption starts a few minutes to a few hours after exposure and lasts for hours to days
Extensor aspects of arms and upper chest are most frequently involved
Despite the diverse clinical presentation , within any one patient, only one clinical form is consistently manifested
Epidermis is mostly unremarkable
Prominent papillary dermal edema with occasional subepidermal separation and extravasated red cells
Superficial and deep perivascular, predominantly lymphocytic infiltrate ( Figure 2.14A )
Noncontributory
Noncontributory
Sweet Syndrome
Superficial and deep perivascular and interstitial predominantly neutrophilic infiltrate with abundant leukocytoclasia, prominent papillary dermal edema, and extravasated erythrocytes
Sweet Syndrome-Like Arthropod Bite Reaction
Superficial and deep dermal inflammation with numerous eosinophils and prominent papillary dermal edema
Pernio
Interface change, superficial and deep dermal perivascular and periadnexal inflammation, increased dermal mucin, prominent papillary dermal edema and extravasated erythrocytes ( Figure 2.14B )
Pityriasis Lichenoides, Acute Form
Parakeratosis with neutrophils, interface change with numerous individually necrotic keratinocytes, papillary dermal edema and extravasated erythrocytes
Cutaneous Lupus Erythematosus
Typically the subacute and tumid forms should be considered in the differential diagnosis
Polymorphous light eruption lacks changes at the dermoepidermal junction, has less prominent periadnexal infiltrate, and lacks interstitial mucin deposits, features typically seen in cutaneous lupus
Papillary dermal edema is more prominent in polymorphous light eruption than in SCLE
Extravasated erythrocytes and prominent papillary dermal edema helpful histopathologic features but not specific for polymorphous light eruption
Extravasated erythrocytes and papillary dermal edema, also observed in pityriasis lichenoides acuta, pernio, Sweet syndrome
Allergic reaction induced by bites from mosquitoes, fleas, and bedbugs
Papules and papulovesicular lesions that are intensely pruritic and often excoriated
Focal epidermal ulceration/excoriation, epidermis, and cornified layer may show changes of excoriation
A superficial and deep, V- or wedge-shaped, perivascular and interstitial, mixed inflammatory cell infiltrate containing numerous eosinophils characteristic ( Figure 2.15A and B )
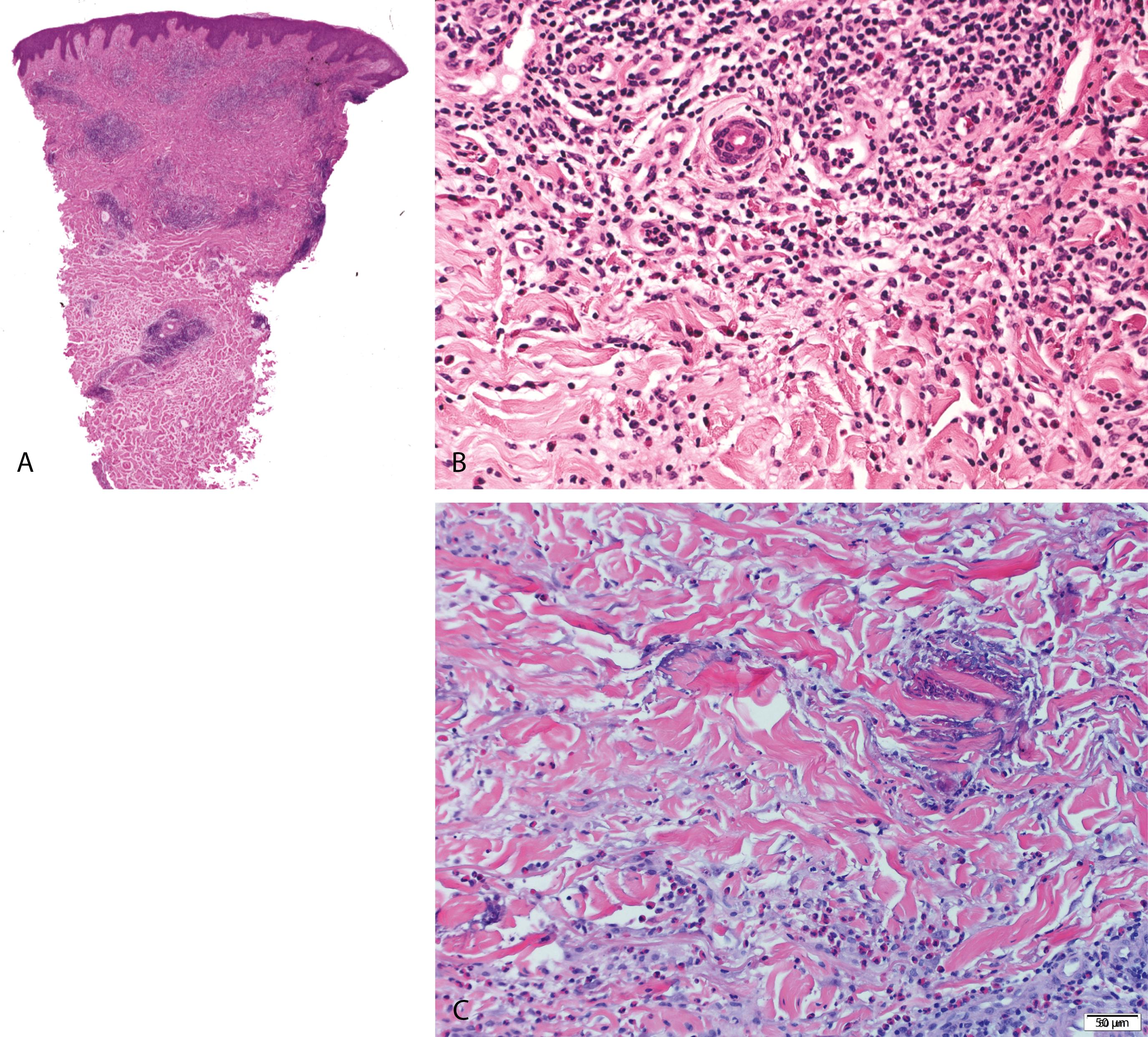
Chronic lesions—characterized by an exuberant inflammatory infiltrate with reactive follicles
Noncontributory
Immunohistochemistry for B- and T-cell lymphoid markers helpful in confirming a reactive process in chronic lesions
Eosinophilic Cellulitis/Wells Syndrome
Pandermal, predominantly eosinophil-rich inflammatory infiltrate with “flame figures” (eosinophilic debris as a result of degranulating eosinophils) ( Figure 2.15C )
Dermal Hypersensitivity Reaction
Hypersensitivity reaction due to drugs while histopathologically indistinguishable from papular urticaria often tends to have an inflammatory infiltrate that is less dense and deep
B-Cell Lymphoproliferative Disease/Follicle Center Lymphoma
Poorly defined follicles with aberrant expression of lineage-specific immunohistochemical markers
Presence of a deep, predominantly eosinophil-rich inflammatory infiltrate helpful but not diagnostic
Identification of foreign body material reminiscent of arthropod parts superficially or within the dermis diagnostic
Reactive lymphoid follicles may be a feature of chronic lesions (persistent arthropod bite reaction)
Ackerman AB, Chongchitnant N, Sanchez J, et al. Histologic Diagnosis of Inflammatory Skin Diseases: An Algorithmic Method Based on Pattern Analysis . 2nd ed. Baltimore: Williams & Wilkins; 1997:202.
Gilliam AC, Wood GS. Cutaneous lymphoid hyperplasias. Semin Cutan Med Surg . 2000;19:133–141.
Howard R, Frieden IJ. Papular urticaria in children. Pediatr Dermatol . 1996;13:246–249.
Self-limited cutaneous eruption of unknown cause that affects young adults and children
Two forms:
Acute or pityriasis lichenoides et varioliformis acuta (PLEVA), more severe form common in children and young adults
Chronic or pityriasis lichenoides chronica (PLC), the milder form more common in adults
Transitional forms with changes between the two extremes occur
In PLEVA, a papular, papulonecrotic, and occasionally vesiculopustular eruption occurs on the trunk and proximal extremities, usually resolves in a few weeks; crops of new lesions can continue to appear, and the disease process itself may have a chronic course
In PLC, recurrent crops of reddish-brown papules with adherent scales occur on the trunk and extremities and resolve in a few weeks
PLEVA
Parakeratosis and scale crust with neutrophils, vacuolar interface change, and individually necrotic keratinocytes ( Figure 2.16A )
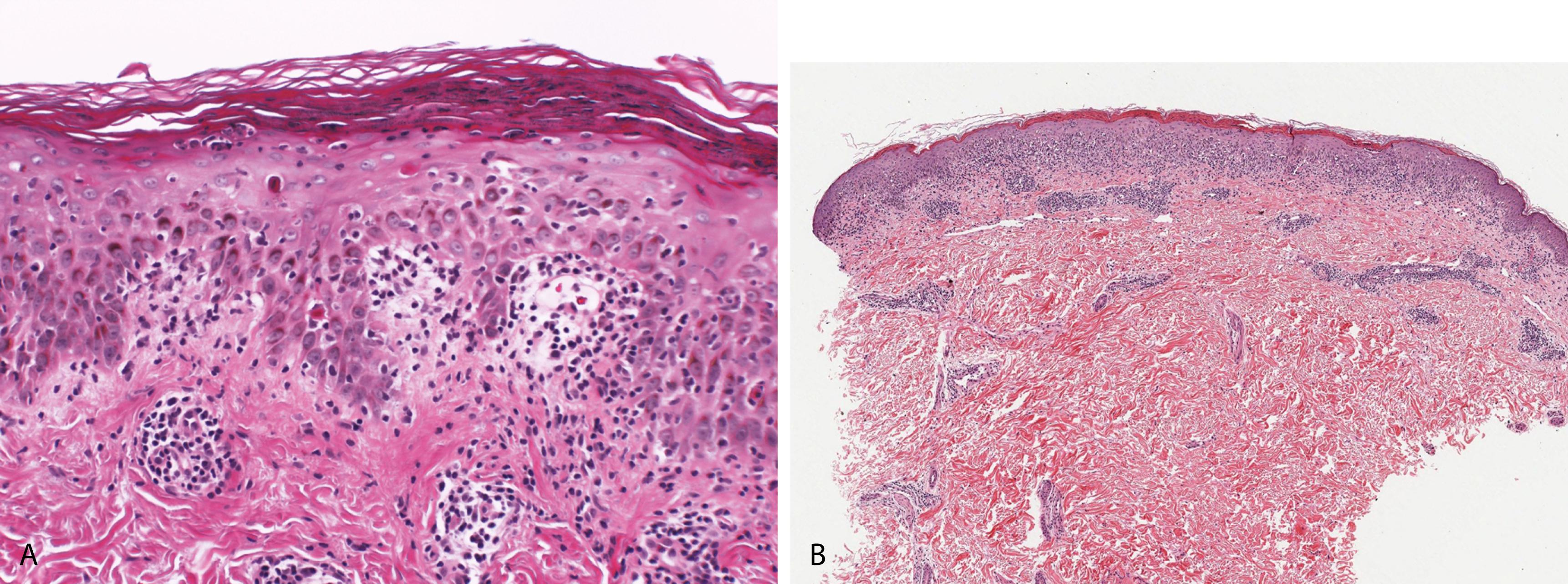
Superficial and deep perivascular and lichenoid, predominantly lymphocytic infiltrate with atypical CD30+ lymphocytes
Lymphocytic vasculitis (especially in the ulceronecrotic variant)
Papillary dermal edema and extravasated erythrocytes
PLC
Hyperkeratosis, parakeratosis, mild epidermal hyperplasia, interface change
Superficial perivascular lymphohistiocytic infiltrate with extravasated erythrocytes ( Figure 2.16B )
Lymphocytes predominantly of the cytotoxic/suppressor T-cell phenotype, presence of CD8+ T lymphocytes confirmed by immunohistochemistry
Immunohistochemical stain for CD30 may reveal scattered CD30+ cells in PLEVA
DIF may reveal IgM and C3 deposits along the basement membrane zone
Lymphomatoid Papulosis
Histopathologic features overlap with PLEVA
Wedge-shaped inflammatory cell infiltrate composed of clusters of atypical CD30+ lymphocytes with admixed eosinophils
Vesicular Insect Bite Reactions
Superficial and deep dermal infiltrate with numerous eosinophils, papillary dermal edema and extravasated erythrocytes helpful histopathologic clues
Density of infiltrate in PLC is less than that observed in PLEVA
Despite the unifying presence of CD30+ lymphocytes in PLEVA and lymphomatoid papulosis they are pathogenetically distinct entities
Bowers S, Warshaw EM. Pityriasis lichenoides and its subtypes. J Am Acad Dermatol . 2006;55:557–572.
Ersoy-Evans S., Greco MF, Mancini AJ, et al. Pityriasis lichenoides in childhood: a retrospective review of 124 patients. J Am Acad Dermatol . 2007;56:205–210.
Kempf W, Kazakov DV, Palmedo G, et al. Pityriasis lichenoides et varioliformis acuta with numerous CD30(+) cells: a variant mimicking lymphomatoid papulosis and other cutaneous lymphomas. A clinicopathologic, immunohistochemical, and molecular biological study of 13 cases. Am J Surg Pathol . 2012;36:1021–1029.
Well-defined, circumscribed patches occur at the same site in response to repeated intake of the drug
Lesions are slightly edematous and erythematous and may develop dusky centers and become bullous and heal with pigmentation
Increasing number of lesions can occur with each successive administration of the offending drug
Interface dermatitis with scattered individually necrotic keratinocytes, (changes identical to those seen in EM) ( Figure 2.17A )
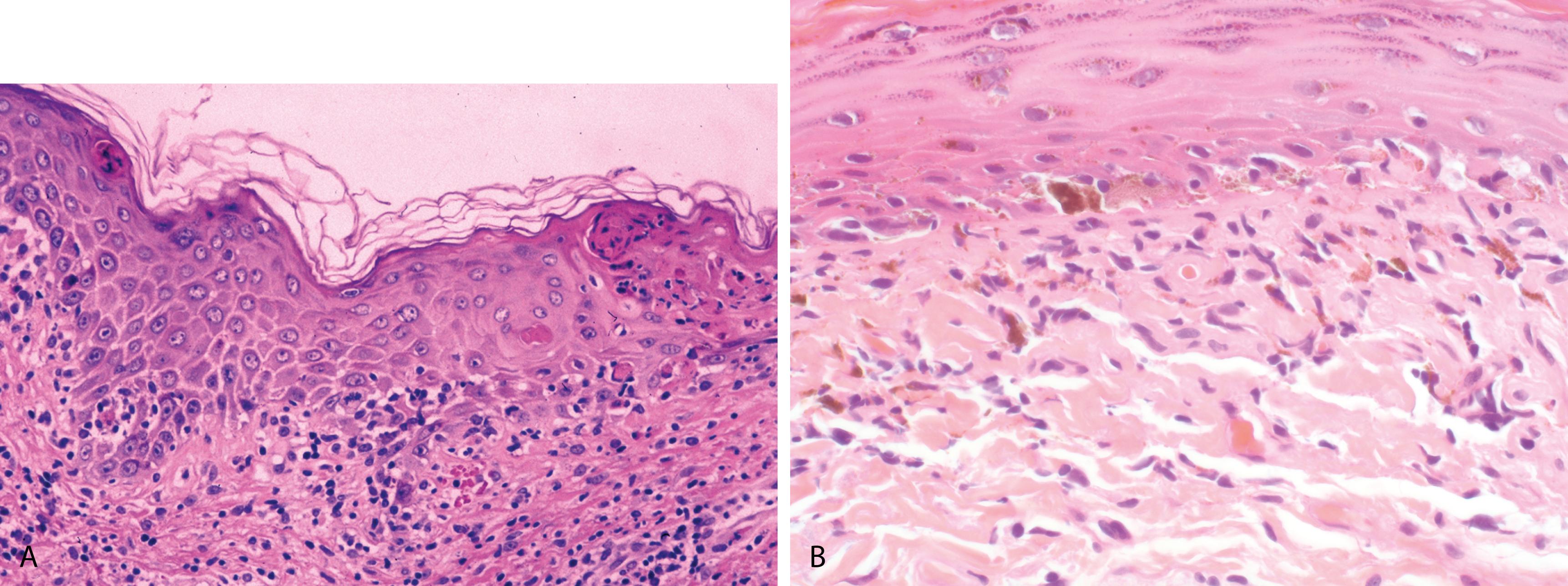
Superficial and deep perivascular and lichenoid inflammatory cell infiltrate composed of lymphocytes, eosinophils, and occasional neutrophils
Prominent pigment incontinence
Bullous variant resulting from more extensive epidermal necrosis has been described
Noncontributory
Noncontributory
Erythema Multiforme
Interface dermatitis with scattered individually necrotic keratinocytes and a predominantly superficial perivascular and patchy lichenoid lymphoid cell infiltrate
Erythema Dyschromicum Perstans/Lichen Planus Pigmentosus
Interface dermatitis with scattered individually necrotic keratinocytes; prominent pigment incontinence may be the only feature in older lesions( Figure 2.17B )
Histopathologic reaction pattern of interface dermatitis not specific or diagnostic and can be exhibited by several unrelated entities
Clinicopathologic correlation key to rendering correct diagnosis
Fixed drug eruptions occur most commonly with trimethoprim-sulfamethoxazole, acetylsalicylic acid, and phenolphthalein
Crowson AN, Magro CM. Recent advances in the pathology of cutaneous drug eruptions. Dermatol Clin . 1999;17:537–560, viii.
Gru AA, Salavaggione AL. Lichenoid and interface dermatoses. Semin Diagn Pathol . 2017;34:237–249.
Sehgal VN, Srivastava G. Fixed drug eruption (FDE): changing scenario of incriminating drugs. Int J Dermatol . 2006;45:897–908.
Multiple, small papules that are most often short lived, heal leaving oval scars, and are usually recurrent
Wedge-shaped superficial and deep perivascular and lichenoid lymphoid cell infiltrate with admixed eosinophils and scattered large and small atypical lymphocytes
Surface ulceration may be present
Reed Sternberg–like cells (in type A) and atypical lymphocytes with cerebriform nuclei resembling mycosis fungoides (in type B) are occasionally seen
Rarely, a dense monomorphous infiltrate of atypical lymphocytes resembling anaplastic large cell lymphoma (type C) can be seen in association with typical clinical features of lymphomatoid papulosis ( Figure 2.18 )
Immunohistochemical stain CD30 highlights the presence of clusters of CD30+ atypical lymphocytes
Clonal rearrangement of T-cell receptor gene may be present
Insect Bite Reaction
Superficial and deep perivascular and interstitial lymphoid cell infiltrate with numerous eosinophils
While reactive CD30+ lymphocytes can be seen these are typically single cells and not clustered
PLEVA
Histologic pattern of interface and lichenoid dermatitis similar to lymphomatoid papulosis
Reactive CD30+ lymphocytes can be seen, however, these are typically scattered single cells and not clustered
Presence of scattered CD30 positive cells not diagnostic of lymphomatoid papulosis as these may be present in reactive conditions
Presence of clusters of CD30 positive atypical lymphoid cells and a wedge-shaped inflammatory infiltrate diagnostic for lymphomatoid papulosis
Clinical correlates of histopathologic subtypes of lymphomatoid papulosis are of no known relevance
Progression of lymphomatoid papulosis to large cell anaplastic lymphoma (CD30 positive) can occur, suggesting that lymphomatoid papulosis may represent the benign end in the spectrum of CD30-positive T-cell lymphoproliferative disorders
Cerroni L. Lymphomatoid papulosis, pityriasis lichenoides et varioliformis acuta, and anaplastic large-cell (Ki-1+) lymphoma. J Am Acad Dermatol . 1997;37:287.
Guitart J, Querfeld C. Cutaneous CD30 lymphoproliferative disorders and similar conditions: a clinical and pathologic prospective on a complex issue. Semin Diagn Pathol . 2009;26:131–140.
LeBoit PE. Lymphomatoid papulosis and cutaneous CD30+ lymphoma. Am J Dermatopathol . 1996;18:221–235.
Werner B, Massone C, Kerl H, Cerroni L. Large CD30-positive cells in benign, atypical lymphoid infiltrates of the skin. J Cutan Pathol . 2008;35:1100–1107.
Hematogenous dissemination of causative organism, Treponema pallidum , results in a cutaneous eruption that can be macular, papular, papulosquamous, or, rarely, pustular
Associated constitutional symptoms such as fever and lymphadenopathy may be present; other manifestations include condyloma lata, syphilis cornee, lues maligna, and alopecia
Patchy/confluent parakeratosis containing neutrophils
Regular psoriasiform epidermal hyperplasia (minimal in macular lesions, prominent in condylomata lata) with focal spongiosis ( Figure 2.19A )
Interface change with scattered individually necrotic keratinocytes and papillary dermal edema
Superficial and deep perivascular, periadnexal, and lichenoid lymphoplasmacytic infiltrate; plasma cells may be present around nerves ( Figure 2.19B )
Infiltrate can be lymphocytic, lymphoplasmacytic, or lymphohistiocytic with rare granuloma formation
Silver stain (Warthin-Starry) may show spirochetes within the epidermis in one third of cases ( not used anymore because of high background associated with this stain )
Immunohistochemistry with a monoclonal antibody to T. pallidum yields better detection rates (organisms are more often found in the dermis compared to the epidermis)
Molecular detection of T. pallidum using PCR the gold standard
Mycosis Fungoides
Shows psoriasiform lichenoid pattern similar to syphilis; however, in mycosis fungoides, atypical lymphoid cells are present within the dermal infiltrate and in the mildly spongiotic epidermis
Plasma cells are not frequent
Subacute and Chronic Spongiotic Dermatitis, Including Photoallergic Dermatitis
May show some psoriasiform hyperplasia and spongiosis
In general, plasma cells are not prominent
Pityriasis Lichenoides
Can simulate secondary syphilis but shows predominantly a lymphocytic infiltrate without plasma cells
Psoriasis and Psoriasiform Drug Eruption and AIDS-Associated Psoriasiform Dermatitis
Inflammatory infiltrate is typically not deep as in secondary syphilis and plasma cells not prominent
Suprapapillary plate thinning is not a feature of secondary syphilis
Sarcoid and Other Conditions With a Prominent Granulomatous Pattern
May appear similar to the granulomatous pattern of secondary syphilis
Clinical correlation and serologic studies are necessary
Increased plasma cells in the inflammatory infiltrate not a constant histopathologic feature
The organism is more commonly seen in the dermis and best detected with immunohistochemistry or PCR-based techniques
Interface change with a perineural plasma cell-rich infiltrate highly suggestive of secondary syphilis
An unusual variant of secondary syphilis is lues maligna, an ulcerative form characterized by thrombotic endarteritis of vessels in the deep dermis resulting in ischemic necrosis
Serologic tests are the most common method of diagnosing syphilis
Hoang MP, High WA, Molberg KH. Secondary syphilis: a histologic and immunohistochemical evaluation. J Cutan Pathol. 2004;31:595-599.
Jeerapaet P, Ackerman AS. Histologic patterns of secondary syphilis. Arch Dermatol. 1973;107:373.
Muller H, Eisendle K, Brauninger W, et al. Comparative analysis of immunohistochemistry, PCR and focus-floating microscopy for the detection of T. pallidum in mucocutaneous lesions of primary, secondary and tertiary syphilis. Br J Dermatol. 2011;165:50-60.
Characterized clinically by fever, arthralgia, leukocytosis, and a cutaneous eruption of painful violaceous plaquelike or nodular lesions involving the extremities and face; truncal lesions are uncommon
Affects adults mainly
An underlying myeloproliferative disorder, most commonly acute myeloid leukemia or inflammatory disease and rarely solid tumors may be present
Sweet syndrome–like eruption reported with many drugs
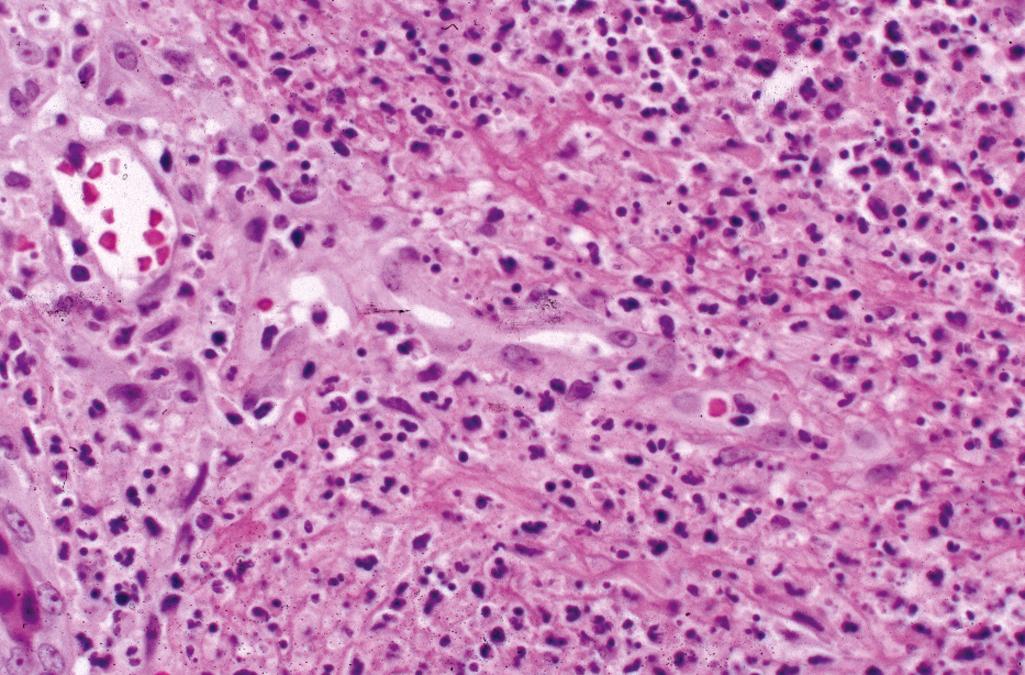
Dense, diffuse, upper dermal predominantly neutrophilic infiltrate, abundant leukocytoclasia, scattered lymphocytes, histiocytes, and eosinophils ( Figure 2.20 )
Marked papillary dermal edema, reactive endothelial cell atypia, and extravasated erythrocytes. Fibrinoid necrosis of vessel walls is not typically seen
Histiocytoid variant-infiltrate composed of immature myeloid cells; lesional cells are not neoplastic
Gram stain useful in excluding an infectious etiology such as cellulitis
Noncontributory
Leukocytoclastic Vasculitis
Vascular damage with fibrin deposition in the vessel wall seen in leukocytoclastic vasculitis is not a feature of Sweet syndrome
Pyoderma Gangrenosum
Inflammatory infiltrate (predominately neutrophilic) is deeper and denser than in Sweet syndrome
Surface ulceration, foci of folliculitis, and secondary vasculitis may be evident
Other Neutrophilic Dermatoses
Behçet disease (pathergic lesion), bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatosis—all characterized by a superficial and deep, predominantly neutrophil-rich inflammatory cell infiltrate without vasculitis and minimal papillary dermal edema
Neutrophil-rich inflammatory infiltrate, leukocytoclasia but NOT vasculitis and marked papillary dermal edema characteristic
Histiocytoid variant of Sweet syndrome is not a variant of leukemia cutis
Potential infectious etiology should be considered and excluded in all cases of neutrophilic dermatoses
Idiopathic ulceronecrotic skin disease that begins as follicular papules and pustules that eventually ulcerate
Lower extremities and trunk are often involved
Fully developed lesions show a necrotic center with a raised, undermined border with a dusky-purple hue
Pyoderma gangrenosum may be the cutaneous manifestation of underlying systemic diseases such as inflammatory bowel disease, connective tissue disease, hematopoietic malignancies, and liver diseases
Histologic features are nonspecific and not diagnostic—vary according to the age of the lesion and the area biopsied
Area biopsied:
Center of the lesion shows ulcer with necrosis, dense diffuse neutrophilic infiltrate, and occasionally secondary vasculitis ( Figure 2.21 )
Undermined border shows a mixed inflammatory cell infiltrate in addition to neutrophils
Periphery of the ulcer shows a lymphocytic and histiocytic inflammatory infiltrate
Age of the lesion
Early lesions show an intradermal microabscess
Advanced lesions show a perivascular lymphoplasmacytic infiltrate, reactive endothelial cell atypia and secondary vasculitis
Special stains and microbiologic cultures help exclude an infectious etiology
Noncontributory
Sweet Syndrome
Neutrophilic infiltrate that is typically more superficial, leukocytoclasia and marked papillary dermal edema
Bacterial Cellulitis
Requires demonstration of bacteria by Gram stain or microbiologic cultures
Histopathology nonspecific and varies with age of the lesion and area biopsied
Clinicopathologic correlation important in making the diagnosis and excluding other ulceronecrotic inflammatory disorders of the skin
Binus AM, Qureshi AA, Li VW, Winterfield LS. Pyoderma gangrenosum: a retrospective review of patient characteristics, comorbidities and therapy in 103 patients. Br J Dermatol . 2011;165:1244.
Ruocco E, Sangiuliano S, Gravina AG, et al. Pyoderma gangrenosum: an updated review. J Eur Acad Dermatol Venereol . 2009;23:1008.
Su WP, Schroeter AL, Perry HO, et al. Histopathologic and immunopathologic study of pyoderma gangrenosum. J Cutan Pathol . 1983;13:323.
Relatively rare, recurrent dermatosis of uncertain pathogenesis, characterized by sudden onset of erythematous patches that evolve into painful plaques
May be idiopathic or associated with insect bites, parasitosis, infections, myeloproliferative disorders, and drug reactions
Associated with peripheral eosinophilia in at least 50% of the patients
Spongiotic intraepidermal vesicles may be present
Dense diffuse dermal infiltrate composed predominantly of eosinophils and occasionally extending into the subcutaneous tissue
“Flame figures” (eosinophilic debris caused by degranulating eosinophils and may impregnate the collagen bundles) more prominent in older lesions ( Figure 2.15C )
Palisading histiocytes with central necrobiosis may be seen in florid lesions
Noncontributory
Noncontributory
Dermal hypersensitivity reactions (to drugs or an arthropod bite)—similar histopathology
Flame figures although a helpful diagnostic clue not specific for Wells syndrome
Eosinophilic cellulitis most likely represents an exaggerated dermal hypersensitivity reaction rather than a specific disease, and a search for inciting stimuli is warranted
Brasileiro LG, Abreu MAMM, Paschoal RS. Wells’ syndrome: the importance of differential diagnosis. An Bras Dermatol . 2019;94:370–372.
Caputo R, Marzano AV, Vezzoli P, Lunardon L. Wells syndrome in adults and children: a report of 19 cases. Arch Dermatol . 2006;142:1157.
Fujii K, Tanabe H, Kanno Y, et al. Eosinophilic cellulitis as a cutaneous manifestation of idiopathic hypereosinophilic syndrome. J Am Acad Dermatol . 2003;49:1174–1177.
Contagious, pruritic, papulovesicular, and pustular eruption most pronounced on the abdomen, buttocks, and anterior axillary folds caused by the mite Sarcoptes scabiei
Eruption believed to be a hypersensitivity reaction
Burrows, produced by the female mite, typically involve the palms, web spaces between fingers, and male genitalia
Persistent pruritic nodules/nodular scabies involving most commonly the scrotum can be seen up to several months after treatment
Sections taken from the burrow show a tunnel-like space (“molting pouches”) between layers of parakeratosis containing the mite/larvae/ova/feces ( Figure 2.22 ).
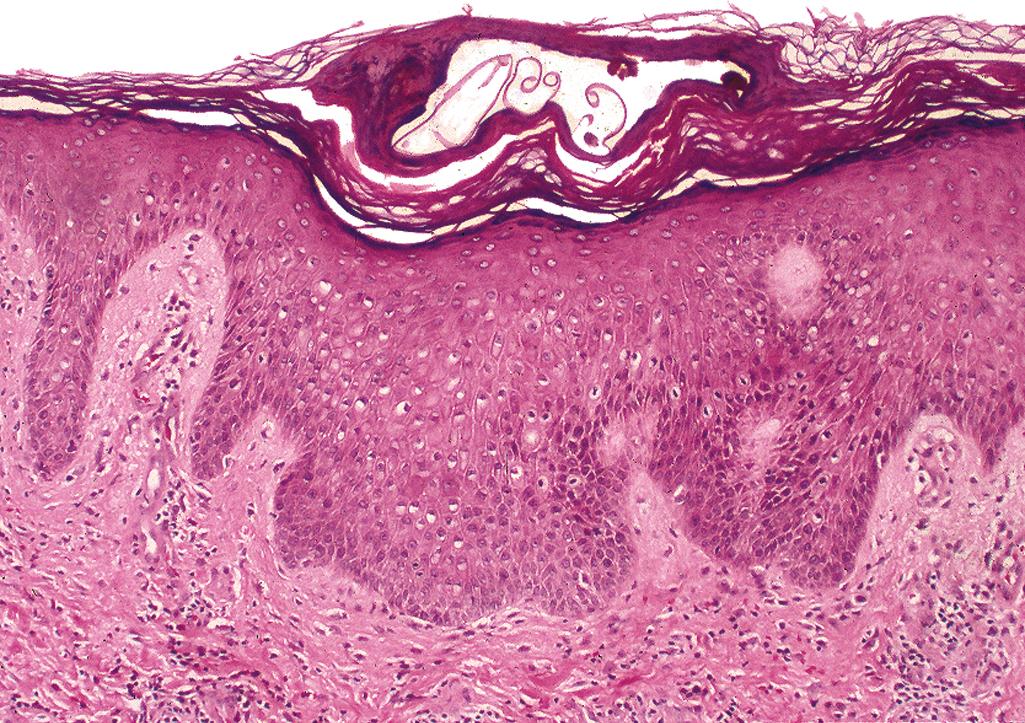
Spongiosis and vesiculation may be present in the epidermis
Superficial and deep dermal infiltrate containing varying numbers of eosinophils
Persistent nodular lesions show dense, diffuse, mixed inflammatory cell infiltrate containing eosinophils, thick-walled blood vessels, and occasionally atypical mononuclear cells, “pseudolymphomatous” pattern may also be seen. Mite is generally absent in these lesions
Noncontributory
A suspected burrow can be shaved, placed on a glass slide, and examined under oil immersion
In the absence of the mite or its products in the cornified layer, the histologic changes cannot be distinguished from other hypersensitivity reactions such as those caused by arthropod bites
Norwegian scabies is a rare variant in which an immeasurable number of mites is present within the cornified layer; generally seen in immunocompromised patients or patients with poor hygiene
Scabetic by-product or “feces” in the stratum corneum may be the only clue
Pink “pigtails” in the stratum corneum representing egg fragments or casings left behind after the mite hatches may be the only clue
Brites C, Weyll M, Pedroso C, et al. Severe and Norwegian scabies are strongly associated with retroviral (HIV-1/HTLV-1) infection in Bahia, Brazil. AIDS . 2002;16:1292.
Chosidow O. Scabies and pediculosis. Lancet . 2000;355:819–826.
Fernandez N, Torres A, Ackerman AB. Pathological findings in human scabies. Arch Dermatol . 1977;113:320.
Benign non-Langerhans cell histiocytosis that most commonly occurs during infancy (within first 6 months of life) but can be seen occasionally in adults (adult xanthogranuloma)
About 20% are congenital
Usually presents as single or multiple tan to pink-red nodules that almost always regress over time to a tan macule or depression
Occasionally found in the deep soft tissue
Eye, particularly the uveal tract, most frequent site of extracutaneous involvement
Distinctive architecture—exophytic lesion with a well-defined epidermal collarette
Lesional cells—uniform-appearing histiocytes with an eosinophilic, vacuolated, or xanthomatous cytoplasm,
Touton giant cells are typically seen ( Figure 2.23 )
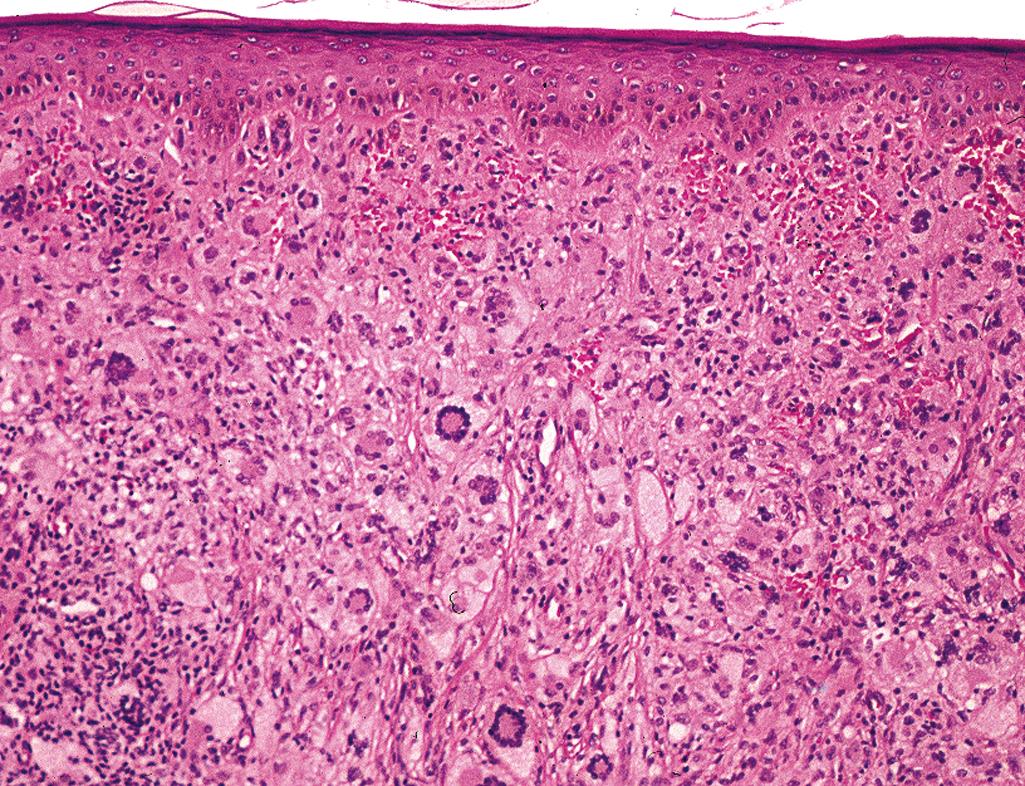
Admixture of neutrophils, eosinophils, and lymphocytes
Resolving lesion resembles dermatofibroma
Oil red O highlights intracytoplasmic neutral lipids
CD68 and factor XIIIa positive
CD1a negative; S-100 protein usually negative
Noncontributory
Langerhans Cell Histiocytosis (Eosinophilic Granuloma)
Presence of histiocytes positive for CD1a and S-100 protein and admixed eosinophils, electron microscopy demonstrates Birbeck granules
Fibrous Histiocytoma (Dermatofibroma)
Spindled fibroblasts and histiocytic cells arranged in a storiform pattern
Peripheral collagen entrapment and overlying epidermal hyperplasia with pigmentation of basal keratinocytes
Xanthoma
Composed of sheets of histiocytes containing abundant intracytoplasmic lipid
Cholesterol clefts and multinucleate giant cells are typical
Pathogenesis remains uncertain; believed to be a reactive rather than neoplastic process
Not associated with a lipid abnormality
Skin lesions almost always regress with time and ultimately appear as a slight depression on the skin surface
Overall prognosis is excellent
Distinctive architecture with epidermal collarette Touton giant cells not specific for juvenile xanthogranuloma—can also be seen in dermatofibroma and xanthoma
Burgdorf WH, Zelger B. JXG, NF1, and JMML: alphabet soup or a clinical issue? Pediatr Dermatol . 2004;21:174.
Dehner LP. Juvenile xanthogranulomas in the first two decades of life: a clinicopathologic study of 174 cases with cutaneous and extracutaneous manifestations. Am J Surg Pathol . 2003;27:579.
Freyer DR, Kennedy R, Bostrom BC, et al. Juvenile xanthogranuloma: forms of systemic disease and their clinical implications. J Pediatr . 1996;129:227–237.
Typically occurs in adults
Most frequently presents as a well-circumscribed red-brown cutaneous nodule with a red-brown to yellow cut surface
May present as localized (giant cell reticulohistiocytoma) or systemic disease (multicentric reticulohistiocytosis)
Cutaneous reticulohistiocytoma (localized form)—may present as single or multiple skin lesions with clinical features similar to xanthogranuloma
Multicentric reticulohistiocytosis (systemic form)—rare, may involve lymph nodes, heart, bone, and joints in addition to widespread skin involvement, may present with progressive erosive arthritis, fever, and weight loss, association with hyperlipidemia, internal malignancies, and autoimmune diseases
Essentially similar features in both localized and systemic forms
Well-defined infiltrate of multinucleate, uniform epithelioid histiocytes with abundant eosinophilic (“oncocytic”) cytoplasm, multinucleate giant cells with “ground-glass” cytoplasm ( Figure 2.24 )
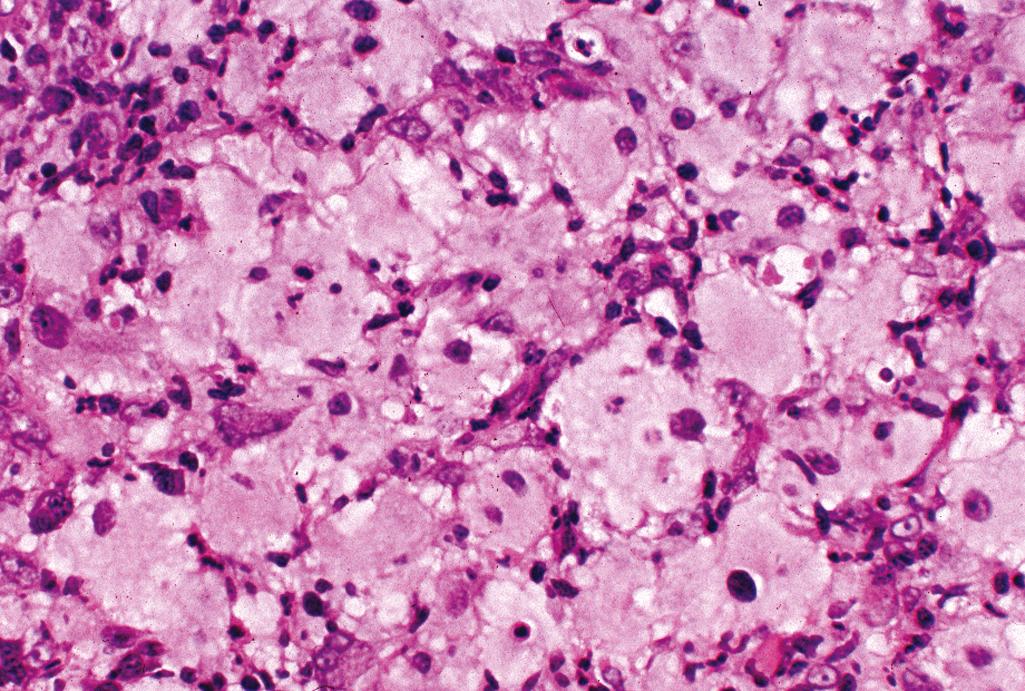
Infrequent mitotic activity
Scattered chronic inflammatory cells
Lesional cells are CD68 positive and S-100 protein and CD1a negative
Noncontributory
Langerhans Cell Histiocytosis
Infiltrate of large histiocytoid CD1a and S100 protein positive cells with nuclear grooves
Juvenile Xanthogranuloma
Distinctive architecture, polymorphous infiltrate, presence of Touton giant cells diagnostic
Solitary and disseminated form of reticulohistiocytoma exhibit identical histopathology and cytomorphology
Predominant population of oncocytic histiocytes and multinucleate giant cells with “ground glass” cytoplasm distinctive
Occasional histiocytes with “ground glass” cytomorphology can be seen in other histiocyte rich entities
Disseminated reticulohistiocytosis is associated with various malignancies (carcinoma of the breast, colon, or lung) or systemic disease (tuberculosis, diabetes, hypothyroidism)
Polyarthritis seen in the disseminated form is due to infiltrate of similar histiocytic cells found in skin around the joints
Jung HD, Kim HS, Lee JY, et al. Multicentric reticulohistiocytosis. Acta Derm Venereol . 2013;93:124–125.
Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol . 2006;30:521.
Snow JL, Muller SA. Malignancy-associated multicentric reticulohistiocytosis: a clinical, histological, and immunophenotypic study. Br J Dermatol . 1995;133:71–76.
Benign granulomatous process of unknown etiology
Occurs most commonly in children and young adults; females more commonly affected than males
Predilection for areas of trauma and exposure, typically the dorsal surface of the hands and feet, ankles, knees, and elbows
Single or multiple annular dermal plaques with central clearing and raised erythematous borders
Spontaneously regress but can recur
Diagnostic histopathologic triad—palisaded lymphohistiocytic infiltrate with occasional multinucleate giant cells (some of which can demonstrate elastophagocytosis) minute foci of necrobiosis (mucinous degeneration of the collagen) and increased dermal mucin ( Figure 2.25 )
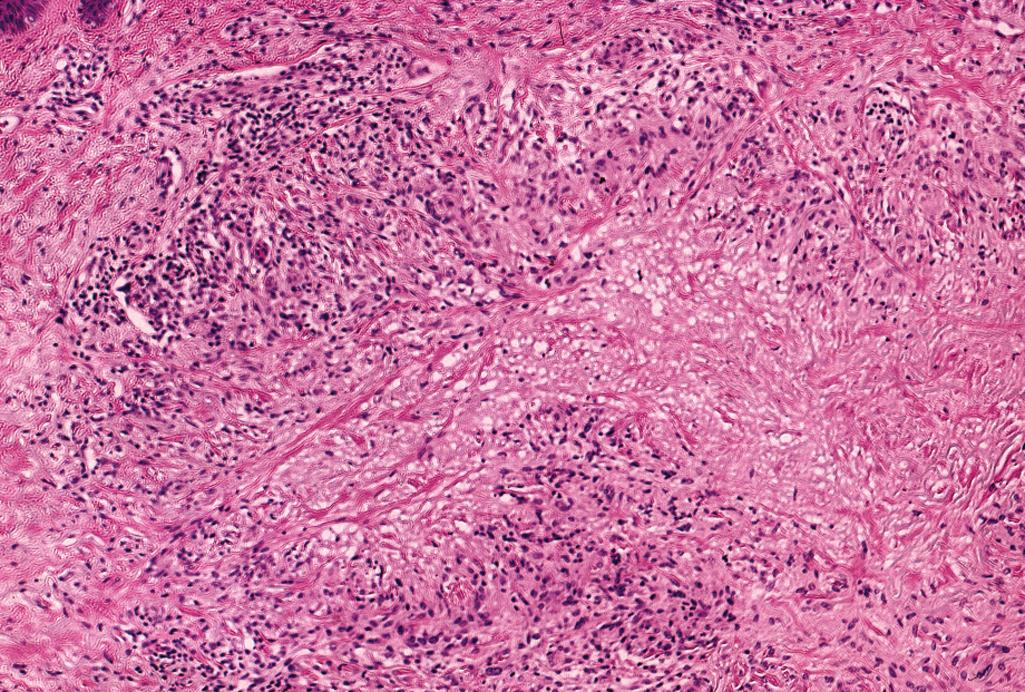
Typically involves upper and middle dermis, occasionally only the upper or deep dermis
Deep variant (subcutaneous granuloma annulare) typically found in a pediatric population
Perivascular infiltrates of lymphocytes; eosinophils in varying numbers may be present
Occasional neutrophils and nuclear fragmentation in areas of mucinous degeneration
Classified as a “blue granuloma” because of the presence of mucinous necrobiosis
Colloidal iron stain highlights mucin
Noncontributory
Rheumatoid Nodule
Characterized by a deeply eosinophilic homogenous foci of fibrinoid degeneration of collagen surrounded by a palisade of lymphohistiocytic infiltrate
Typically seen in the deep dermis or subcutis
Necrobiosis Lipoidica
Biopsy specimens are usually rectangular
Foci of basophilic degeneration of collagen stratified between layers of an inflammatory cell infiltrate (“sandwich appearance”) that typically contains plasma cells
Involvement of deep dermis is typical
Diagnostic histopathologic triad (palisaded lymphohistiocytic infiltrate, necrobiosis and increased dermal mucin)
Subcutaneous variant of granuloma annulare (pseudorheumatoid nodule) typically presenting in children with deep-seated nodules in the dermis or subcutaneous fat may be difficult to distinguish from rheumatoid nodule
A well-known diagnostic pitfall is diagnosing epithelioid sarcoma as granuloma annulare, and vice versa
Barren DF, Cootauco MH, Cohen BA. Granuloma annulare: a clinical review. Prim Care Pract . 1997;1:33–39.
Gunes P, Goktay F, Mansur AT, et al. Collagen-elastic tissue changes and vascular involvement in granuloma annulare: a review of 35 cases. J Cutan Pathol . 2009;36:838.
Magro CM, Crowson AN, Regauer S. Granuloma annulare and necrobiosis lipoidica tissue reactions as a manifestation of systemic disease. Hum Pathol . 1996;27:50–56.
Degenerative cutaneous disease of unknown etiology
More common in women than men
Characteristically affects the anterior tibial surface but also has a predilection for the thighs, popliteal areas, and dorsum of the feet and arms
Indurated yellow-brown oval plaques with a violaceous border
Center of the plaque may later become atrophic with a distinctive yellow waxy hue
Very small proportion occur in patients with diabetes mellitus
Rectangular contour of biopsy sample, alternating horizontal layers of basophilic degeneration of collagen and an inflammatory infiltrate of histiocytes, lymphocytes, and plasma cells ( Figure 2.26 ) diagnostic (“sandwich” or tiered appearance at scanning magnification)
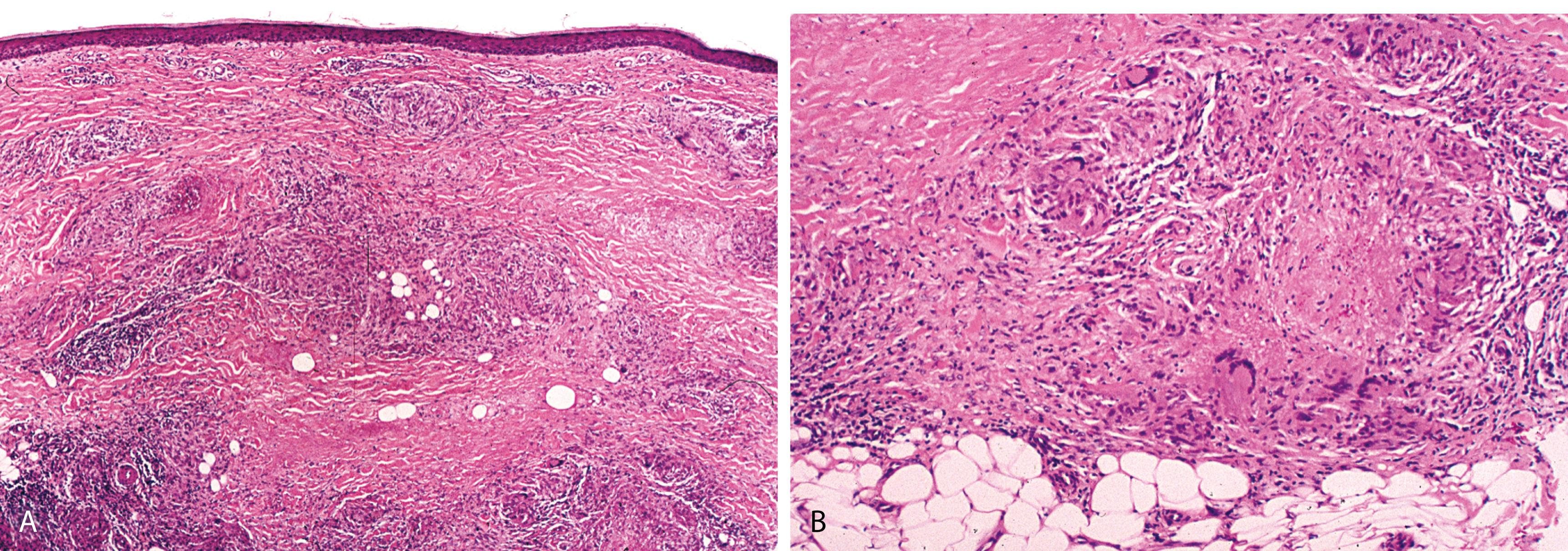
Vascular changes (reactive endothelial cell atypia and thickened vessel walls) are features shared with diabetic microangiopathy
Zones of dermal sclerosis
Sarcoidal type granulomas are seen on occasion
Noncontributory
DIF may reveal IgM, IgA, C3 and fibrinogen in thickened vessels
Rheumatoid Nodule
Deeply eosinophilic homogenous foci of fibrinoid degeneration of collagen surrounded by a palisade of lymphohistiocytic infiltrate typically seen in the deep dermis or subcutis
Granuloma Annulare
Demarcated zones of necrobiosis with mucin surrounded by a palisade of lymphohistiocytic infiltrate, typically involving the upper half of the dermis
Necrobiotic Xanthogranuloma
Atrophic epidermis, granulomatous inflammation with Touton giant cells of the deep dermis and subcutis, necrobiosis with cholesterol clefts
Characteristic tiered appearance at scanning magnification—zones of necrobiosis alternating with layers of an inflammatory infiltrate
Presence of plasma cells in the inflammatory infiltrate a helpful clue
Less than 1% of the patients with diabetes develop necrobiosis lipoidica
Lowitt MH, Dover JS. Necrobiosis lipoidica. J Am Acad Dermatol . 1991;25:735–748.
Magro CM, Crowson AN, Regauer S. Granuloma annulare and necrobiosis lipoidica tissue reactions as a manifestation of systemic disease. Hum Pathol . 1996;27:50–56.
O’Toole EA, Kennedy U, Nolan JJ, et al. Necrobiosis lipoidica: only a minority of patients have diabetes mellitus. Br J Dermatol . 1999;140:283–286.
Chronic deep-seated inflammatory nodule that occurs in patients with rheumatoid arthritis and occasionally in patients with SLE
Seen during the course of the disease in 30% to 40% of patients with rheumatoid arthritis
Predilection for areas subject to mechanical trauma, typically in para-articular locations, including metacarpophalangeal and proximal interphalangeal joints
Solitary or multiple, firm, nontender, freely mobile, large subcutaneous nodules
Central areas of homogeneous eosinophilic degeneration of collagen surrounded by a peripheral palisade of histiocytes and lymphocytes ( Figure 2.27 ), located in the subcutaneous tissue and deep dermis
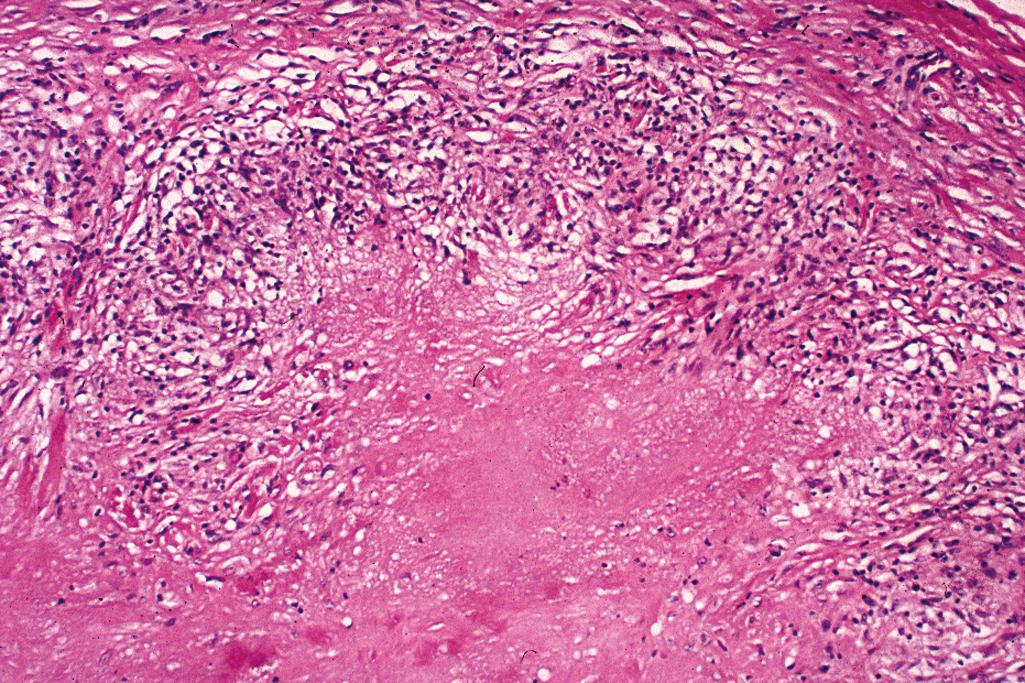
Classified as a “red granuloma” because of the central homogenous eosinophilic areas
Noncontributory
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here