Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Salivary gland infections are in general not a common condition. The incidence, complications, and mortality rates of salivary gland infections have decreased largely due to improved oral hygiene and advances in health care. There remains a risk of mortality. Bacterial infection of the salivary glands is believed to be due to retrograde spread through the papillae of the ducts, salivary stasis, and/or obstruction of salivary flow.
Retrograde spread can arise from dental infections or tonsillitis. Nosocomial organisms can enter the oral cavity after oral instrumentation. Neonates can develop salivary gland infections secondary to transitory bacteremia. Salivary stasis may occur as a result of dehydration or reduced salivary production. Hospitalization, the post-operative state, medically prescribed reduced fluid intake, advanced age, radiation therapy, medications (anticholinergics, diuretics, antidepressants), Sjögren syndrome, depression, and anorexia all contribute as risk factors to stasis. The most common cause of obstruction to saliva flow is a calculus within the salivary gland or duct ( Fig. 7.1.1 ).
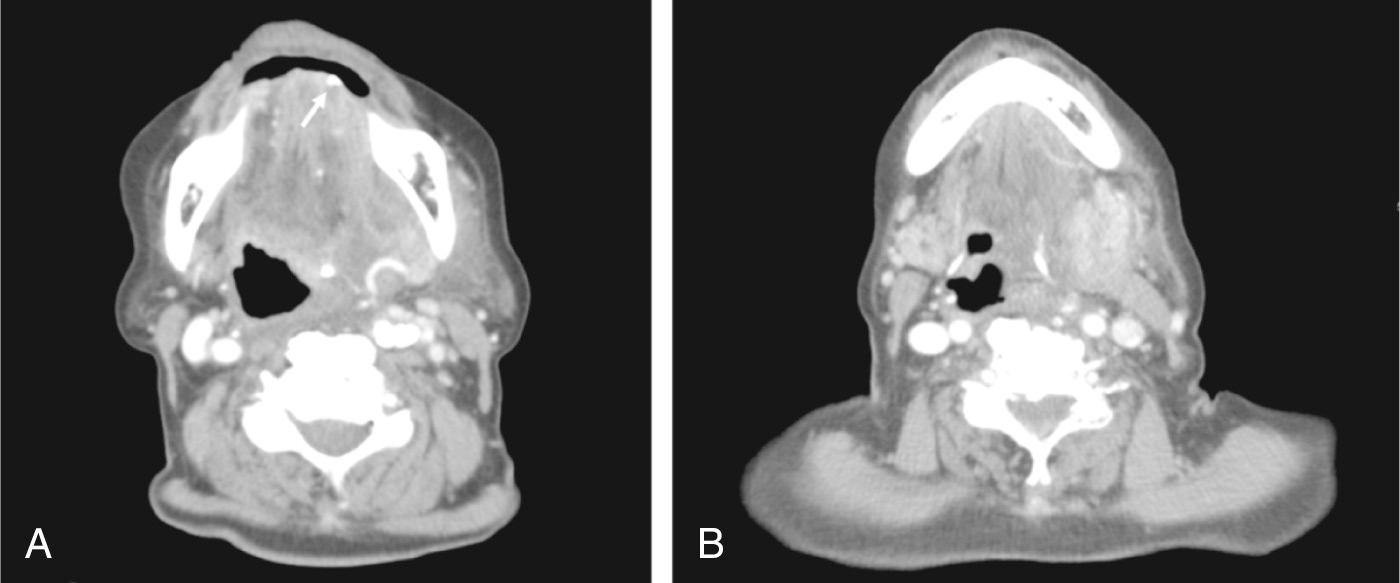
The parotid gland is most often involved in bacterial infections. Parotid gland saliva contains fewer lysozymes, and lower concentrations of secretory immunoglobulin A (IgA), and sialic acids which inhibit bacterial adherence by causing agglutination of bacteria and glycoproteins. Infrequently, seeding of bacteria into the gland during fine needle aspiration cytology (FNAC) of a pre-existing lesion such as Warthin tumor may result in local inflammation.
Staphylococcus aureus is by far the most predominant organism cultured, followed by anaerobic organisms such as Peptostreptococcus spp., Prevotella spp. and Fusobacterium spp. Other aerobic and facultative pathogens sometimes implicated include Streptococcus spp. (including S. pneumoniae and S. pyogenes ) and Haemophilus influenzae . There is roughly an equal proportion of aerobic and facultative bacteria compared with anaerobic bacteria, with a mean of 1.6 isolates per specimen. Klebsiella pneumoniae and Escherichia coli are common in hospital-acquired parotitis. Adherence of Gram-negative organisms at the duct papillae can occur in immunocompromised or debilitated patients from a deficiency of fibronectin, a glycoprotein in the extracellular matrix. Children in Southeast Asia may develop parotitis secondary to Burkholderia pseudomallei , an aerobic organism found in soil and surface water. Lastly, methicillin-resistant Staphylococcus aureus (MRSA) parotitis can occur in neonates.
Bacterial parotitis is predominantly unilateral. Patients present with a diffuse painful swelling in the cheek or angle of jaw that is tender and warm. The thick fascial layer around the parotid often contains the infection within the gland, hence it is uncommon for parotid abscesses to erupt through the skin. Intraorally, the papilla may appear inflamed. Pus can sometimes be milked out of or be seen from the duct. The floor of the mouth may be elevated and tense in submandibular gland infections. An abscess involving the deep parotid lobe and deep neck spaces may manifest as trismus, neck swelling and medialization of the oropharyngeal wall or tonsil ( Fig. 7.1.2 ). Rare cases of facial nerve dysfunction are caused by nerve compression or neurotoxins causing neuritis. Neoplasm, however, needs to be excluded.
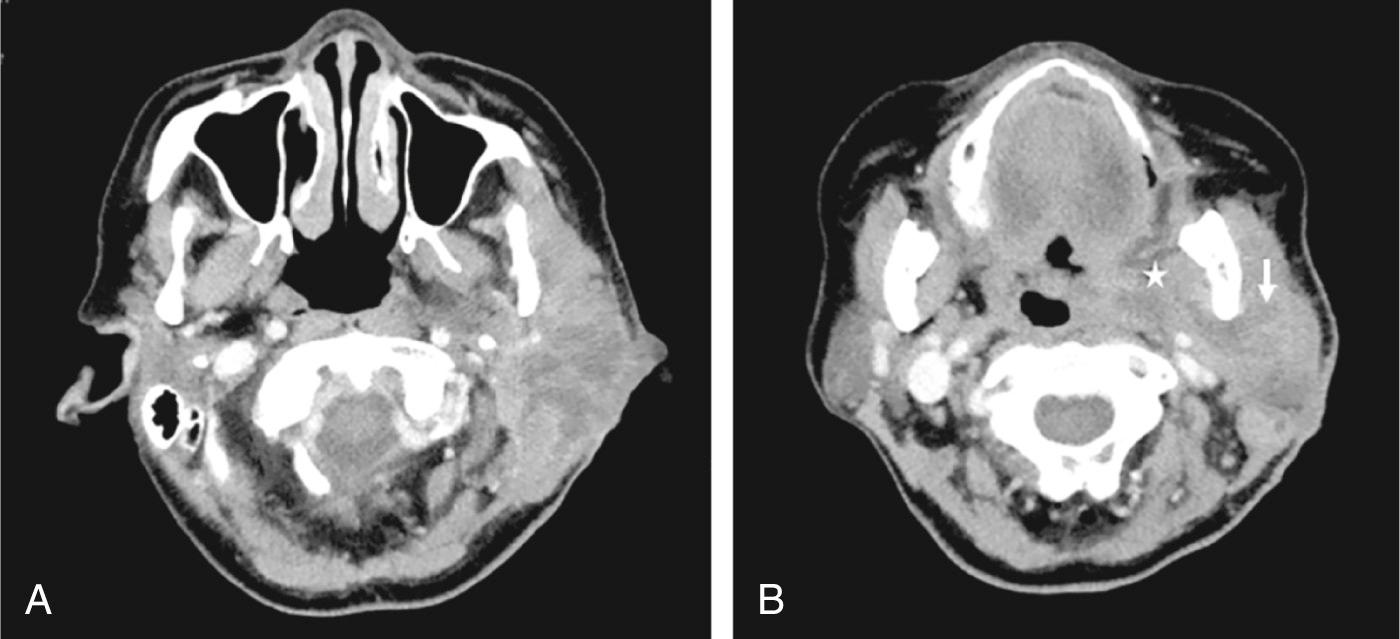
Imaging is often not necessary at the initial presentation for most infections. Ultrasound, although operator dependent, can be performed initially. Computed tomography (CT) or magnetic resonance imaging (MRI) of the salivary glands offer much greater detail and can detect an underlying calculus, abscess or neoplasm. Confirming the size of the abscess, sepsis, and failure to improve are indications for imaging.
An infection may develop into a frank abscess. Inflammation may spread to the surrounding muscles such as the temporalis and pterygoids, resulting in trismus. Involvement of the deep neck spaces may lead to sepsis, carotid artery aneurysm, jugular vein thrombosis, airway compromise, and even mediastinitis. Elderly patients are more susceptible to sepsis from a poorly controlled salivary gland infection.
Conservative measures include adequate hydration, warm packs, massage, and sialogogues. Antibiotic choice is often empirical. Amoxicillin/clavulanic acid targets Gram-positive and anaerobic flora. Clindamycin can be selected if the patient is allergic to penicillin. If hospital-acquired, Gram-negative organisms should be covered. Intravenous antibiotics can be initiated if there is no improvement after 24–48 hours.
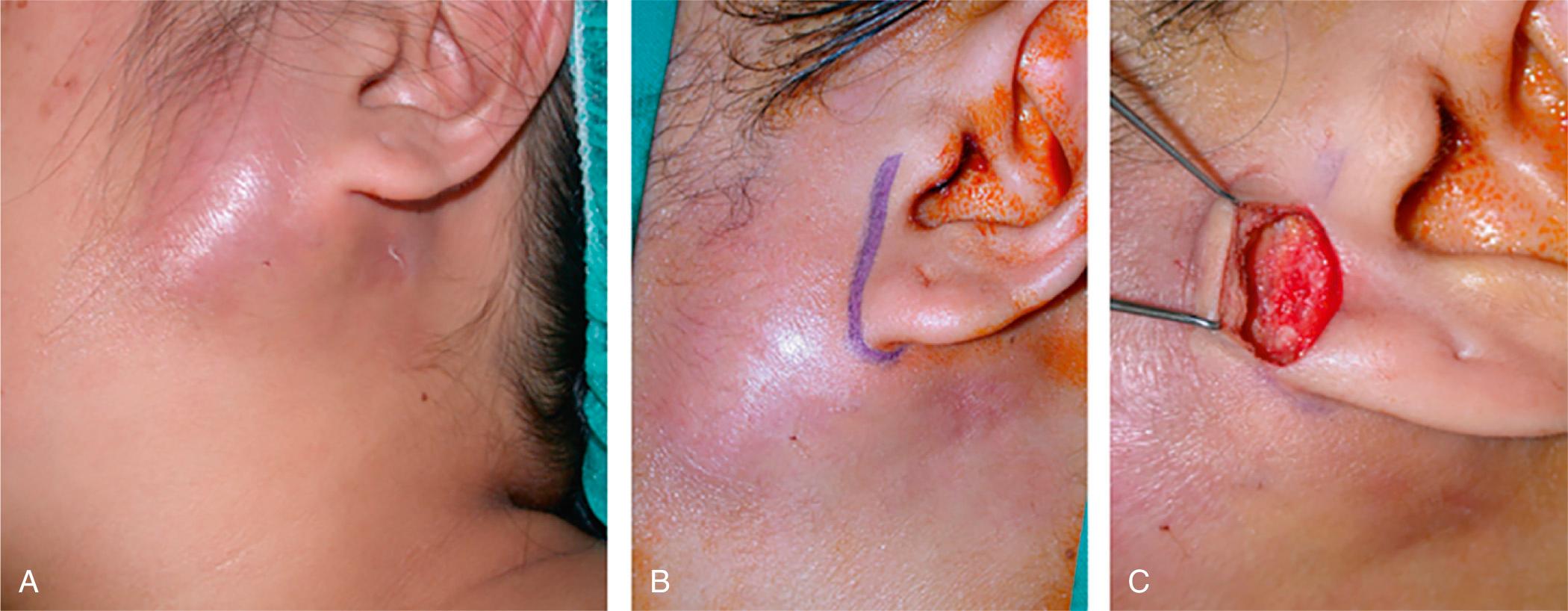
Surgery may be necessary if the gland contains a sizable abscess. A linear incision in the form of the superior limb of a modified Blair incision and a skin crease incision should be employed for parotid and submandibular gland abscesses, respectively ( Fig. 7.1.3 ). A skin flap should be raised to expose the gland. Needle aspiration prior to making an incision into the gland may be helpful. Incision into the parotid gland should be made parallel to the direction of the facial nerve fibers. The facial nerve is not dissected or exposed deliberately. Blunt dissection is used to explore the pocket, followed by irrigation and placement of a passive drain. The drain can guide insertion of a cannula to facilitate saline flushing. Serial aspiration with ultrasound is a method of managing a discrete abscess in patients who refuse or are unfit for surgery under general anesthesia.
Chronic bacterial parotitis, 10 times more common in children (see Chapter 8.4 ), can manifest as either intermittent parotid swelling or a continuous discomfort within the gland. MRI sialography reveals salivary duct strictures and dilatations that have a “sausage-like appearance”.
The parotid gland is the most commonly infected salivary gland, followed by the submandibular gland.
Bacterial infection of the salivary glands is due to retrograde spread through the papillae of the ducts, salivary stasis, and/or obstruction of salivary flow.
Staphylococcus aureus is the most common organism cultured in acute bacterial infections, followed by anaerobic organisms.
Conservative management including antibiotics is usually adequate for acute salivary gland infections, failing which surgery can be performed.
Sjögren syndrome (SS) is a chronic autoimmune disease characterized by hypofunction of salivary and lacrimal glands, and possible systemic multiorgan manifestations. The hallmark of the disease is dry eyes, dry mouth, fatigue, and joint pain. The clinical features of SS can be divided into two broad categories of exocrine glandular features and extraglandular features. SS is defined as primary when it presents alone and secondary when it is in the presence of another autoimmune disease. The autoimmune diseases associated with SS include rheumatoid arthritis, systemic lupus erythematosus, etc. SS is most common in women in their 50s but can affect men, adolescents, and young adults. The female to male ratio is 9 : 1, with a prevalence of 0.03–0.1% in the adult population.
Although there are defined classification criteria for SS, practical challenges remain for the clinician in making an accurate diagnosis because there is no single gold standard test for diagnosing SS. The investigators from the Sjögren's International Collaborative Clinical Alliance (SICCA) and the European League Against Rheumatism (EULAR) Sjögren's Task Force formed the International Sjögren's Syndrome Criteria Working Group. In 2016, this working group endorsed the American College of Rheumatology/European League Against Rheumatism classification criteria for primary SS ( Table 7.2.1.1 ). The classification of primary SS applies to any individual who meets the inclusion criteria, does not have any of the conditions listed as exclusion criteria, and has a score of ≥4 when the weights from the five criteria items are summed. The inclusion criteria are applicable to any patient with at least one symptom of ocular or oral dryness, defined as a positive response to at least one of the following questions: (1) Have you had daily, persistent, troublesome dry eyes for more than 3 months? (2) Do you have a recurrent sensation of sand or gravel in the eyes? (3) Do you use tear substitutes more than three times a day? (4) Have you had a daily feeling of dry mouth for more than 3 months? (5) Do you frequently drink liquids to aid swallowing dry food? The exclusion criteria that are also considered as the differential diagnosis of SS include: (a) history of head and neck radiation treatment; (b) active hepatitis C infection (with confirmation by polymerase chain reaction [PCR]); (c) AIDS; (d) sarcoidosis; (e) amyloidosis; (f) graft-versus-host disease; (g) IgG4-related disease. Focal lymphocytic sialadenitis with a focus score of at least 1 focus/4 mm 2 of tissue and anti-SSA/Ro antibody positivity have the highest weight in the criteria set, each scoring 3 points.
| Item | Weight/Score |
|---|---|
| Labial salivary gland with focal lymphocytic sialadenitis and focus score of ≥1 foci/4 mm 2 . a | 3 |
| Anti-SSA/Ro-positive. | 3 |
| Ocular staining score ≥5 (or van Bijsterveld score ≥4) in at least one eye. b,c | 1 |
| Schirmer test ≤5 mm/5 min in at least one eye. b | 1 |
| Unstimulated whole saliva flow rate ≤0.1 mL/min. b,d | 1 |
a The histopathologic examination should be performed by a pathologist with expertise in the diagnosis of focal lymphocytic sialadenitis and focus score count, using the protocol described by Daniels et al.
b Patients who are normally taking anticholinergic drugs should be evaluated for objective signs of salivary hypofunction and ocular dryness after a sufficient interval without these medications in order for these components to be a valid measure of oral and ocular dryness.
c Ocular staining score described by Whitcher et al. ; van Bijsterveld score described by van Bijsterveld.
d Unstimulated whole saliva flow rate measurement described by Navazesh and Kumar.
Patients suspected of SS should undergo a thorough medical history and physical examination with attention to the characteristic features of SS and other systemic manifestations of rheumatic diseases, e.g., rheumatic arthritis, systemic lupus erythematosus, and conditions that may resemble SS.
Salivary hypofunction can be tested by sialometry and scintigraphy, but these two tests are often not available in routine clinical setting. Minor salivary gland biopsy with focal lymphocytic sialadenitis and a focus score of ≥1 foci/4 mm 2 provides a histologic ground for the diagnosis. The focus score is defined as the number of lymphocytic foci adjacent to normal-appearing mucous acini and containing >50 lymphocytes/4 mm 2 of glandular tissue. However, many patients are reluctant to undergo this procedure since it carries a certain risk of complications.
There are a growing number of studies supporting the use of salivary gland imaging for the diagnosis of SS. The two most often used imaging modalities are magnetic resonance imaging (MRI) and ultrasonography. MRI of the parotid glands in SS is characterized by inhomogeneity of the parenchyma on both T1 and T2 weighted sequences. There is typically a nodular pattern, characterized by multiple hypo- and hyperintense areas of varying size. This “honeycomb” or “salt-and-pepper” pattern is thought to arise from fatty infiltration, fibrosis, ductal dilation, and lymphoid infiltration of parotid lobules. MRI results correlate well with those of salivary gland biopsy.
In recent years, there has been rapid advancement in the ultrasonography of the salivary gland. The ultrasonographic images are characterized by multiple hypoechoic areas with convex borders. Hyperechoic linear bands, cysts, and calcifications may be evident in more advanced disease. In contrast to MRI, ultrasound is more accessible and can be readily used. Recent data have shown a good to excellent diagnostic accuracy of B-mode ultrasound. However, it should also be kept in mind that ultrasound is operator-dependent. Further studies are required to validate and standardize the ultrasound definition, diagnostic procedures, and scoring system of salivary gland in SS.
All patients should be evaluated by an ophthalmologist for the presence of dry eye on a formal examination. The examinations include the Schirmer test, a slit-lamp examination for assessment of tear break-up time, and ocular surface staining.
Patients with primary SS often possess antibodies to the Ro/SSA (anti-Ro/anti-SSA) or La/SSB antigens (anti-La/anti-SSB), and many patients possess both. The frequency of anti-Ro/SSA and/or anti-La/SSB antibodies has varied between studies, but generally 60–80% of patients with primary SS exhibit one or both of these autoantibodies.
Anti-Ro/SSA antibody reactivity is directed at two distinct polypeptide antigens with molecular weights of 60 kDa and 52 kDa that are encoded by different genes. Patients with SS may express activity against both the 60 kDa and the 52 kDa proteins or against either protein alone. The majority show reactivity against both antigens. The presence of anti-Ro52 antibodies has been associated with more severe disease.
In considering the diagnosis of SS, it is important to bear in mind that anti-Ro/SSA is not a specific antibody to SS. Other conditions such as systemic lupus erythematosus, systemic sclerosis, inflammatory myopathies, autoimmune liver diseases, etc., could also be characterized by the presence of anti-Ro/SSA.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here