Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
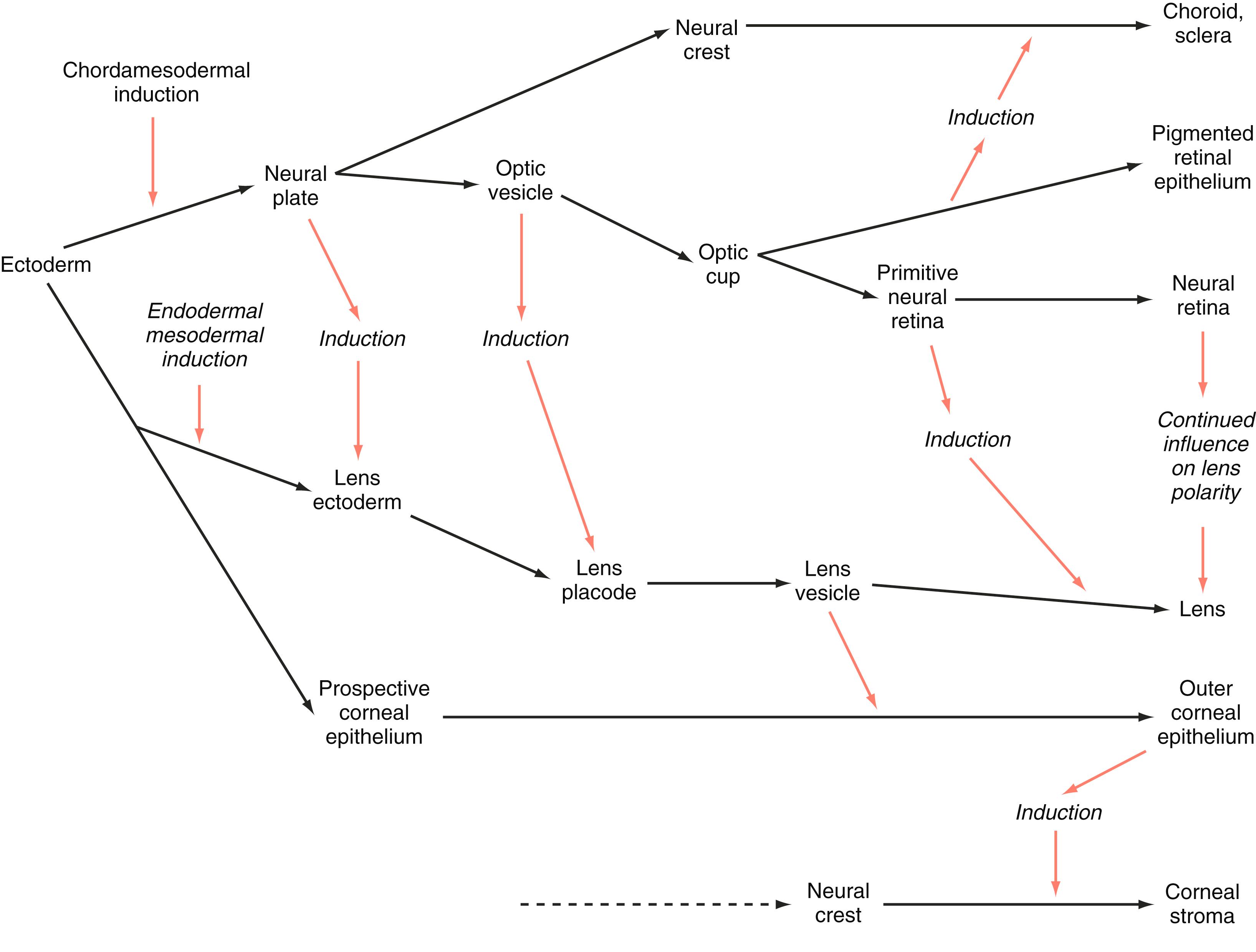
The major sense organs arise in large measure from the thickened ectodermal placodes that appear lateral to the neural plate in the early embryo (see Figure 6.10 ). The following descriptions begin with the most cranial placodes and continue to the most caudal. The midline adeno hypophyseal placode , located just rostral to the anterior neural ridge (see Figure 6.10D ), becomes the primordium of Rathke’s pouch (the precursor of the adenohypophysis). This structure arises adjacent to the neural tissue that ultimately forms the neurohypophysis. Arising also from the anterior neural region, bilateral olfactory placodes (see Figure 6.10D ) are the precursors of the olfactory epithelium. They give rise to olfactory neurons and their supporting cells and to glial cells and neuroendocrine cells that migrate from the placode into the brain. Closely associated with the olfactory placodes is the preneural tissue that forms the functionally associated olfactory bulbs of the brain. The bilateral lens placodes , associated with the optic vesicles (future retina) extending outward from the secondary prosencephalon, are the lens precursors.
Next in line are the paired trigeminal placodes (cranial nerve V), each of which arises from two placodal precursors—ophthalmic and maxillomandibular (see Figure 6.10D ). The otic placodes (precursors of the inner ear) in the human are the remaining representatives of the dorsolateral series of placodes, all of which produce vibration-detecting organs. In fish and some amphibians, the other members of the dorsolateral series give rise to the lateral line organs, which serve as vibration and electroreceptors in aquatic vertebrates.
The caudalmost group constitutes the epibranchial placodes , which are located just dorsal to the region where the first through third pharyngeal pouches abut the cervical ectoderm (see Figure 6.10D ). Their specification depends on signals (fibroblast growth factor [FGF] and bone morphogenetic protein [BMP]) emanating from the pharyngeal pouch endoderm. These placodes produce sensory neurons that supply visceral structures. The first epibranchial placode produces neurons (geniculate ganglion of cranial nerve VII) ( Figure 13.1B ) that innervate taste buds. Similarly, neurons arising from the second epibranchial placode (inferior [petrosal] ganglion of cranial nerve IX) innervate taste buds and the heart and other visceral organs. The third epibranchial placode contributes to the inferior (nodose) ganglion of the vagus nerve (cranial nerve X), and its neurons innervate the heart, stomach, and other viscera. Of cranial nerves V, VII, IX, and X, the proximal sensory ganglia are derived largely from neural crest cells, and the distal ganglia are principally placodal in origin (see Figure 13.1B ). The placode-derived neurons (those in the distal ganglia) begin to establish peripheral and central connections before axons emerge in the neural crest–derived neuronal precursors in the proximal ganglia.
In addition to odorant receptor cells, the olfactory placode also produces hypothalamic gonadotropic-releasing hormone (GnRH) neurons that undergo an unusual migration from the periphery into the central nervous system. Similarly, the otic placode gives rise to bipolar neurons that connect hair cells in the inner ear to their central targets.
A major function of both the epibranchial and trigeminal placodes is to produce neurons. The conversion of epithelial cells to neuroblasts in the placodes is accomplished in much the same manner as that which occurs within the neural tube (see Figure 11.4 ). Through the process called interkinetic nuclear migration , actual cell division occurs at the apical (in this case, outer) end of the tall epithelial placodal cells. Then the nuclei migrate toward the basal (inner) surface of the cells while they become committed to a neuronal fate as sensory neuroblasts . They then pass through breaches in the basal lamina and migrate internally. There they join other neuroblasts from the same origin to form precursors of the appropriate sensory ganglia.
All the placodes arise from a single preplacodal region that encircles the cranial neural plate. The preplacodal region is induced by cranial mesoderm, with the neural tube playing a supporting role. The inductive process involves activating the FGF pathway , along with inhibiting Wnt and BMP by their natural antagonists (see p. 97). Characteristic of the induced preplacodal tissue is the expression of the transcription factors, Six and Eya , which promote a generic placodal fate to the cells within the preplacodal region. This is followed by specific secondary inductive signals from different sources that specify the formation of individual placodes ( Table 13.1 ).
| Placode | Origin | Inducing Tissue | Inducers |
|---|---|---|---|
| Adenohypophysis | Anterior neural ridge | Anterior neural ridge | Shh |
| Nonneural ectoderm | Mesoderm | Nodal | |
| Lens | Anterior neural ridge | Neural plate | FGFR |
| Nonneural ectoderm | Mesoderm | BMP | |
| Olfactory | Anterior neural ridge | Anterior neural ridge | FGF |
| Nonneural ectoderm | Neural crest | ||
| Otic | Neural folds | Hindbrain | FGF |
| Nonneural ectoderm | Mesoderm | FGFR | |
| Retinoic acid | |||
| Trigeminal | Nonneural ectoderm | Dorsal neural tube | PDGF |
| Wnt | |||
| Epibranchial | Nonneural ectoderm | Hindbrain | FGF |
| Mesoderm | FGFR |
After their specification, the preplacodal cells migrate toward the areas where the placodes will form. During these migrations, the preplacodal cells interact with migrating neural crest cells, which may play a role in shaping the form and locations of the placodes. After their initial formation, the sensory placodes (olfactory, optic, and otic) and the adenohypophyseal placode develop further as invaginations from the surface ectoderm. They then break off from that ectoderm and form epithelially lined vesicles before they differentiate into their unique structures.
This chapter concentrates on the development of the eyes and ears, the most complex and important sense organs in humans. The organs of smell and taste are discussed in Chapter 14 because their development is intimately associated with the development of the face and pharynx. The sensory components of the cranial nerves are discussed in Chapter 11 .
The eye is a very complex structure that originates from constituents derived from several sources, including the wall of the secondary prosencephalon, the overlying surface ectoderm, and migrating cranial neural crest mesenchyme. Two basic themes characterize early ocular development. One is an ongoing series of inductive signals that result in the initial establishment of the major components of the eye. The other is the coordinated differentiation of many of these components.
For normal vision to occur, many complex structures within the eye must properly relate to neighboring structures. The cornea and lens must both become transparent and properly aligned to provide an appropriate pathway for light to reach the retina. The retina must be configured to receive concrete visual images and to transmit patterned visual signals to the proper parts of the brain through neural processes extending from the retina into the optic nerve.
A single continuous eye field begins to take shape around the area of the prechordal plate during late gastrulation. Cells in the eye field express RAX (retina and anterior neural fold homeobox). Mutations in RAX are the basis for anophthalmia , a rare condition characterized by the absence of any ocular structures in humans. Other prominent markers are Pax-6 and Lhx-2 , which are heavily involved in patterning the eye fields ( Figure 13.2 ). With the secretion of sonic hedgehog (shh) by the prechordal plate and the ventral midline of the diencephalon, Pax-6 expression in the midline is repressed, and the single eye field splits into two separate eye fields, located on either side of the secondary prosencephalon. Splitting of the eye field involves the migration of cells from the region of the future hypothalamus into the middle of the original eye field. Rax and another important transcription factor, Six-3 , protect the ability of the forebrain to secrete shh by suppressing Wnt activity. If Wnt is not suppressed, the anterior region of the developing brain becomes posteriorized and is unable to secrete shh. The absence of Six-3 activity results in the loss of shh secretion and prevents the splitting of the eye fields and leads to the condition of holoprosencephaly and the formation of only one eye (see p. 313 and Figure 8.21 ). The expression of Six-3, Pax-6, and Rax downregulates the expression of Otx-2, the transcription factor that characterizes the earliest forebrain. Downregulation of Otx-2 is required for the establishment of retinal identity.
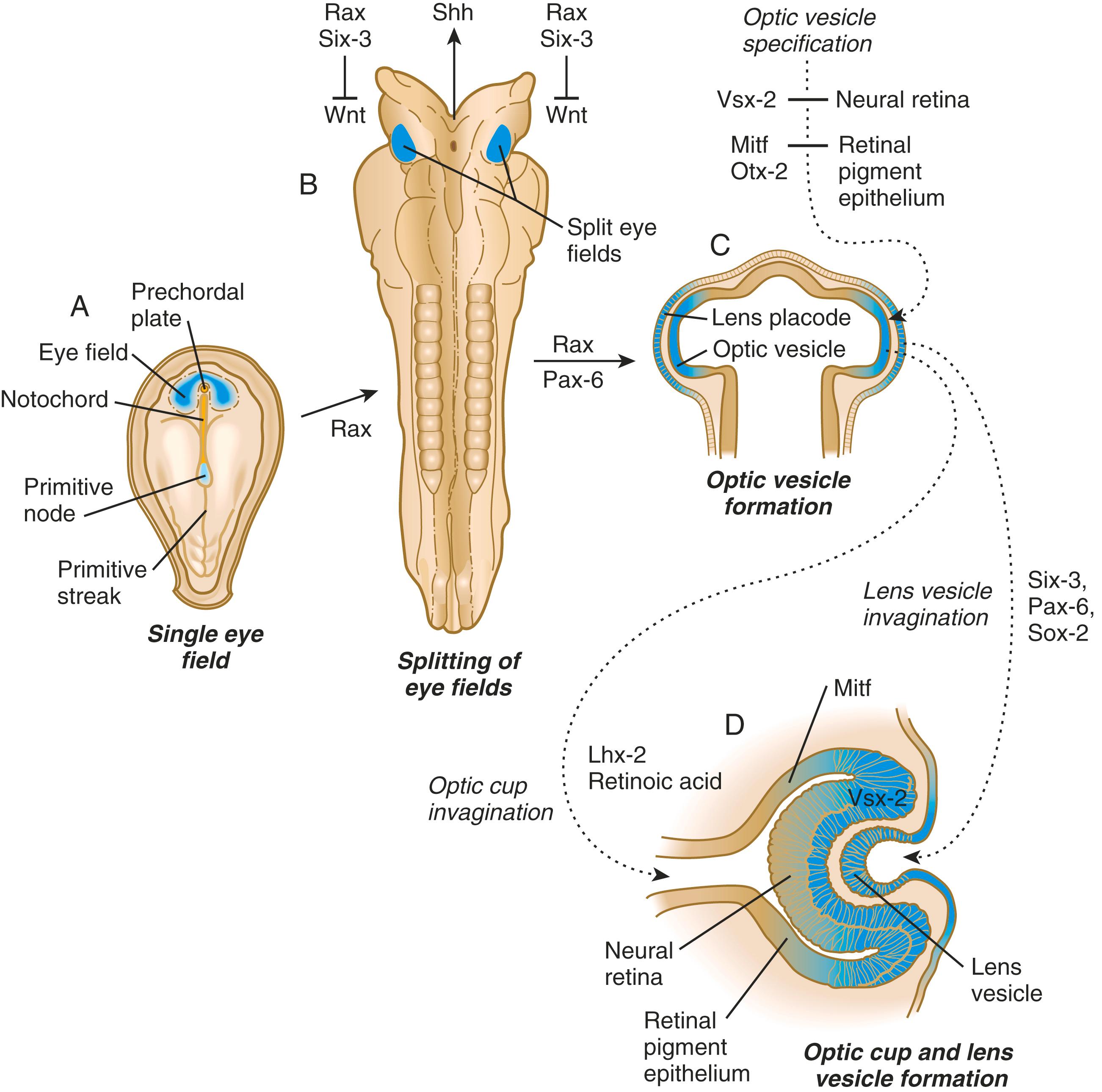
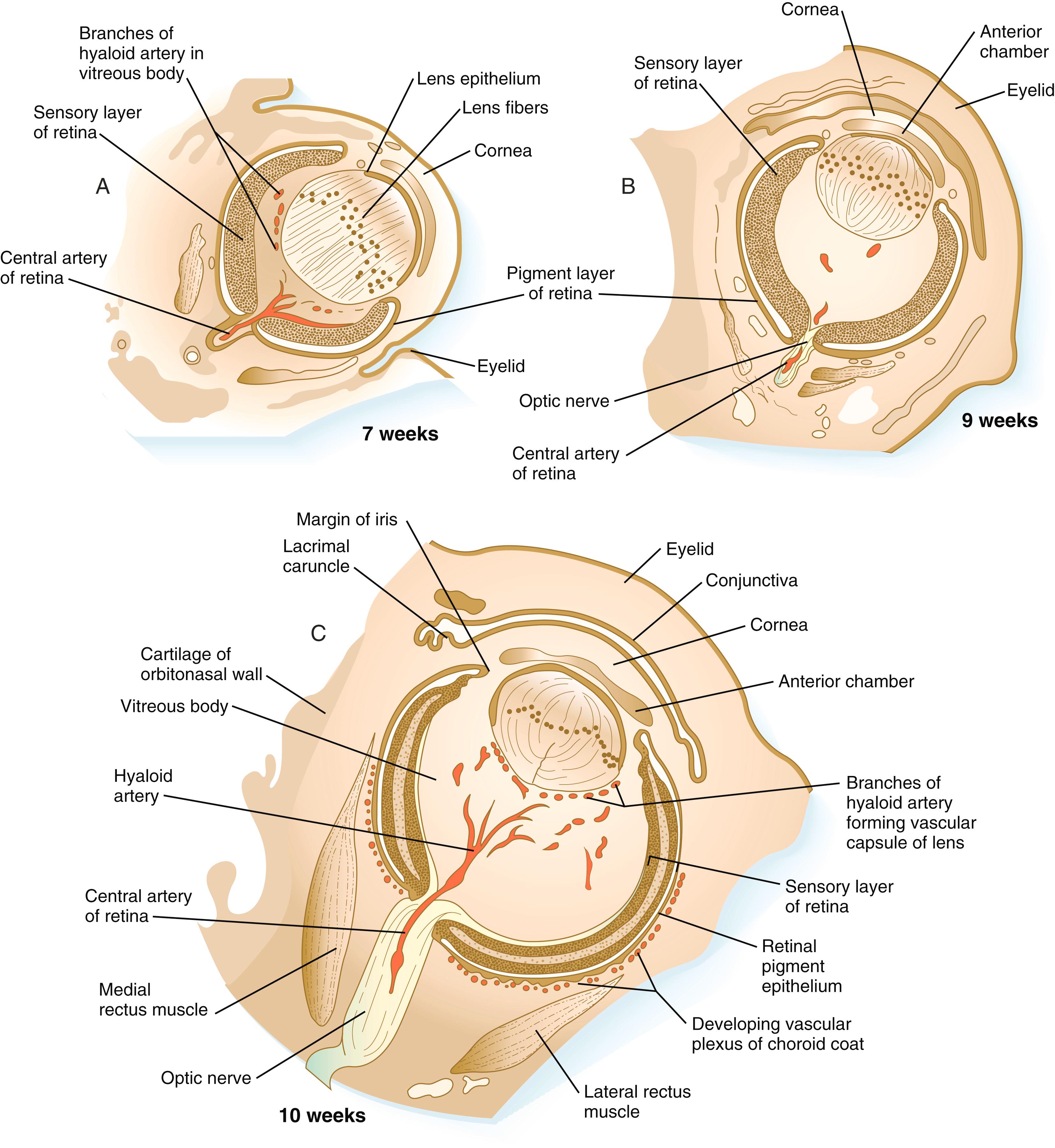
Development of the eye is first evident at approximately 22 days’ gestation, when the lateral walls of the forebrain begin to bulge out as optic grooves ( Figure 13.3 ). Within a few days, the optic grooves enlarge to form optic vesicles , which terminate very close to the overlying lens placode in the surface ectoderm. A local gradient of shh is involved in outgrowth of the optic vesicle. High concentrations of shh at the base induce formation of the optic stalk, whereas lower concentrations permit development of the optic cup. While the optic vesicle expands, the pattern for the future neural retina and retinal pigment epithelium is laid down. Initially, the transcription factor Mitf is expressed throughout the optic vesicle, but the subsequent expression of Vsx-2 in the distal optic vesicle (future neural retina) confines Mitf to more proximal regions, which will become the retinal pigment epithelium. Apposition of the outer wall of the optic vesicle to the surface ectoderm is essential for the transmission of an important inductive message that stimulates the cells of the lens placode to thicken and begin forming the lens (see Figures 13.2 and 13.4 ). This apposition displaces the periocular mesenchyme, which in earlier stages acts to inhibit lens induction.
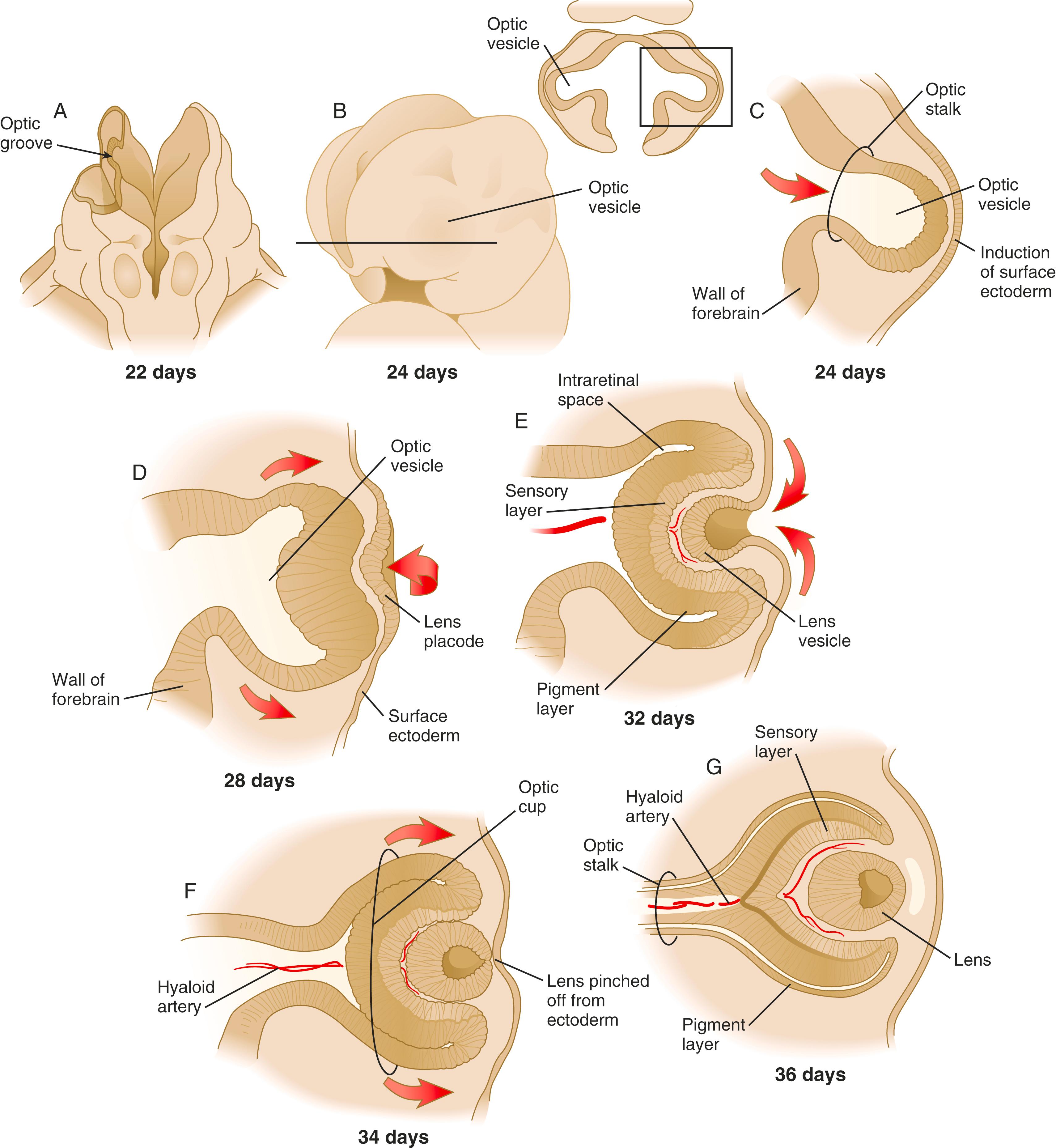
The interaction between the optic vesicle and the overlying ectoderm was one of the first recognized inductive processes. It was initially characterized by deletion and transplantation experiments conducted on amphibian embryos. When the optic vesicles were removed early, the surface ectoderm differentiated into ordinary ectodermal cells instead of lens fibers. Conversely, when optic vesicles were combined with certain types of ectoderm other than eye, the ectoderm was stimulated to form lens fibers. Subsequent research on amphibian embryos has shown that series of preparatory inductions from neural plate and underlying mesoderm condition the ectoderm for its final induction into lens by the optic vesicle. In mammals, an important mechanism underlying the severe microphthalmia (tiny eyes) or anophthalmia (absence of eyes) seen in the small eye and fidget mutants is a disturbance in the apposition of optic vesicles and surface ectoderm that interferes with lens induction.
The paired box gene Pax6 plays a prominent role throughout early eye development and at certain later stages of development of the retina and lens. Pax-6 and Dlx-5 are initially expressed in the common lens and nasal placodes and much of the ventral secondary prosencephalon. In Drosophila, Pax6 has been called a master gene for eye development; that is, it can turn on the cascade of genes that guide development of the eye. The power of Pax-6 is shown by the formation of ectopic eyes on antennae and legs in Drosophila when the gene is improperly expressed. In the absence of Pax-6 expression (eyeless mutant), eyes do not form. In the small eye mutation, the mammalian equivalent of eyeless , the early optic vesicle forms, but, as previously noted, eye development does not progress because the surface ectoderm is unable to respond to the inductive signal emitted by the optic vesicle.
The identification in humans of two genes ( Eya [eyes absent] and Six [sine oculis]) that are activated by Pax-6 in Drosophila strongly suggests that despite major differences in the structure and development of the vertebrate and insect eye, the basic genetic apparatus has been conserved throughout phylogeny. In mice, Eya-1 and Eya-2 are expressed in the lens placodes and are required for placodal induction and early differentiation, but in the absence of Pax-6 function, they are not expressed, and eye development fails to proceed.
While the process of lens induction occurs, the surface ectoderm stimulates the outer face of the optic vesicle to flatten and ultimately to become concave. This results in the transformation of the optic vesicle to the optic cup (see Figure 13.3F ). The progression from optic vesicle to optic cup requires the expression of Lhx-2 and the action of retinoic acid. In their absence, eye development is arrested at the optic vesicle stage (see Figure 13.2 ). Meanwhile, the induced lens ectoderm thickens and invaginates to form a lens vesicle , which detaches from the surface epithelium from which it originated ( Figure 13.5 ; see Figure 13.3 ). Then the lens vesicle takes over and becomes the primary agent of a new inductive reaction by acting on the overlying surface ectoderm and causing it to begin corneal development (see Figure 13.4 ).
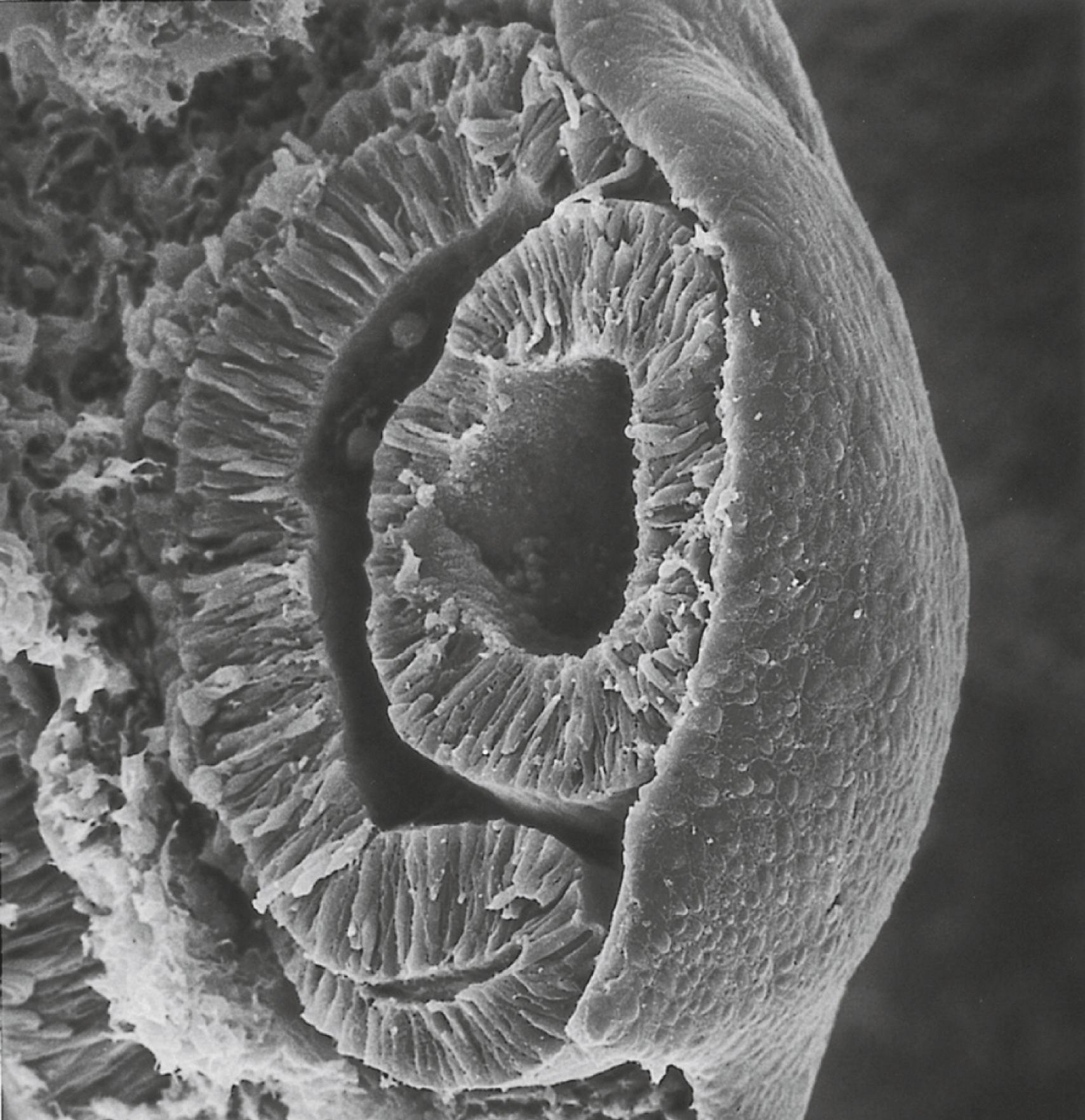
Formation of the optic cup is an asymmetric process that occurs at the ventral margin of the optic vesicle, rather than at its center. This results in the formation of a gap called the choroid fissure, which is continuous with a groove in the optic stalk ( Figure 13.6 ). During much of early ocular development, the choroid fissure and optic groove form a channel through which the hyaloid artery passes into the posterior chamber of the eye. Differential expression of Pax genes determines which cells become optic cup (future retina), and which cells become optic stalk (future optic nerve). Through exposure to high concentrations of shh, the expression of Pax-6 is inhibited and Pax-2 is induced in the optic stalk, whereas a lower concentration of shh more distally permits the expression of Pax-6 in the optic vesicle, thus paving the way toward formation of the retina.
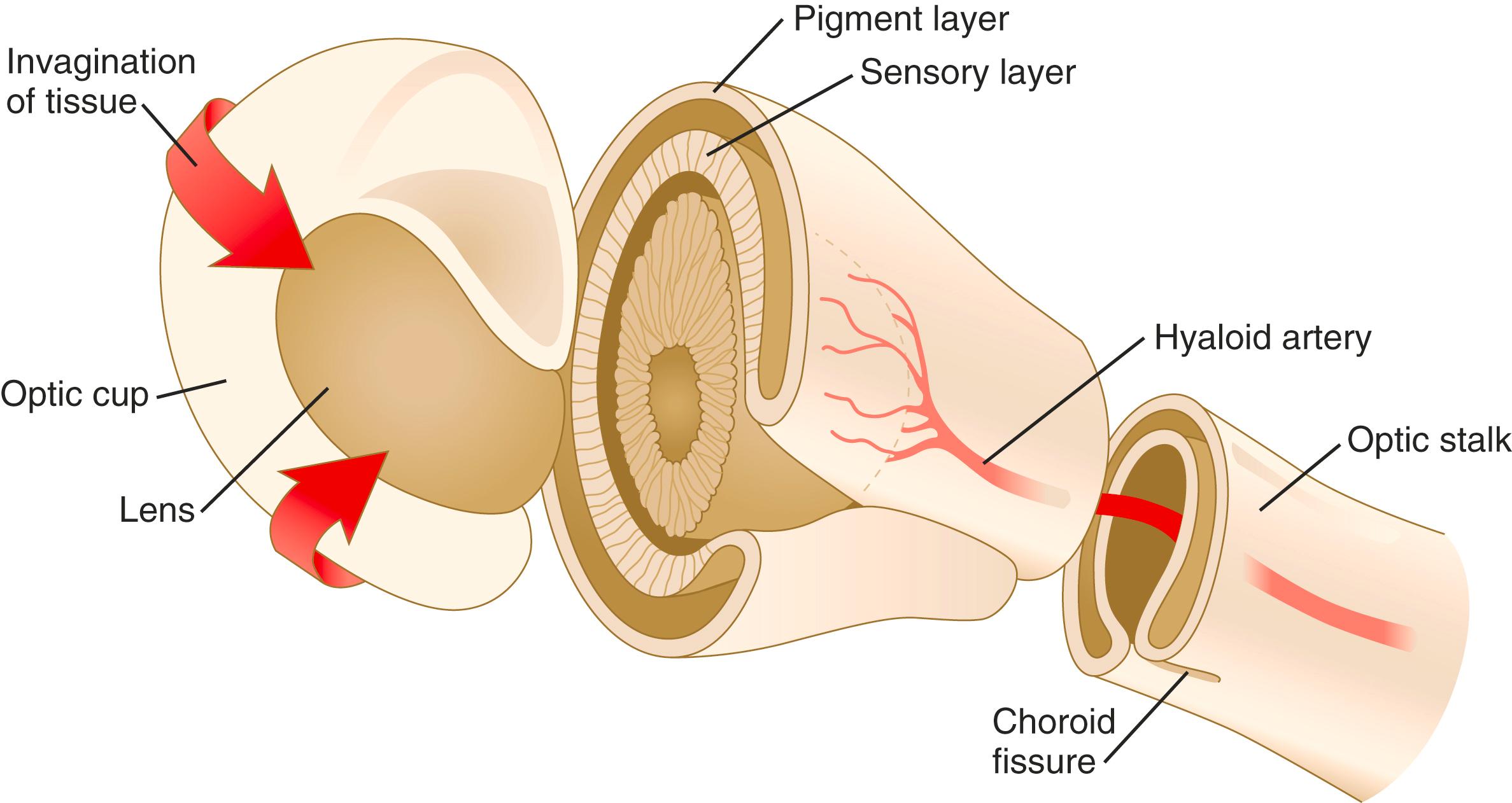
The optic stalk initially represents a narrow neck that connects the optic cup to the diencephalon, but as development progresses, it is invaded by neuronal processes emanating from the ganglion cells of the retina. Pax-2–expressing cells in the optic stalk provide guidance cues to outgrowing retinal axons that pass through the optic nerve and optic chiasm and enter the contralateral optic tract. After the neuronal processes have made their way to the appropriate regions of the brain, the optic stalk is properly known as the optic nerve .
Later in development, the choroid fissure closes, and no trace of it is seen in the normal iris. An unusual feature of closure of the choroid fissure is that the two opposing ridges of tissue consist of the basal surfaces of the epithelial cells. These surfaces are covered by basal lamina material, which must be removed before proper fusion of the ridges of the choroid fissure. Nonclosure of the choroid fissure results in the anomaly of coloboma (see Figure 13.20B ). In certain forms of coloboma, especially forms that are associated with anomalies of the kidneys, mutations of Pax-2 genes are seen. In Pax-2–mutant mice, retinal axons do not cross the midline through the optic chiasm, but rather remain in the ipsilateral optic tract.
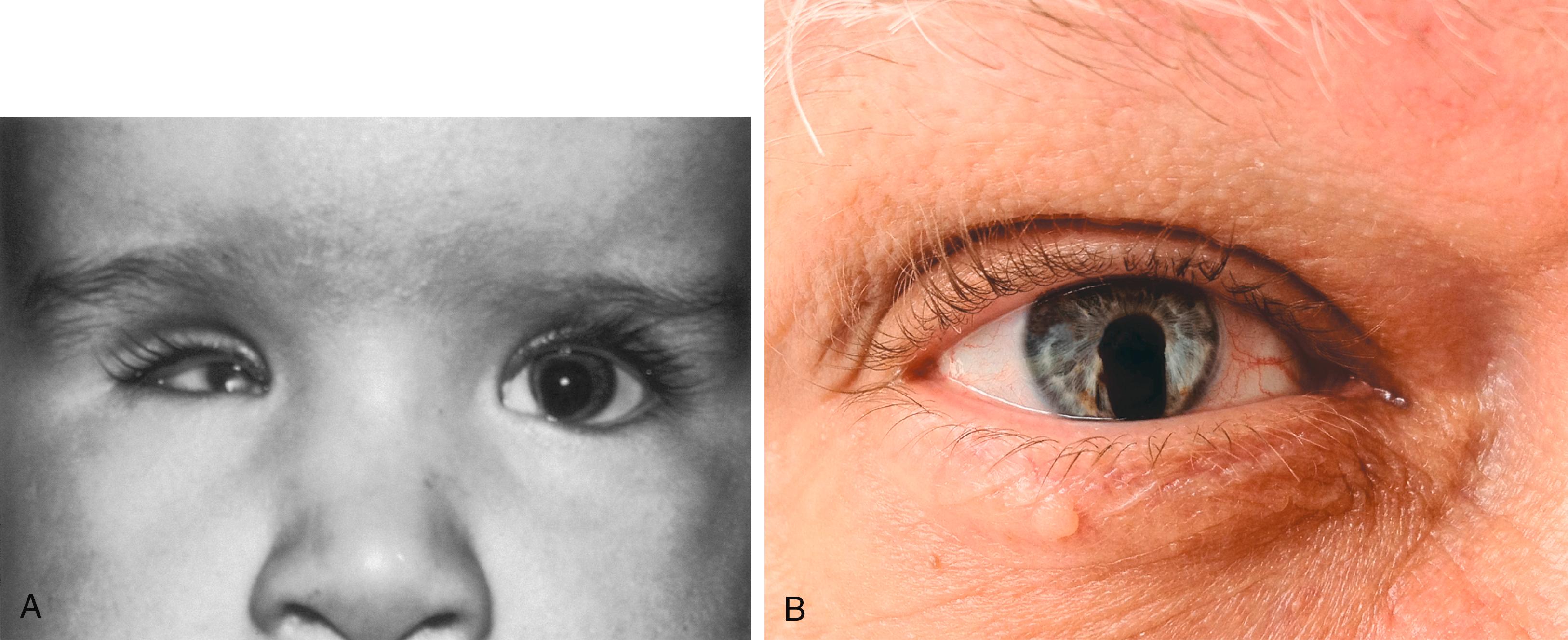
The development of the lens involves four phases of activity: (1) specification of future lens-forming cells from preplacodal ectoderm; (2) invagination of elongated epithelial cells to form a lens pit, followed by a lens vesicle; (3) elongation and differentiation of lens fibers; and (4) functional fine-tuning of lens fiber cells.
The lens is derived from cells in the generic preplacodal region, as discussed earlier. Within the preplacodal region is a bipotential stem cell that can give rise to cells of either the olfactory or lens placode. There is evidence that the earliest prelens cells form in a salt-and-pepper manner and ultimately sort themselves out to form a cellularly homogeneous lens placode. The bipotential stem cell expresses both Pax-6 and Dlx-2. With the separation of the definitive lens and nasal placodes, cells of the former express Pax-6 and the latter, Dlx-5.
From the earliest stages, formation of the lens depends on genetic instructions provided by Pax-6. Pax-6 expression is required for the surface ectoderm to respond to inductive signals (FGF and BMP) from the underlying optic vesicle by activating and combining with another transcription factor, Sox-2. This leads to the thickening of the surface ectoderm to form the lens placode (see Figure 13.3D ). At the same time, migrating neural crest cells, which do not penetrate the space between optic vesicle and prospective lens, give off signals that inhibit cells in other areas of the preplacodal region from forming lens. Inhibition of lens-forming capacity is signaled by the downregulation of Pax-6 by these cells. Pax-6 and Six-3 expression continues as the lens placode invaginates to form the lens vesicle , which ultimately breaks off from the surface ectoderm. At this point, Pax-6 plays a new role in regulating the activity of the genes governing the formation of the lens crystallin proteins.
When it is breaking off from the surface ectoderm, the lens vesicle is roughly spherical and has a large central cavity (see Figure 13.3E ). At the end of the sixth week, the cells at the inner pole of the lens vesicle begin to elongate in an early step toward their transformation into the long, transparent cells called lens fibers ( Figure 13.7A ). The influence of the transcriptional regulator Foxe-3 , which operates downstream of Pax-6, facilitates the breaking off of the lens vesicle from the surface ectoderm and the transformation of posterior cells into lens fibers.
Differentiation of the lens is a very precise and well-orchestrated process, initiated by BMP, FGF, and Wnt signaling and involving several levels of organization. At the cellular level, relatively nonspecialized lens epithelial cells, under the influence of Sox-2, Pax-6, and other proteins paired with an oncogene called Maf , undergo a profound transformation into transparent, elongated cells that contain large quantities of specialized crystallin proteins . At the tissue level, the entire lens is responsive to signals from the retina and other structures of the eye so that its shape and overall organization are best adapted for the transmission of undistorted light rays from the corneal entrance to the light-receiving cells of the retina.
At the cellular level, cytodifferentiation of the lens consists of the transformation of mitotically active lens epithelial cells into elongated postmitotic lens fiber cells. Later development of the elongated lens fibers involves their polarization, with apical ends facing toward the future cornea, and their regular organization into an onionskin-like arrangement around a central lens nucleus (see below). The apical end of each lens fiber contains a single primary cilium—a major component of the planar cell polarity function that helps organize the developing lens. As much as 90% of the soluble protein in the postmitotic lens fibers consists of crystallin proteins. The mammalian lens contains three major crystallin proteins: α, β, and γ.
The formation of crystallin-containing lens fibers begins with the elongation of epithelial cells from the inner pole of the lens vesicle (see Figure 13.3 ). These cells become the fibers of the lens nucleus ( Figure 13.8 ). The remaining lens fibers arise from the transformation of the cuboidal cells of the anterior lens epithelium. During embryonic life, mitotic activity is spread throughout the outer lens epithelial cells. Around the time of birth, mitotic activity ceases in the central region of this epithelium, leaving a germinative ring of mitotically active cells surrounding the central region. Daughter cells from the germinative region move into the equatorial region of cellular elongation, where they cease to divide and take on the cytological characteristics of RNA-producing cells and begin to form crystallin mRNAs. These cells soon elongate tremendously, fill up with crystallins, and transform into secondary lens fibers that form concentric layers around the primary fibers of the lens nucleus. The midline region where secondary lens fibers from opposite points on the equator join is recognized as the anterior and posterior lens suture (see Figure 13.7D ). With this arrangement, the lens fibers toward the periphery are successively younger. As long as the lens grows, new secondary fibers move in from the equator onto the outer cortex of the lens.
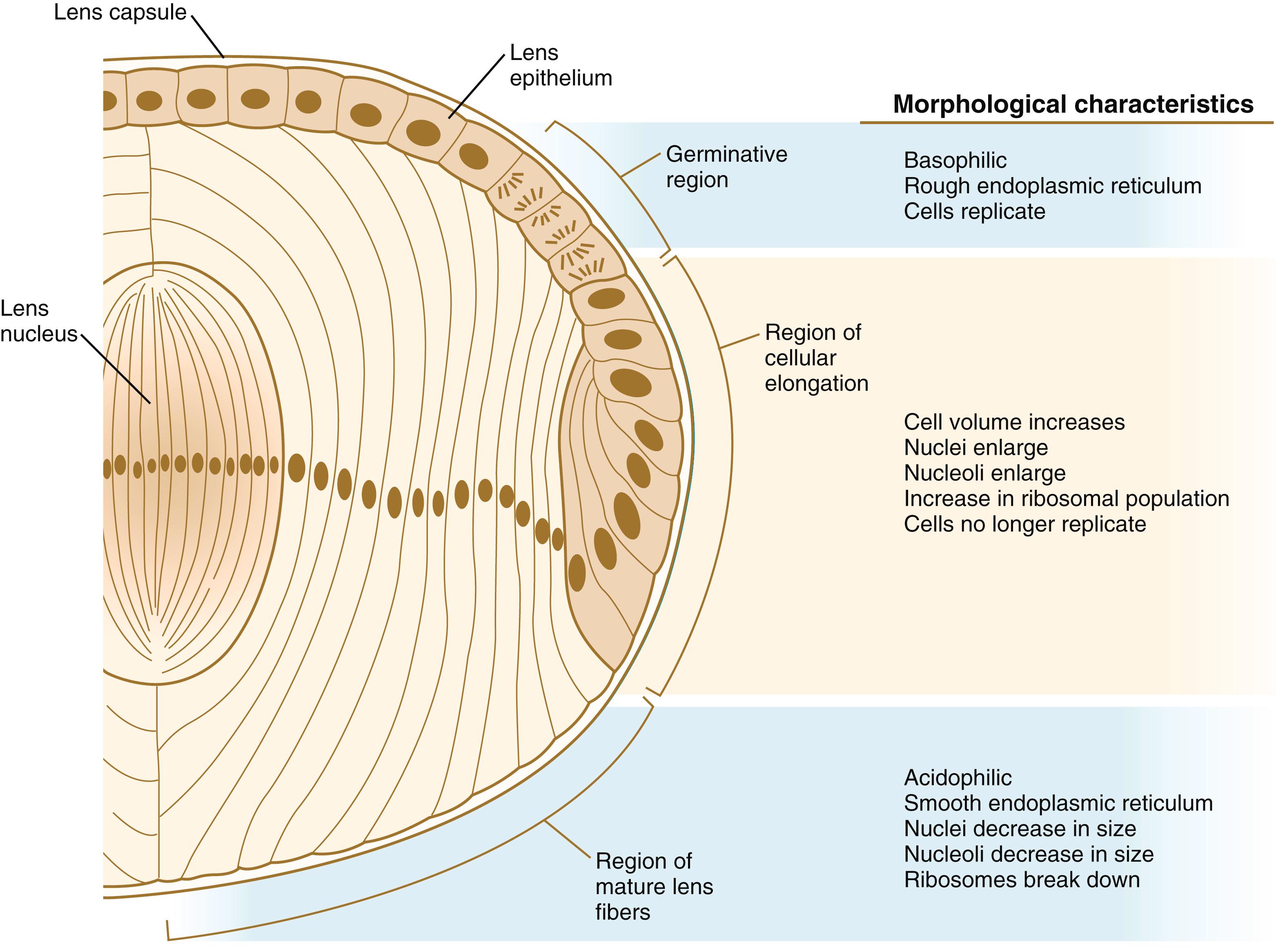
The crystallin proteins show a very characteristic pattern and sequence of appearance, with the α-crystallins appearing first in the morphologically undifferentiated epithelial cells. Synthesis of β-crystallins is seen when the lens fibers begin to elongate, whereas the expression of γ-crystallins is restricted to terminally differentiated lens fiber cells. Each of the crystallin protein families contains several members. They show different patterns of activation (some members of a family are coordinately activated) and different patterns of accumulation. These patterns facilitate the optical clearing of the lens to allow the efficient transmission of light. Another major process that adds to the transparency of lens fiber is the clearing of most organelles through autophagic mechanisms.
Throughout much of its life, the lens is under the influence of the retina. After induction of the lens, secretions of the retina, of which FGF is a major component, accumulate in the vitreous humor behind the lens and stimulate the formation of lens fibers. A striking example of the continued influence of the retina on lens morphology is seen after a developing lens is rotated so that its outer pole faces the retina. Very rapidly, under the influence of retinal secretions, the low epithelial cells of the former outer pole begin to elongate and form an additional set of lens fibers ( Figure 13.9 ). A new lens epithelium forms on the corneal side of the rotated lens. Such structural adaptations are striking evidence of a mechanism that ensures correct alignment between the lens and the rest of the visual system throughout development.
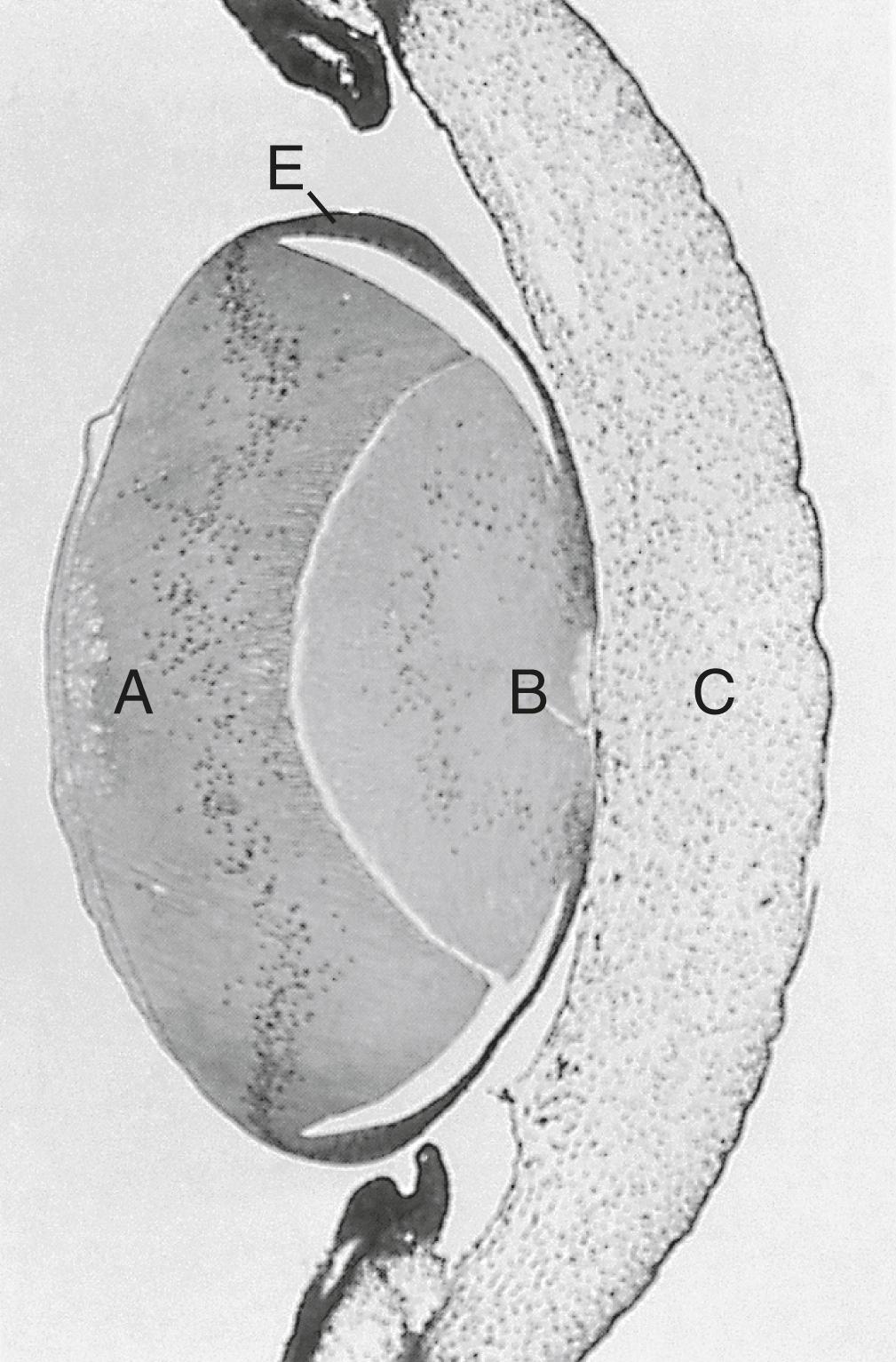
Formation of the cornea is the result of the last of the series of major inductive events in eye formation (see Figure 13.4 ), with the lens vesicle acting on the overlying surface ectoderm. This induction results in the transformation of a typical surface ectoderm, consisting of a basal layer of cuboidal cells and a superficial periderm, to a transparent, multilayered structure with a complex extracellular matrix and cellular contributions from several sources. In keeping with its multifaceted role at almost all stages of eye development, Pax-6 expression in the surface ectoderm is a requirement for corneal induction. While the early cornea begins to take shape, DKK-2 suppression of Wnt signaling prevents the ectoderm of the cornea from becoming hair-producing skin.
The inductive influence of the lens stimulates a change in the basal ectodermal cells. These cells increase in height, largely as a result of the elaboration of secretory organelles (e.g., Golgi apparatus) on the basal ends of the cells. While these changes are completed, the cells begin to secrete epithelially derived collagen types I, II, and IX to form the primary stroma of the cornea ( Figure 13.10 ).
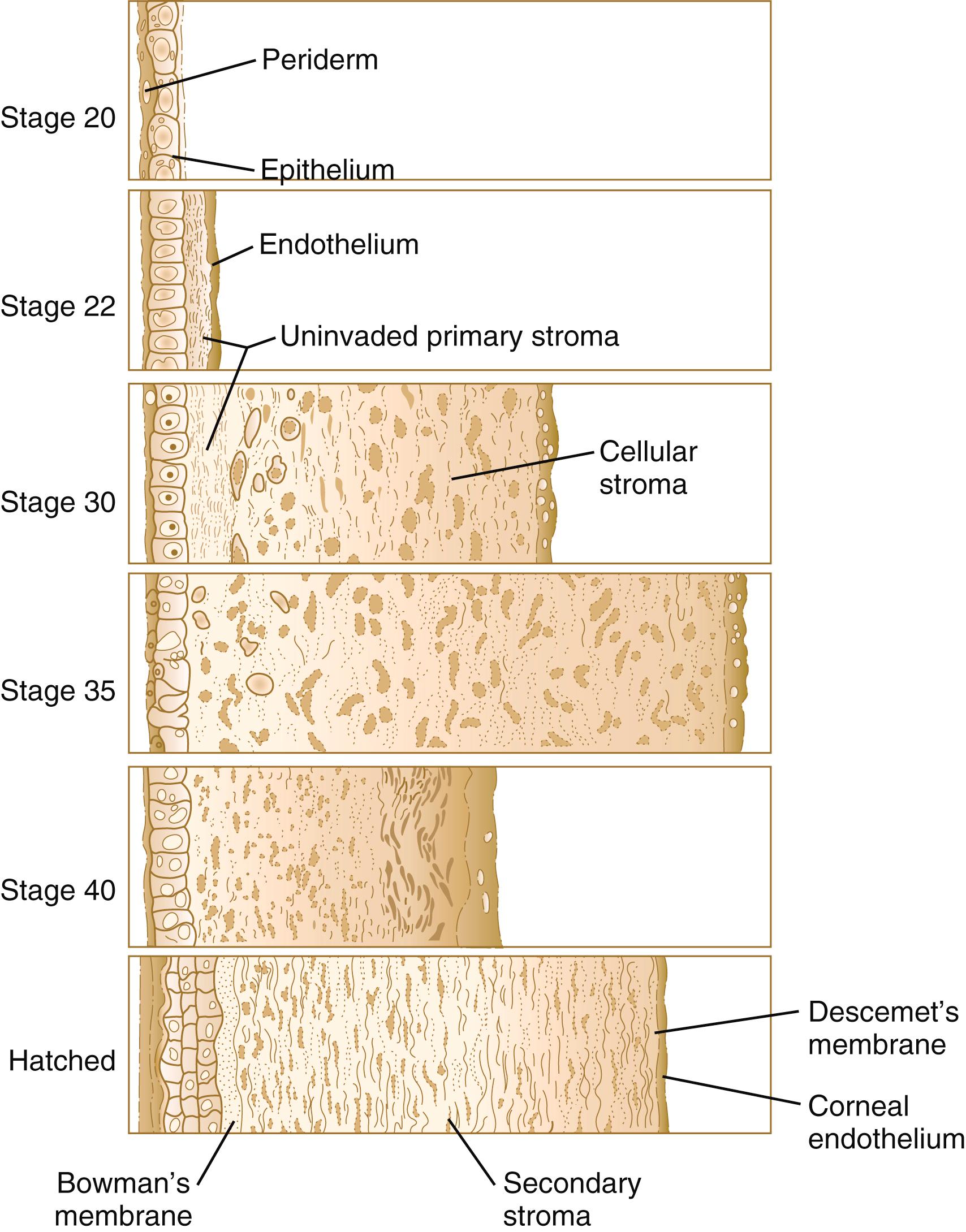
Using the primary stroma as a basis for migration, neural crest cells around the lip of the optic cup migrate centrally between the primary stroma and the lens capsule as an initial wave of neural crest migration. Although mesenchymal in morphology during their migration, these cells become transformed into a cuboidal epithelium called the corneal endothelium when their migration is completed. At this point, the early cornea consists of (1) an outer epithelium, (2) a still acellular primary stroma, and (3) an inner endothelium.
The migration of neural crest cells between the lens and overlying ectoderm is subject to tight developmental control. A positive stimulus for migration is the production of transforming growth factor-β (TGF-β) by the lens. Modulating this influence is the presence of semaphorin 3A on the lens. The periocular neural crest cells express neuropilin-1 , which, when combined with semaphorin, inhibits migration. At a critical time in development, a subpopulation of these neural crest cells ceases to express neuropilin. These neural crest cells then migrate into the space between the corneal epithelium and endothelium as precursors of the primary corneal stroma. A similar mechanism allows the penetration of sensory nerve fibers into the cornea.
After the corneal endothelium has formed a continuous layer, its cells synthesize large amounts of hyaluronic acid and secrete it into the primary stroma. Because of its pronounced water-binding capacities, hyaluronic acid causes the primary stroma to swell greatly. This provides a proper substrate for the second wave of cellular migration into the developing cornea ( Figure 13.11 ). These cells, also of neural crest origin, are fibroblastic. They migrate and proliferate in the hyaluronate-rich spaces between layers of collagen in the primary corneal stroma. The migratory phase of cellular seeding of the primary corneal stroma ceases when these cells begin to produce large amounts of hyaluronidase , which breaks down much of the hyaluronic acid in the primary stroma. In other parts of the embryo (e.g., limb bud), there is also a close correlation between high amounts of hyaluronic acid and cellular migration and a cessation of migration with its removal. With the removal of hyaluronic acid, the cornea decreases in thickness. When the migratory fibroblasts have settled, the primary corneal stroma is considered to have been transformed into the secondary stroma .
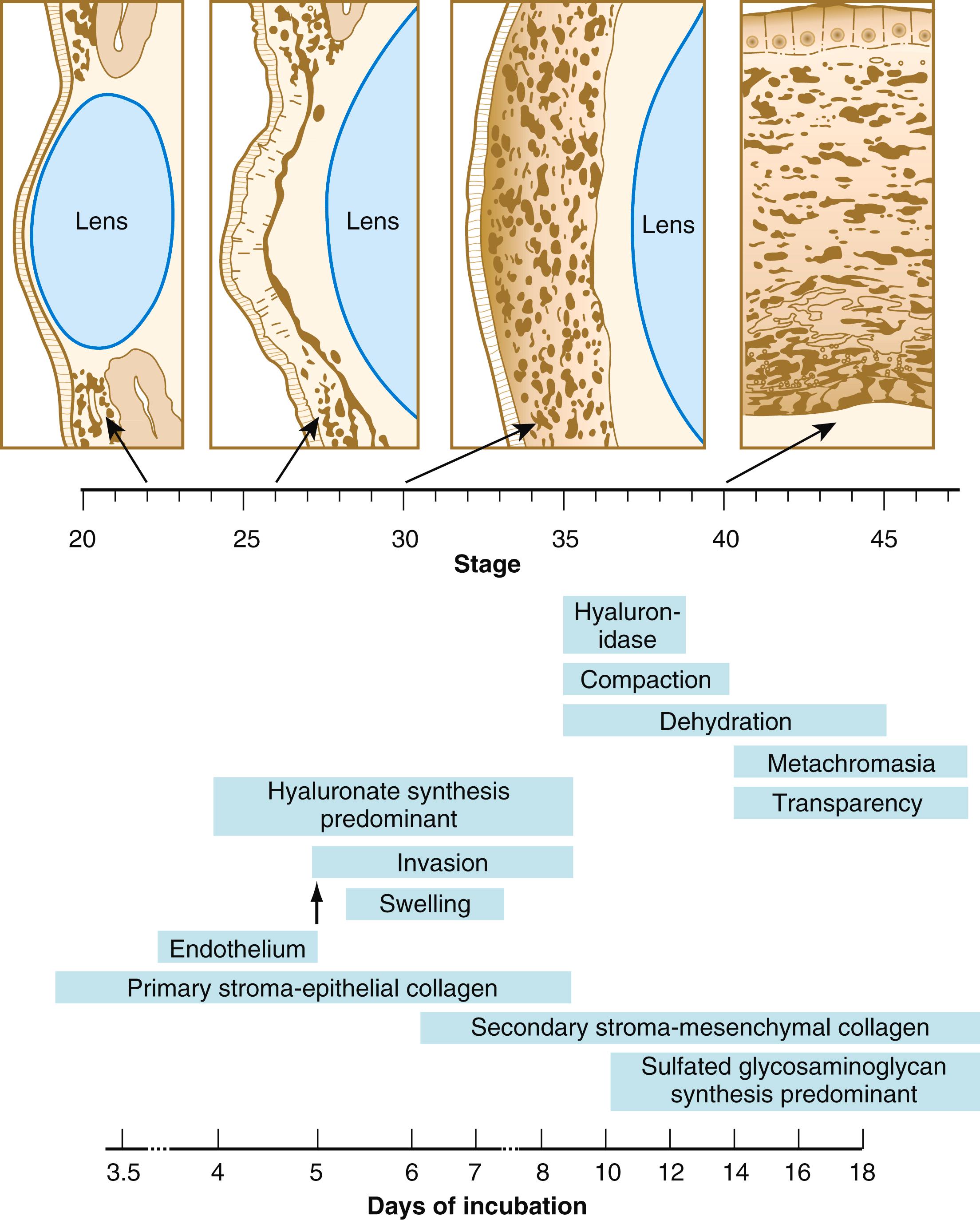
The fibroblasts of the secondary stroma contribute to its organization by secreting coarse collagen fibers to the stromal matrix. The corneal collagen fibers are laid down in a highly regular orthogonal manner ( Figure 13.12 ). Nevertheless, prominent layers of acellular matrix continue to be secreted by epithelial and endothelial cells of the cornea. These secretions provide the remaining layers that constitute the mature cornea. Listed from outside in, they are (1) the outer epithelium, (2) Bowman’s membrane , (3) the secondary stroma, (4) Descemet’s membrane , and (5) the corneal endothelium (see Figure 13.10 ).
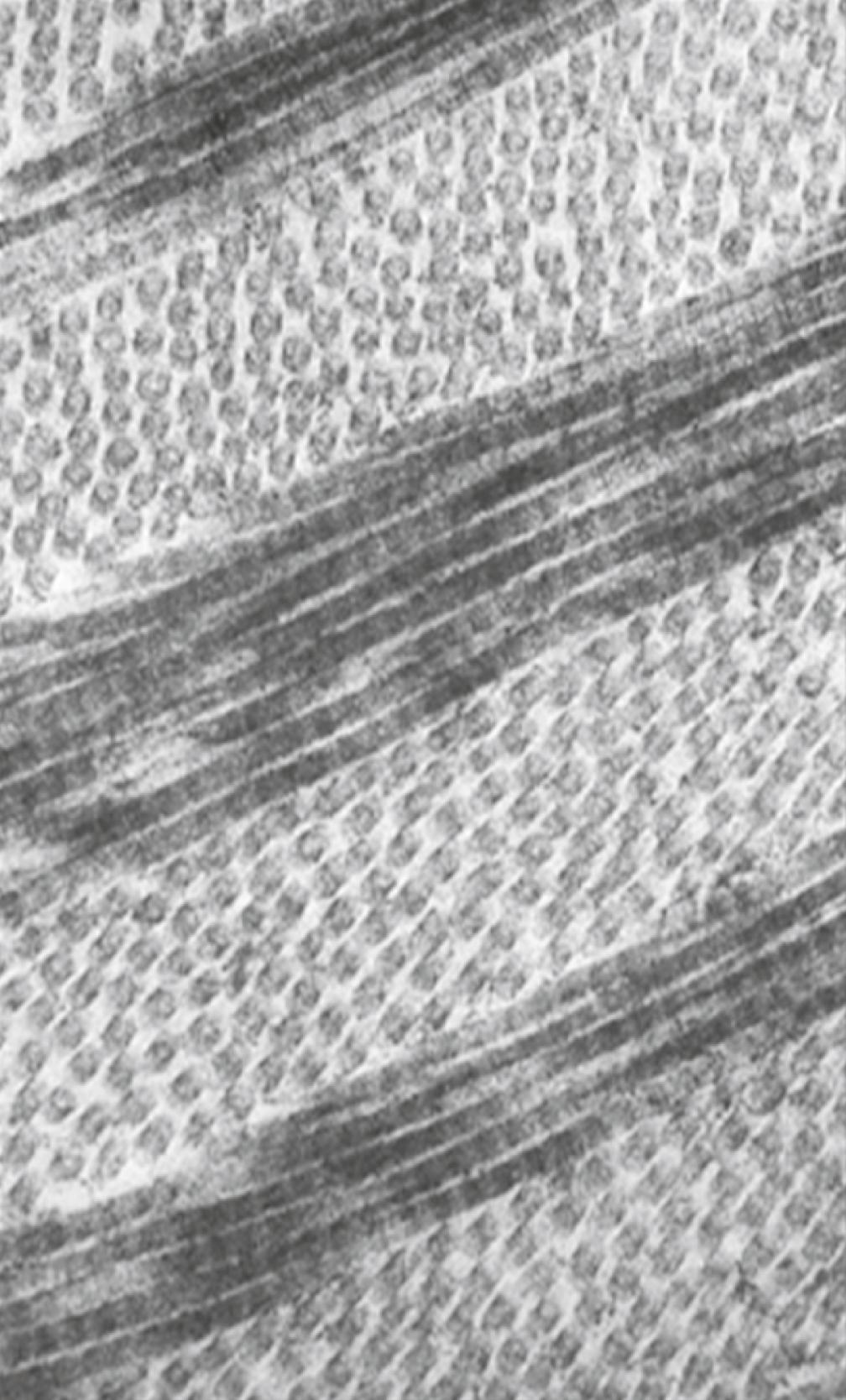
The final developmental changes in the cornea involve the formation of a transparent pathway free from optical distortion, through which light can enter the eye. A major change is a great increase in transparency, from approximately 40% to 100% transmission of light. This increase is accomplished by removing much of the water from the secondary stroma. The initial removal of water occurs with the degradation of much of the water-binding hyaluronic acid. The second phase of dehydration is mediated by thyroxine , which is secreted into the blood by the maturing thyroid gland. Thyroxine acts on the corneal endothelium by causing it to pump sodium from the secondary stroma into the anterior chamber of the eye. Water molecules follow the sodium ions, thus effectively completing the dehydration of the corneal stroma. The role of the thyroid gland in this process was shown in two ways. When relatively mature thyroid glands were transplanted onto the extraembryonic membranes of young chick embryos, thereby allowing thyroid hormone to gain access to the embryonic circulation via the blood vessels supplying the membrane ( chorioallantoic membrane ), premature dehydration of the cornea occurred. Conversely, the application of thyroid inhibitors retarded the clearing of the cornea.
The other late event in the cornea is a pronounced change in its radius of curvature in relation to that of the eyeball as a whole. This morphogenetic change, which involves several mechanical events, including intraocular fluid pressure, allows the cornea to work with the lens in bringing light rays into focus on the retina. If irregularities develop in the curvature of the cornea during its final morphogenesis, astigmatism develops in the individual, which causes distortions in the visual image.
While the lens and cornea are taking shape, profound changes are also occurring in the optic cup (see Figure 13.3 ). The inner layer of the optic cup thickens, and the epithelial cells begin a long process of differentiation into neurons and light receptor cells of the neural retina . The outer layer of the optic cup remains thin and ultimately becomes transformed into the retinal pigment epithelium (see Figure 13.7 ). Cells of the retinal pigment epithelium do not differentiate into neurons during normal embryogenesis, but in postnatal life some of these cells maintain stem cell properties and can differentiate into multiple cell types. At the same time, the outer lips of the optic cup undergo a quite different transformation into the iris and ciliary body, which are involved in controlling the amount of light that enters the eye (iris) and the curvature of the lens (ciliary body).
Formation of the optic cup and the distinction between neural retina and retinal pigment epithelium depend on molecular events occurring early in eye formation. Stimulated by FGFs from the overlying surface ectoderm, a cooperative interaction between Pax-2 and Pax-6 subdivides the optic vesicle into distal (closest to the surface ectoderm) and proximal (closest to the optic stalk) regions. Under the influence of Pax-6, the distal part of the optic vesicle invaginates to form the inner wall of the optic cup and begins to express the paired-like homeodomain transcription factor Vsx-2 , which characterizes future neural retina. The helix-loop-helix transcription factor Mitf (microphthalmia-associated transcription factor) is initially expressed throughout the entire optic vesicle, but through the inductive effect of BMP from the surrounding neural crest mesenchyme and the action of Pax-2 and Pax-6, its expression becomes restricted to the proximal part of the optic vesicle, which is the future retinal pigment epithelium.
The neural retina is a multilayered structure; its embryonic development can be appreciated only after its adult organization is understood ( Figure 13.13 ). When seen in cross section under a microscope, the neural retina consists of alternating light-staining and dense-staining strips that correspond to layers rich in nuclei or cell processes. The direct sensory pathway in the neural retina is a chain of three neurons that traverse the thickness of the retina. The first element of the chain is the light receptor cell, either a rod or a cone . A light ray that enters the eye passes through the entire thickness of the neural retina until it impinges on the outer segment of a rod or cone cell (photoreceptor) in the extreme outer layer of the retina. The nucleus of the stimulated rod and cone cell is located in the outer nuclear layer . The photoreceptor cell sends a process toward the outer plexiform layer , where it synapses with a process from a bipolar cell located in the inner nuclear layer . The other process from the bipolar neuron leads into the internal plexiform layer and synapses with the third neuron in the chain, the ganglion cell . The bodies of the ganglion cells, which are located in the ganglion cell layer , send out long processes that course through the innermost nerve fiber layer toward their exit site from the eye, the optic nerve, through which they reach the brain.
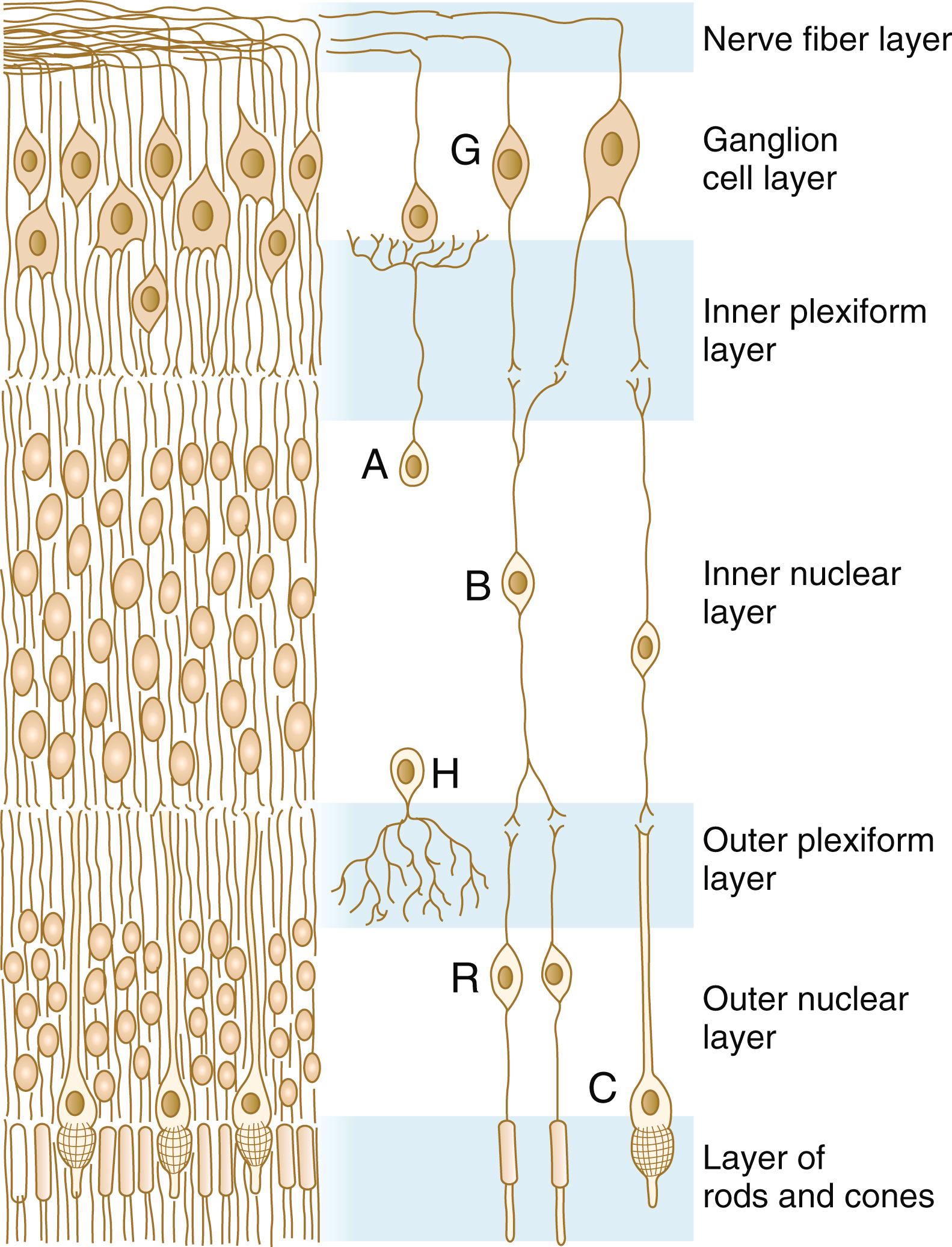
If all light signals were processed only through the simple three-link series of neurons in the retina, visual acuity would be much less than it actually is. Many levels of integration have occurred by the time a visual pattern is stored in the visual cortex of the brain. The first is in the neural retina. At synaptic sites in the inner and the outer plexiform layers of the retina, other cells, such as horizontal and amacrine cells (see Figure 13.13 ), are involved in the horizontal redistribution of the simple visual signal. This redistribution facilitates the integration of components of a visual pattern. Another prominent cell type in the retina is the Müller glial cell , which sends processes to almost all layers of the retina and seems to play a role similar to that of astrocytes in the central nervous system.
From the original columnar epithelium of the inner sensory layer of the optic cup (see Figure 13.7 ), the primordium of the neural retina takes on the form of a mitotically active, thickened pseudostratified columnar epithelium organized in a manner similar to that of the early neural tube. During the early stages of development of the retina, its polarity becomes fixed according to the same axial sequence as that seen in the limbs (see Chapter 10 ). The nasotemporal (anteroposterior) axis is fixed first; this is followed by fixation of the dorsoventral axis. Finally, radial polarity is established.
To process distinct visual signals, the retina must develop according to a well-defined pattern, and that pattern must be carried on to the brain so that distinct visual images are produced. Despite its cuplike shape, patterning in the retina is often described in a two-dimensional manner, with dorsoventral and nasotemporal gradients serving as the basis for retinal pattern formation and differentiation.
Dorsoventral patterning is initiated through the presence of BMP-4 dorsally and shh ventrally ( Figure 13.14A ). The presence of shh ventrally stimulates the production of Otx-2 in the outer layer of the optic vesicle and differentiation of the retinal pigment epithelium. Within the inner layer of the optic vesicle, shh and a protein, ventroptin , both antagonists of BMP-4, stimulate the expression of the transcription factors Vax-2 and Pax-2 in the ventral retina. In the dorsal part of the future retina, BMP-4 signals the expression of Tbx-5 , the transcription factor involved in the formation of the forelimb (see p. 195). Although many molecules are unequally distributed along the two axes of the retina, opposing gradients of specific ephrins and their receptors are heavily involved in the characterization of the retinal axes (see Figure 13.14A ).
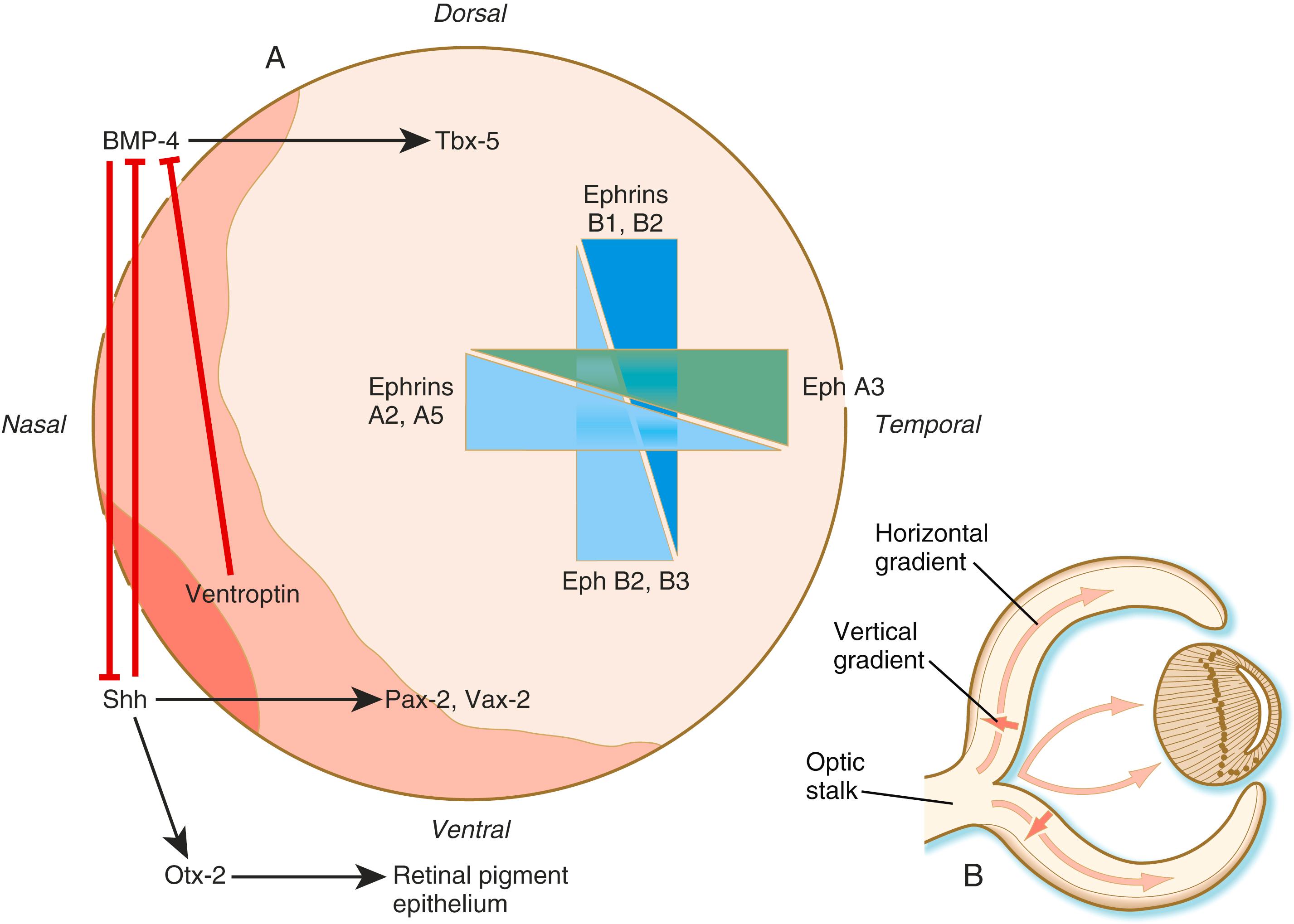
As the number of cells in the early retina increases, the differentiation of cell types begins. The switch to neurogenesis within the retina requires FGF-3 and -8 signaling from the ventral midline. There are two major gradients of differentiation in the retina. The first proceeds roughly vertically from the inner to the outer layers of the retina. The second moves horizontally from the center toward the periphery of the retina ( Figure 13.14B ).
Evidence obtained on fish retinas suggests that the horizontal gradient of neurogenesis may be driven by an initial point source of shh in a manner similar to a better-understood process that occurs in Drosophila . Differentiation in the horizontal gradient begins with the appearance of ganglion cells and the early definition of the ganglion cell layer ( Figure 13.15 ). As the ganglion cells differentiate, the surrounding cells are prevented from premature differentiation by the activity of the Notch gene. A major function of Notch is to maintain populations of cells in the nondifferentiated state until the appropriate local cues for their further differentiation appear. With the later differentiation of the horizontal and amacrine cells, the inner and outer nuclear layers take shape. While the cells within the nuclear layers send out processes, the inner and outer plexiform layers become better defined. The bipolar neurons and cone cells differentiate last, thus completing the first gradient.
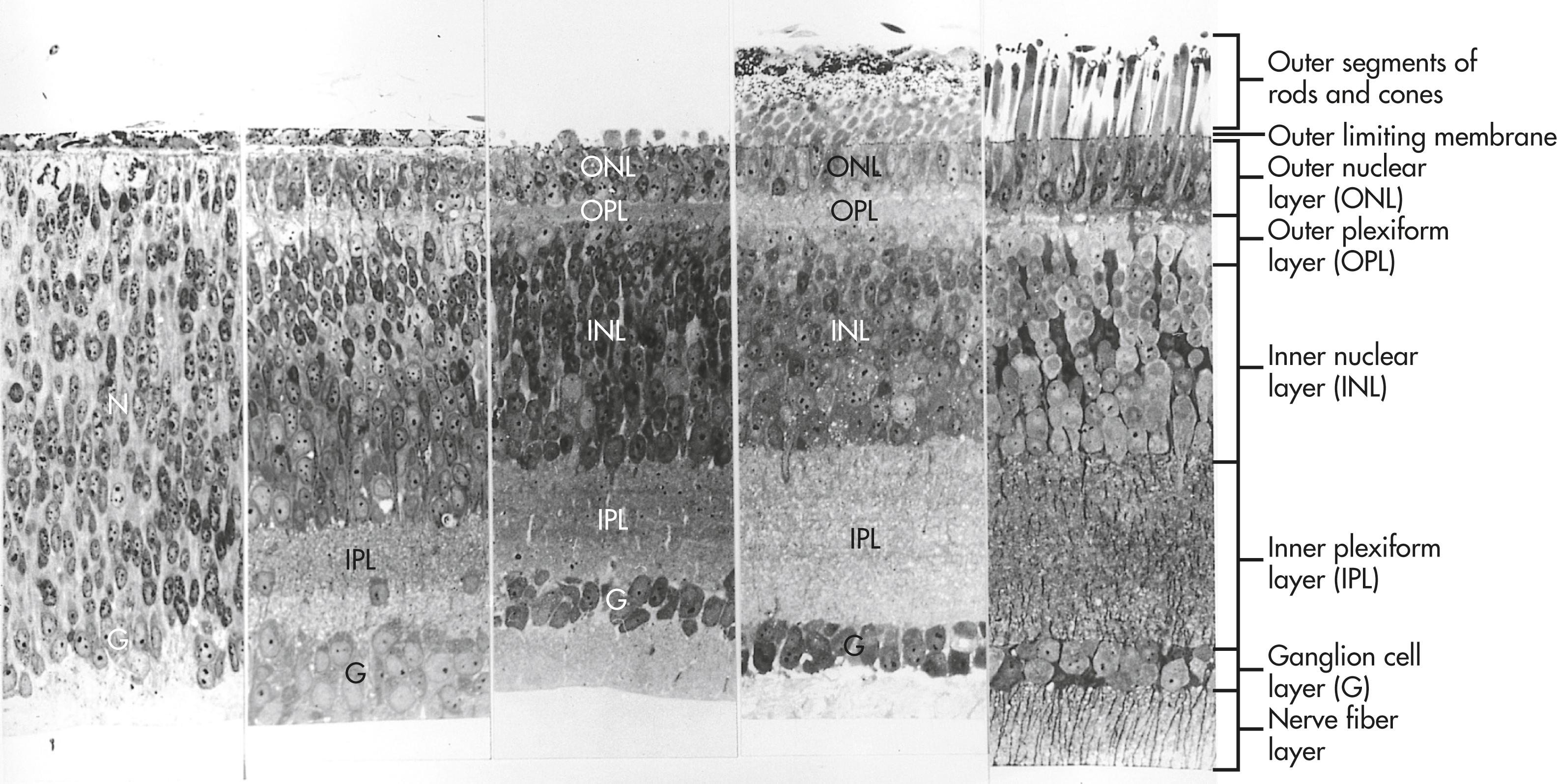
The horizontal gradient of differentiation of the neural retina is based on the outward spread of the first vertical gradient from the center to the periphery of the retina. The retina cannot grow from within. During the phase of growth in the human eye (or throughout life in the case of continuously growing animals such as fish), developmentally immature retinal precursor cells along the edge of the retina undergo mitosis as an ever-expanding concentric ring on the periphery of the retina. Just inside the ring of mitosis, cellular differentiation occurs in a manner corresponding roughly to that of the vertical gradient.
Cell lineage experiments involving the use of retroviral or other tracers (e.g., horseradish peroxidase) introduced into neuronal precursors in the early retina have revealed two significant cellular features of retinal differentiation. First, progeny of a single labeled cell are distributed in a remarkably straight radial pattern following the vertical axis of retinal differentiation. There seems to be little lateral mixing among columns of retinal cells ( Figure 13.16 ). The second cellular feature of retinal differentiation is that a single labeled precursor cell can give rise to more than one type of differentiated retinal cell. A single tripotential type of Otx-2-expressing retinal precursor cell gives rise to first cones, then rods, and finally, bipolar cells.
A later stage in retinal differentiation is the growth of axons from the ganglion cells along the innermost layer of the retina toward and into the optic stalk. Cells near the exit point express netrin-1 , which acts as an attractor for the outgrowing nerve processes. When the axons reach the optic stalk, they grow into it, following cues provided by Pax-2–expressing cells. As the axons reach the area of the optic chiasm, they undergo a segregation so that the axons that arise from the temporal half of the retina remain on the same side, and the axons from the nasal half of the retina decussate through the optic chiasm over to the other side. The choice of pathways involves precise local signaling, with netrin-1 again serving as a growth cone attractor and shh playing a new role as a repulsive signal to axonal outgrowth. During this phase of retinal axonal outgrowth, the precise retinal map is maintained in the organization of the optic nerve, and it is ultimately passed on to the visual centers of the brain.
Mediated by a still poorly defined influence of the lens, differentiation of the iris and ciliary body occurs at the lip of the optic cup, where the developing neural and pigmented retinal layers meet. Rather than being sensory in function, these structures are involved in modulating the amount and character of light that ultimately impinges on the retina. In addition, the ciliary body is the source of the aqueous humor that fills the anterior chamber of the eye. The iris partially encircles the outer part of the lens and through contraction or relaxation, it controls the amount of light passing through the lens. The iris contains an inner unpigmented epithelial layer and an outer pigmented layer, which are continuous with the neural and pigmented layers of the retina ( Figure 13.17 ). The stroma of the iris , which is superficial to the outer pigmented layer of the iris, is of neural crest origin and secondarily migrates into the iris. Within the stroma of the iris lie the primordia of the sphincter pupillae and dilator pupillae muscles. These muscles are unusual because they are of neuroectodermal origin; they arise from the anterior epithelial layer of the iris through the transdifferentiation of pigmented cells into smooth muscle.
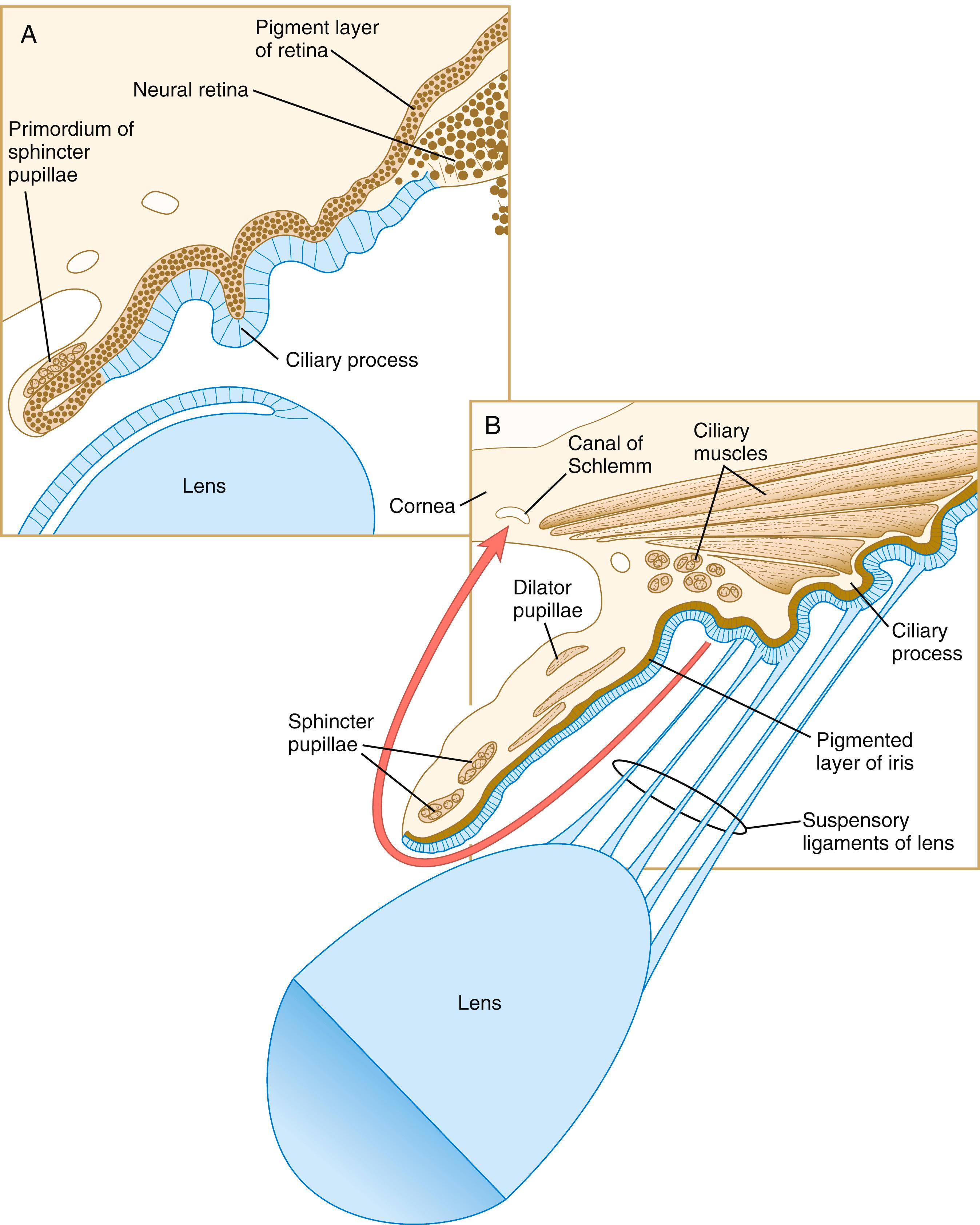
Eye color results from levels and distribution of pigmentation in the iris. Much of the bluish color of the iris in most newborns and many adults is due to the scattering of light rays from bundles of collagen within the stroma of the iris. Pigment cells also appear in the iridial stroma in front of the pigmented epithelium. The greater the density of pigment cells is in this area, the browner the eye color will be. Definitive pigmentation of the eye gradually develops over the first 6 to 10 months of life.
Between the iris and the neural retina lies the ciliary body, a muscle-containing structure that is connected to the lens by radial sets of fibers called the suspensory ligament of the lens . By contractions of the ciliary musculature acting through the suspensory ligament, the ciliary body modulates the shape of the lens in focusing light rays on the retina. Factors leading to the formation of the ciliary body are uncertain, but it seems to be induced by the lens (acting through FGFs) and surrounding neural crest mesenchyme (acting through BMPs). Expression of the transcription factor Otx-1 is important for the development of both the ciliary body and the iris. Cranial neural crest cells migrate into both the developing ciliary body and the iris to form part of the stroma of these structures.
The ciliary body of the eye secretes aqueous humor into the posterior chamber of the eye. The fluid passes in front of the lens through the pupil into the anterior chamber, where it maintains outward pressure on the cornea. It is then resorbed through a trabecular meshwork and into the canal of Schlemm in the angle of the eye (see Figure 13.17 ). This outflow area arises from the organization of neural crest cells into a trabecular meshwork of lamellae covered by flat endothelium-like cells, which abut onto an enlarged venous sinus, designated the canal of Schlemm. Development of this network in humans is not completed until approximately the time of birth.
During early development of the retina, a loose mesenchyme invades the cavity of the optic cup and forms a loose fibrillar mesh along with a gelatinous substance that fills the space between the neural retina and lens. This material is called the vitreous body .
During much of embryonic development, the vitreous body is supplied by the hyaloid artery and its branches ( Figure 13.18 ). The hyaloid artery enters the eyeball through the choroid fissure of the optic stalk (see Figure 13.6 ), passes through the retina and vitreous body, and terminates in branches to the posterior wall of the lens. As development of the retinal vasculature progresses, the portions of the hyaloid artery (and its branches supplying the lens) in the vitreous body regress through apoptosis of their endothelial cells and leave a hyaloid canal . The more proximal part of the hyaloid arterial system persists as the central artery of the retina and its branches.
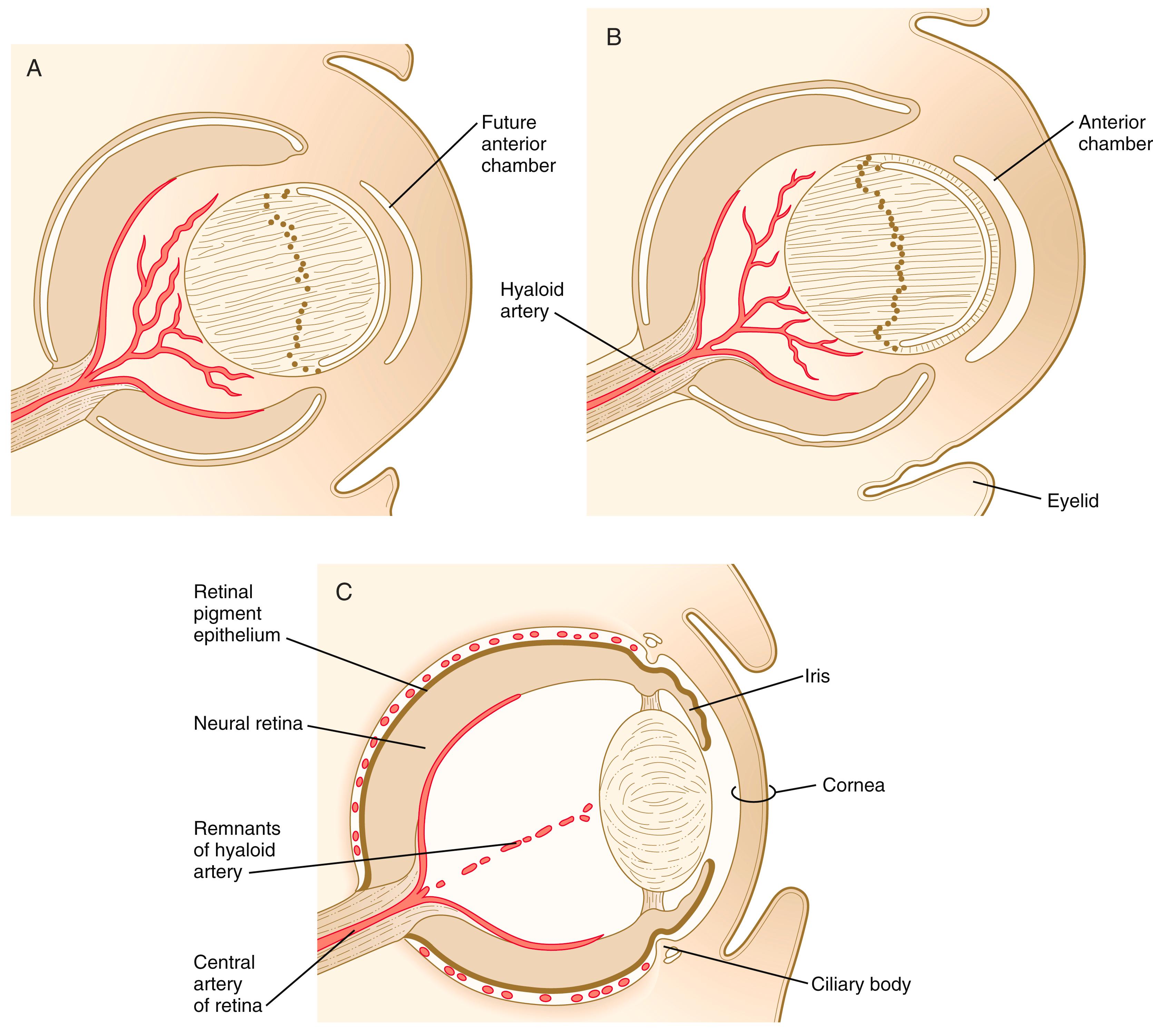
Outside the optic cup lies a layer of mesenchymal cells, largely of neural crest origin. Reacting to an inductive influence from the pigment epithelium of the retina, these cells differentiate into structures that provide vascular and mechanical support for the eye. The innermost cells of this layer differentiate into a highly vascular tunic called the choroid coat (see Figure 13.7C ), and the outermost cells form a white, densely collagenous covering known as the sclera . The opaque sclera, which serves as a tough outer coating of the eye, is continuous with the cornea. The extraocular muscles, which are derived from cranial mesoderm and provide gross movements to the eyeball, attach to the sclera.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here