Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter will briefly consider the examination of the recipient’s liver, removed at transplantation, and then focus on the role of the pathologist in the interpretation of the liver allograft biopsy.
Complications following liver transplantation have been described in other chapters of the text, as summarized in Fig. 24.1 . Biopsy manifestations of these complications are comprehensively addressed elsewhere and will be described here with an emphasis on the pediatric setting. It is stressed that biopsy alone is rarely sufficient to identify the precise cause of graft injury. This requires multidisciplinary communication and integration with other diagnostic modalities. It should also be remembered that damage to a graft can be multifactorial, and therefore the biopsy may reflect more than one complication. In cases where more than one etiological factor is present, liver biopsy can help to identify the predominant cause of graft injury.
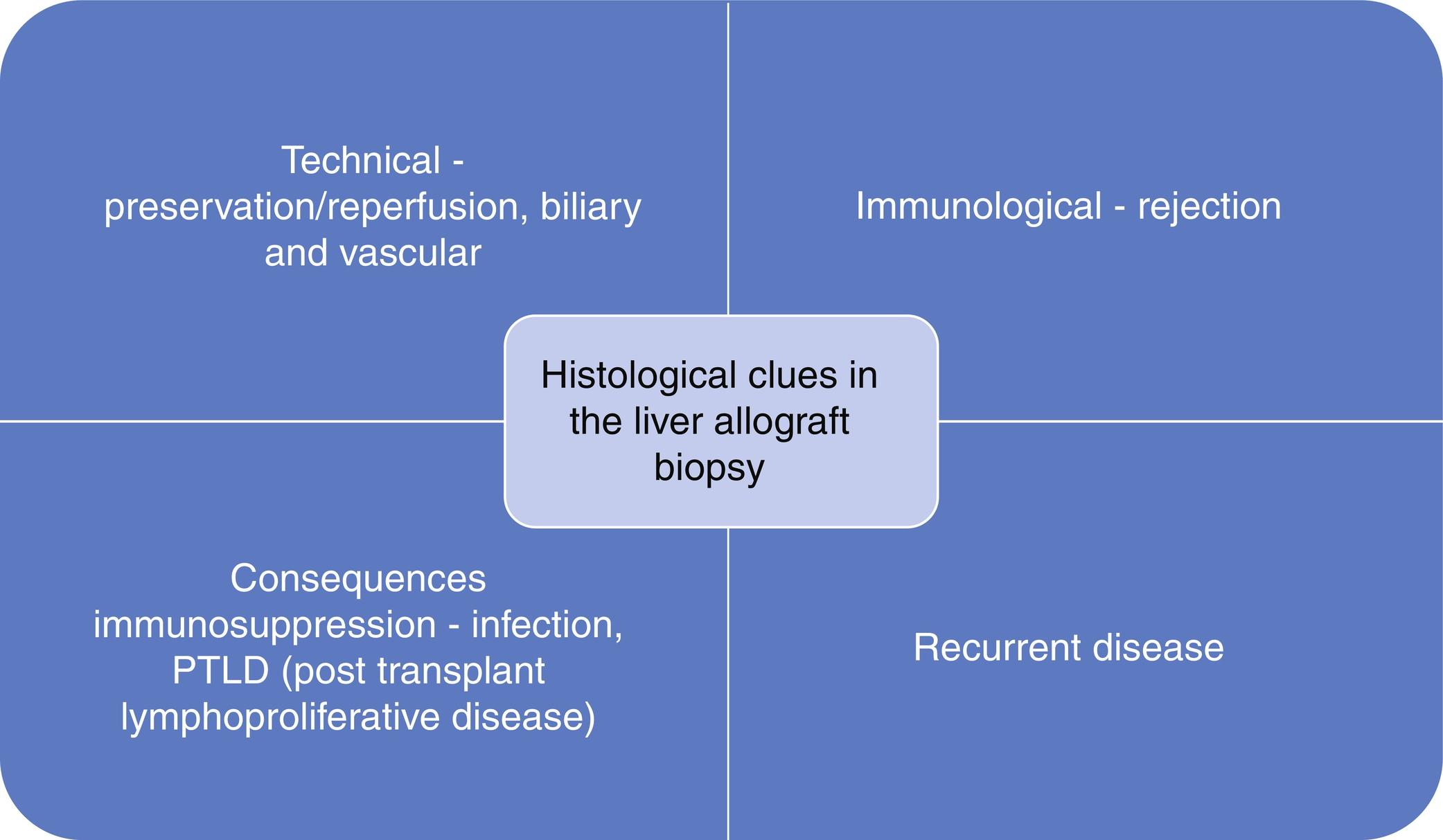
In the context of early post-operative complications, biopsies are taken with a clinical indication rather than as a protocol procedure. The pathologist cannot answer the clinical question if they do not know what the question is! The request form must say more than “abnormal liver function tests post–liver transplant.” Date of, and indication for, transplantation and the reason for the biopsy are minimum requirements if the biopsy is going to contribute to effective management and justify the risk to the patient of taking it. Major complications of biopsy may be less than 1%, but this cannot be ignored. Timing is important; it is very difficult to interpret findings after blind treatment of suspected rejection, for example.
The first role the pathologist has following liver transplantation is the examination of the recipient’s own liver, sometimes referred to as the “explant.” Defining the original disease is important for future management because some conditions can recur in the graft. Discovery of an unsuspected hepatocellular carcinoma (HCC) in a background of cirrhosis is less frequent in pediatric, as compared with adult, practice but remains an important consideration. The liver explant should be finely sliced to discover any dominant nodules. Certain pediatric conditions do predispose to HCC, for example, tyrosinemia and ATPB11 disease (bile salt export pump [BSEP] deficiency/progressive familial intrahepatic cholestasis type 2). Particular care should be taken in these settings to identify and sample all nodules that stand out from the cirrhotic background. Many of these will transpire to be regenerative/dysplastic nodules, but some may be HCCs. The distinction between pre-neoplastic and neoplastic nodules in the cirrhotic liver may be difficult. Among other criteria, features favoring a diagnosis of HCC include reticulin fiber depletion and diffuse CD34 expression on sinusoidal endothelium. Young adults with primary sclerosing cholangitis are occasionally only discovered to have hilar cholangiocarcinoma once the explant is examined after transplantation; the hilum should therefore be extensively sampled in these patients.
In some centers, a post-reperfusion biopsy of the newly implanted liver (also referred to as a “time zero” biopsy) accompanies the explant. Examination of this specimen provides a baseline and may also occasionally reveal an unsuspected liver disease in the donor. Iron deposition related to genetic hemochromatosis and globules of alpha 1 antitrypsin are occasionally seen in this setting. Large-droplet macrovesicular steatosis is a more frequent donor-related lesion and is discussed further in the preservation/reperfusion injury section.
Under this heading are conditions arising from the logistics and practicalities of transplantation, including procurement, transport and preservation, and the necessity of anastomoses of venous inflow and outflow, hepatic artery, and bile duct. There is an imbalance between the number of children awaiting transplantation and the availability of size-matched donors. Children may therefore be in receipt of a reduced or split graft, the single lobe transplanted in the living related donor setting, for example.
This occurs when the allograft liver volume is insufficient for the recipient and presents as early dysfunction or non-function of the graft, often with ascites. Small-for-size grafts have an inferior outcome in children. The postulated mechanism is portal venous hyperperfusion and reactive arterial vasospasm. Endothelial denudation of portal veins and hemorrhage into portal areas may be a clue to this in early biopsies. Later changes can occur secondary to arterial compromise, including ischemic cholangiopathy, which may lead to biliary manifestations in the peripheral liver biopsy, or an obliterative portal venopathy may develop with nodular regenerative hyperplasia. At this stage, the pathologist would not be able to implicate “small for size” specifically as the mechanism of damage.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here