Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Retinopathy of prematurity (ROP), which was first described as retrolental fibroplasia in 1942, is a vasoproliferative disease of extremely preterm infants and is a leading cause of preventable childhood blindness worldwide. Low gestational age (GA) and low birth weight (BW) contribute to ROP development, and evidence supports oxygen-induced toxic effects on immature retinal vasculature. Improved oxygen supplementation protocols in preterm neonates have resulted in a dramatic fall in the incidence of ROP. Despite major advances in identification and treatment, the Global Burden of Disease study estimated that 257,000 years lived with disability worldwide in 2010 were associated with visual impairment secondary to ROP, and the worldwide incidence will likely increase due to improved infant survival rates at increasingly younger gestational ages.
This chapter reviews retinal vascular development as it relates to ROP pathogenesis, natural history and risk factors of ROP, current screening recommendations, telemedicine programs, and advances in artificial intelligence for ROP diagnosis. Further, the challenges for ROP screening and diagnosis using current technology are discussed.
The choroid meets the metabolic needs of the retina in early fetal life with vascularization by 6 weeks gestational age. As the retina becomes increasingly complex and metabolically active, this single circulation becomes insufficient, which leads to a state of physiological retinal hypoxia. This hypoxia stimulates vascular endothelial growth factor (VEGF) with subsequent development of a retinal circulation at approximately 13–14 weeks GA. Centrifugal migration occurs by vascular endothelial cells, microglia, pericytes, and astrocytes from the optic disc. Vasculogenesis is the process of these cells aligning into vascular cords and differentiating into capillaries. This is followed by angiogenesis in which vessels extend out from existing vessels, remodel, and form a mature retinal vascular network with capillary-free areas. Fetal retinal vascular development transitions from a process of vasculogenesis to angiogenesis after 20 weeks GA.
The developing retina responds to excessive or insufficient oxygen by trimming or inducing growth in the microvasculature to match the retina's metabolic requirements. While oxygen-dependent VEGF plays a critical part in all stages of vascular development, other factors are also involved, such as insulin-like growth factor 1 (IGF-1) and erythropoietin. In normal development, the retinal vessels reach the ora serrata nasally at 36 weeks GA and temporally soon after birth at approximately 40 weeks GA.
There are two phases in ROP development, and both occur postnatally.
Premature birth requires considerable physiological adaptation given the fetus is removed from a milieu uniquely suited to growth. Specifically, cardiopulmonary adaptations associated with birth include a rise in the partial pressure of oxygen ( P aO 2 ), a measurement of oxygen pressure in arterial blood, from <25 mmHg to 80–95 mmHg within 6 hours. This directly influences the oxygen saturation ( S pO 2 ), which similarly increases from 60% to 70% maintained in utero (i.e., physiologic hypoxia) to >88% postnatally. Despite being in a new state of hyperoxia, preterm babies often require high levels of supplemental oxygen for prevention of serious sequelae, including death. These high and variable oxygen levels in the neonatal intensive care unit (NICU) and lack of maternal protection lead to decreased levels of intraocular growth factors (e.g. IGF-1 and VEGF), vasoconstriction, and, ultimately, vaso-obliteration, which is defined as the cessation of retinal vascular development with or without retraction and/or loss of retinal vessels. Vaso-obliteration was first observed in 1992 using the oxygen-induced retinopathy (OIR) ROP model. Eyes of infants initially vascularized into zones II and III exhibited obliteration of formed vessels and regression into zone I following exposure to unblended oxygen.
Akt, also known as protein kinase B (PKB), regulates endothelial cell proliferation, differentiation, and metabolism. The complex Akt endothelial cell survival pathway also influences the expression and activity of various pro- and anti-angiogenic factors. In utero, maternal IGF-1 controls VEGF activation of the Akt pathway. Thus, low levels of IGF-1 postnatally result in reduced survival and growth of vascular endothelial cells. Hyperoxia-induced downregulation of hypoxia-inducible factors (HIF) leads to decreased VEGF expression, and HIF-1 stabilization protects the retina in the OIR model. Omega-3 long chain polyunsaturated fatty acids, which have a role in vascular and neuronal survival, also become deficient following premature birth, and supplementation of these fatty acids prevents ROP in the experimental animal and in preterm infants. The roles of beta-adrenergic receptors, adenosine, apelin, nitric oxide, and gene mutations and single-nucleotide polymorphisms of ROP-related factors are actively being investigated.
The transition from phase I to phase II ROP is more dependent on the infant’s postmenstrual age (PMA) rather than chronological age, suggesting disease pathogenesis is related to the programmed timing of development. As the infant ages, usually around 32 weeks GA, the VEGF and IGF-1 drive increases proportionally to the amount of avascular retina present and the degree of hypoxia. This proliferative phase (i.e., phase II ROP) can lead to arteriovenous shunts, retinal neovascularization, vitreoretinal traction, retinal detachments, and blindness. Screening of preterm babies prior to phase II ROP at approximately 31 weeks GA is imperative for achieving good outcomes as ROP often gets worse prior to improving. Screening guidelines and current limitations of ROP diagnosis are discussed later and new antivasculogenic therapies are described in Chapter 44 .
Extremely preterm infants are at risk of developing ROP given their low gestational age (≤30 GA) and low BW (≤1500 g), which are major risk factors. The multicenter Early Treatment for Retinopathy of Prematurity (ETROP) study found that 68% of infants born <1251 g developed mild ROP or worse. The Cryotherapy for ROP (CRYO-ROP) study found that every additional gestational week led to a 19% decrease in developing severe ROP requiring treatment (i.e., threshold ROP), whereas each 100 g increase in BW was associated with a 27% decrease in threshold ROP.
Although ROP may develop in preterm infants who never receive oxygen, studies have confirmed the relationship between early postnatal oxygen administration, prolonged oxygen use, and high arterial oxygen levels with ROP development. Flynn et al. found that the odds ratio of developing severe ROP was 1.9 for every 12-hour period of P aO 2 >80 mmHg. Larger, more mature infants are at risk in less developed countries. This is due to increased ROP disease and severity in countries with lack of oxygen saturation monitors and little to no advanced training of neonatal staff and/or ophthalmologists. Both hyperoxia and hypoxia contribute to ROP pathogenesis, thus it is not surprising that there is also a relationship between neonatal hypoxia, especially recurrent apnea, and ROP. Surfactant deficiency leads to respiratory distress syndrome, and the need for surfactant therapy and prolonged mechanical ventilation are independent ROP risk factors.
The Surfactant, Positive Airway Pressure, Pulse Oximetry Randomized Trial (SUPPORT) and Benefits of Oxygen Saturation Targeting Study II (BOOST-II) reported significantly higher mortality and necrotizing enterocolitis in infants targeted to 85%–89% S pO 2 compared to those targeted to 91%–95% S pO 2 , with significantly more ROP in the latter cohort. These data confirm that lower oxygen levels reduce the incidence of ROP blindness but at the expense of reduced survival and higher morbidity. The Supplemental Therapeutic Oxygen for Prethreshold Retinopathy of Prematurity (STOP-ROP) trial found no difference in ROP incidence in infants receiving 89%–94% S pO 2 compared to 96%–99% S pO 2 .
Based on the STOP-ROP Trial, one group set baseline target oxygen saturations to 85%–95% and increased to >97% after diagnosis of prethreshold or worse ROP; this protocol showed inhibition of further progression of stage 2 ROP or worse in premature infants. A meta-analysis concluded that high supplemental oxygen given at or after 32 weeks PMA was associated with reduced incidence of severe ROP. In a recent study, biphasic oxygen targets (e.g. 85% to 92% at <34 weeks and >95% at >34 weeks) have shown a decrease in ROP incidence and severity without increasing mortality. Overall, the ideal oxygen range remains controversial, but age should guide management with S pO 2 targets of 85%–89% considered unsafe in all extremely preterm infants.
Other risk factors for ROP have been suggested, including prenatal and perinatal factors, demographics, medical interventions, comorbidities of prematurity, nutrition, and genetic factors. Acidosis is a risk factor for ROP, and the combination of hypercarbia (i.e., high P CO 2 ), P aO 2 , and low pH in the first three days after birth is associated with severe ROP. Ventilation days and seizure occurrence have been associated with severe ROP. Male gender has been associated with ROP in numerous, but not all, studies; this is hypothesized to be due to increased maternal levels of pro-inflammatory and pro-angiogenic factors. There is also likely a genetic predisposition, as variants in BDNF encoding brain-derived neurotrophic factor have been linked to retinopathy severity in preterm infants. Race and ethnicity have been shown to impact ROP incidence ; however, specific findings have differed between regions. Assisted reproductive technology, especially in vitro fertilization, has been associated with increased incidence of severe ROP in a recent meta-analysis ; larger, high-quality studies are needed to confirm these preliminary findings.
There are many conflicting studies regarding other independent risk factors; multiple births, maternal preeclampsia-eclampsia, hyperglycemia, and inflammation (e.g. sepsis, chorioamnionitis) have not been definitively confirmed as ROP risk factors despite individual, mostly retrospective, studies suggesting associations may exist. The Light Reduction in Retinopathy of Prematurity (Light-ROP) trial found no association between ROP and the ambient light in hospitals. Recombinant erythropoietin, a cytokine angiogenic factor that regulates fetal erythropoiesis, is used to treat anemia or prematurity. The use of erythropoietin and blood transfusions have been reported to increase the risk of ROP; however, these associations are not supported by systematic analyses. Lastly, a meta-analysis demonstrated that consumption of mainly human milk was associated with lower ROP incidence, whereas prolonged parenteral nutrition has been suggested as an ROP risk factor. Kim et al. reviewed over 300 studies related to ROP risk factors, and variability of study quality and presence of confounders (e.g. factors that influence BW) were common limitations for making significant associations.
ROP is classified based on the location (zone), severity (stage and vascular changes), and extent of disease according to the International Classification of Retinopathy of Prematurity (ICROP) guidelines developed in 1984 and 1987, refined in 2005, and revised in 2021 ( Table 43.1 ). The normal fundus of the extremely premature baby can be difficult to visualize in detail ( Figs. 43.1 and 43.2 ) since the retinal blood vessels are thin and straight. Later, they increase in caliber and tortuosity, abnormally so with active ROP.
| Severity | |
|---|---|
| Stage of ROP |
|
| Aggressive ROP (A-ROP) |
|
| Anterior–posterior location |
|
| Extent of disease (in clock hours) | 30-degree sectors of ROP along circumference of the vascularized retina |
| Plus disease | Abnormally dilated and tortuous vessels within the central retina |
| Pre-plus disease | Abnormal vascular dilation and tortuosity insufficient for diagnosis of plus disease |
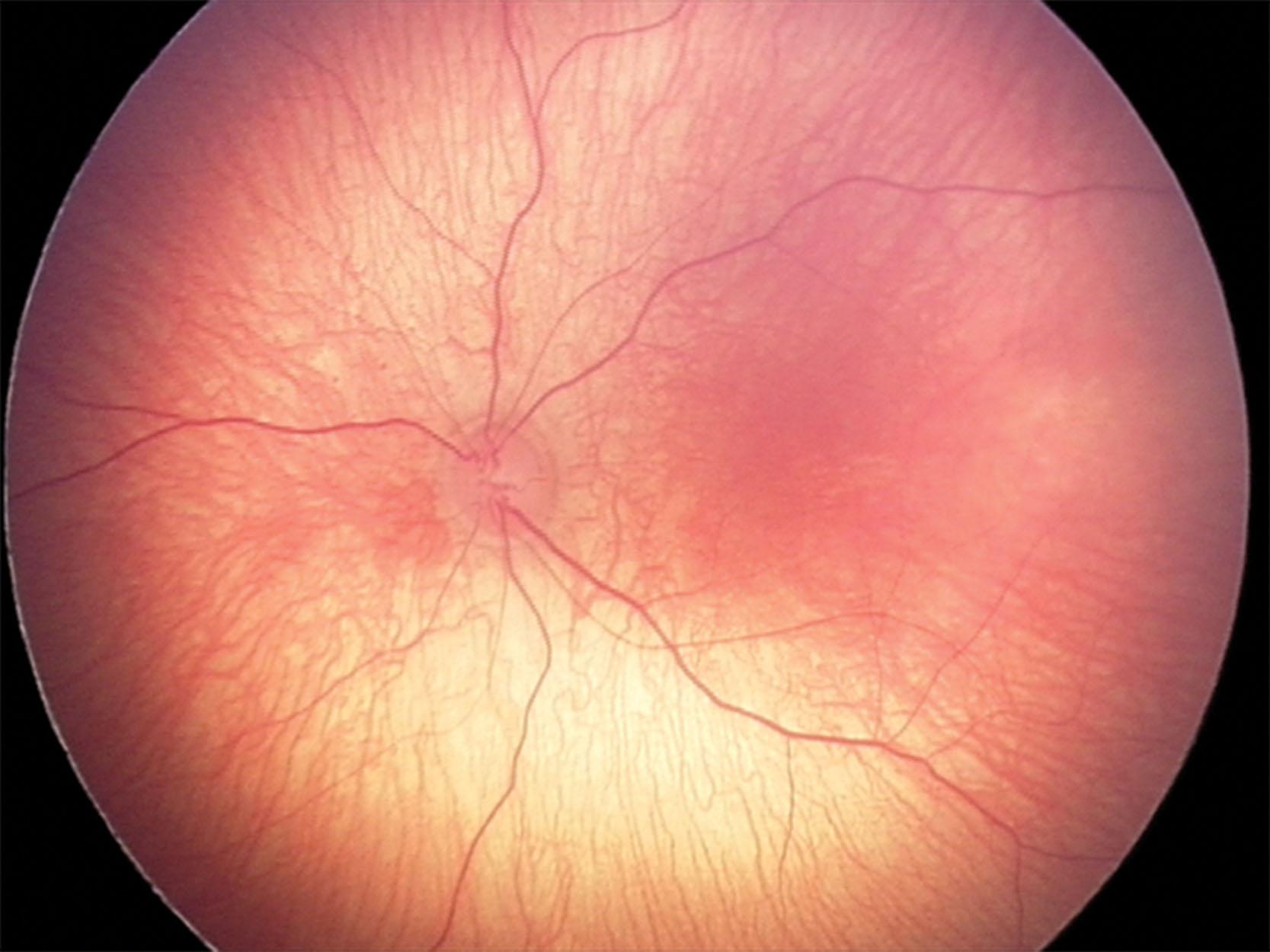
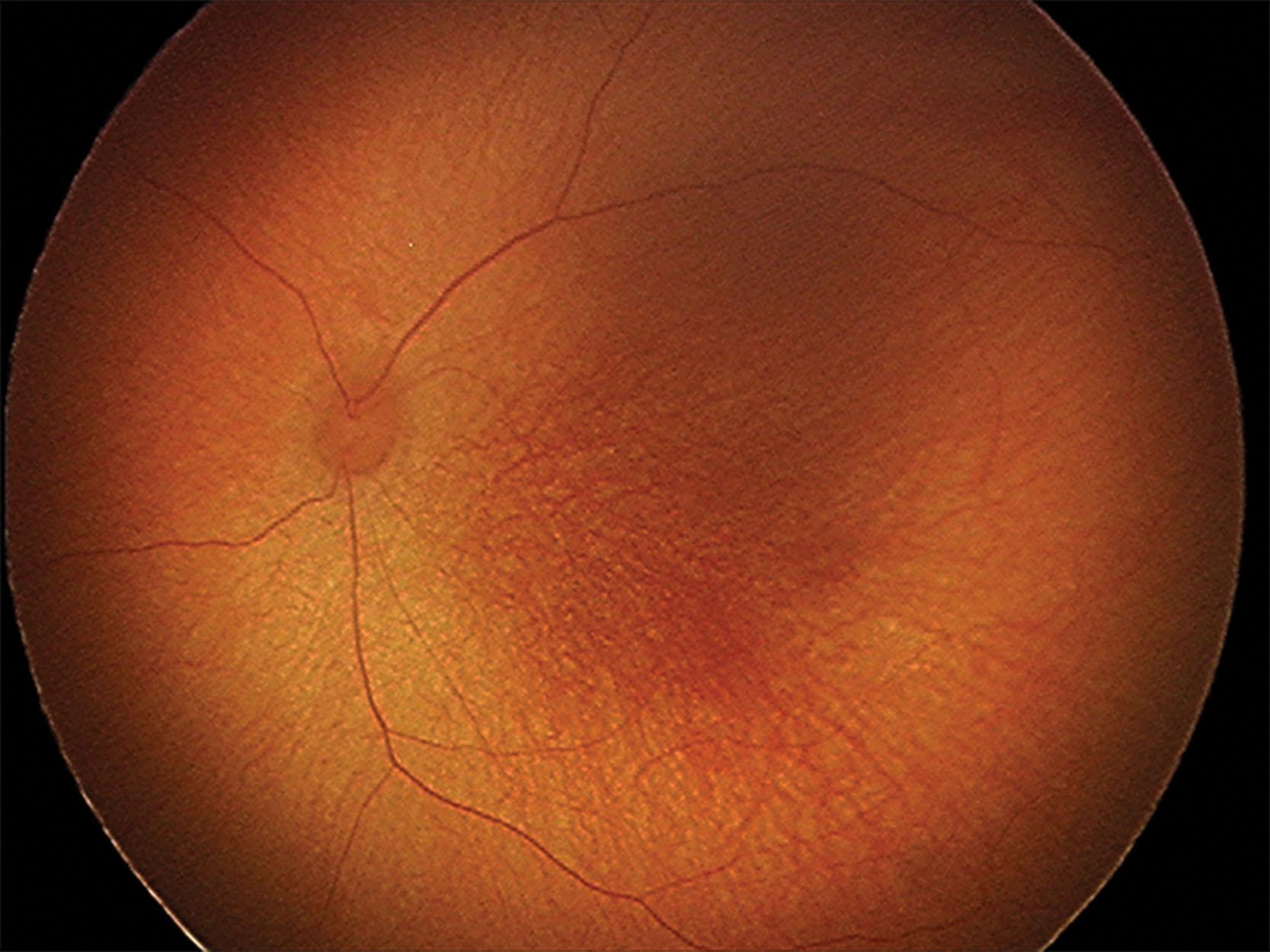
Vascular abnormalities generally increase with more posterior disease and with higher stage and extent of peripheral disease. Plus disease is defined as venous dilation and arteriolar tortuosity within the central retina. An intermediate level, pre-plus disease, was introduced to the 2005 ICROP classification reflecting the fact that vascular changes present on a continuum. In rare cases, infants may develop aggressive ROP (A-ROP) with progressive ROP.The third edition of ICROP added definitions of notch and posterior zone II (see Table 43.1 ).
The retina is divided into three concentric zones centered on the optic disc, with zone I defined as a circle that subtends 30 degrees and is centered around the optic disc with a radius twice the disc–foveal distance. Zone II is doughnut shaped and extends from zone I to the temporal equator and to a tangential point on the nasal ora serrata. The remaining crescent, zone III, includes all retina anterior to zone II. With no anatomic landmarks identifying zone III, only when the direct nasal retina is fully vascularized can it be stated during clinical ophthalmoscopic examination that zone III has been entered.
The peripheral changes at the leading edge of the developing retinal vessels have been divided into five stages. The overall stage is determined by the individual clock hour with the worse stage. Avascular retina (i.e., immature vessels) in zone I or II without other pathology is typically seen in the developing retina of premature infants, and needs to be observed carefully to monitor for development of ROP.
A flat gray-white demarcation line, comprised of differentiated vascular endothelial cells and precursors cells, separates the vascularized from the anterior avascular retina without dimension. Often faint, this line can be difficult to identify. Retinal vessels extend to the line but do not cross it ( Figs. 43.3 and 43.4A ).
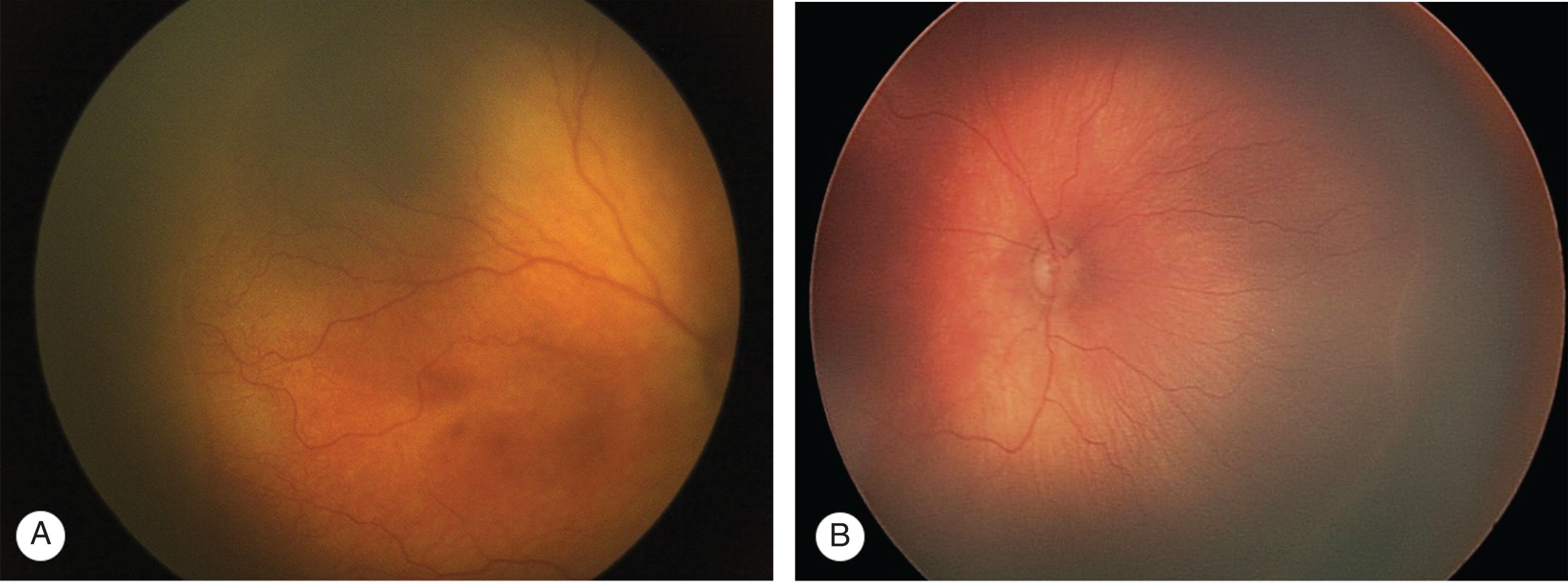
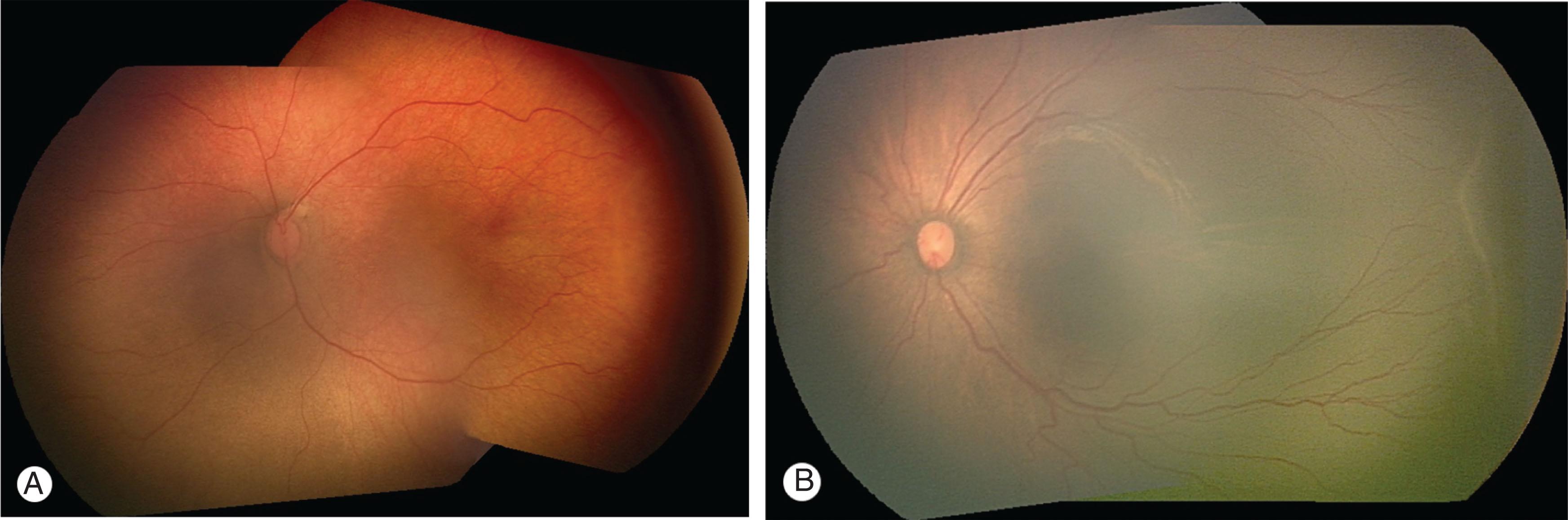
Progression leads to stage 2 ROP where the demarcation line increases in volume and extends out of the plane of the retina ( Figs. 43.4B and 43.5 ). The color of the ridge may be white or pink, and small neovascular tufts may be seen posterior to the ridge. Differentiating stage 1 and early stage 2 is difficult but usually not prognostically important.
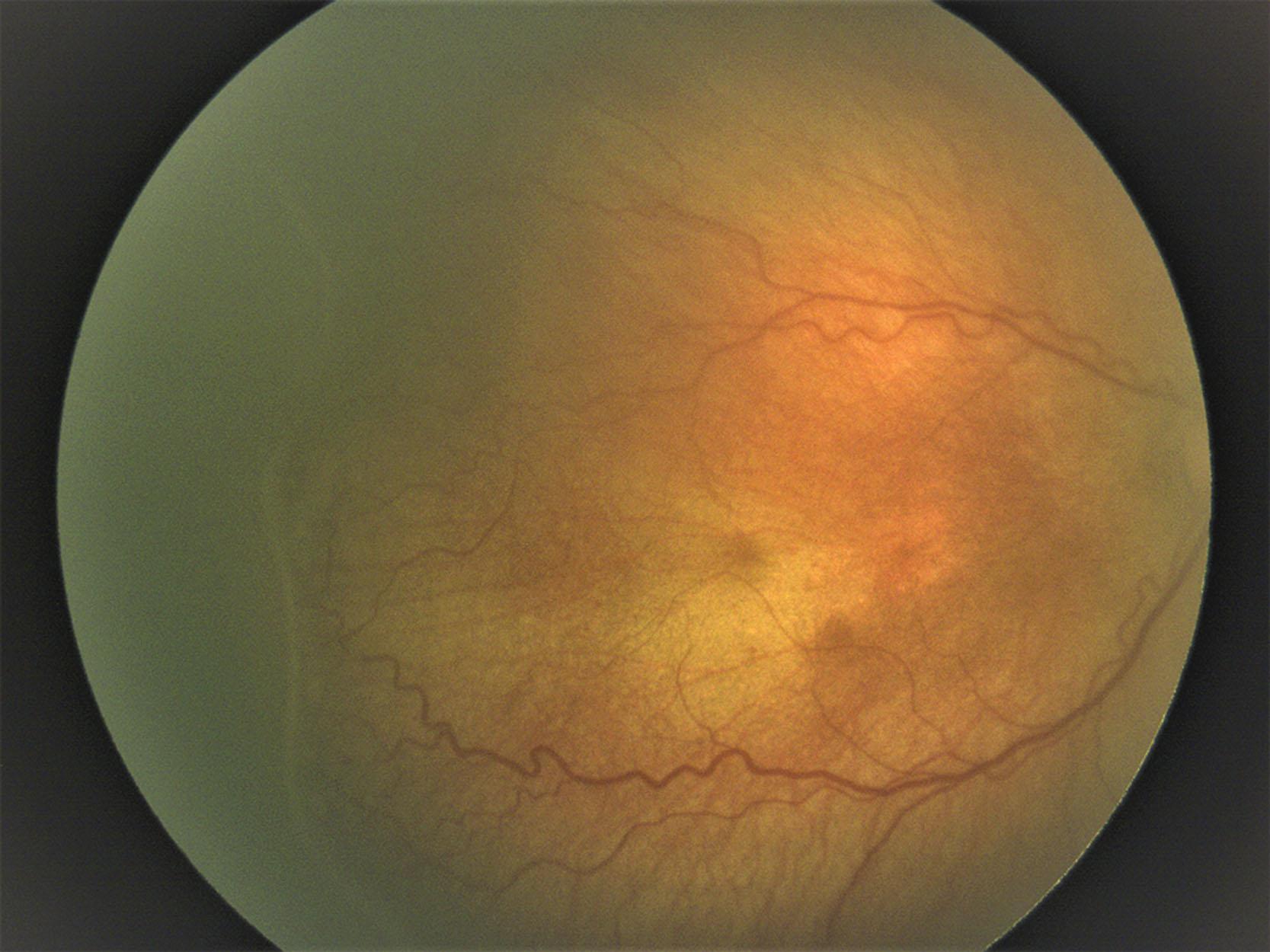
Stage 3 has the features of stage 2 but is characterized by extraretinal fibrovascular proliferation along the ridge ( Figs. 43.6 and 43.7 ). The new vessels may be continuous with, or disconnected from, the posterior border of the ridge. The extraretinal neovascularization may extend from the region of the ridge into the vitreous or, typically in more posterior disease, may extend across the surface of the vascularized retina.

Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here