Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Children affected by retinal degeneration associated with extraocular manifestations define the inherited systemic retinitis pigmentosa (RP) syndromes (retinal dystrophies related to inborn errors of metabolism are covered in Chapter 65 ) ( Tables 46.1 and 46.2 ). The gene or the group of genes involved in the syndrome belong to a specific biologic network whose mutations lead to retinal degeneration associated with manifestations in other organs defined by the specific biologic role of the protein. Retinal degeneration occurs in these syndromes by virtue of the biologic complexity of the retina. The photoreceptor cell is remarkably sensitive to alterations in many biologic pathways. The clinical presentation is often indistinguishable between syndromes with the classical features of retinal degeneration: night blindness, visual field constriction, and reduced visual acuity. In the early phases of the retinal degeneration, the fundus sometimes appears normal although the electroretinogram (ERG) is altered. Later, the fundus will acquire usual aspects observed in retinal degeneration: retinal pigmentary changes such as hypopigmentation and/or hyperpigmentation with clumping or formation of bone-spicules, retinal atrophy, optic disc pallor, narrowed vessels, and macular changes that are prominent and early in the cone–rod dystrophies.
| Extraocular clinical feature associated with RP in a child | Key feature found in systemic syndrome | |
|---|---|---|
| Stature |
|
|
| Skeletal abnormality |
|
|
| Deafness | Usher syndrome (variable severity according to types), Alström syndrome (progressive starting in childhood) | |
| CNS malformations | Cerebellar vermis agenesis (molar tooth) | Joubert syndrome |
| Kidney dysfunction |
|
BBS, Alström syndrome, Jeune syndrome, Senior–Løken syndrome, Joubert syndrome |
| Endocrine dysfunction |
|
|
| Cardiomyopathy | Alström syndrome | |
| Cognitive impairment | Classical: Cohen syndrome, Joubert syndromeOccasional: BBS, Alström | |
| Teeth |
|
|
| Skin/hair |
|
|
| Name of the syndrome with retinal degeneration | Main diagnostic extraocular signs | Identified gene(s) (main genes – not exhaustive) | Pathogenesis |
|---|---|---|---|
| Usher syndrome type 1 |
|
MYO7A , CDH23 , USH1C , PCDH15 , SANS , CIB2 | Stereocilia and photoreceptor development and function. Network of Usher proteins |
| Usher syndrome type 2 |
|
GPR98 , USH2A , WHRN , PDZD7 (modifier) | Stereocilia and photoreceptor development and function. Network of Usher proteins |
| Usher syndrome type 3 |
|
USH3A ( CLRN1 ), HRAS1 | Stereocilia and photoreceptor development and function. Network of Usher proteins |
| Bardet–Biedl syndrome |
|
BBS1 to BBS 24 | Ciliopathy |
| Alström syndrome |
|
ALSM1 | Ciliopathy |
| MORM |
|
INPP5E | Ciliopathy |
| Senior–Løken syndrome |
|
NPHP1 to 6 , SDCCAG8 | Ciliopathy |
| Mainzer–Saldino syndrome |
|
IFT140 | Ciliopathy |
| Joubert syndrome |
|
INPP5E , TMEM216 , NPHP1 , CEP290 , TMEM67 , RPGRIP1L , CC2D2A , CXORF5 , TTC21B , KIF7 , TCTN1 , TMEM237 , CEP41 , TMEM138 , C5ORF42 , TCTN3 , ZNF423 , TMEM231 , CSPP1 , PDE6D , KIAA0586 , TCTN2 , CEP104 , KIAA0556 , B9D1 , MKS1 , TMEM107 , ARMC9 , CEP120 , SUFU , PIBF1 , B9D2 , ARL3 , FAM149B1 | Ciliopathy |
| Jeune syndrome(related to short rib thoracic dysplasia) |
|
IFT80 , DYNC2H1 , TTC21B , WDR19 , NEK1 , WDR35 , WDR60 , IFT140 , IFT172 , WDR34 , CEP120 , KIAA0586 , DYNC2LI1 , IFT52 , TCTEX1D2 , IFT81 , INTU | Ciliopathy |
| Cohen syndrome |
|
VPS13B | Presumed vesicular transport |
| Cockayne syndrome (and overlapping with XP genes) |
|
CSA ( ERCC8 ), CSB ( ERCC6 ) and XPG/CS = ERCC5 ; XPB/CS = ERCC3 ; XPF/CS = ERCC4 |
|
| Jalili syndrome | Amelogenesis imperfecta | CNNM4 | Metal transport protein |
| Hypotrichosis with juvenile macular dystrophy | Early hair loss | CDH3 | Cadherin family |
| Ectodermal dysplasia, ectrodactyly, macular dystrophy (EEM) syndrome |
|
CDH3 | Cadherin family |
| Oliver–McFarlane syndrome |
|
PNPLA6 | |
| Boucher–Neuhauser syndrome |
|
PNPLA6 | |
| Spondylometaphyseal dysplasia or spondyloepiphyseal dysplasia associated to cone dystrophy |
|
PCYT1A |
All these syndromic conditions are rare inherited diseases; the most frequent syndromes are Usher syndrome (USH) and Bardet–Biedl syndrome (BBS).
Over the past two decades, most of the genes responsible for these genetically highly heterogenous conditions have been identified. This field will continue to greatly benefit from new genome exploration, especially high-throughput sequencing with next-generation sequencing (NGS) technologies, panel targeted sequencing, whole exome sequencing or whole genome sequencing). Molecular diagnosis and genetic counseling are constantly improving, especially for the syndromes which are highly heterogeneous. The classical syndromes illustrating this group of diseases are described, but a growing number of overlapping phenotypes have become evident as molecular investigations improve. Moreover, different pathogenic variants in the same gene may lead to different syndromes on the same clinical spectrum (see section “The ciliopathies”). Major clinical variability in each syndrome has been described. For some genes, specific mutations may lead to isolated features such as isolated RP (e.g. Usher syndrome gene USH2A or Bardet–Biedl genes BBS8 and BBS3 ). A child presenting with RP should be systematically evaluated for extraocular associated features (unless he/she is already known to be affected by a non-syndromic pathogenic variant). These conditions require multidisciplinary follow-up and management.
Holistic care is advocated taking into account the multiple associated disabilities. Many research approaches to alleviate the retinal degeneration are underway, including gene transfer and gene editing, pharmacological compounds, and cell therapies.
In Usher syndrome, sensorineural deafness is associated with progressive retinal degeneration. USH occurs in 1 of every 10 deaf children (5% of all cases of congenital deafness). It is the most frequently inherited syndrome with deafness and is the most common syndrome among the deaf–blind. The syndrome is subdivided into three groups: Usher type 1 (USH1), type 2 (USH2), and type 3 (USH3). USH1 and 2 are usually diagnosed in early or late childhood, respectively, but may be diagnosed later in life as the visual impairment may be overlooked in a deaf child. Usher type 3 is much rarer; the onset of deafness is in late childhood and is followed by visual loss. It is more common in patients with a Finnish descent or those of Askenazi Jewish origin. Vestibular dysfunction is classically associated with USH1. Each type of Usher syndrome is autosomal recessive but genetically heterogeneous. For each type, a number of genes have been identified (see Table 46.1 ). Molecular studies have shown clinical overlap between the classical types USH1 and USH2. Some Usher genes can be involved in either isolated deafness ( MYO7A or CI2N for example) or isolated RP ( USH2A for example); therefore variants from genetic screening should be interpreted cautiously with respect to their prognostic implications. Cochlear implantation is highly recommended for these children (especially in USH1). No therapy is yet available for the retinal degeneration ( Fig. 46.1A–C ).
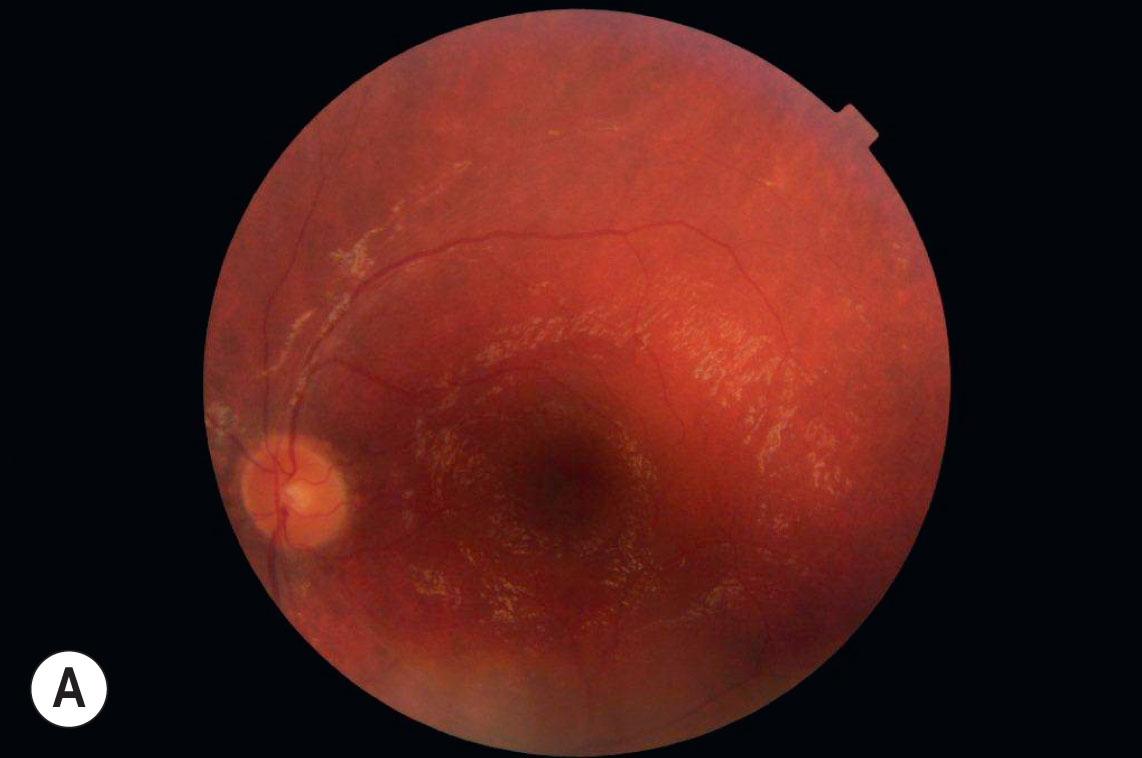
The child with USH1 is affected with congenital severe to profound sensorineural hearing loss associated with delayed sitting and walking due to abnormal vestibular function. Retinal degeneration occurs early in childhood. As early as 2 or 3 years old, the ERG can confirm the diagnosis of retinal degeneration although the fundus appears to be normal. Five causative genes are known and mutated in various proportions according to the studied population: myosin 7 A gene ( MYO7A or USH1A ) is recognized to be the main USH1 gene mutated in half of the cases, harmonin gene ( USH1C ), cadherin-related 23 gene ( CDH23 ; USH1D ), protocadherin-related 15 gene ( PCDH15 or USH1F ), sans gene ( USH1G ), and calcium and integrin family member 2 gene ( CIB2 or USH1J ). Although always severe, the course of disease may vary according to the type of variant.
Congenital mild to severe sensorineural stable hearing loss with major impairment in high frequencies and normal vestibular function are the main characteristics of USH2. The retinal degeneration occurs later than in USH1, usually during or after puberty. USH2 and USH3 are not usually symptomatic in the first decade and initially have a normal fundus appearance. Many patients retain good visual acuity in spite of constricted visual fields. The ERG may initially be normal; however, ERG changes may be detected in pre-symptomatic young children. During the second decade, night blindness and loss of peripheral vision become evident and inexorably progress ( Fig. 46.1D ). Three main genes have been identified: usherin ( USH2A , and is also a common cause of non-syndromic RP), G-protein coupled receptor 98 ( GPR98 also named ADGRV1 , VLGR1 , USH2B or USH2C ), and whirlin ( WHRN , also named USH2D ) (NB: in addition PDZD7 has been named as as a modifier).
The onset of RP in USH3 is variable and is often recognized after the second decade of life, but can be confused with the two other types. Sensory hearing loss is postlingual after the acquisition of speech (as opposed to the prelingual loss in USH1 and USH2), but it evolves progressively to profound deafness. Vestibular dysfunction of variable intensity occurs in half of the patients. Usher type 3 is mostly due to pathogenic variants in the clarin 1 gene ( CLRN1 , also named UHS3A ), particularly in Ashkenazi Jews and Finns. Much rarer biallelic pathogenic variants were found in HRAS1 (USH3B) in Amish families.
Progress has been made in understanding the pathogenesis of this disease, which affects the stereocilia of the inner ear and photoreceptor cells of the retina. Overall, 10 genes are implicated at the present time. The USH1 gene products form a network, the Usher interactome, which plays a key role in the early hair bundle development. They are also key components of the mechanoelectrical transduction machinery necessary for hearing. In the retina, the Usher interactome acts at the level of the ciliary/periciliary region of the connecting cilia of the photoreceptor cells. These are important for the transport of proteins between the outer and the inner segments. Alteration of the USH proteins leads to early dysfunction of the inner ear and of the retina simultaneously ( Fig. 46.1E ).
In addition to Usher syndrome, other syndromic non-Usher retinal dystrophies with deafness as main symptoms can occur in childhood such as: autosomal dominant Leber congenital amaurosis with early-onset deafness with variants in the TUBB4B gene, autosomal recessive cone rod dystrophy, and hearing loss with variants in the CEP250 gene.
A single non-motile cilium (“primary cilium”) is present in almost every vertebrate cell; motile cilia are present only in specific organs such as in the respiratory or the reproductive systems (for review see Brown et al. ) ( Fig. 46.2A ). The primary cilium is the central “antenna” of the cell, allowing transduction of sensorial information from the extracellular environment to the cell. The photoreceptor cell has a connecting cilium and is, therefore, a ciliated cell.
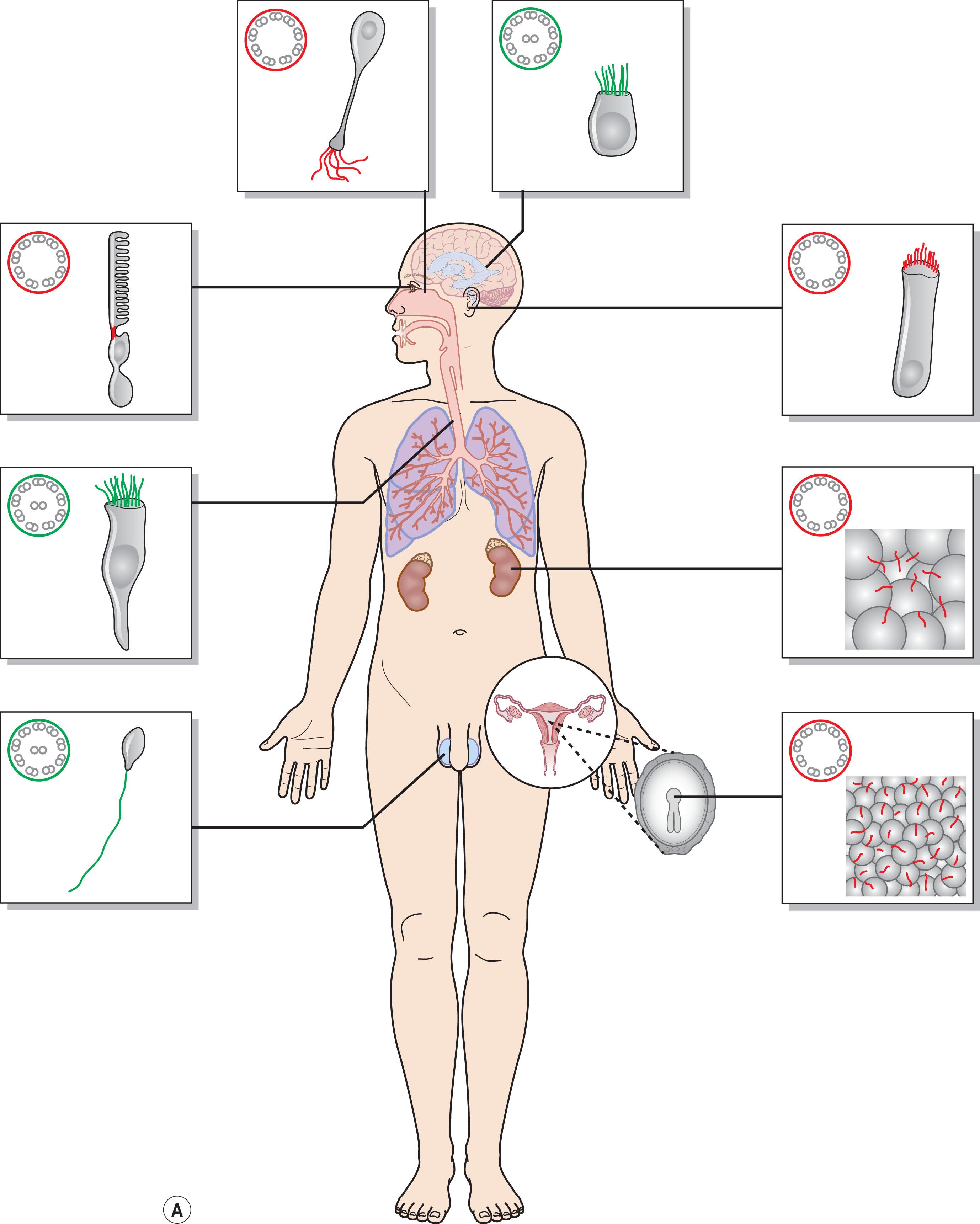
Ciliopathies are rare genetic disorders characterized by primary cilium dysfunction, frequently affecting photoreceptors and causing retinal degeneration either as an isolated condition (e.g. Leber congenital amaurosis with CEP290 mutations) or as a ciliopathy syndrome affecting more than one organ system. Ciliopathies involve many target organs that have in common an important role of the primary cilia. Overlapping phenotypes have been reported, as well as great clinical variability in the number and degree of severity of affected organs among patients with the same syndrome. The most common ciliopathies are schematically represented in Fig. 46.2B .
Molecular investigations have revealed major genetic heterogeneity for each syndrome and also allelic variability (different mutations in the same gene may lead to different syndromes). For instance, CEP290 – NPHP6 truncating variants have been identified in Joubert syndrome and hypomorphic variants in Leber congenital amaurosis (LCA). Moreover, CEP290 – NPHP6 is also mutated in several distinct disorders (nephronophthisis, Senior–Løken syndrome, Joubert syndrome, BBS, and the lethal Meckel syndrome).
Supplementary genetic factors such as a third mutated allele in a BBS gene (known as oligogenism – a third pathogenic variant occurring in addition to the two main variants found in recessive inheritance) or other genetic modifiers can influence the phenotype.
Annual multidisciplinary follow-up is recommended for these children, paying special attention to renal function, endocrine status, and prevention of obesity.
The cardinal features of the Bardet–Biedl syndrome (BBS) are early-onset retinal degeneration, polydactyly, obesity, renal dysfunction, and hypogonadism ( Fig. 46.3A–D ). Secondary features may include intellectual disability, anosmia, diabetes, cardiac anomalies, hepatic fibrosis, brachydactyly, and Hirschprung disease. The retinal dystrophy is early in onset, and leads to severe visual handicap before adulthood. Profound ERG abnormalities can be detected as early as 3 years of age but may occur later. Legal blindness usually results before the second decade of life. Retinal dystrophies in BBS are mainly a rod–cone dystrophy or a cone–rod dystrophy, usually classified as a global retinal degeneration because both rods and cones are affected ( Fig. 46.3E ).
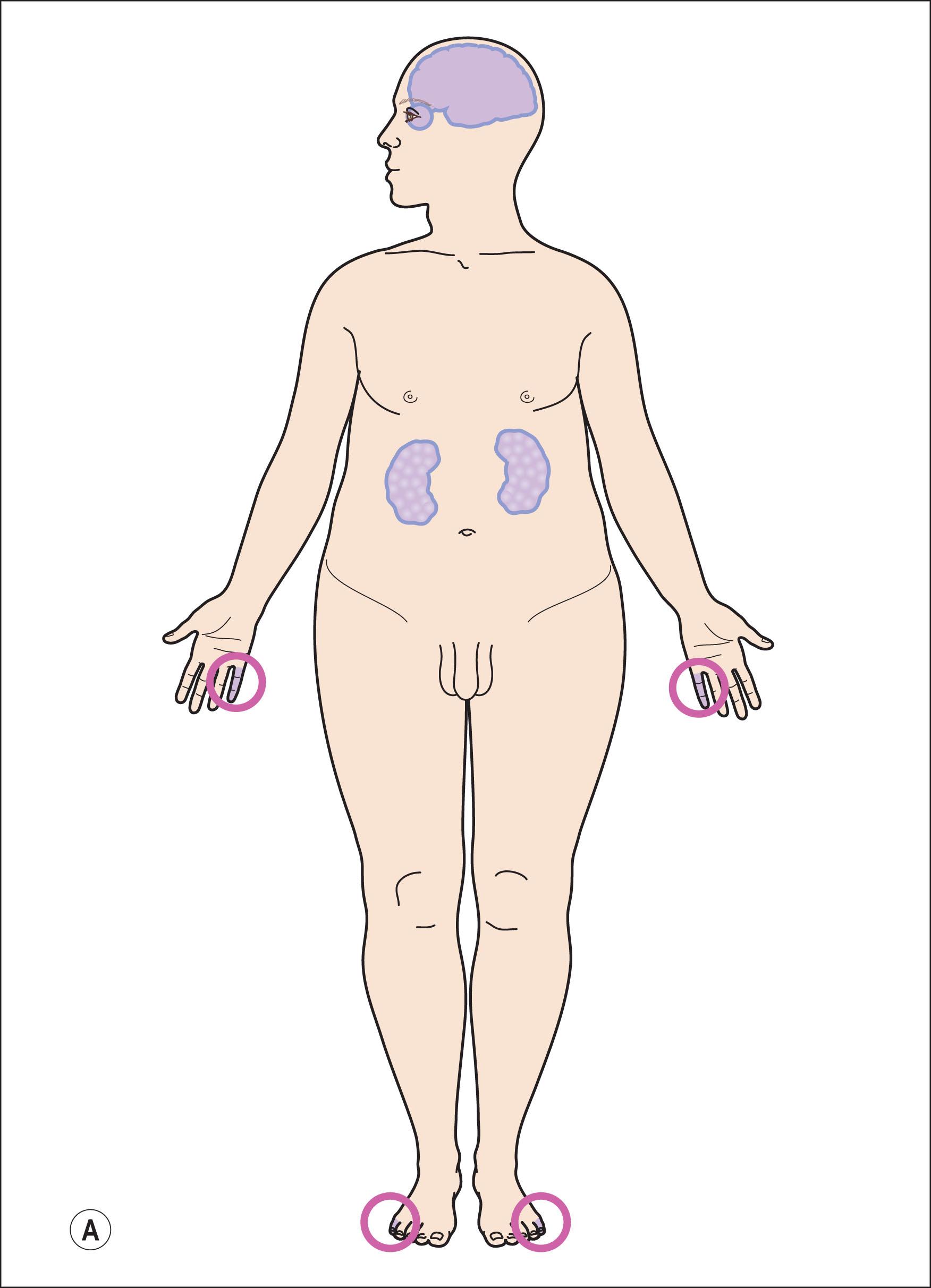
Obesity is the second major feature, present in 72%–96% of BBS patients. It usually begins in early childhood and becomes increasingly severe with age. Its origin is both central (hypothalamic eating control) and peripheral (adipose tissue).
Limb anomalies are found in almost 95% of BBS patients, usually a postaxial polydactyly (69%). Other limb malformations such as brachydactyly or syndactyly are frequently reported for both hands and feet and have a diagnostic value. Abnormalities of the genitalia can occur: hypogonadism in males or vaginal atresia in females. Rarely, there is hydrometrocolpos, a neonatal vaginal malformation leading to a massive abdominal tumor. Renal dysfunction can occur in late childhood and may lead to kidney failure. Neuropsychiatric symptoms can include developmental delay, intellectual disability, learning difficulties, speech deficit, and behavioral problems. Intellectual function ranges from severe intellectual disability through low intelligence to normal intelligence. Slow ideation and hyperemotive status are common.
BBS is an autosomal recessive heterogeneous condition; more than 24 genes have been identified, which account for the molecular diagnosis of about 85% of cases.
All the genes have been related to cilium biogenesis and/or function. BBS1 and BBS10 are the two most common, each accounting for about 20% of the cases. Implication of the other BBS genes ranges from a unique family to a few percent of mutated families. The classical autosomal recessive inheritance model has been challenged by molecular and functional investigations with the oligogenic model and the effect of additional genetic modulators on the phenotype.
Alström syndrome (ALMS) manifestations include retinal degeneration in early childhood, sensory hearing impairment, and metabolic defects leading to insulin resistance with type 2 diabetes mellitus and obesity in childhood. Polydactyly does not occur. Cardiomyopathy can occur in infancy within the first months of life that can be recurrent or appear de novo with juvenile or adult onset that may lead to congestive heart failure. Renal dysfunction is reported in half of ALMS cases. Developmental or motor delay can occur (46%).
The first manifestation of ALMS is severe early-onset cone–rod retinal dystrophy, sometimes mimicking Leber congenital amaurosis with very early visual impairment, intense photophobia, and nystagmus ( Fig. 46.4 ). Children usually become truncally obese during their first year. Sensorineural hearing loss presents in the first decade in up to 70%; it may progress to the moderately severe range (40–70 dB) by the end of the first to second decade. Insulin-resistant type 2 diabetes mellitus often presents in the second decade and is accompanied by acanthosis nigricans (pigmentation mostly in body folds). Other endocrine and metabolic abnormalities include hypothyroidism, diabetes insipidus, dyslipidemia, growth hormone deficiency, hyperuricemia, hyperlipidemia, hypothyroidism, and hypogonadotrophic hypogonadism. Hepatic and renal dysfunction can be present in the second decade. Restrictive lung disease may represent a high risk during anesthesia. This syndrome requires a multidisciplinary follow-up to detect complications once the diagnosis is confirmed.
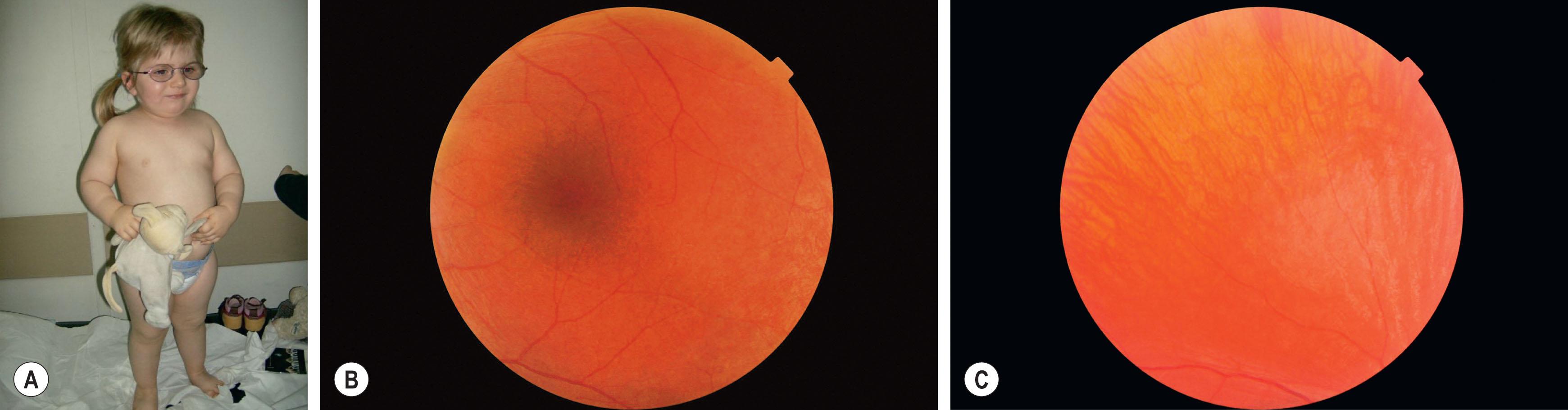
A single gene is involved in all the cases, ALMS1 , in which mainly nonsense and frame shift mutations occur (96%). ALMS1 protein has been found to be located at the base of cilia, however the precise pathogenesis remains to be determined.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here