Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Vaccines are one of the most significant achievements of science and public health. As a result of successful vaccination programs and campaigns, many vaccine-preventable diseases are now uncommon in the United States. Vaccines for the prevention of infectious diseases are regulated by the U.S. Food and Drug Administration (FDA), and the legal framework for regulation is derived from Section 351 of the Public Health Service Act and certain sections of the federal Food, Drug, and Cosmetic Act (FD&C Act). , The FD&C Act defines drugs, in part, by their intended use as “articles intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease.” Thus, vaccines are a unique class of pharmaceutical products that meet the statutory definition of both a drug and biological product. Prophylactic vaccines differ from many other drugs and biologicals primarily in how they are administered to a large population, in particular young, healthy people, to prevent rather than treat disease, their mechanism of action, and their risk/benefit profile. Although subject to the same regulations as other biological products, vaccines are inherently more difficult to develop, characterize, and manufacture than most pharmaceutical products. Current U.S. licensed vaccines are listed in Tables 80.1 and 80.2 .
| Vaccine | Manufacturer | |
|---|---|---|
| Anthrax vaccine, adsorbed | Emergent Biodefense Operations Lansing, Inc. | |
| BCG vaccine | Organon Teknika Corporation | |
| Cholera vaccine, live, oral | Pax Vax Bermuda Ltd. | |
| COVID-19 Vaccine, mRNA | ModernaTX, Inc. | |
| COVID-19 Vaccine, mRNA | Pfizer-BioNTech | |
| Diphtheria and tetanus toxoids adsorbed | Sanofi Pasteur, Inc. | |
| Diphtheria and tetanus toxoids and acellular pertussis vaccine, adsorbed | Sanofi Pasteur, Inc., a Sanofi Pasteur, Ltd., GlaxoSmithKline Biologicals | |
| Diphtheria and tetanus toxoids and acellular pertussis vaccine adsorbed, hepatitis B (recombinant) and inactivated poliovirus vaccine combined | GlaxoSmithKline Biologicals | |
| Diphtheria and tetanus toxoids and acellular pertussis adsorbed and inactivated poliovirus vaccine | Sanofi Pasteur, Ltd., GlaxoSmithKline Biologicals | |
| Diphtheria and tetanus toxoids and acellular pertussis adsorbed, inactivated poliovirus and Haemophilus influenzae type b conjugate (tetanus toxoid conjugate) vaccine | Sanofi Pasteur, Ltd. | |
| Diphtheria and tetanus toxoids and acellular pertussis adsorbed, inactivated poliovirus, haemophilus b conjugate [meningococcal protein conjugate] and hepatitis b [recombinant] vaccine | MSP Vaccine Company | |
| Tetanus and diphtheria toxoids, adsorbed | Massachusetts Public Health Biological Laboratories | |
| Tetanus and diphtheria toxoids, adsorbed for adult use | Sanofi Pasteur, Inc., Sanofi Pasteur, Ltd. | |
| Tetanus toxoid | Sanofi Pasteur, Inc. | |
| Tetanus toxoid, reduced diphtheria toxoid and acellular pertussis vaccine, adsorbed | Sanofi Pasteur, Ltd., GlaxoSmithKline Biologicals | |
| Haemophilus influenzae type b conjugate vaccine (meningococcal protein conjugate) | Merck Sharp and Dohme Corp. | |
| Haemophilus influenzae type b conjugate vaccine (meningococcal protein conjugate) and hepatitis B (recombinant) vaccine | Merck Co., Inc. | |
| Haemophilus influenzae type b conjugate vaccine (tetanus toxoid conjugate) | GlaxoSmithKline Biologicals, Sanofi Pasteur S.A. | |
| Meningococcal (groups A, C, Y, and W-135), oligosaccharide diphtheria CRM 197 conjugate vaccine | Novartis Vaccines and Diagnostics, Inc. | |
| Meningococcal (Groups A, C, Y, W) Conjugate Vaccine | Sanofi Pasteur, Inc. | |
| Meningococcal group B vaccine | Wyeth Pharmaceuticals Inc., Novartis Vaccines and Diagnostics, Inc. | |
| Meningococcal groups C and Y and Haemophilus influenzae type b tetanus toxoid conjugate vaccine | GlaxoSmithKline Biologicals | |
| Meningococcal (groups A, C, Y and W-135) polysaccharide diphtheria toxoid conjugate vaccine | Sanofi Pasteur, Inc. | |
| Meningococcal polysaccharide vaccine, A, C, Y, W135 combined | Sanofi Pasteur, Inc. | |
| Pneumococcal polysaccharide vaccine, polyvalent | Merck Co., Inc. | |
| Pneumococcal 7-valent conjugate vaccine (diphtheria CRM 197 protein) | Wyeth Pharmaceuticals Inc. | |
| Pneumococcal 13-valent conjugate vaccine (diphtheria CRM 197 protein) | Wyeth Pharmaceuticals Inc. | |
| Typhoid vaccine, live oral, Ty21a | Berna Biotech | |
| Typhoid Vi polysaccharide vaccine | Sanofi Pasteur S.A. |
a License for the pertussis component of this product is held by the Research Foundation for Microbial Diseases of Osaka University (Aventis Pasteur Laboratories, Inc.).
| Vaccine | Manufacturer a | |
|---|---|---|
| Adenovirus type 4 and type 7 vaccine, live | Barr Labs Inc. | |
| COVID-19 Vaccine, mRNA | ModernaTX, Inc. | |
| COVID-19 Vaccine, mRNA | Pfizer-BioNTech | |
| Dengue Tetravalent Vaccine, Live | Sanofi Pasteur Inc. | |
| Diphtheria and tetanus toxoids and acellular pertussis vaccine adsorbed, hepatitis B (recombinant) and inactivated poliovirus vaccine combined | GlaxoSmithKline Biologicals | |
| Diphtheria and tetanus toxoids and acellular pertussis adsorbed and inactivated poliovirus vaccine | Sanofi Pasteur, Ltd., GlaxoSmithKline Biologicals | |
| Diphtheria and tetanus toxoids and acellular pertussis adsorbed, inactivated poliovirus, haemophilus b conjugate [meningococcal protein conjugate] and hepatitis b [recombinant] vaccine | MSP Vaccine Company | |
| Ebola Zaire Vaccine, Live | Merck Sharp & Dohme Corp. | |
| Hepatitis A vaccine, inactivated | Merck & Co., Inc., GlaxoSmithKline Biologicals | |
| Hepatitis B vaccine, recombinant | Merck & Co., Inc., GlaxoSmithKline Biologicals | |
| Hepatitis B Vaccine (Recombinant), Adjuvanted | Dynavax Technologies Corporation | |
| Hepatitis A vaccine, inactivated and hepatitis B (recombinant) vaccine | GlaxoSmithKline Biologicals | |
| Haemophilus influenzae type b conjugate vaccine (meningococcal protein conjugate) and hepatitis B recombinant vaccine | Merck & Co., Inc. | |
| Human papillomavirus (types 6, 11, 16, 18) recombinant vaccine | Merck & Co., Inc. | |
| Human papillomavirus bivalent (types 16 and 18) vaccine, recombinant | GlaxoSmithKline Biologicals | |
| Human papillomavirus 9-valent vaccine, recombinant | Merck & Co., Inc. | |
| Influenza virus vaccine, trivalent, types A and B | Sanofi Pasteur, Inc., Novartis Vaccines and Diagnostics Ltd., GlaxoSmithKline Biologicals, ID Biomedical Corporation of Quebec, CSL, Ltd., Protein Sciences Corporation | |
| Influenza A (H1N1) 2009 monovalent | Sanofi Pasteur, Inc., Novartis Vaccines and Diagnostics Ltd., ID Biomedical Corporation of Quebec, MedImmune Vaccines, Inc., CSL, Ltd. | |
| Influenza virus vaccine, H5N1 (for national stockpile) | Sanofi Pasteur, Inc. | |
| Influenza A (H5N1) virus monovalent vaccine, adjuvanted | ID Biomedical Corporation of Quebec, Seqirus Inc. | |
| Influenza vaccine, adjuvanted | Novartis Vaccines and Diagnostics | |
| Influenza virus vaccine, quadrivalent, types A and B | Sanofi Pasteur, Inc., GlaxoSmithKline Biologicals, ID Biomedical Corporation of Quebec, MedImmune Vaccines, Inc. | |
| Influenza virus vaccine, intranasal | MedImmune Vaccines, Inc. | |
| Japanese encephalitis vaccine, inactivated | The Research Foundation for Microbial Diseases of Osaka University | |
| Japanese encephalitis vaccine, inactivated, adsorbed | Valneva Austria GmbH | |
| Measles, mumps, and rubella virus vaccine, live | Merck & Co., Inc. | |
| Measles, mumps, rubella, and varicella vaccine, live | Merck & Co., Inc. | |
| Poliovirus vaccine inactivated, monkey kidney cell | Sanofi Pasteur, S.A. | |
| Rabies vaccine | Novartis Vaccines and Diagnostics Ltd., Sanofi Pasteur, S.A. | |
| Rotavirus vaccine, live, oral | GlaxoSmithKline Biologicals | |
| Rotavirus vaccine, live, oral, pentavalent | Merck & Co., Inc. | |
| Rubella virus vaccine, live | Merck & Co., Inc. | |
| Smallpox and monkeypox vaccine, live, nonreplicating | Bavarian Nordic | |
| Smallpox (vaccinia) vaccine, live | Sanofi Pasteur Biologics | |
| Varicella virus vaccine live | Merck & Co., Inc. | |
| Yellow fever vaccine | Sanofi Pasteur, Inc. | |
| Zoster vaccine, live | Merck & Co., Inc. | |
| Zoster vaccine, recombinant, adjuvanted | GlaxoSmithKline Biologicals |
a These are the names of the license holders. Company names may be different.
Regulation of biologics has historically been initiated in response to issues of safety. Over time, legislative authorities have evolved to strengthen and modernize the regulation of vaccines and other biologics. Prior to 1902, manufacturing and product standards for biologics were not federally mandated. However, in 1902, the U.S. Congress passed an act to regulate the sale of viruses, serums, toxins, and analogous products (later referred to as the Biologics Control Act) following the deaths of 20 children who received contaminated products. This Act authorized the Hygienic Laboratory of the Public Health and Marine Hospital Service to issue regulations that governed all aspects of commercial production of vaccines, serums, toxins, and antitoxins, and similar products with the objective of ensuring their safety and purity. The regulations under this legislation contained the primary concepts for the regulation of biologicals, such as labeling, mandatory facility inspections, and batch-certification guidelines. In 1930, the Hygienic Laboratory was reorganized, expanded, and renamed the National Institutes of Health (NIH).
In 1944, Congress recodified the 1902 Biologics Control Act as part of the U.S. Public Health Service Act (PHS Act) of 1944. The PHS Act incorporated the 1902 Biologics Control Act into Section 351 of the PHS Act (42 U.S.C. 262). As with the 1902 Act, the 1944 PHS Act focused primarily on extensive control over manufacturing methods to ensure safety and purity. Unique to the 1944 PHS Act was Congress’s explicit addition of the requirement that biologics manufacturers demonstrate potency as a measure of clinical usefulness. The PHS Act created the Laboratory of Biologics Control to facilitate testing and licensure of biologicals products and manufacturing establishments. After 1944, the authority of the Laboratory of Biologics Control was derived from Section 351 of the PHS Act and certain sections of the 1938 FD&C Act. In 1948, the Laboratory of Biologics Control joined the NIH Division of Infectious Diseases and Division of Tropical Diseases to form the National Microbiological Institute (later renamed the National Institute of Allergy and Infectious Diseases). The administrative authority for the regulation of biologics was originally granted to the National Microbiological Institute.
Although important regulations had been enacted to improve product safety, by the 1950s, the only legal requirement for vaccine licensing was the submission of written protocols for vaccine production and safety testing to the Laboratory of Biologics Control. Regulations were dramatically expanded in 1955, when more than 200 cases of polio were attributed to incompletely inactivated polio vaccine manufactured by the Cutter Laboratories. As a result of the “Cutter Incident,” administrative authority for the regulation of biologicals was transferred by Congress to the Division of Biologics Standards, a newly created division within the NIH. Regulations were strengthened that required more precise experimental testing to assess the safety of vaccines.
Congress passed the Consumer Safety Act of 1972 and transferred regulatory authority from NIH to FDA for the administration of the 1944 PHS Act. In 1972, the Division of Biologics Standards, which was charged with administering and enforcing Section 351 of the PHS Act, was transferred by the Secretary of Health, Education, and Welfare to the FDA, and became the Bureau of Biologics. Once administrative responsibility for the regulation of biologicals was transferred from NIH to FDA, the FDA announced its intention to require that all new biologicals satisfy the additional standards of safety and efficacy mandated in the Drug Amendments Act of 1962. This resulted in the transfer of the regulations pertaining to biologics from Part 73 of Chapter I of Title 42 (USC §262) to Chapter I of Title 21 of the CFR. In 1982, the Bureau of Biologics was renamed the Office of Biologics Research and Review and combined with the Office of Drugs Research and Review to form the Center for Drugs and Biologics. In 1987, following a series of organizational changes within the FDA, the Bureau of Biologics was ultimately transformed into the Center for Biologics Evaluation and Research (CBER). A chronology of the development of the U.S. Biologicals Control Authority is outlined in Fig. 80.1 .
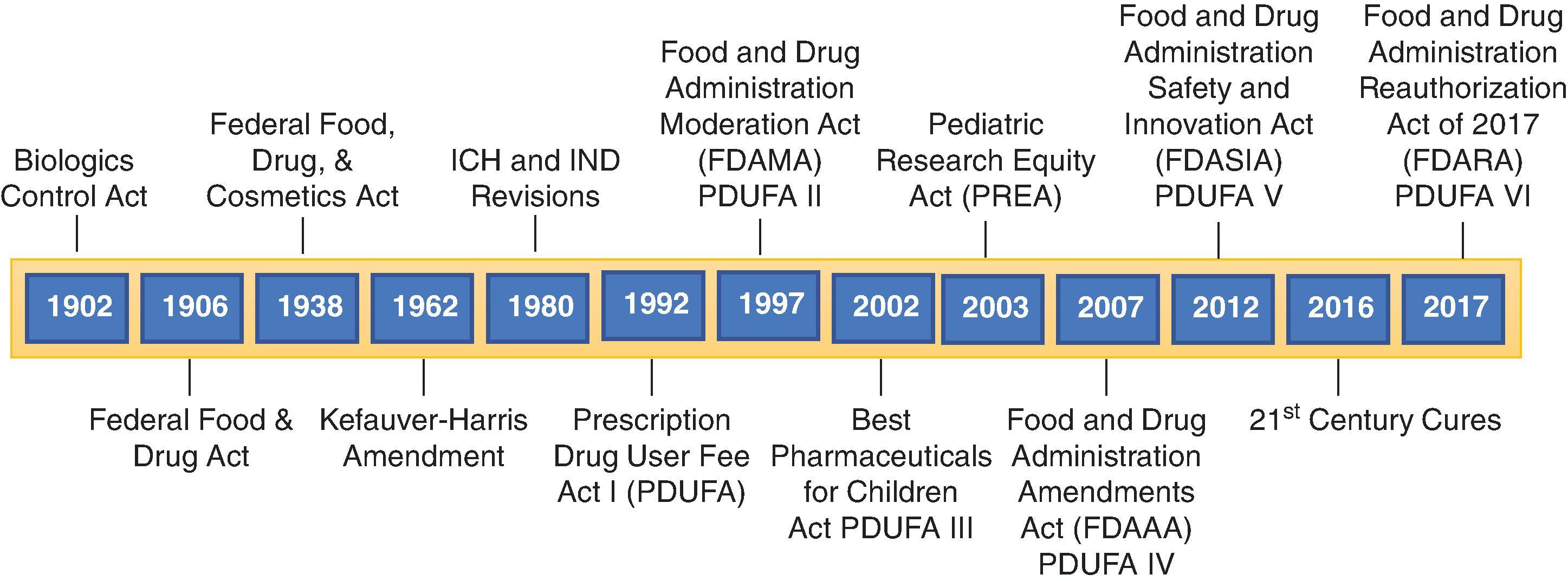
Since its inception, the FD&C Act has been amended by Congress several times, including the FDA Modernization Act (FDAMA) of 1997, the Pediatric Research Equity Act of 2003, the FDA Amendments Act (FDAAA) of 2007, the FDA Safety and Innovation Act (FDASIA) of 2012, the 21st Century Cures Act passed in 2016, and the FDA Reauthorization Act of 2017 (FDARA).
Among other things, the FDAMA of 1997 included measures to modernize the regulation of biologics by synchronizing their review process with that of drugs and eliminating the requirement for an establishment license for biologics. Expedited approval mechanisms for life-threatening conditions also were authorized under FDAMA.
The Pediatric Research Equity Act (PREA) of 2003 amended the FD&C Act by adding Section 505(B) to address product development for pediatric subjects from birth to 16 years of age. It requires that manufacturers submit a pediatric assessment with every application submitted under Section 505 of the FD&C Act or section 351 of the PHS Act for a new active ingredient, new indication, new dosage form, new dosing regimen, or new route of administration unless the applicant has obtained a waiver or deferral from the FDA. The pediatric assessment must contain data adequate to assess the safety and effectiveness of the drug or the biological product for the claimed indications in all relevant pediatric subpopulations and data to support dosing and administration for each pediatric subpopulation for which the product is safe and effective.
The FDAAA of 2007 includes 11 titles that added many new provisions to the FD&C Act. It reauthorized and amended several drug and medical device provisions and provided the FDA with additional responsibilities and new authorities. The provisions of FDAAA that have had a significant impact on the regulations of vaccines and the review process are contained in Title IV, the PREA, and Title IX, Enhanced Authorities Regarding Postmarket Safety of Drugs. The FDAAA reauthorized and revised the PREA, primarily to enhance FDA oversight and applicant accountability for the agreed-upon pediatric assessments. Of note, a new provision directed the FDA to establish the Pediatric Review Committee (PeRC), an internal review committee with pediatric expertise. This committee is required to provide consultation to FDA review divisions on all pediatric plans and assessments and on all deferral and waiver requests. Thus, early in the application review process the review team must assess whether PREA applies. If PREA applies, then a pediatric assessment must be presented to the PeRC.
Section 901 of Title IX of the FDAAA authorizes the FDA to require certain postmarketing studies and clinical trials for prescription drug and biological products approved under Section 505 of the FD&C Act or Section 351 of the PHS Act (42 USC §262). Section 901 of the FDAAA also created new Sections 505-1 and 505(o)(4) of the FD&C Act, authorizing the FDA, under certain circumstances, to require risk evaluation and mitigation strategies and safety-related labeling changes, respectively. The FDAAA also specifies adverse event reporting requirements for products with labeling changes that are a result of a pediatric assessment. Specifically, during the 12 months from the date that such a labeling change is made, all adverse event reports are reviewed by the FDA Pediatric Advisory Committee. Following review, the Pediatric Advisory Committee makes recommendations regarding whether the FDA should take action in response to such reports and whether the current pharmacovigilance plan is adequate.
The FDASIA was signed into law in 2012 and expanded the FDA’s authority by strengthening the agency’s ability to safeguard and advance public health by promoting innovation, increasing stakeholder involvement in FDA processes, and enhancing the safety of the drug supply chain. In addition to reauthorizing the prescription drug and medical device user fee programs, FDASIA established new user fee programs for generic drugs and biosimilar biological products. The provisions of FDASIA that impact the regulation of vaccines are contained in Pediatric Drugs and Devices (Title V) and Drug Approval and Patient Access (Title IX) . Title IX expands the scope of products that qualify for accelerated approval and creates a new “breakthrough therapy” program, among other things (see below). FDASIA also revised PREA to include a provision that requires vaccine manufacturers to submit a Pediatric Study Plan early in the drug development process. This initial Pediatric Study Plan must contain an outline of the pediatric study or studies that the sponsor plans to conduct, including, to the extent practicable, study objectives and design, age groups, relevant end points, and statistical approach, as well as any request for a deferral, partial waiver, or waiver. The FDA internal PeRC must be consulted for the review of the initial study plan, the agreed initial pediatric plan, and certain amendments to such plans. Both sponsors and the FDA must comply with prescribed timelines regarding submission, review, responses, and agreements reached regarding the Pediatric Study Plan, which are also described in applicable FDA guidance.
The 21st Century Cures Act (Cures Act) was signed into law in 2016 and strengthened the FDA’s ability to accelerate product development and promote innovative tools. The provisions of the Cures Act that impact the regulation of vaccines are contained in Title III (Development).
For example, the Cures Act revised and codified FDA’s qualification process to expedite the development of publicly available drug development tools (DDTs) and clinical outcome assessments. DDTs are methods, materials, or measures that can aid drug development and regulatory review, such as biomarkers. Once qualified, a DDT can be widely used across multiple drug and biologic development programs, including vaccines. The 21st Century Cures Act amends Chapter V of the Federal Food, Drug, and Cosmetic Act indicating that FDA “shall establish a program to evaluate the potential use of real world evidence (REW) (1) to help to support the approval of a new indication for a drug approved under section 505(c), and (2) to help to support or satisfy post-approval study requirements.” Although vaccines are licensed under the Public Health Service Act, the Office of Vaccines Research and Review (OVRR) at CBER has been contacted by regulated industry about the use of RWE to confirm the clinical benefit of vaccines and to support changes in labeling to include new comparative claims of vaccine effectiveness. In addition, evaluation of RWE may be essential as confirmatory effectiveness data to support accelerated approval of certain vaccines against other rare or emerging infectious diseases.
The 21st Century Cures Act defines “real-world evidence” as “data regarding the usage, or the potential benefits or risks, of a drug derived from sources other than randomized clinical trials.” This can include data derived from prospective and retrospective observational studies and registries and may include use of electronic health records (EHRs) or claims data.
The FDA Reauthorization Act of 2017 (FDARA), signed into law on August 18, 2017, amends the Federal Food, Drug, and Cosmetic Act to revise and extend the user-fee programs for drugs, medical devices, generic drugs, and biosimilar biological products. It contains nine titles, including those reauthorizing the four user fee programs through FY2022. Many provisions of FDARA are not directly applicable to preventive vaccines and thus, are not further described here. Of note, FDARA also amended Section 505A the FD&C Act to require the Secretary to review and act on a proposed pediatric study request or proposed amendment to a written request within 120 days of submission and to provide to the internal review committee any response to a proposed pediatric study request.
Public Law 115-92, enacted December 12, 2017, amended section 564 of the FD&C Act to allow for emergency uses of medical products for threats in addition to biological, chemical, radiological, or nuclear agent(s), to include other agents that may cause or are associated with, an imminently life-threatening and specific risk to the U.S. military forces. The legislation also authorized the Department of Defense (DoD) to request and FDA to provide assistance to expedite development and the FDA’s review of products to diagnose, treat, or prevent serious or life-threatening diseases or conditions pertaining to American military personnel. A joint program was launched by FDA and DoD on January 16, 2018, to prioritize the expedited development of safe and effective medical products including vaccines intended for deployed American military personnel.
Of note, these amendments to the FD&C Act also renewed the Prescription Drug User Fee Act (PDUFA) that was first enacted in 1992 and authorized the FDA to collect user fees from companies. These fees enabled FDA to hire additional reviewers and support staff and upgrade its information technology systems. In return for these additional resources, the FDA agreed to certain review performance goals, such as completing reviews of new drug applications and biologics license applications (BLAs) and taking regulatory actions on them in predictable timeframes. These changes revolutionized the drug approval process in the United States and enabled FDA to speed the application review process for new drugs and biologics without compromising the FDA’s high standards for demonstration of safety, efficacy, and quality. The PDUFA program has been reauthorized every 5 years, 1997 (PDUFA II), 2002 (PDUFA III), 2007 (PDUFA IV), 2012 (PDUFA V), and 2017 (PDUFA VI). It includes the 5-year review performance goals for drug and BLAs, supplements and resubmissions, meeting management goals, clinical holds, major dispute resolution, special protocol question assessment and agreement, electronic applications and submissions, discipline review, and complete response letters.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here